MiR-222 Targeted PUMA to Improve Sensitization of UM1 Cells to Cisplatin
Abstract
:1. Introduction
2. Results
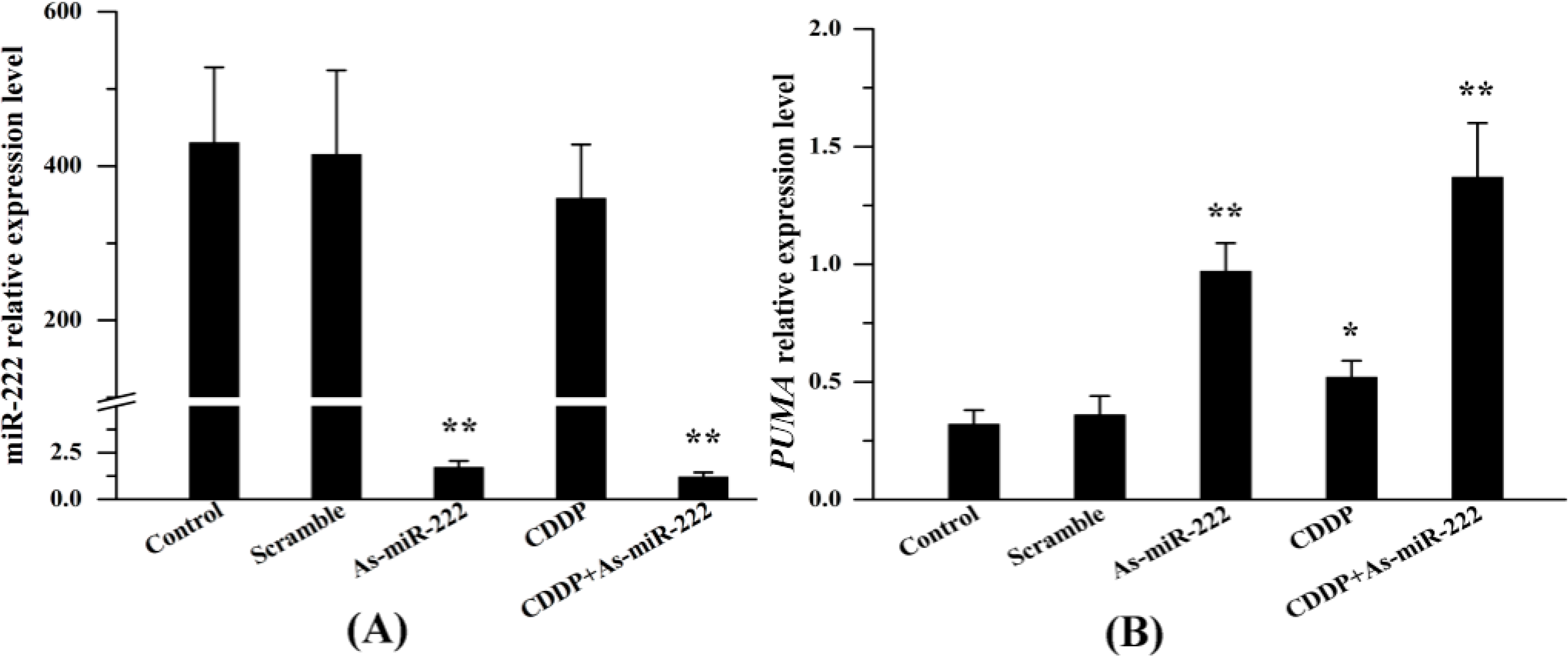
2.1. miR-222 and PUMA Expression in UM1 Cells Treated with Combination Therapy
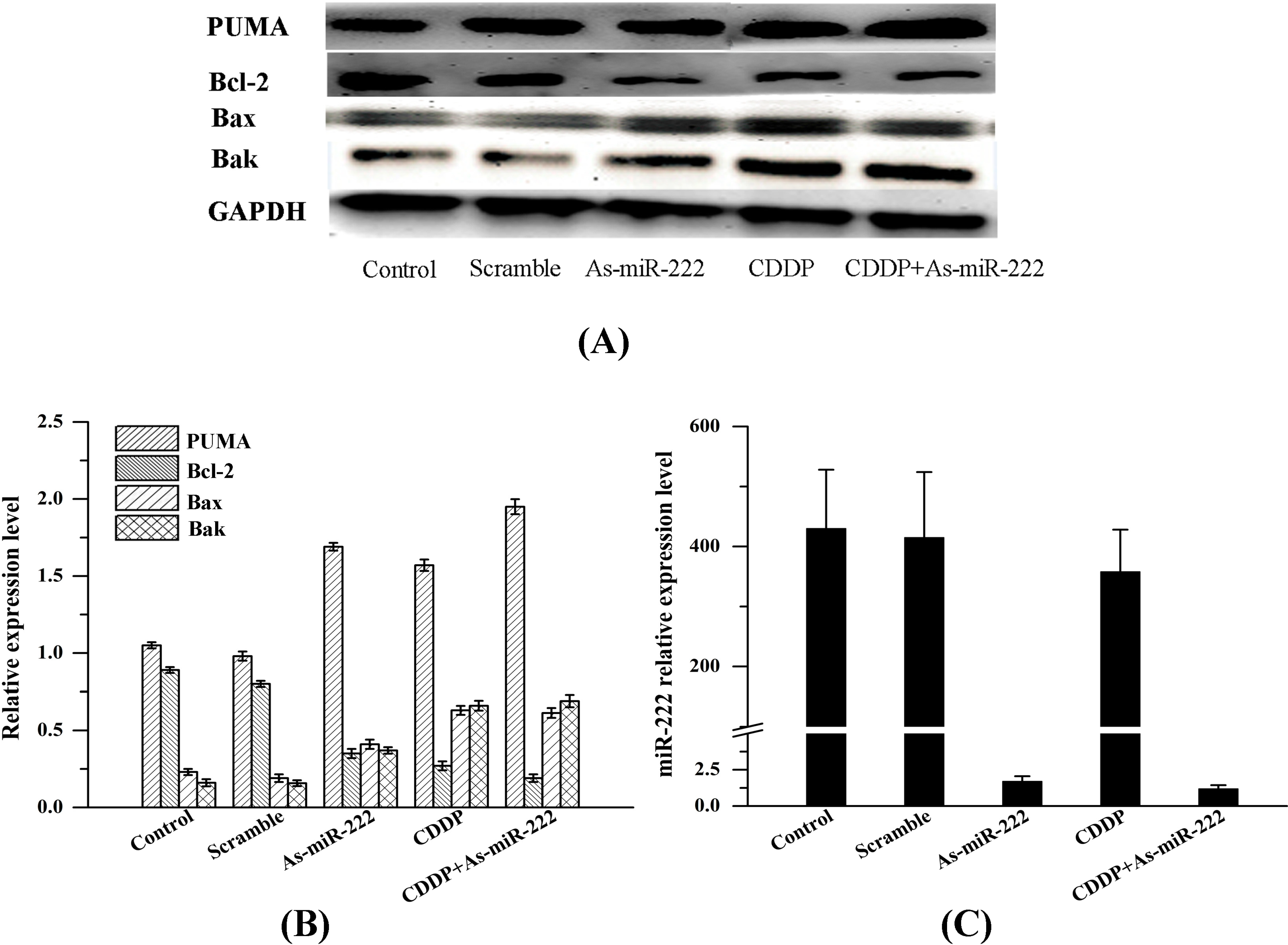
2.2. As-miR-222 and CDDP Alters Apoptotic Protein Expression
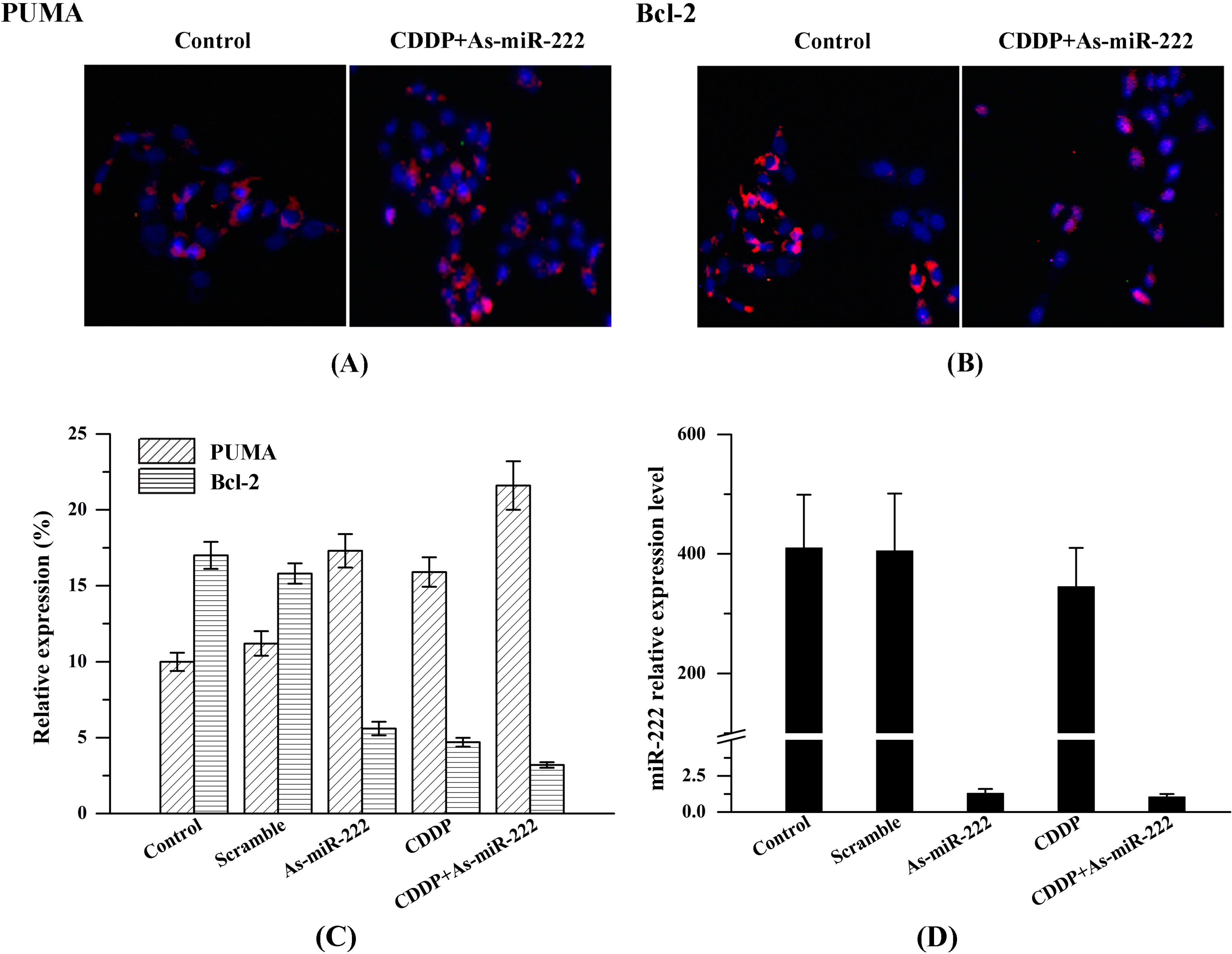
2.3. Determination of PUMA and Bcl-2 Expression in UM1 Cells
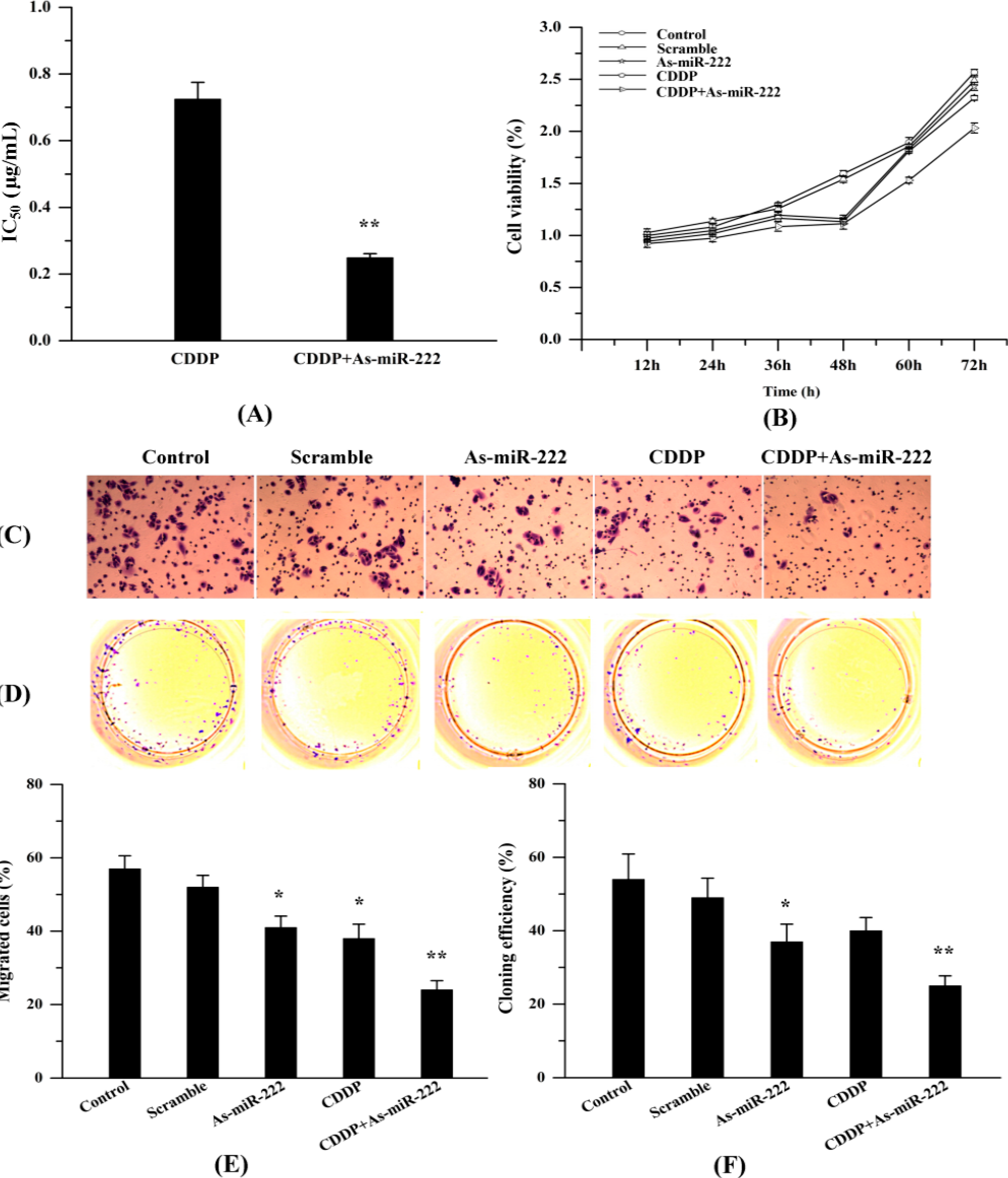
2.4. As-miR-222 Increases the Cytotoxicity of CDDP on UM1 Cells and Inhibited Cell Proliferation and Invasion
2.5. As-miR-222 and CDDP Induced UM1 Cell Apoptosis

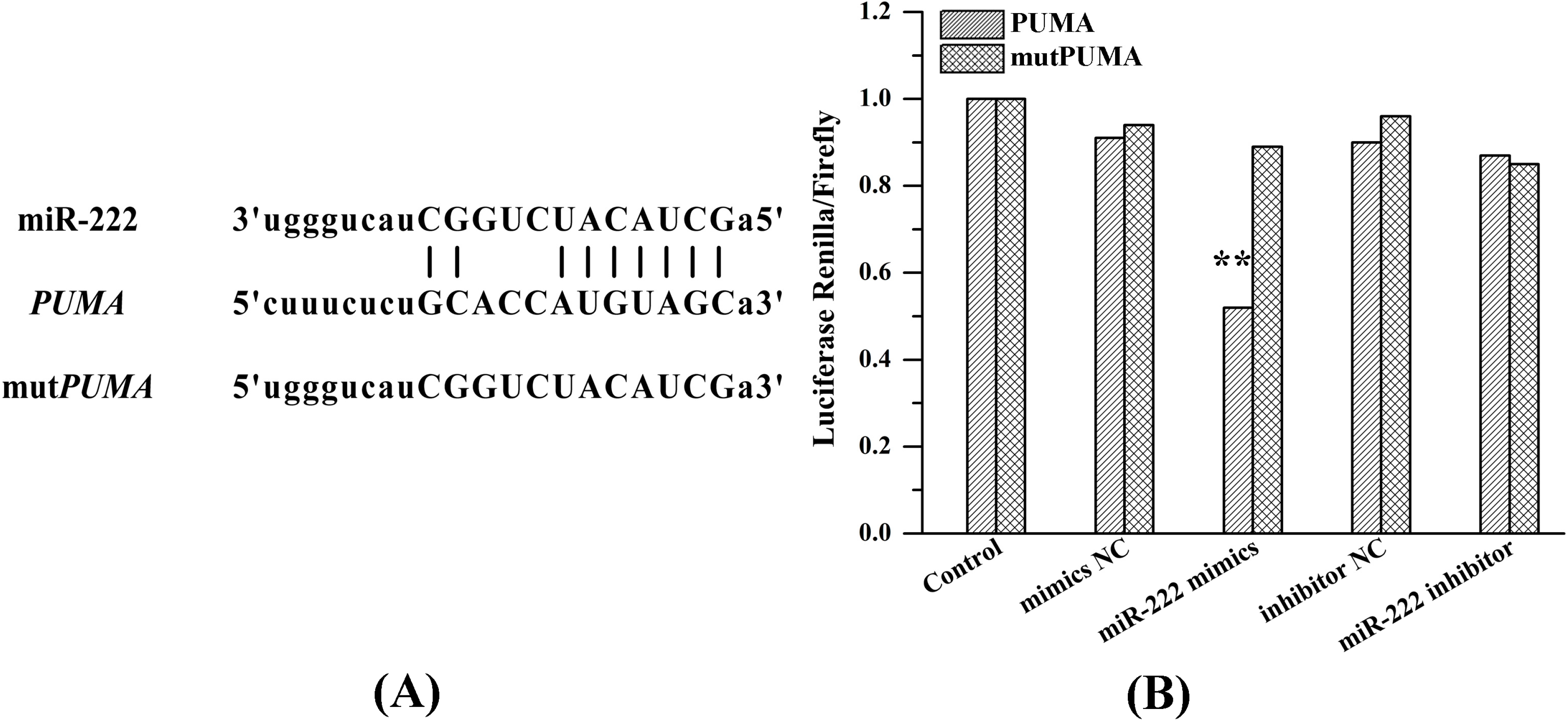
2.6. miR-222 Acts Directly on PUMA mRNA 3'UTR
3. Discussion
4. Experimental Section
4.1. Cell Culture and Transfection
4.2. RT-PCR
4.3. Western Blot Analysis
4.4. Immunofluorescence
4.5. Cell Viability Assay
4.6. Cell Migration Assay
4.7. Flow Cytometry
4.8. Luciferase Assay
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tran, N.; O’Brien, C.J.; Clark, J.; Rose, B. Potential role of micro-RNAs in head and neck tumorigenesis. Head Neck 2010, 32, 1099–1111. [Google Scholar] [CrossRef] [PubMed]
- Chiang, W.F.; Hung, P.S.; Liu, S.Y.; Yuan, T.C.; Chang, K.W.; Chen, Y.P.; Liu, Y.C.; Lin, S.C. Increase of ZASC1 gene copy number in recurrent oral carcinoma. Oral Dis. 2011, 17, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.A.; Dreicer, R. Systemic chemotherapy for advanced bladder cancer: Update and controversies. J. Clin. Oncol. 2006, 24, 5545–5551. [Google Scholar] [CrossRef] [PubMed]
- Childs, G.; Fazzari, M.; Kung, G.; Kawachi, N.; Brandwein-Gensler, M.; McLemore, M.; Chen, Q.; Burk, R.D.; Smith, R.V.; Prystowsky, M.B.; et al. Low-level expression of microRNAs let-7d and miR-205 are prognostic markers of head and neck squamous cell carcinoma. Am. J. Pathol. 2009, 174, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Tsai, M.M.; Hung, P.S.; Kao, S.Y.; Liu, T.Y.; Wu, K.J.; Chiou, S.H.; Lin, S.C.; Chang, K.W. miR-31 ablates expression of the HIF regulatory factor FIH to activate the HIF pathway in head and neck carcinoma. Cancer Res. 2010, 70, 1635–1644. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, L.; Hwang, P.M.; Kinzler, K.W.; Vogelstein, B. PUMA induces the rapid apoptosis of colorectal cancer cells. Mol. Cell 2001, 7, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Altamemi, I.; Murphy, E.A.; Catroppo, J.F.; Zumbrun, E.E.; Zhang, J.; McClellan, J.L.; Singh, U.P.; Nagarkatti, P.S.; Nagarkatti, M. Role of microRNAs in resveratrol-mediated mitigation of colitis-associated tumorigenesis in ApcMin/+mice. J. Pharmacol. Exp. Ther. 2014, 350, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Skaftnesmo, K.O.; Prestegarden, L.; Micklem, D.R.; Lorens, J.B. MicroRNAs in tumorigenesis. Curr. Pharm. Biotechnol. 2007, 8, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Cheng, Z.; Zhang, J.; Li, R.; Zhao, P.; Fu, Z.; You, Y. hsa-mir-181a and hsa-mir-181b function as tumor suppressors in human glioma cells. Brain Res. 2008, 1236, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Le Sage, C.; Nagel, R.; Egan, D.A.; Schrier, M.; Mesman, E.; Mangiola, A.; Anile, C.; Maira, G.; Mercatelli, N.; Ciafre, S.A.; et al. Regulation of the p27Kip1 tumor suppressor by miR-221 and miR-222 promotes cancer cell proliferation. EMBO J. 2007, 26, 3699–3708. [Google Scholar]
- Zhang, C.; Kang, C.; You, Y.; Pu, P.; Yang, W.; Zhao, P.; Wang, G.; Zhang, A.; Jia, Z.; Han, L.; et al. Co-suppression of miR-221/222 cluster suppresses human glioma cell growth by targeting p27kip1 in vitro and in vivo. Int. J. Oncol. 2009, 34, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Z.; Han, L.; Zhang, A.L.; Fu, Y.C.; Yue, X.; Wang, G.X.; Jia, Z.F.; Pu, P.Y.; Zhang, Q.Y.; Kang, C.S. MicroRNA-221 and microRNA-222 regulate gastric carcinoma cell proliferation and radioresistance by targeting PTEN. BMC Cancer 2010, 10, 367. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Zhao, W.; Zhou, L.; Zhang, L.; Liu, Z.; Yu, D. miR-222 regulates the cell biological behavior of oral squamous cell carcinoma by targeting PUMA. Oncol. Rep. 2014, 31, 1255–1262. [Google Scholar] [PubMed]
- Nakano, K.; Vousden, K.H. PUMA, a novel proapoptotic gene, is induced by p53. Mol. Cell 2001, 7, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Avila, J.L.; Grundmann, O.; Burd, R.; Limesand, K.H. Radiation-induced salivary gland dysfunction results from p53-dependent apoptosis. Int. J. Radiat. Oncol. Biol. Phys. 2009, 73, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Labi, V.; Villunger, A. PUMA-mediated tumor suppression: A tale of two stories. Cell Cycle 2010, 9, 4269–4275. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Willis, S.N.; Wei, A.; Smith, B.J.; Fletcher, J.I.; Hinds, M.G.; Colman, P.M.; Day, C.L.; Adams, J.M.; Huang, D.C. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol. Cell 2005, 17, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, J.; Zhang, A.; Wang, Y.; Han, L.; You, Y.; Pu, P.; Kang, C. PUMA is a novel target of miR-221/222 in human epithelial cancers. Int. J. Oncol. 2010, 37, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
- Joladarashi, D.; Thandavarayan, R.A.; Babu, S.S.; Krishnamurthy, P. Small engine, big power: MicroRNAs as regulators of cardiac diseases and regeneration. Int. J. Mol. Sci. 2014, 15, 15891–15911. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Coukos, G.; Zhang, L. miRNAs in human cancer. Methods Mol. Biol. 2012, 822, 295–306. [Google Scholar] [PubMed]
- Liao, J.; Liu, R.; Yin, L.; Pu, Y. Expression profiling of exosomal miRNAs derived from human esophageal cancer cells by Solexa high-throughput sequencing. Int. J. Mol. Sci. 2014, 15, 15530–15551. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.J.; Sakai, R. Production of avaroferrin and putrebactin by heterologous expression of a deep-sea metagenomic DNA. Mar. Drugs 2014, 12, 4799–4809. [Google Scholar] [CrossRef] [PubMed]
- Tahiri, A.; Leivonen, S.K.; Luders, T.; Steinfeld, I.; RagleAure, M.; Geisler, J.; Makela, R.; Nord, S.; Riis, M.L.; Yakhini, Z.; et al. Deregulation of cancer-related miRNAs is a common event in both benign and malignant human breast tumors. Carcinogenesis 2014, 35, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.E.; Ghoshal, K.; Ramaswamy, B.; Roy, S.; Datta, J.; Shapiro, C.L.; Jacob, S.; Majumder, S. MicroRNA-221/222 confers tamoxifen resistance in breast cancer by targeting p27Kip1. J. Biol. Chem. 2008, 283, 29897–29903. [Google Scholar] [CrossRef] [PubMed]
- Kovalchuk, O.; Filkowski, J.; Meservy, J.; Ilnytskyy, Y.; Tryndyak, V.P.; Chekhun, V.F.; Pogribny, I.P. Involvement of microRNA-451 in resistance of the MCF-7 breast cancer cells to chemotherapeutic drug doxorubicin. Mol. Cancer Ther. 2008, 7, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.Z.; Morris, M.E.; Yu, A.M. MicroRNA-328 negatively regulates the expression of breast cancer resistance protein (BCRP/ABCG2) in human cancer cells. Mol. Pharmacol. 2009, 75, 1374–1379. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, M.; di Leva, G.; Romano, G.; Nuovo, G.; Suh, S.S.; Ngankeu, A.; Taccioli, C.; Pichiorri, F.; Alder, H.; Secchiero, P.; et al. miR-221&222 regulate TRAIL resistance and enhance tumorigenicity through PTEN and TIMP3 downregulation. Cancer Cell 2009, 16, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Han, L.; Zhang, A.; Yang, W.; Zhou, X.; Pu, P.; Du, Y.; Zeng, H.; Kang, C. Global changes of mRNA expression reveals an increased activity of the interferon-induced signal transducer and activator of transcription (STAT) pathway by repression of miR-221/222 in glioblastoma U251 cells. Int. J. Oncol. 2010, 36, 1503–1512. [Google Scholar] [PubMed]
- Zhang, C.Z.; Zhang, J.X.; Zhang, A.L.; Shi, Z.D.; Han, L.; Jia, Z.F.; Yang, W.D.; Wang, G.X.; Jiang, T.; You, Y.P.; et al. MiR-221 and miR-222 target PUMA to induce cell survival in glioblastoma. Mol. Cancer 2010, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- John-Aryankalayil, M.; Palayoor, S.T.; Cerna, D.; Simone, C.B.; Falduto, M.T.; Magnuson, S.R.; Coleman, C.N. Fractionated radiation therapy can induce a molecular profile for therapeutic targeting. Radiat. Res. 2010, 174, 446–458. [Google Scholar] [CrossRef] [PubMed]
- De Tullio, G.; de Fazio, V.; Sgherza, N.; Minoia, C.; Serrati, S.; Merchionne, F.; Loseto, G.; Iacobazzi, A.; Rana, A.; Petrillo, P.; et al. Challenges and opportunities of microRNAs in lymphomas. Molecules 2014, 19, 14723–14781. [Google Scholar]
- Marignol, L.; Robson, T.; McCarthy, H.O.; Worthington, J.; Murray, M.M.; Hollywood, D.; Lawler, M.; Hirst, D.G. The tissue plasminogen activator gene promoter: A novel tool for radiogenic gene therapy of the prostate? J. Gene Med. 2008, 10, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Granados Lopez, A.J.; Lopez, J.A. Multistep model of cervical cancer: Participation of miRNAs and coding genes. Int. J. Mol. Sci. 2014, 15, 15700–15733. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, F.; Zhao, W.; Zhou, L.; Liu, Z.; Li, W.; Yu, D. MiR-222 Targeted PUMA to Improve Sensitization of UM1 Cells to Cisplatin. Int. J. Mol. Sci. 2014, 15, 22128-22141. https://doi.org/10.3390/ijms151222128
Jiang F, Zhao W, Zhou L, Liu Z, Li W, Yu D. MiR-222 Targeted PUMA to Improve Sensitization of UM1 Cells to Cisplatin. International Journal of Molecular Sciences. 2014; 15(12):22128-22141. https://doi.org/10.3390/ijms151222128
Chicago/Turabian StyleJiang, Fangfang, Wei Zhao, Lijie Zhou, Zifeng Liu, Wenqing Li, and Dongsheng Yu. 2014. "MiR-222 Targeted PUMA to Improve Sensitization of UM1 Cells to Cisplatin" International Journal of Molecular Sciences 15, no. 12: 22128-22141. https://doi.org/10.3390/ijms151222128



