Antimicrobial and Hypoglycemic Activities of Novel N-Mannich Bases Derived from 5-(1-Adamantyl)-4-substituted-1,2,4-triazoline-3-thiones
Abstract
:1. Introduction
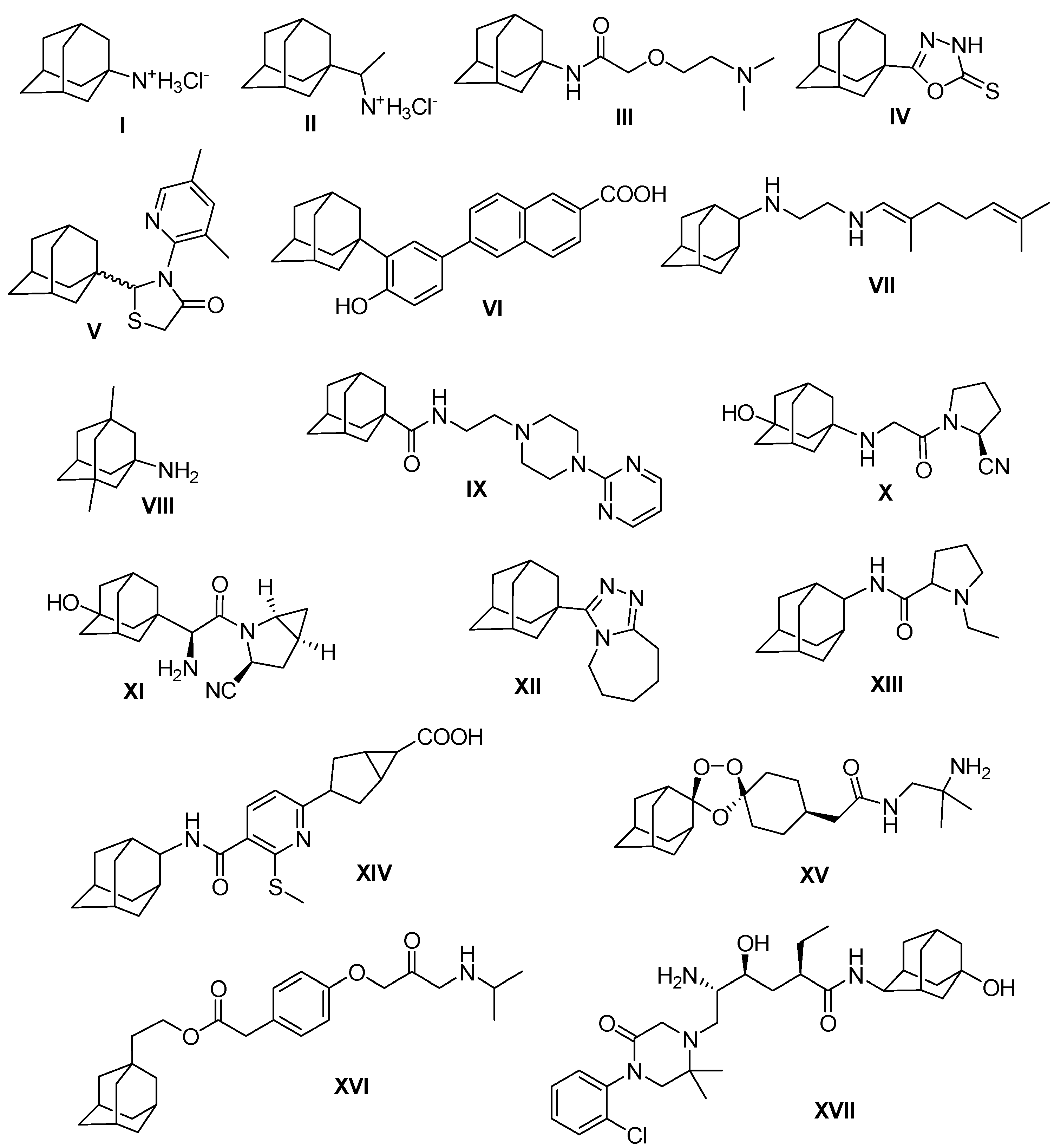
2. Results and Discussion
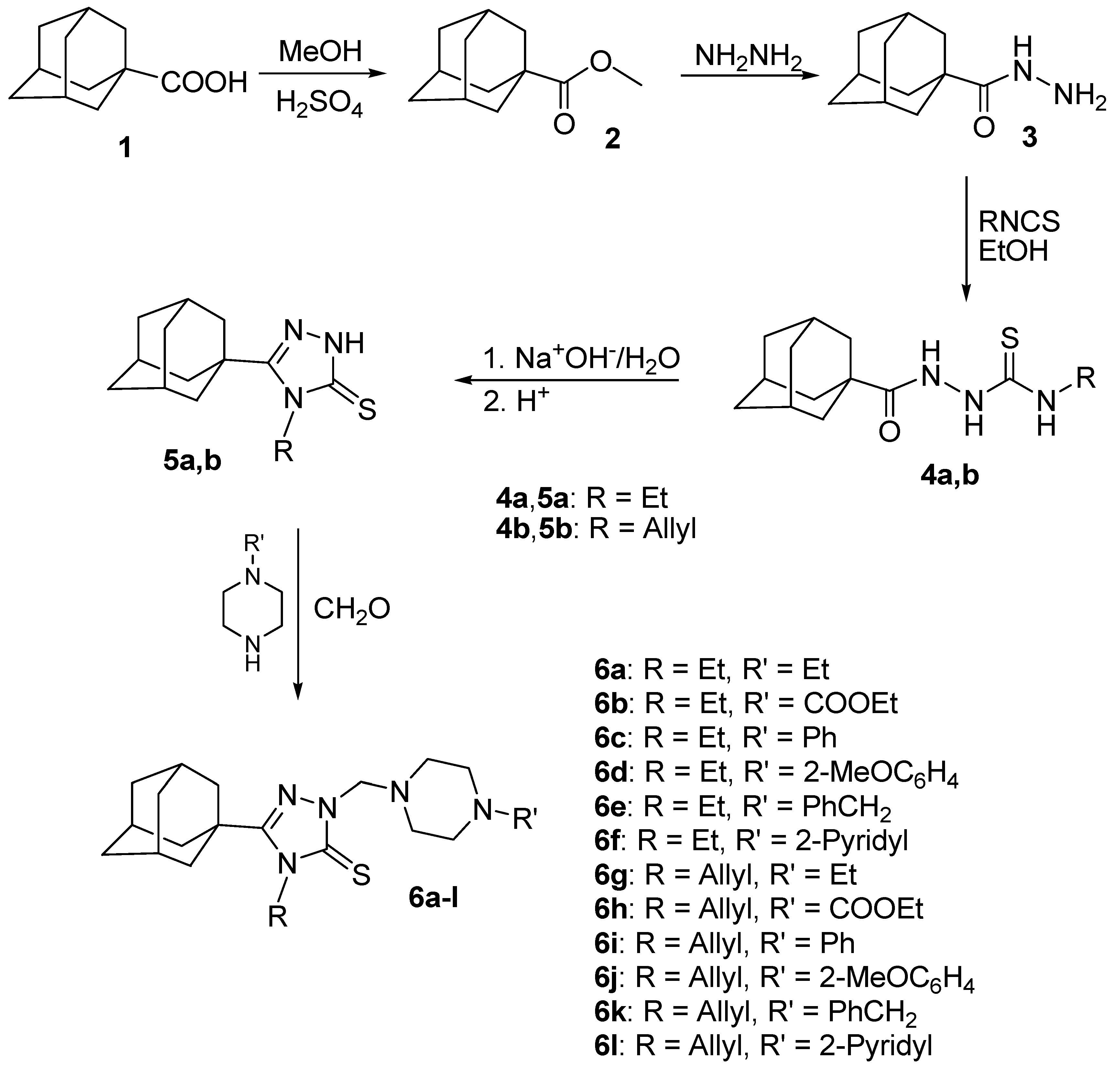
2.1. Chemistry
| Comp. No. | R | R' | Cryst. Solv. | m.p. (°C) | Yield (%) | Molecular Formula (Mol. Wt.) |
|---|---|---|---|---|---|---|
| 6a | C2H5 | C2H5 | EtOH/H2O | 158–160 | 77 | C21H35N5S (389.6) |
| 6b | C2H5 | COOC2H5 | EtOH/H2O | 153–155 | 72 | C22H35N5O2S (433.61) |
| 6c | C2H5 | C6H5 | EtOH | 139–141 | 88 | C25H35N5S (437.64) |
| 6d | C2H5 | 2-CH3OC6H4 | EtOH | 204–206 | 92 | C26H37N5OS (467.67) |
| 6e | C2H5 | C6H5CH2 | EtOH | 168–170 | 81 | C26H37N5S (451.67) |
| 6f | C2H5 | 2-Pyridyl | EtOH/H2O | 144–146 | 92 | C24H34N6S (438.63) |
| 6g | CH2=CHCH2 | C2H5 | EtOH/H2O | 109–111 | 66 | C22H35N5S (401.61) |
| 6h | CH2=CHCH2 | COOC2H5 | EtOH/H2O | 114–116 | 60 | C23H35N5O2S (445.62) |
| 6i | CH2=CHCH2 | C6H5 | EtOH | 161–163 | 69 | C26H35N5S (449.65) |
| 6j | CH2=CHCH2 | 2-CH3OC6H4 | EtOH | 208–210 | 74 | C27H37N5OS (479.68) |
| 6k | CH2=CHCH2 | C6H5CH2 | EtOH/H2O | 109–111 | 59 | C27H37N5S (463.68) |
| 6l | CH2=CHCH2 | 2-Pyridyl | EtOH/H2O | 133–135 | 66 | C25H34N6S (450.64) |
2.2. In Vitro Antimicrobial Activity
| Comp. No. | Clog p | Diameter of Growth Inhibition Zone (mm) a | |||||
|---|---|---|---|---|---|---|---|
| SA | BS | ML | EC | PA | CA | ||
| 6a | 3.96 | – | – | – | – | – | – |
| 6b | 5.51 | – | – | – | – | – | – |
| 6c | 5.67 | – | – | – | – | – | – |
| 6d | 5.69 | 15 | 17 | 11 | – | – | – |
| 6e | 6.59 | 21 (8) b | 20 (4) b | 17 | 14 | – | – |
| 6f | 4.72 | 15 | 18 (8) b | 11 | – | – | – |
| 6g | 4.21 | 11 | 13 | – | – | – | – |
| 6h | 5.76 | – | 12 | – | – | – | – |
| 6i | 5.92 | 14 | 16 | 12 | – | – | – |
| 6j | 5.82 | 19 (8) b | 18 (16) b | 18 (16) b | – | – | – |
| 6k | 6.84 | 28 (0.5) b | 30 (0.5) b | 19 (8) b | 18 (16) b | 12 | – |
| 6l | 4.97 | 17 | 19 (8) b | 10 | – | – | – |
| Gentamicin | 26 (2) b | 25 (2) b | 18 (2) b | 20 (0.5) b | 19 (1) b | NT | |
| Ampicillin | 23 (2) b | 21 (0.5) b | 19 (2) b | 17 (2) b | 16 (2) b | NT | |
| Clotrimazole | NT | NT | NT | NT | NT | 21 (2) b | |
2.3. In Vivo Hypoglycemic Activity
| Treatment | Results | ||
|---|---|---|---|
| C0 (mg/dL) a | C24 (mg/dL) a | % Glucose Reduction b | |
| Group 1 c | 302.6 ± 11.64 | 287.2 ± 16.85 | 5.09% |
| Group 2 d | 295.4 ± 17.52 | 183.0 ± 13.38 * | 38.05% |
| 6a (10 mg/kg) | 289.0 ± 18.05 | 204.4 ± 7.37 * | 29.27% (76.93%) |
| 6a (20 mg/kg) | 290.4 ± 10.60 | 188.0 ± 8.6 * | 35.26% (46.34%) |
| 6c (10 mg/kg) | 285.8 ± 14.31 | 241.6 ± 19.2 | 15.47% (40.66%) |
| 6c (20 mg/kg) | 297.6 ± 18.57 | 245.4 ± 11.54 | 17.54% (23.05%) |
| 6f (10 mg/kg) | 289.0 ± 14.35 | 197.8 ± 6.13 * | 31.56% (82.94%) |
| 6f (20 mg/kg) | 292.6 ± 10.33 | 189.0 ± 7.42 * | 35.41% (46.53%) |
| 6g (10 mg/kg) | 291.0 ± 5.58 | 190.6 ± 9.05 * | 34.50% (90.67%) |
| 6g (20 mg/kg) | 283.4 ± 10.45 | 184.0 ± 15.28 * | 35.07% (46.09%) |
| 6j (10 mg/kg) | 278.4 ± 15.07 | 228.4 ± 11.41 | 17.96% (47.20%) |
| 6j (20 mg/kg) | 297.4 ± 17.94 | 203.4 ± 15.28 * | 31.61% (41.54%) |
| 6l (10 mg/kg) | 290.4 ± 18.37 | 199.0 ± 18.75 * | 31.47% (82.71%) |
| 6l (20 mg/kg) | 295.4 ± 12.58 | 200.0 ± 18.78 * | 32.30% (42.45%) |
2.4. Oral Acute Toxicity Testing
| Comp. No. | LD50 * | LD50 (95% Confidence Limit) |
|---|---|---|
| 6a | 892 ± 38.70 | 892 (844–1089) |
| 6f | 787 ± 41.05 | 787 (721–923) |
| 6g | 833 ± 23.80 | 833 (765–891) |
| 6l | 741 ± 22.50 | 741 (624–806) |
3. Experimental Section
3.1. General
3.2. General Procedure for the Preparation of 5-(1-Adamantyl)-4-ethyl or allyl-2-(4-substituted piperazine-1-ylmethyl)-1,2,4-triazoline-3-thiones 6a–l
3.3. Determination of the in Vitro Antimicrobial Activity (Agar Disc-Diffusion Method)
3.4. Determination of the Minimal Inhibitory Concentration (MIC)
3.5. Determination of the in Vivo Hypoglycemic Activity
3.6. Determination of the Oral Acute Toxicity of Compounds 6a, 6f, 6g and 6l
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lamoureux, G.; Artavia, G. Use of the adamantane structure in medicinal chemistry. Curr. Med. Chem. 2010, 17, 2967–2978. [Google Scholar]
- Liu, J.; Obando, D.; Liao, V.; Lifa, T.; Codd, R. The many faces of the adamantyl group in drug design. Eur. J. Med. Chem. 2011, 46, 1949–1963. [Google Scholar]
- Davies, W.L.; Grunnert, R.R.; Haff, R.F.; McGahen, J.W.; Neumeyer, E.M.; Paulshock, M.; Watts, J.C.; Wood, T.R.; Hermann, E.C.; Hoffmann, C.E. Antiviral activity of 1-adamantamine (amantadine). Science 1964, 144, 862–863. [Google Scholar]
- Wendel, H.A.; Snyder, M.T.; Pell, S. Trial of amantadine in epidemic influenza. Clin. Pharmacol. Ther. 1966, 7, 38–43. [Google Scholar]
- Vernier, V.G.; Harmon, J.B.; Stump, J.M.; Lynes, T.E.; Marvel, J.P.; Smith, D.H. The toxicologic and pharmacologic properties of amantadine hydrochloride. Toxicol. Appl. Pharmacol. 1969, 15, 642–665. [Google Scholar]
- Schwab, R.S.; England, A.C., Jr.; Poskanzer, D.C.; Young, R.R. Amantadine in the treatment of Parkinson’s disease. J. Am. Med. Assoc. 1969, 208, 1168–1170. [Google Scholar]
- Hayden, F.G.; Gwaltney, J.M., Jr.; van de Castle, R.L.; Adams, K.F.; Giordani, B. Comparative toxicity of amantadine hydrochloride and rimantadine hydrochloride in healthy adults. Antimicrob. Agents Chemother. 1981, 19, 226–233. [Google Scholar]
- Scherm, A.; Peteri, D. Antiviral 1-(aminoethoxyacetylamino)adamantanes. Chem. Abstr. 1971, 74, 99516. [Google Scholar]
- Van Derpoorten, K.; Balzarini, J.; de Clercq, E.; Poupaert, J.H. Anti-HIV activity of N-1-adamantyl-4-aminophthalimide. Biomed. Pharmacother. 1997, 51, 464–468. [Google Scholar]
- Burstein, M.E.; Serbin, A.V.; Khakhulina, T.V.; Alymova, I.V.; Stotskaya, L.L.; Bogdan, O.P.; Manukchina, E.E.; Jdanov, V.V.; Sharova, N.K. Inhibition of HIV-1 replication by newly developed adamantane-containing polyanionic agents. Antivir. Res. 1999, 41, 135–144. [Google Scholar]
- El-Emam, A.A.; Al-Deeb, O.A.; Al-Omar, M.A.; Lehmann, J. Synthesis, antimicrobial, and anti-HIV-1 activity of certain 5-(1-adamantyl)-2-substituted thio-1,3,4-oxadiazoles and 5-(1-adamantyl)-3-substituted aminomethyl-1,3,4-oxadiazoline-2-thiones. Bioorg. Med. Chem. 2004, 12, 5107–5113. [Google Scholar]
- Balzarini, J.; Orzeszko, B.; Mauri, J.K.; Orzeszko, A. Synthesis and anti-HIV studies of 2-adamantyl-substituted thiazolidin-4-ones. Eur. J. Med. Chem. 2007, 42, 993–1003. [Google Scholar]
- Sun, S.Y.; Yue, P.; Chen, X.; Hong, W.K.; Lotan, R. The synthetic retinoid CD437 selectively induces apoptosis in human lung cancer cells while sparing normal human lung epithelial cells. Cancer Res. 2002, 62, 2430–2436. [Google Scholar]
- Wang, J.J.; Wang, S.S.; Lee, C.F.; Chung, M.A.; Chern, Y.T. In vitro antitumor and antimicrobial activity of N-substituents of maliemide by adamantane and diamantane. Chemotherapy 1997, 43, 182–189. [Google Scholar]
- Antoniadou-Vyza, E.; Tsitsa, P.; Hytiroglou, E.; Tsantili-Kakoulidou, A. New adamantan-2-ol and adamantan-1-methanol derivatives as potent antibacterials, synthesis, antibacterial activity and lipophilicity studies. Eur. J. Med. Chem. 1996, 31, 105–110. [Google Scholar]
- Omar, K.; Geronikaki, A.; Zoumpoulakis, P.; Camoutsis, C.; Soković, M.; Ćirić, A.; Glamoćlija, J. Novel 4-thiazolidinone derivatives as potential antifungal and antibacterial drugs. Bioorg. Med. Chem. 2010, 18, 426–432. [Google Scholar]
- Scherman, M.S.; North, E.J.; Jones, V.; Hess, T.N.; Grzegorzewicz, A.E.; Kasagami, T.; Kim, I.H.; Merzlikin, O.; Lenaerts, A.J.; Lee, R.E.; et al. Screening a library of 1600 adamantyl ureas for anti-Mycobacterium tuberculosis activity in vitro and for better physical chemical properties for bioavailability. Bioorg. Med. Chem. 2012, 20, 3255–3262. [Google Scholar]
- El-Emam, A.A.; Al-Tamimi, A.-M.S.; Al-Omar, M.A.; Al-Rashood, K.A.; Habib, E.E. Synthesis and antimicrobial activity of novel 5-(1-adamantyl)-2-aminomethyl-4-substituted-1,2,4-triazoline-3-thiones. Eur. J. Med. Chem. 2013, 68, 96–102. [Google Scholar]
- Al-Deeb, O.A.; Al-Omar, M.A.; El-Brollosy, N.R.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activities of novel 2-[3-(1-adamantyl)-4-substituted-5-thioxo-1,2,4-triazolin-1-yl]acetic acids, 2-[3-(1-adamantyl)-4-substituted-5-thioxo-1,2,4-triazolin-1-yl]propionic acids and related derivatives. Arzneim.-Forsch. 2006, 56, 40–47. [Google Scholar]
- Kadi, A.A.; El-Brollosy, N.R.; Al-Deeb, O.A.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activities of novel 2-(1-adamantyl)-5-substituted-1,3,4-oxadiazoles and 2-(1-adamantylamino)-5-substituted-1,3,4-thiadiazoles. Eur. J. Med. Chem. 2007, 42, 235–242. [Google Scholar]
- Kadi, A.A.; Al-Abdullah, E.S.; Shehata, I.A.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial and anti-inflammatory activities of novel 5-(1-adamantyl)-1,3,4-thiadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5006–5011. [Google Scholar]
- Al-Abdullah, E.S.; Asiri, H.H.; Lahsasni, S.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activity, of novel S-substituted and N-substituted 5-(1-adamantyl)-1,2,4-triazole-3-thiols. Drug Des. Dev. Ther. 2014, 8, 505–518. [Google Scholar]
- Jia, L.; Tomaszewski, J.E.; Hanrahan, C.; Coward, L.; Noker, P.; Gorman, G.; Nikonenko, B.; Protopopova, M. Pharmacodynamics and pharmacokinetics of SQ109, a new diamine-based antitubercular drug. Br. J. Pharmacol. 2005, 144, 80–87. [Google Scholar]
- Protopopova, M.; Hanrahan, C.; Nikonenko, B.; Samala, R.; Chen, P.; Gearhart, J.; Einck, L.; Nacy, C.A. Identification of a new antitubercular drug candidate, SQ109, from a combinatorial library of 1,2-ethylenediamines. J. Antimicrob. Chemother. 2005, 56, 968–974. [Google Scholar]
- Bormann, J. Memantine is a potent blocker of N-methyl-d-aspartate (NMDA) receptor channels. Eur. J. Pharmacol. 1989, 166, 591–592. [Google Scholar]
- Abou-Gharbia, M.A.; Childers, W.E.; Fletcher, H.; McGaughey, G.; Patel, U.; Webb, M.B.; Yardley, J.; Andree, T.; Boast, C.; Kucharik, R.J.; et al. Synthesis and SAR of adatanserin: Novel adamantly aryl- and heteroarylpiperazines with dual serotonin 5-HT1A and 5-HT2 activity as potential anxiolytic and antidepressant agents. J. Med. Chem. 1999, 42, 5077–5094. [Google Scholar]
- Villhauer, E.B.; Brinkman, J.A.; Naderi, G.B.; Burkey, B.F.; Dunning, B.E.; Prasad, K.; Mangold, B.L.; Russell, M.E.; Hughes, T.E. 1-(3-Hydroxy-1-adamantyl)aminoacetyl-2-cyano-(S)-pyrrolidine: A potent, selective, and orally bioavailable dipeptidyl peptidase IV inhibitor with antihyperglycemic properties. J. Med. Chem. 2003, 46, 2774–2789. [Google Scholar]
- Augeri, D.J.; Robl, J.A.; Betebenner, D.A.; Magnin, D.R.; Khanna, A.; Robertson, J.G.; Wang, A.; Simpkins, L.M.; Taunk, P.; Huang, Q.; et al. Discovery and preclinical profile of saxagliptin (BMS-477118): A highly potent, long-acting, orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. J. Med. Chem. 2005, 48, 5025–5037. [Google Scholar]
- Ahrén, B.; Landin-Olsson, M.; Jansson, P.A.; Svensson, M.; Holmes, D.; Schweizer, A. Inhibition of dipeptidyl peptidase-4 reduces glycemia, sustains insulin levels, and reduces glucagon levels in type 2 diabetes. J. Clin. Endocrinol. Metab. 2004, 89, 2078–2084. [Google Scholar]
- Joharapurkar, A.; Dhanesha, N.; Shah, G.; Kharul, R.; Jain, M. 11β-Hydroxysteroid dehydrogenase type 1: Potential therapeutic target for metabolic syndrome. Pharmacol. Rep. 2012, 64, 1055–1065. [Google Scholar]
- Anagnostis, P.; Katsiki, N.; Adamidou, F.; Athyros, V.G.; Karagiann, A.; Kita, M.; Mikhailidis, D.P. 11β-Hydroxysteroid dehydrogenase type 1 inhibitors: Novel agents for the treatment of metabolic syndrome and obesity-related disorders. Metabolism 2013, 62, 21–33. [Google Scholar]
- Olson, S.; Aster, S.D.; Brown, K.; Carbin, L.; Graham, D.W.; Hermanowski-Vosatka, A.; LeGrand, C.B.; Mundt, S.S.; Robbins, M.A.; Schaeffer, J.M.; et al. Adamantyl triazoles as selective inhibitors of 11β-hydroxysteroid dehydrogenase type 1. Bioorg. Med. Chem. Lett. 2005, 15, 4359–4362. [Google Scholar]
- Cheng, H.; Hoffman, J.; Le, P.; Nair, S.K.; Cripps, S.; Matthews, J.; Smith, C.; Yang, M.; Kupchinsky, S.; Dress, K.; et al. The development and SAR of pyrrolidine carboxamide 11β-HSD1 inhibitors. Bioorg. Med. Chem. Lett. 2010, 20, 2897–2902. [Google Scholar]
- Cott, J.S.; Choormun, J. 11β-Hydroxysteroid dehydrogenase type 1 (11β-HSD-1) inhibitors in developmen. In New Therapeutic Strategies for Type-2 Diabetes, Small Molecule Approaches; Jones, R.N., Ed.; Royal Society Chemistry: Cambridge, UK, 2012; pp. 109–133. [Google Scholar]
- El-Emam, A.A.; Ibrahim, T.M. Synthesis, anti-inflammatory and analgesic activity of certain 3-(1-adamantyl)-4-substituted-5-mercapto-1,2,4-triazole derivatives. Arzneim.-Forsch. 1991, 41, 1260–1264. [Google Scholar]
- Kouatly, O.; Geronikaki, A.; Kamoutsis, C.; Hadjipavlou-Litina, D.; Eleftheriou, P. Adamantane derivatives of thiazolyl-N-substituted amide, as possible non-steroidal anti-inflammatory agents. Eur. J. Med. Chem. 2009, 44, 1198–1204. [Google Scholar]
- Baxter, A.; Bent, J.; Bowers, K.; Braddock, M.; Brough, S.; Fagura, M.; Lawson, M.; McInally, T.; Mortimore, M.; Robertson, M.; et al. Hit-to-lead studies: The discovery of potent adamantane amide P2X7 receptor antagonists. Bioorg. Med. Chem. Lett. 2003, 13, 4047–4050. [Google Scholar]
- Lee, W.G.; Lee, S.D.; Cho, J.H.; Jung, Y.; Kim, J.H.; Hien, T.T.; Kang, K.W.; Ko, H.; Kim, Y.C. Structure-activity relationships and optimization of 3,5-dichloropyridine derivatives as novel P2X7 receptor antagonists. J. Med. Chem. 2012, 55, 3687–3698. [Google Scholar]
- Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S.A.; Charman, W.N.; Scheurer, C.; Urwyler, H.; Tomas, J.S.; Snyder, C.; et al. The structure-activity relationship of the antimalarial ozonide arterolane (OZ277). J. Med. Chem. 2010, 53, 481–491. [Google Scholar]
- Boder, N.; Elkoussi, A.; Zuobi, K.; Kovacs, P. Synthesis and pharmacological activity of adaprolol enantiomers: A new soft drug for treating glaucoma. J. Ocul. Pharmacol. Ther. 1996, 12, 115–122. [Google Scholar]
- Nakamura, Y.; Fujimoto, T.; Ogawa, Y.; Namiki, H.; Suzuki, S.; Asano, M.; Sugita, C.; Mochizuki, A.; Miyazaki, S.; Tamaki, K.; et al. Lead optimization of 5-amino-6-(2,2-dimethyl-5-oxo-4-phenylpiperazin-1-yl)-4-hydroxyhexanamides to reduce a cardiac safety issue: Discovery of DS-8108b, an orally active renin inhibitor. Bioorg. Med. Chem. 2013, 21, 3175–3196. [Google Scholar]
- El-Emam, A.A.; Al-Abdullah, E.S.; Al-Tuwaijri, H.M.; Said-Abdelbaky, M.; García-Granda, S. 3-(Adamantan-1-yl)-4-ethyl-1-[(4-phenylpiperazin-1-yl)methyl]-1H-1,2,4-triazole-5(4H)-thione. Acta Cryst. 2012, E68, o2380–o2381. [Google Scholar]
- El-Emam, A.A.; Al-Tuwaijri, H.M.; Al-Abdullah, E.S.; Chidan Kumar, C.S.; Fun, H.K. 3-(Adamantan-1-yl)-4-ethyl-1-{[(4-(2-methoxyphenylpiperazin-1-yl)]methyl}-1H-1,2,4-triazole-5(4H)-thione. Acta Cryst. 2014, E70, o25–o26. [Google Scholar]
- Al-Abdullah, E.S.; Al-Tuwaijri, H.M.; El-Emam, A.A.; Chidan Kumar, C.S.; Fun, H.K. 3-(Adamantan-1-yl)-1-[(4-benzylpiperazin-1-yl)]methyl]-4-ethyl-1H-1,2,4-triazole-5(4H)-thione. Acta Cryst. 2013, E69, o1813–o1814. [Google Scholar]
- Woods, G.L.; Washington, J.A. Antibacterial susceptibility tests: Dilution and disk diffusion methods. In Manual of Clinical Microbiology; Murray, P.R., Baron, E.J., Pfaller, M.A., Tenover, F.C., Yolken, R.H., Eds.; American Society of Microbiology: Washington, DC, USA, 1995. [Google Scholar]
- National Committee for Clinical Laboratory Standards. In Approved Standard Document M-7A; NCCLS: Villanova, PA, USA, 1985.
- Rossini, A.A.; Like, A.A.; Chick, W.L.; Appel, M.C.; Cahill, G.F. Studies of streptozotocin-induced insulitis and diabetes. Proc. Nat. Accad. Sci. USA 1977, 74, 2485–2489. [Google Scholar]
- Olajide, O.A.; Awe, S.O.; Makinde, J.M.; Morebise, O. Evaluation of the antidiabetic property of Morinda lucida leaves in streptozotocin diabetes rats. J. Pharm. Pharmacol. 1999, 51, 1321–1324. [Google Scholar]
- Litchfield, J.T.; Wilcoxon, T. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 1949, 96, 99–104. [Google Scholar]
- Serradas, P.; Bailbe, D.; Portha, B. Long-term gIiclazide treatment improves the in vitro glucose-induced insulin release in rats with type 2 (non-insulin-dependent) diabetes induced by neonatal streptozotocin. Diabetologia 1989, 55, 55–57. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Abdullah, E.S.; Al-Tuwaijri, H.M.; Hassan, H.M.; Haiba, M.E.; Habib, E.E.; El-Emam, A.A. Antimicrobial and Hypoglycemic Activities of Novel N-Mannich Bases Derived from 5-(1-Adamantyl)-4-substituted-1,2,4-triazoline-3-thiones. Int. J. Mol. Sci. 2014, 15, 22995-23010. https://doi.org/10.3390/ijms151222995
Al-Abdullah ES, Al-Tuwaijri HM, Hassan HM, Haiba ME, Habib EE, El-Emam AA. Antimicrobial and Hypoglycemic Activities of Novel N-Mannich Bases Derived from 5-(1-Adamantyl)-4-substituted-1,2,4-triazoline-3-thiones. International Journal of Molecular Sciences. 2014; 15(12):22995-23010. https://doi.org/10.3390/ijms151222995
Chicago/Turabian StyleAl-Abdullah, Ebtehal S., Hanaa M. Al-Tuwaijri, Hanan M. Hassan, Mogedda E. Haiba, Elsayed E. Habib, and Ali A. El-Emam. 2014. "Antimicrobial and Hypoglycemic Activities of Novel N-Mannich Bases Derived from 5-(1-Adamantyl)-4-substituted-1,2,4-triazoline-3-thiones" International Journal of Molecular Sciences 15, no. 12: 22995-23010. https://doi.org/10.3390/ijms151222995
APA StyleAl-Abdullah, E. S., Al-Tuwaijri, H. M., Hassan, H. M., Haiba, M. E., Habib, E. E., & El-Emam, A. A. (2014). Antimicrobial and Hypoglycemic Activities of Novel N-Mannich Bases Derived from 5-(1-Adamantyl)-4-substituted-1,2,4-triazoline-3-thiones. International Journal of Molecular Sciences, 15(12), 22995-23010. https://doi.org/10.3390/ijms151222995




