Isolation and Structural Elucidation of Antiproliferative Compounds of Lipidic Fractions from White Shrimp Muscle (Litopenaeus vannamei)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Lipidic Extraction and Partition
Antiproliferative Activity of Fractions Obtained by Partition
| Fraction | 400 µg/mL | 200 µg/mL | 100 µg/mL |
|---|---|---|---|
| Hexanic | 80.69 ± 4.62 a | 86.78 ± 10.13 a | 96.59 ± 6.94 a |
| Methanolic | 23.29 ± 5.61 b | 33.61 ± 13.8 b | 90.44 ± 8.27 a |
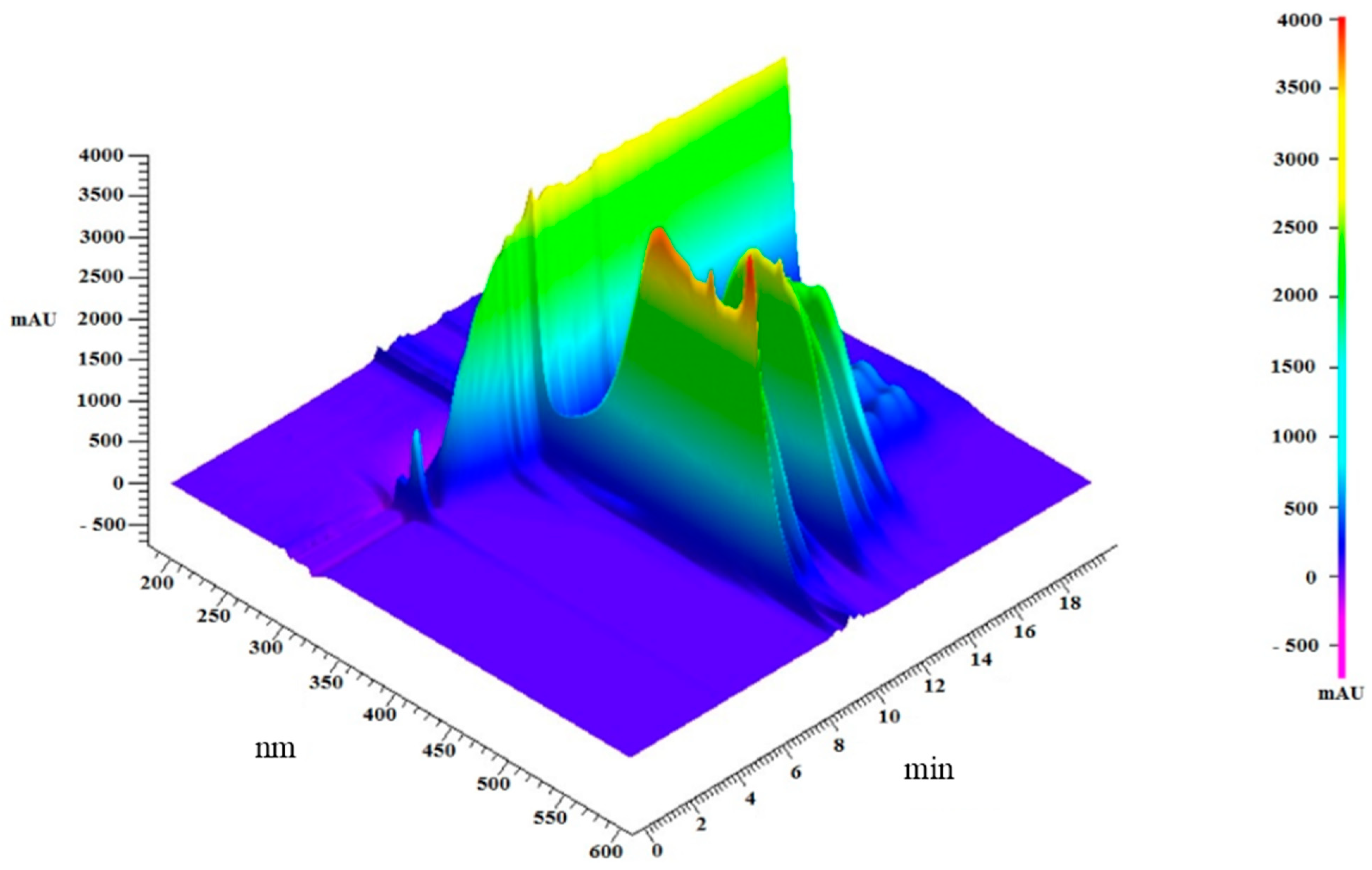
2.2. Analysis of Lipidic Composition by RP-HPLC
Antiproliferative Activity of Methanolic Sub-Fractions Obtained by Semi-Preparative RP-HPLC
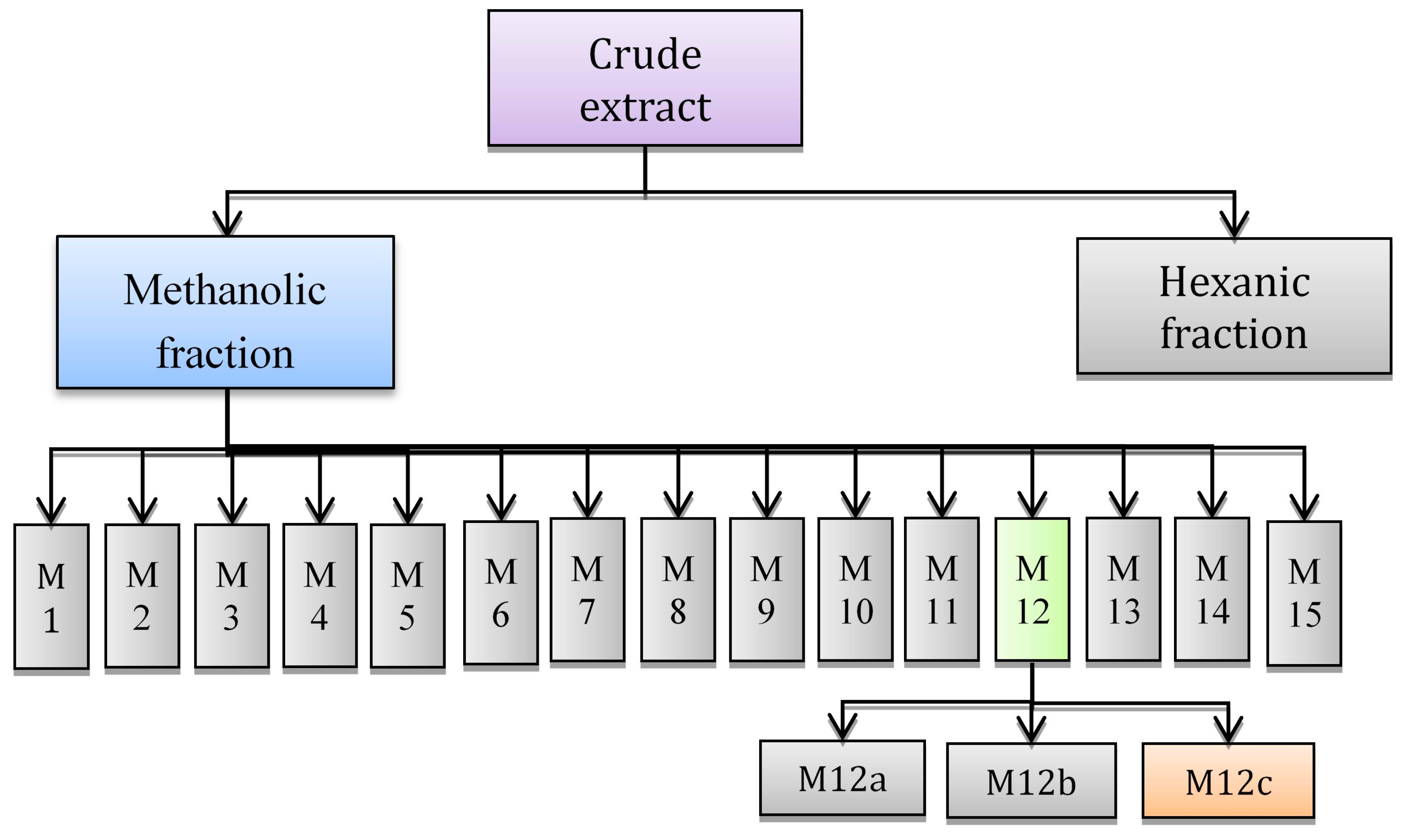
2.3. Fractioning by Open Column Chromatography

2.3.1. Chemical/Structural Characterization of M12 Fractions
| Fraction | 200 µg/mL | 100 µg/mL |
|---|---|---|
| M1 | 129.22 ±16.75 a | 113.60 ± 13.22 b |
| M2 | 133.53 ± 11.62 a | 118.22 ± 8.77 b |
| M3 | 129.76 ± 0.51 a | 120.17 ± 10.67 b |
| M4 | 98.06 ± 3.60 b | 117.86 ± 9.05 b |
| M5 | 2.004 ± 2.05 e | 74.61 ± 5.92 c,d |
| M6 | 3.402 ± 3.31 e | 69.14 ± 10.94 c,d |
| M7 | 57.72 ± 6.02 c,d | 171.56 ± 8.20 a |
| M8 | 6.379 ± 4.48 e | 16.16 ± 4.19 e,f |
| M9 | 2.19 ± 0.63 e | 3.28 ± 1.26 f |
| M10 | 6.20 ± 1.48 e | 16.76 ± 4.55 f |
| M11 | 13.00 ± 8.505 e | 44.83 ± 12.21 d,e |
| M12 | 2.67 ± 0.92 e | 2.79 ± 2.84 f |
| M13 | 8.51 ± 3.79 e | 22.84 ± 2.59 e,f |
| M14 | 67.63 ± 8.75 d | 105.95 ± 13.78 b,c |
| M15 | 97.93 ± 5.67 b | 136.19 ± 2.69 a,b |
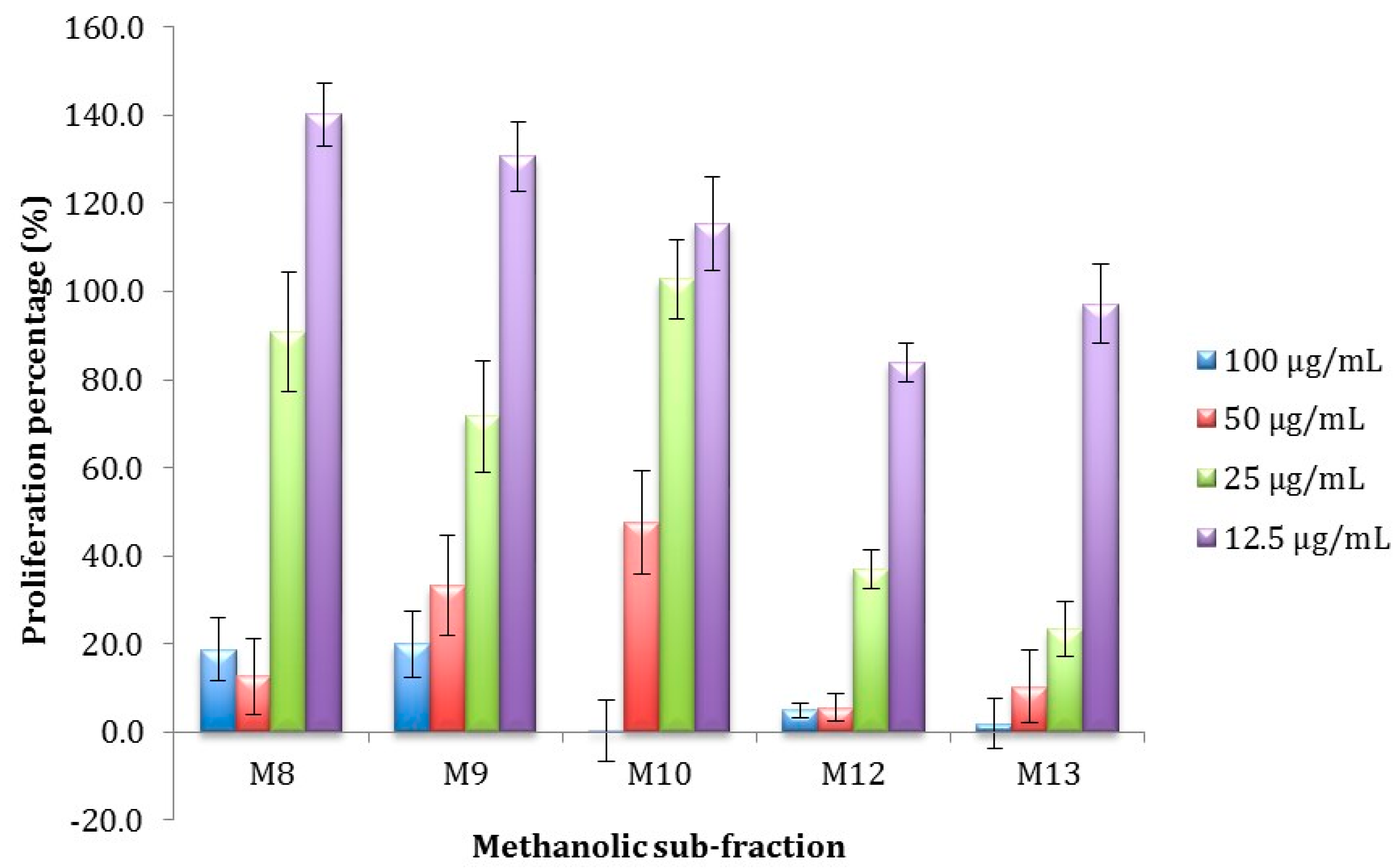
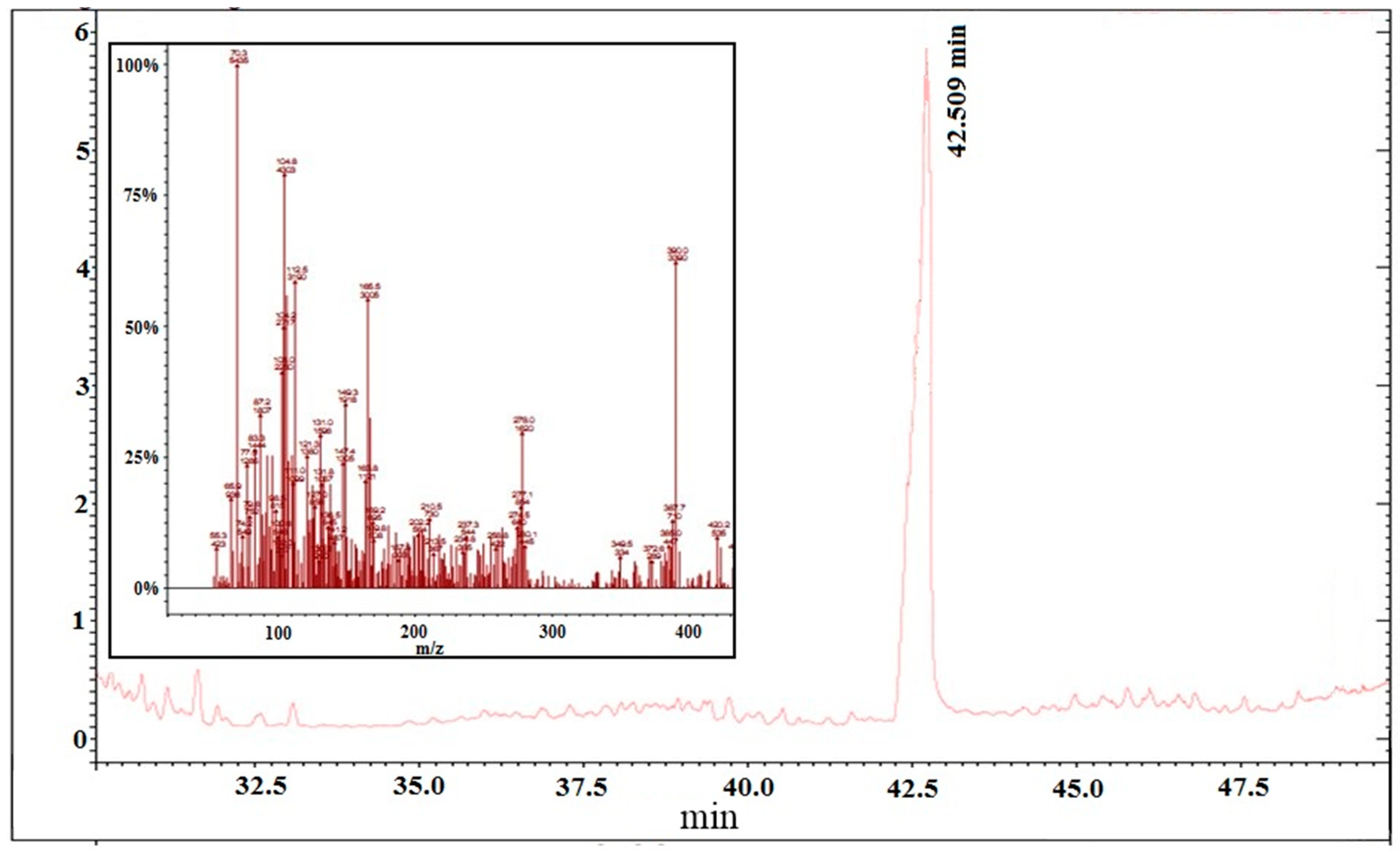
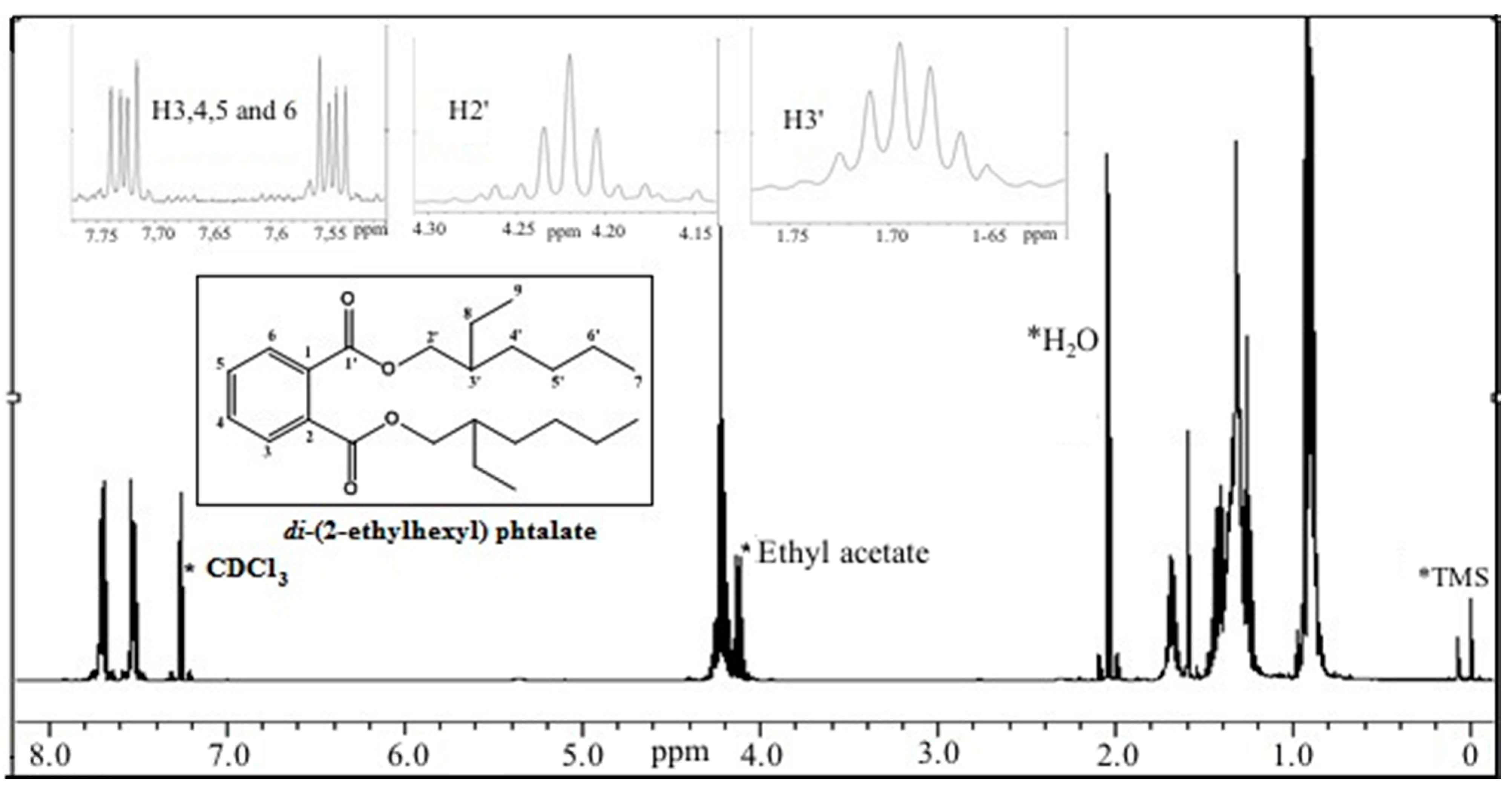
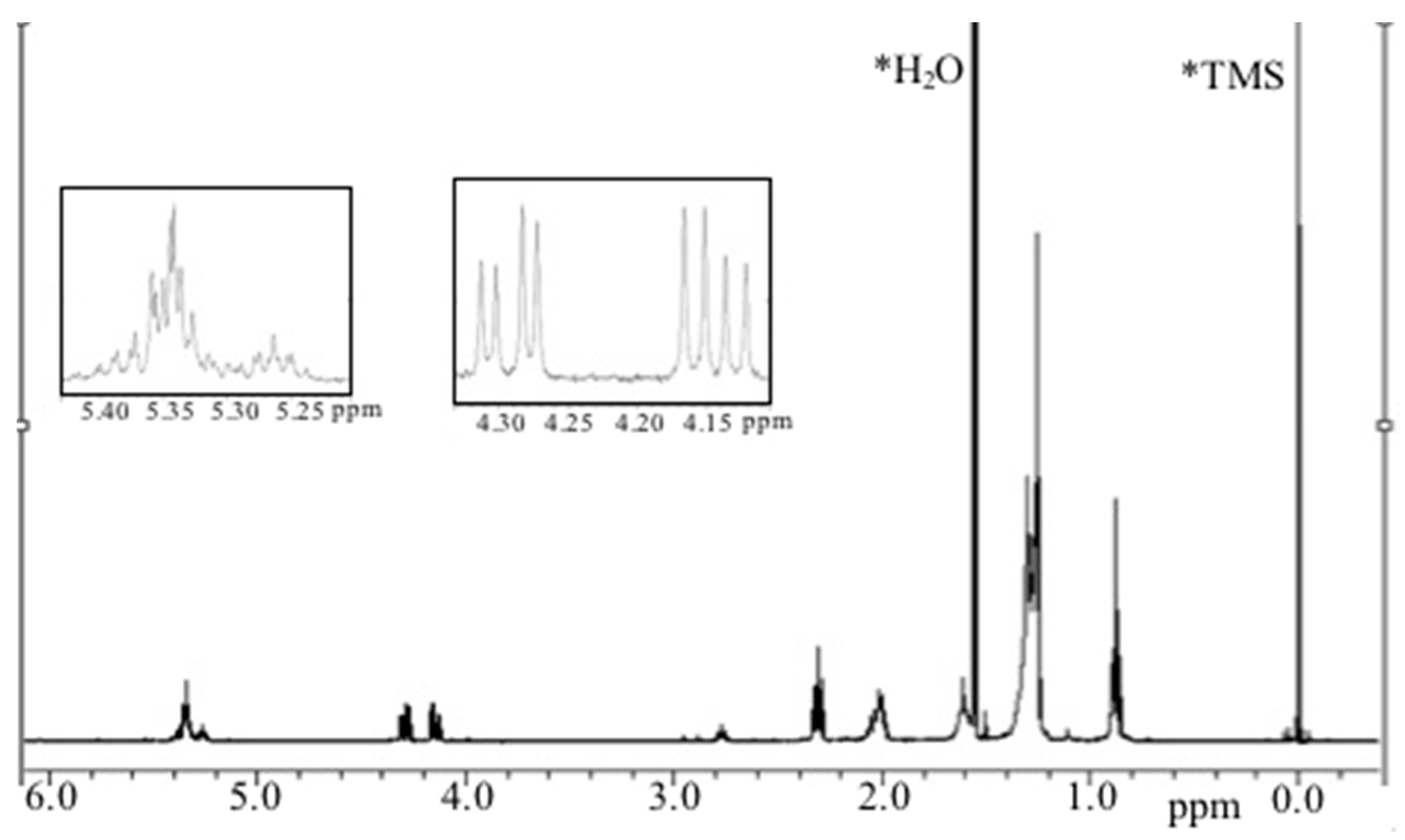
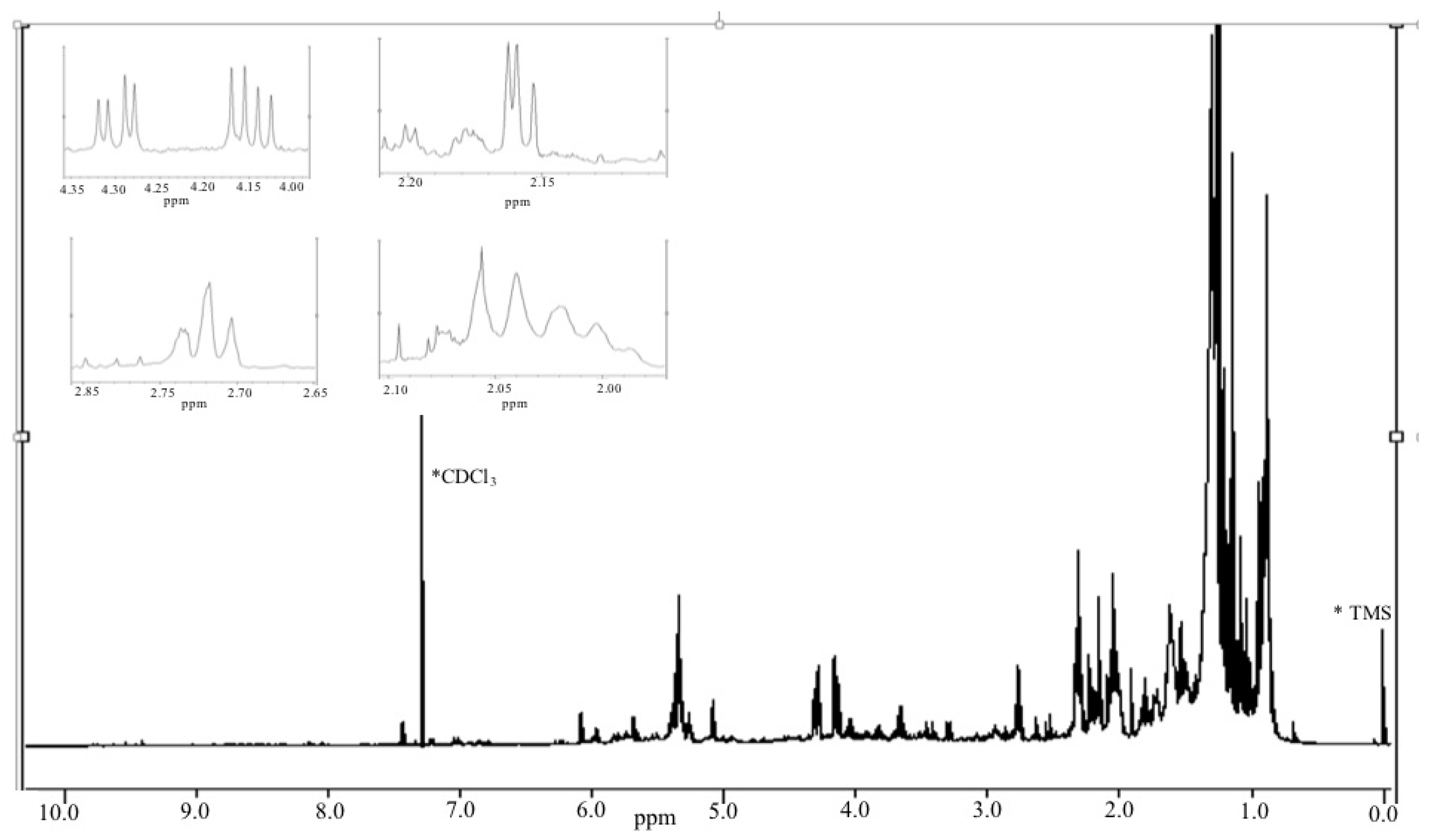
2.3.2. Antiproliferative Activity of M12 Fractions

3. Experimental Section
3.1. Testing Species
3.2. Analysis of Lipidic Composition by RP-HPLC
3.3. Isolation of Bioactive Component by Column Chromatography
3.4. Cell Lines
3.5. Antiproliferation Assay
3.6. 1H NMR Analysis
3.7. GC-MS Analysis
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nerurkar, P.; Ray, R.B. Bitter melon: Antagonist to cancer. Pharm. Res. 2010, 27, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; He, H.L.; Wang, G.F.; Wu, H.; Zhou, B.C.; Chen, X.L.; Zhang, Y.Z. Oyster (Crassostrea gigas) hydrolysates produced on a plant scale have antitumor activity and immunostimulating effects in BALB/c mice. Mar. Drugs 2010, 8, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Pelayo-Zaldivar, C. Las frutas y hortalizas como alimentos funcionales. Contactos 2003, 47, 12–19. [Google Scholar]
- Sporn, M.B.; Dunlop, N.M.; Newton, D.L.; Smith, J.M. Prevention of chemical carcinogenesis by vitamin a and its synthetic analogs (retinoids). Fed. Proc. 1976, 35, 1332–1338. [Google Scholar] [PubMed]
- Jemal, A.; Bray, F.; Center, M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Oksuz, A.; Ozylmaz, A.; Aktas, M.; Gercek, G.; Motte, J. A comparative study on proximate, mineral and fatty acid compositions of deep seawater rose shrimp (Parapenaus longirostris, Lucas 1846) and red shrimp (Plesionika martia, A. Milne-Edwards 1883). J. Anim. Vet. Adv. 2009, 8, 183–189. [Google Scholar]
- De Oliveira e Silva, E.R.; Seidman, C.E.; Tian, J.J.; Hudgins, L.C.; Sacks, F.M.; Breslow, J.L. Effects of shrimp consumption on plasma lipoproteins. Am. J. Clin. Nutr. 1996, 64, 712–717. [Google Scholar]
- Ezquerra-Brauer, J.M.; Brignas-Alvarado, L.; Burgos-Hernández, A.; Rouzaud-Sández, O. Control de la composición química y atributos de calidad de camarones cultivados. In Avances en Nutrición Acuícola VII; Universidad Autónoma de Nuevo León: Monterrey, Mexico, 2004; pp. 441–462. [Google Scholar]
- Cheng, H.C.; Chien, H.; Liao, C.H.; Yang, Y.Y.; Huang, S.Y. Carotenoids suppress proliferating cell nuclear antigen and cyclin D1 expression in oral carcinogenic models. J. Nutr. Biochem. 2007, 18, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Palozza, P.; Serini, S.; Maggiano, N.; Angelini, M.; Boninsegna, A.; di Nicuolo, F.; Ranelletti, F.O.; Calviello, G. Induction of cell cycle arrest and apoptosis in human colon adenocarcinoma cell lines by β-carotene through down-regulation of cyclin A and Bcl-2 family proteins. Carcinogenesis 2002, 23, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Sacha, T.; Zawada, M.; Hartwich, J.; Lach, Z.; Polus, A.; Szostek, M.; Zdziƚowska, E.; Libura, M.; Bodzioch, M.; Dembińska-Kieć, A.; et al. The effect of β-carotene and its derivatives on cytotoxicity, differentiation, proliferative potential and apoptosis on the three human acute leukemia cell lines: U-937, HL-60 and TF-1. Biochim. Biophys. Acta 2005, 1740, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Guruvayoorappan, C.; Kuttan, G. β-Carotene down-regulates inducible nitric oxide synthase gene expression and induces apoptosis by suppressing Bcl-2 expression and activating caspase-3 and p53 genes in B16F-10 melanoma cells. Nutr. Res. 2007, 27, 336–342. [Google Scholar] [CrossRef]
- Cui, Y.; Lu, Z.; Bai, L.; Shi, Z.; Zhao, W.; Zhao, B. β-Carotene induces apoptosis and up-regulates peroxisome proliferator-activated receptor γ expression and reactive oxygen species production in MCF-7 cancer cells. Eur. J. Cancer 2007, 43, 2590–2601. [Google Scholar] [CrossRef] [PubMed]
- Stivala, L.A.; Savio, M.; Cazzalini, O.; Pizzala, R.; Rehak, L.; Bianchi, L.; Vannini, V.; Prosperi, E. Effect of β-carotene on cell cycle progression of human fibroblasts. Carcinogenesis 1996, 17, 2395–2401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, W.; Hu, L.; Zhao, L.; Huang, J. Carotenoids inhibit proliferation and regulate expression of peroxisome proliferators-activated receptor gamma (PPARγ) in K562 cancer cells. Arch. Biochem. Biophys. 2011, 512, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Kumlin, M.; Ingelman-Sundberg, M.; Wolk, A. Dietary long-chain n-3 fatty acids for the prevention of cancer: A review of potential mechanisms. Am. J. Clin. Nutr. 2004, 79, 935–945. [Google Scholar] [PubMed]
- Mangelsdorf, D.J.; Thummel, C.; Beato, M.; Herrlich, P.; Schütz, G.; Umesono, K.; Blumberg, B.; Kastner, P.; Mark, M.; Chambon, P.; et al. The nuclear receptor superfamily: The second decade. Cell 1995, 83, 835–839. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.M.; Gonzalez, F.J. Regulation of squamous cell carcinoma carcinogenesis by peroxisome proliferator-activated receptors. In Signaling Pathways in Squamous Cancer; Glick, A.B., van Maes, C., Eds.; Springer: New York, NY, USA, 2011; pp. 223–240. [Google Scholar]
- Sánchez-Camargo, A.P.; Almeida Meireles, M.Â.; Lopes, B.L.F.; Cabral, F.A. Proximate composition and extraction of carotenoids and lipids from brazilian redspotted shrimp waste (Farfantepenaeus paulensis). J. Food Eng. 2011, 102, 87–93. [Google Scholar] [CrossRef]
- García, F.; González-Baró, M.; Pollero, R. Transfer of lipids between hemolymph and hepatopancreas in the shrimp Macrobrachium borellii. Lipids 2002, 37, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves Abreu, V.K.; Fernandes Pereira, A.L.; Fontoura Vidal, T.; Fuentes Zapata, J.F.; de Sousa Neto, M.A.; de Freitas, E.F. Fatty acids, cholesterol, oxidative rancidity, and color of irradiated shrimp. Food Sci. Technol. 2010, 30, 969–973. [Google Scholar]
- Johnston, J.J.; Ghanbari, H.A.; Wheeler, W.B.; Kirk, J.R. Characterization of shrimp lipids. J. Food Sci. 1983, 48, 33–35. [Google Scholar] [CrossRef]
- Chew, B.P.; Mathison, B.D.; Hayek, M.G.; Massimino, S.; Reinhart, G.A.; Park, J.S. Dietary astaxanthin enhances immune response in dogs. Vet. Immunol. Immunopathol. 2011, 140, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Arredondo-Figueroa, J.L.; Pedroza-Islas, R.; Ponce-Palafox, J.T.; Vernon-Carter, E.J. Pigmentation of pacific white shrimp (Litopenaeus vannamei, Boone 1931) with esterified and saponified carotenoids from red chili (Capsicum annuum) in comparison to astaxanthin. Revista Mexicana de Ingeniería Química 2003, 2, 101–108. [Google Scholar]
- Mezzomo, N.; Maestri, B.; dos Santos, R.L.; Maraschin, M.; Ferreira, S.R.S. Pink shrimp (P. brasiliensis and P. paulensis) residue: Influence of extraction method on carotenoid concentration. Talanta 2011, 85, 1383–1391. [Google Scholar]
- Babu, C.M.; Chakrabarti, R.; Surya Sambasivarao, K.R. Enzymatic isolation of carotenoid-protein complex from shrimp head waste and its use as a source of carotenoids. Food Sci. Technol. 2008, 41, 227–235. [Google Scholar]
- El-Sayed, M.H. di-(2-Ethylhexyl) phtalate, a major bioactive metabolite with antimicrobial and cytotoxic activity isolated from the culture filtrate of newly isolated soil streptomycin (Streptomices mirabilis strain NSQu-25). World Appl. Sci. J. 2012, 20, 1202–1212. [Google Scholar]
- Moushumi Priya, A.; Jayachandran, S. Induction of apoptosis and cell cycle arrest by bis-(2-ethylhexyl) phthalate produced by marine Bacillus pumilus MB 40. Chem. Biol. Interact. 2012, 195, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.N.; Laskar, S.; Sen, S.K. Dibutyl phthalate, the bioactive compound produced by Streptomyces albidoflavus 321.2. Microbiol. Res. 2006, 161, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Kim, J.H.; Lim, D.S.; Kim, C.H. Anti-leukaemic and anti-mutagenic effects of di-(2-ethylhexyl) phthalate isolated from Aloe vera linne. J. Pharm. Pharmacol. 2000, 52, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, M.; Abd-Alla, H.I.; Hassan, A.Z.; Aly, H.F.; Ghani, M.A. Chemical characterization, antioxidant and inhibitory effects of some marine sponges against carbohydrate metabolizing enzymes. Org. Med. Chem. Lett. 2012, 2, 30. [Google Scholar] [CrossRef] [PubMed]
- Bu, T.; Liu, M.; Zheng, L.; Guo, Y.; Lin, X. α-Glucosidase inhibition and the in vivo hypoglycemic effect of butyl-isobutyl-phthalate derived from the Laminaria japonica rhizoid. Phytother. Res. 2010, 24, 1588–1591. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.J.; Kang, K.H.; Kim, S.K. Isolation and antioxidant activity evaluation of two new phthalate derivatives from seahorse, Hippocampus kuda bleeler. Biotechnol. Bioprocess Eng. 2012, 17, 1031–1040. [Google Scholar] [CrossRef]
- Peropadre, A.; Fernández Freire, P.; Herrero, O.; Pérez Martín, J.M.; Hazen, M.J. Cytotoxic effects of di-(2-ethylhexyl) phthalate on cultured mammalian cells. Curr. Top. Toxicol. 2013, 9, 35–42. [Google Scholar]
- Krüger, T.; Cao, Y.; Kjærgaard, S.K.; Knudsen, L.E.; Bonefeld-Jørgensen, E.C. Effects of phthalates on the human corneal endothelial cell line B4G12. Int. J. Toxicol. 2012, 31, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Giam, C.S.; Chan, H.S.; Neff, G.S.; Atlas, E.L. Phthalate ester plasticizers: A new class of marine pollutant. Science 1978, 199, 419–421. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.M.; Green, P.G.; Higashi, R.M.; Young, T.M. Tidal salt marsh sediment in california, USA. Part 2: Occurrence and anthropogenic input of trace metals. Chemosphere 2006, 64, 1899–1909. [Google Scholar] [CrossRef] [PubMed]
- Adeniyi, A.A.; Okedeyi, O.O.; Yusuf, K.A. Flame ionization gas chromatographic determination of phthalate esters in water, surface sediments and fish species in the Ogun river catchments, Ketu, Lagos, Nigeria. Environ. Monit. Assess. 2011, 172, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Tyl, C.; Brecker, L.; Wagner, K. H-1 NMR spectroscopy as tool to follow changes in the fatty acids of fish oils. Eur. J. Lipid Sci. Technol. 2008, 110, 141–148. [Google Scholar] [CrossRef]
- Mika, A.; Gołębiowski, M.; Skorkowski, E.F.; Stepnowski, P. Composition of fatty acids and sterols composition in brown shrimp Crangon crangon and herring Clupea harengus membras from the Baltic sea. Int. J. Oceanogr. Hydrobiol. 2012, 41, 57–64. [Google Scholar] [CrossRef]
- Mao, X.; Liu, P.; He, S.; Xie, J.; Kan, F.; Yu, C.; Li, Z.; Xue, C.; Lin, H. Antioxidant properties of bio-active substances from shrimp head fermented by bacillus licheniformis OPL-007. Appl. Biochem. Biotechnol. 2013, 171, 1240–1252. [Google Scholar] [CrossRef] [PubMed]
- Sowmya, R.; Sachindra, N.M. Evaluation of antioxidant activity of carotenoid extract from shrimp processing byproducts by in vitro assays and in membrane model system. Food Chem. 2012, 134, 308–314. [Google Scholar] [CrossRef]
- Sindhu, S.; Sherief, P.M. Extraction, characterization, antioxidant and anti-inflammatory properties of carotenoids from the shell waste of arabian red shrimp Aristeus alcocki, ramadan 1938. Open Conf. Proc. J. 2011, 2, 95–103. [Google Scholar] [CrossRef]
- Mehrabian, S.; Shirkhodaei, E. Modulation of mutagenicity of various mutagens by shrimp flesh and skin extracts in salmonella test. Pak. J. Biol. Sci. 2006, 9, 598–600. [Google Scholar] [CrossRef]
- Wilson-Sanchez, G.; Moreno-Félix, C.; Velazquez, C.; Plascencia-Jatomea, M.; Acosta, A.; Machi-Lara, L.; Aldana-Madrid, M.L.; Ezquerra-Brauer, J.M.; Robles-Zepeda, R.; Burgos-Hernandez, A. Antimutagenicity and antiproliferative studies of lipidic extracts from white shrimp (Litopenaeus vannamei). Mar. Drugs 2010, 8, 2795–2809. [Google Scholar] [CrossRef] [PubMed]
- Chiu, L.C.; Wan, J.M. Induction of apoptosis in HL-60 cells by eicosapentaenoic acid (EPA) is associated with downregulation of Bcl-2 expression. Cancer Lett. 1999, 145, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.B.; Ross, J.A.; Fearon, K.C.; Anderson, J.D.; Carter, D.C. Cell cycle arrest and induction of apoptosis in pancreatic cancer cells exposed to eicosapentaenoic acid in vitro. Br. J. Cancer 1996, 74, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Hernandez, A.; Peña-Sarmiento, M.; Moreno-Ochoa, F. Mutagencity and antimutagencity studies of lipidic extracts from yellowtail fish (Seriola lalandi), lisa fish (Mugil cephalus) and cazón fish (Mustelus lunulatus). Food Chem. Toxicol. 2002, 40, 1469–1474. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.W.; Anke, H.; Davoli, P. Simple method for the extraction and reversed-phase high-performance liquid chromatographic analysis of carotenoid pigments from red yeasts (Basidiomycota, Fungi). J. Chromatogr. 2007, 1145, 118–122. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Saiz, C.-M.; Velázquez, C.; Hernández, J.; Cinco-Moroyoqui, F.-J.; Plascencia-Jatomea, M.; Robles-Sánchez, M.; Machi-Lara, L.; Burgos-Hernández, A. Isolation and Structural Elucidation of Antiproliferative Compounds of Lipidic Fractions from White Shrimp Muscle (Litopenaeus vannamei). Int. J. Mol. Sci. 2014, 15, 23555-23570. https://doi.org/10.3390/ijms151223555
López-Saiz C-M, Velázquez C, Hernández J, Cinco-Moroyoqui F-J, Plascencia-Jatomea M, Robles-Sánchez M, Machi-Lara L, Burgos-Hernández A. Isolation and Structural Elucidation of Antiproliferative Compounds of Lipidic Fractions from White Shrimp Muscle (Litopenaeus vannamei). International Journal of Molecular Sciences. 2014; 15(12):23555-23570. https://doi.org/10.3390/ijms151223555
Chicago/Turabian StyleLópez-Saiz, Carmen-María, Carlos Velázquez, Javier Hernández, Francisco-Javier Cinco-Moroyoqui, Maribel Plascencia-Jatomea, Maribel Robles-Sánchez, Lorena Machi-Lara, and Armando Burgos-Hernández. 2014. "Isolation and Structural Elucidation of Antiproliferative Compounds of Lipidic Fractions from White Shrimp Muscle (Litopenaeus vannamei)" International Journal of Molecular Sciences 15, no. 12: 23555-23570. https://doi.org/10.3390/ijms151223555




