Effects of Low Doses of Ionizing Radiation Exposures on Stress-Responsive Gene Expression in Human Embryonic Stem Cells
Abstract
:1. Introduction
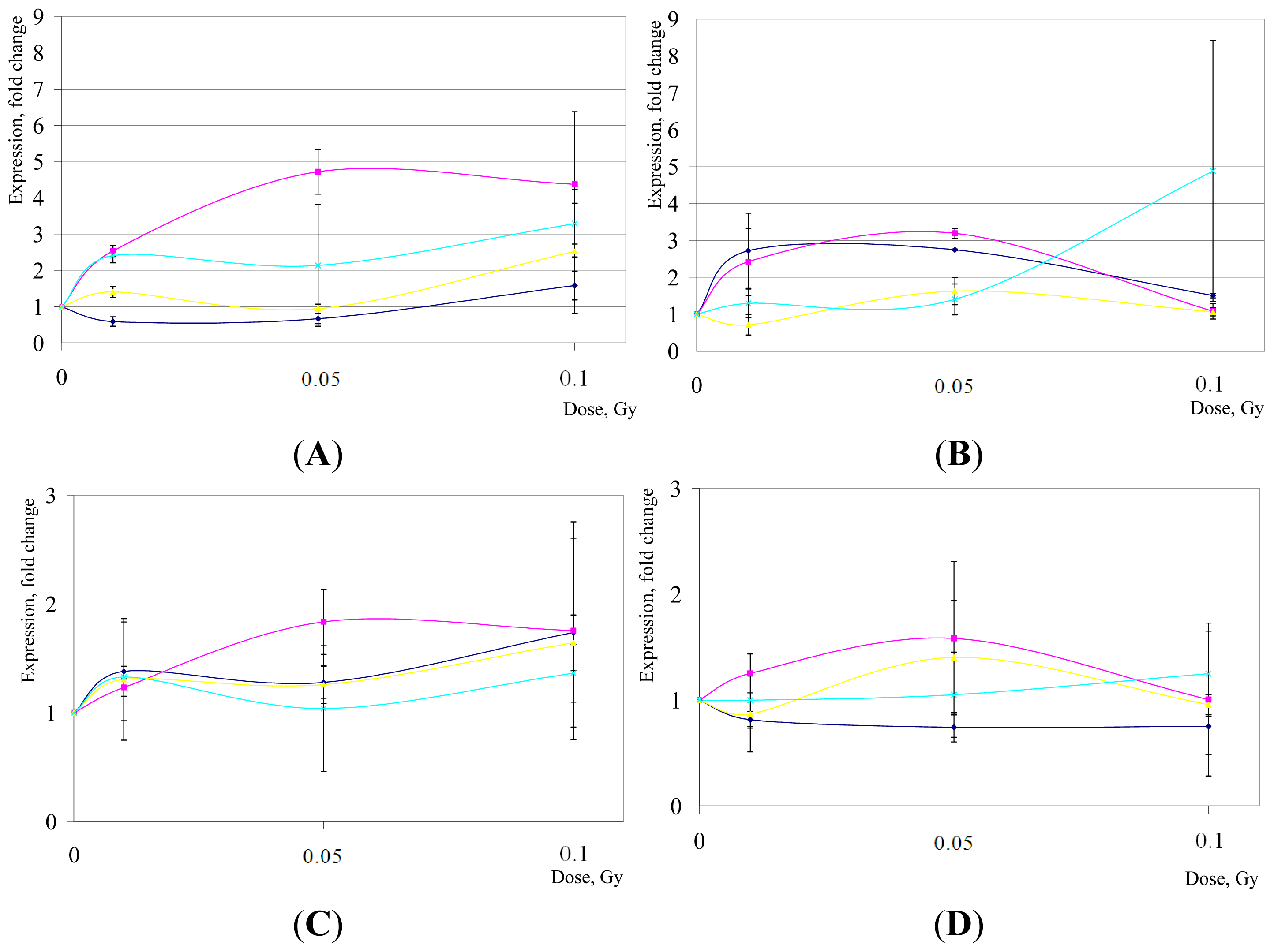
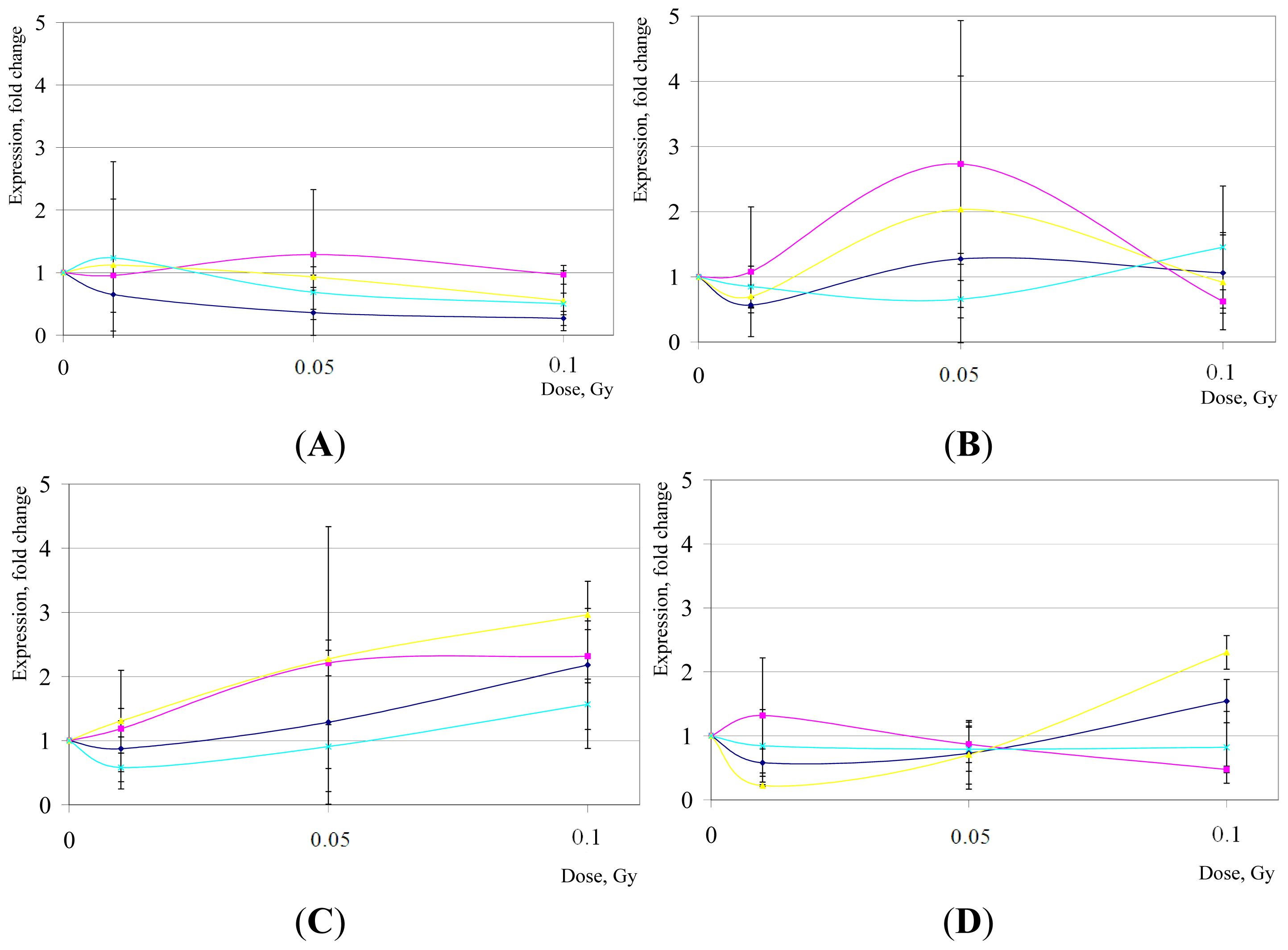
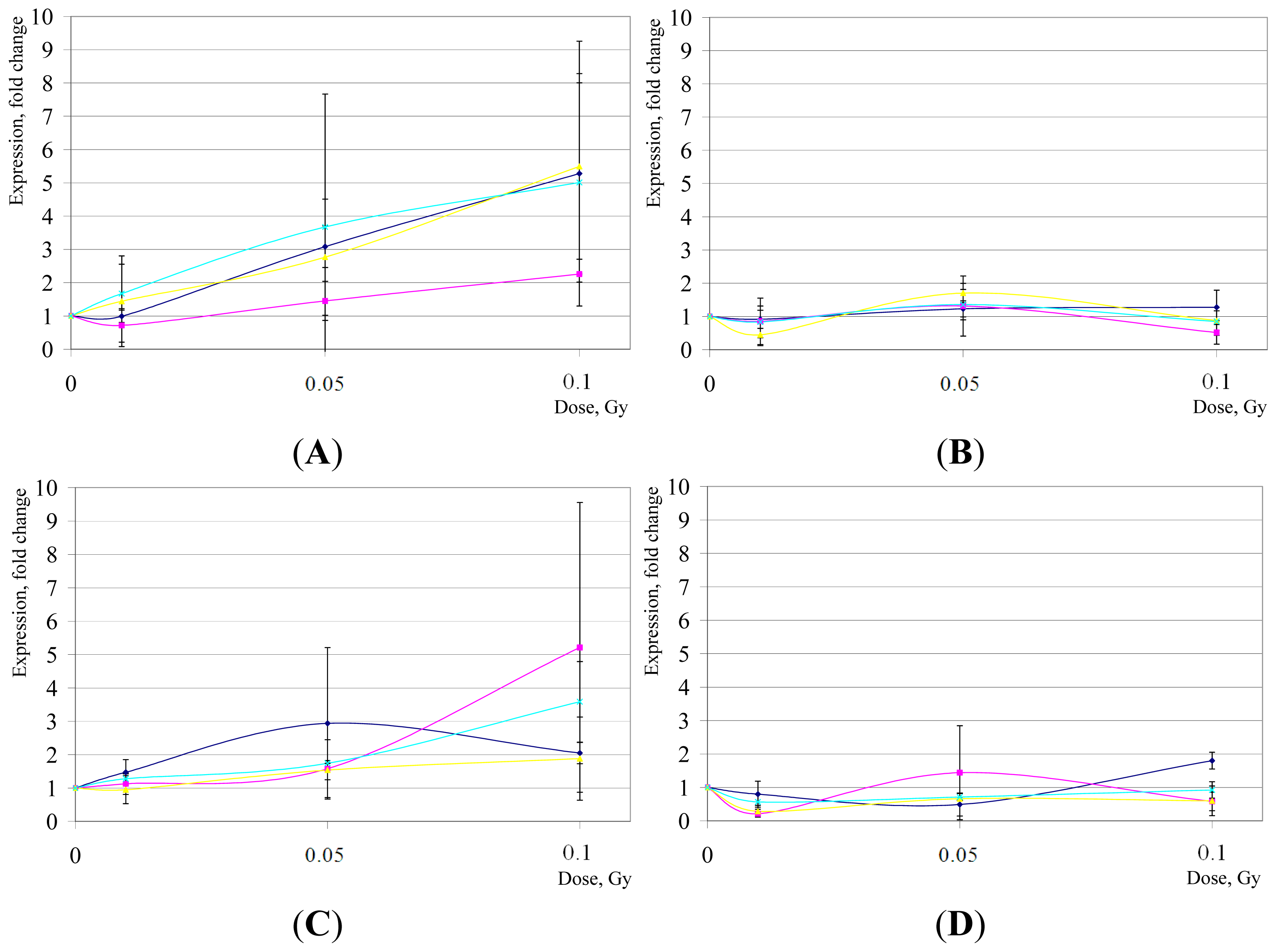
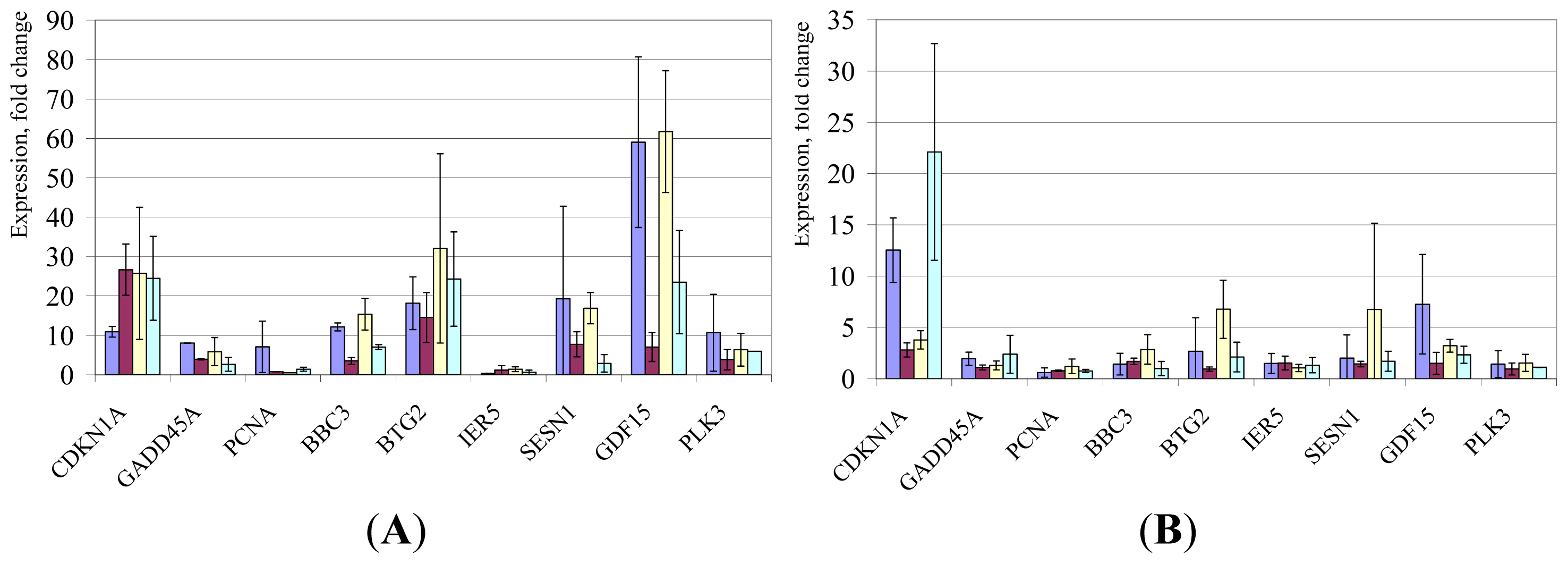
2. Results and Discussion
3. Experimental Section
3.1. Cell Cultures and Irradiation
3.2. Immunocytochemistry
3.3. Quantitative Real-Time PCR and Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Dauer, L.T.; Brooks, A.L.; Hoel, D.G.; Morgan, W.F.; Stram, D.; Tran, P. Review and evaluation of updated research on the health effects associated with low-dose ionising radiation. Radiat. Prot. Dosim 2010, 140, 103–136. [Google Scholar]
- Morgan, W.F.; Bair, W.J. Issues in low dose radiation biology: the controversy continues. A perspective. Radiat. Res 2013, 179, 501–510. [Google Scholar]
- Brenner, D.J.; Hall, E.J. Computed tomography—An increasing source of radiation exposure. N. Engl. J. Med 2007, 357, 2277–2284. [Google Scholar]
- Brenner, D.J.; Mossman, K.L. Do radiation doses below 1 cGy increase cancer risks? Radiat. Res 2005, 163, 692–693. [Google Scholar]
- Cleaver, J.E. Biology and genetics in the biological effects of ionizing radiation (BEIR VII) report. Health Phys 2005, 89, S32. [Google Scholar]
- Tubiana, M.; Aurengo, A.; Averbeck, D.; Masse, R. Recent reports on the effect of low doses of ionizing radiation and its dose-effect relationship. Radiat. Environ. Biophys 2006, 44, 245–251. [Google Scholar]
- Feinendegen, L.E. Evidence for beneficial low level radiation effects and radiation hormesis. Brit. J. Radiol 2005, 78, 3–7. [Google Scholar]
- Calabrese, E.J. Hormesis: Improving predictions in the low-dose zone. In Molecular, Clinical and Environmental Toxicology; Springer: Basel, Switzerland, 2012; Volume 101, pp. 551–564. [Google Scholar]
- Mothersill, C.; Seymour, C. Implications for human and environmental health of low doses of ionising radiation. J. Environ. Radioact 2013. [Google Scholar] [CrossRef]
- Kadhim, M.; Salomaa, S.; Wright, E.; Hildebrandt, G.; Belyakov, O.V.; Prise, K.M.; Little, M.P. Non-targeted effects of ionising radiation-Implications for low dose risk. Mutat. Res 2013, 752, 84–98. [Google Scholar]
- Rodel, F.; Frey, B.; Gaipl, U.; Keilholz, L.; Fournier, C.; Manda, K.; Schollnberger, H.; Hildebrandt, G.; Rodel, C. Modulation of inflammatory immune reactions by low-dose ionizing radiation: Molecular mechanisms and clinical application. Curr. Med. Chem 2012, 19, 1741–1750. [Google Scholar]
- Amundson, S.A.; Do, K.T.; Fornace, A.J., Jr. Induction of stress genes by low doses of γ rays. Radiat. Res 1999, 152, 225–231. [Google Scholar]
- Sokolov, M.V.; Smirnova, N.A.; Camerini-Otero, R.D.; Neumann, R.D.; Panyutin, I.G. Microarray analysis of differentially expressed genes after exposure of normal human fibroblasts to ionizing radiation from an external source and from DNA-incorporated iodine-125 radionuclide. Gene 2006, 382, 47–56. [Google Scholar]
- Sokolov, M.V.; Neumann, R.D.; Panyutin, I.G. Effects of DNA-targeted ionizing radiation produced by 5-[125I]iodo-2′-deoxyuridine on global gene expression in primary human cells. BMC Genomics 2007, 8, 192. [Google Scholar]
- Ding, L.H.; Shingyoji, M.; Chen, F.; Hwang, J.J.; Burma, S.; Lee, C.; Cheng, J.F.; Chen, D.J. Gene expression profiles of normal human fibroblasts after exposure to ionizing radiation: A comparative study of low and high doses. Radiat. Res 2005, 164, 17–26. [Google Scholar]
- Franco, N.; Lamartine, J.; Frouin, V.; Le Minter, P.; Petat, C.; Leplat, J.J.; Libert, F.; Gidrol, X.; Martin, M.T. Low-dose exposure to gamma rays induces specific gene regulations in normal human keratinocytes. Radiat. Res 2005, 163, 623–635. [Google Scholar]
- Gruel, G.; Voisin, P.; Vaurijoux, A.; Roch-Lefevre, S.; Gregoire, E.; Maltere, P.; Petat, C.; Gidrol, X.; Roy, L. Broad modulation of gene expression in CD4+ lymphocyte subpopulations in response to low doses of ionizing radiation. Radiat. Res 2008, 170, 335–344. [Google Scholar]
- Jin, Y.W.; Na, Y.J.; Lee, Y.J.; An, S.; Lee, J.E.; Jung, M.; Kim, H.; Nam, S.Y.; Kim, C.S.; Yang, K.H.; et al. Comprehensive analysis of time- and dose-dependent patterns of gene expression in a human mesenchymal stem cell line exposed to low-dose ionizing radiation. Oncol. Rep 2008, 19, 135–144. [Google Scholar]
- Mezentsev, A.; Amundson, S.A. Global gene expression responses to low- or high-dose radiation in a human three-dimensional tissue model. Radiat. Res 2011, 175, 677–688. [Google Scholar]
- Albrecht, H.; Durbin-Johnson, B.; Yunis, R.; Kalanetra, K.M.; Wu, S.; Chen, R.; Stevenson, T.R.; Rocke, D.M. Transcriptional response of ex vivo human skin to ionizing radiation: Comparison between low- and high-dose effects. Radiat. Res 2012, 177, 69–83. [Google Scholar]
- Knops, K.; Boldt, S.; Wolkenhauer, O.; Kriehuber, R. Gene expression in low- and high-dose-irradiated human peripheral blood lymphocytes: possible applications for biodosimetry. Radiat. Res 2012, 178, 304–312. [Google Scholar]
- Amundson, S.A.; Grace, M.B.; McLeland, C.B.; Epperly, M.W.; Yeager, A.; Zhan, Q.; Greenberger, J.S.; Fornace, A.J., Jr. Human in vivo radiation-induced biomarkers: Gene expression changes in radiotherapy patients. Cancer Res 2004, 64, 6368–6371. [Google Scholar]
- Bosi, A.; Olivieri, G. Variability of the adaptive response to ionizing radiations in humans. Mutat. Res 1989, 211, 13–17. [Google Scholar]
- MacCallum, D.E.; Hall, P.A.; Wright, E.G. The Trp53 pathway is induced in vivo by low doses of gamma radiation. Radiat. Res 2001, 156, 324–327. [Google Scholar]
- Mothersill, C.; Lyng, F.; Seymour, C.; Maguire, P.; Lorimore, S.; Wright, E. Genetic factors influencing bystander signaling in murine bladder epithelium after low-dose irradiation in vivo. Radiat. Res. 2005, 163, 391–399. [Google Scholar]
- Warters, R.L.; Packard, A.T.; Kramer, G.F.; Gaffney, D.K.; Moos, P.J. Differential gene expression in primary human skin keratinocytes and fibroblasts in response to ionizing radiation. Radiat. Res 2009, 172, 82–95. [Google Scholar]
- Snijders, A.M.; Marchetti, F.; Bhatnagar, S.; Duru, N.; Han, J.; Hu, Z.; Mao, J.H.; Gray, J.W.; Wyrobek, A.J. Genetic differences in transcript responses to low-dose ionizing radiation identify tissue functions associated with breast cancer susceptibility. PLoS One 2012, 7, e45394. [Google Scholar]
- Von Neubeck, C.; Shankaran, H.; Karin, N.J.; Kauer, P.M.; Chrisler, W.B.; Wang, X.; Robinson, R.J.; Waters, K.M.; Tilton, S.C.; Sowa, M.B. Cell type-dependent gene transcription profile in a three-dimensional human skin tissue model exposed to low doses of ionizing radiation: implications for medical exposures. Environ. Mol. Mutagen 2012, 53, 247–259. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Onyshchenko, M.I.; Panyutin, I.G.; Neumann, R.D. Expression of pluripotency-associated genes in the surviving fraction of cultured human embryonic stem cells is not significantly affected by ionizing radiation. Gene 2010, 455, 8–15. [Google Scholar]
- Wilson, K.D.; Sun, N.; Huang, M.; Zhang, W.Y.; Lee, A.S.; Li, Z.; Wang, S.X.; Wu, J.C. Effects of ionizing radiation on self-renewal and pluripotency of human embryonic stem cells. Cancer Res 2010, 70, 5539–5548. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Panyutin, I.G.; Neumann, R.D. Dynamics of the transcriptome response of cultured human embryonic stem cells to ionizing radiation exposure. Mutat. Res 2011, 709–710, 40–48. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Neumann, R.D. Unraveling the global microRNAome responses to ionizing radiation in human embryonic stem cells. PLoS One 2012, 7, e31028. [Google Scholar]
- Di Leonardo, A.; Linke, S.P.; Clarkin, K.; Wahl, G.M. DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Develop 1994, 8, 2540–2551. [Google Scholar]
- Zhan, Q.; Eldeiry, W.; Bae, I.; Alamo, I.; Kastan, M.; Vogelstein, B.; Fornace, A. Similarity of the DNA-damage responsiveness and growth-suppressive properties of waf1/cip1 and gadd45. Int. J. Oncol 1995, 6, 937–946. [Google Scholar]
- Daino, K.; Ichimura, S.; Nenoi, M. Early induction of CDKN1A (p21) and GADD45 mRNA by a low dose of ionizing radiation is due to their dose-dependent post-transcriptional regulation. Radiat. Res 2002, 157, 478–482. [Google Scholar]
- Fornace, A.J., Jr.; Amundson, S.A.; Do, K.T.; Meltzer, P.; Trent, J.; Bittner, M. Stress-gene induction by low-dose gamma irradiation. Milit. Med 2002, 167, 13–15. [Google Scholar]
- Amundson, S.A.; Lee, R.A.; Koch-Paiz, C.A.; Bittner, M.L.; Meltzer, P.; Trent, J.M.; Fornace, A.J., Jr. Differential responses of stress genes to low dose-rate γ irradiation. Mol. Cancer Res 2003, 1, 445–452. [Google Scholar]
- Kis, E.; Szatmari, T.; Keszei, M.; Farkas, R.; Esik, O.; Lumniczky, K.; Falus, A.; Safrany, G. Microarray analysis of radiation response genes in primary human fibroblasts. Int. J. Radiat. Oncol. Biol. Phys 2006, 66, 1506–1514. [Google Scholar]
- Paul, S.; Amundson, S.A. Development of gene expression signatures for practical radiation biodosimetry. Int. J. Radiat. Oncol. Biol. Phys 2008, 71, 1236–1244. [Google Scholar]
- El-Saghire, H.; Thierens, H.; Monsieurs, P.; Michaux, A.; Vandevoorde, C.; Baatout, S. Gene set enrichment analysis highlights different gene expression profiles in whole blood samples X-irradiated with low and high doses. Int. J. Radiat. Biol 2013, 89, 628–638. [Google Scholar]
- Zhan, Q.; Bae, I.; Kastan, M.B.; Fornace, A.J., Jr. The p53-dependent gamma-ray response of GADD45. Cancer Res 1994, 54, 2755–2760. [Google Scholar]
- Kearsey, J.M.; Coates, P.J.; Prescott, A.R.; Warbrick, E.; Hall, P.A. Gadd45 is a nuclear cell cycle regulated protein which interacts with p21Cip1. Oncogene 1995, 11, 1675–1683. [Google Scholar]
- Jin, S.; Fan, F.; Fan, W.; Zhao, H.; Tong, T.; Blanck, P.; Alomo, I.; Rajasekaran, B.; Zhan, Q. Transcription factors Oct-1 and NF-YA regulate the p53-independent induction of the GADD45 following DNA damage. Oncogene 2001, 20, 2683–2690. [Google Scholar]
- Tavakoli, H.; Manoochehri, M.; Modarres Mosalla, S.M.; Ghafori, M.; Karimi, A.A. Dose-dependent and gender-related radiation-induced transcription alterations of Gadd45a and Ier5 inhuman lymphocytes exposed to gamma ray emitted by (60)Co. Radiat. Prot. Dosim 2013, 154, 37–44. [Google Scholar]
- Rouault, J.P.; Falette, N.; Guehenneux, F.; Guillot, C.; Rimokh, R.; Wang, Q.; Berthet, C.; Moyret-Lalle, C.; Savatier, P.; Pain, B.; et al. Identification of BTG2, an antiproliferative p53-dependent component of the DNA damage cellular response pathway. Nat. Genet 1996, 14, 482–486. [Google Scholar]
- Tsai, M.H.; Chen, X.; Chandramouli, G.V.; Chen, Y.; Yan, H.; Zhao, S.; Keng, P.; Liber, H.L.; Coleman, C.N.; Mitchell, J.B.; et al. Transcriptional responses to ionizing radiation reveal that p53R2 protects against radiation-induced mutagenesis in human lymphoblastoid cells. Oncogene 2006, 25, 622–632. [Google Scholar]
- Kabacik, S.; Mackay, A.; Tamber, N.; Manning, G.; Finnon, P.; Paillier, F.; Ashworth, A.; Bouffler, S.; Badie, C. Gene expression following ionising radiation: Identification of biomarkers for dose estimation and prediction of individual response. Int. J. Radiat. Biol 2011, 87, 115–129. [Google Scholar]
- Turtoi, A.; Schneeweiss, F.H. Effect of (211)At alpha-particle irradiation on expression of selected radiation responsive genes in human lymphocytes. Int. J. Radiat. Biol 2009, 85, 403–412. [Google Scholar]
- Shan, B.; Xu, J.; Zhuo, Y.; Morris, C.A.; Morris, G.F. Induction of p53-dependent activation of the human proliferating cell nuclear antigen gene in chromatin by ionizing radiation. J. Biol. Chem 2003, 278, 44009–44017. [Google Scholar]
- Ding, K.K.; Shang, Z.F.; Hao, C.; Xu, Q.Z.; Shen, J.J.; Yang, C.J.; Xie, Y.H.; Qiao, C.; Wang, Y.; Xu, L.L.; et al. Induced expression of the IER5 gene by gamma-ray irradiation and its involvement in cell cycle checkpoint control and survival. Radiat. Environ. Biophys 2009, 48, 205–213. [Google Scholar]
- Nakamura, S.; Nagata, Y.; Tan, L.; Takemura, T.; Shibata, K.; Fujie, M.; Fujisawa, S.; Tanaka, Y.; Toda, M.; Makita, R.; et al. Transcriptional repression of Cdc25B by IER5 inhibits the proliferation of leukemic progenitor cells through NF-YB and p300 in acute myeloid leukemia. PLoS One 2011, 6, e28011. [Google Scholar]
- Okazaki, R.; Moon, Y.; Norimura, T.; Eling, T. Ionizing radiation enhances the expression of the nonsteroidal anti-inflammatory drug-activated gene (NAG1) by increasing the expression of TP53 in human colon cancer cells. Radiat. Res 2006, 165, 125–130. [Google Scholar]
- Chang, J.T.; Chan, S.H.; Lin, C.Y.; Lin, T.Y.; Wang, H.M.; Liao, C.T.; Wang, T.H.; Lee, L.Y.; Cheng, A.J. Differentially expressed genes in radioresistant nasopharyngeal cancer cells: gp96 and GDF15. Mol. Cancer Ther 2007, 6, 2271–2279. [Google Scholar]
- Bahassi, E.M.; Myer, D.L.; McKenney, R.J.; Hennigan, R.F.; Stambrook, P.J. Priming phosphorylation of Chk2 by polo-like kinase 3 (Plk3) mediates its full activation by ATM and a downstream checkpoint in response to DNA damage. Mutat. Res 2006, 596, 166–176. [Google Scholar]
- Wang, L.; Dai, W.; Lu, L. Stress-induced c-Jun activation mediated by Polo-like kinase 3 in corneal epithelial cells. J. Biol. Chem 2007, 282, 32121–32127. [Google Scholar]
- Nosel, I.; Vaurijoux, A.; Barquinero, J.F.; Gruel, G. Characterization of gene expression profiles at low and very low doses of ionizing radiation. DNA Repair 2013, 12, 508–517. [Google Scholar]
- Budworth, H.; Snijders, A.M.; Marchetti, F.; Mannion, B.; Bhatnagar, S.; Kwoh, E.; Tan, Y.; Wang, S.X.; Blakely, W.F.; Coleman, M.; et al. DNA repair and cell cycle biomarkers of radiation exposure and inflammation stress in human blood. PLoS One 2012, 7, e48619. [Google Scholar]
- Filion, T.M.; Qiao, M.; Ghule, P.N.; Mandeville, M.; van Wijnen, A.J.; Stein, J.L.; Lian, J.B.; Altieri, D.C.; Stein, G.S. Survival responses of human embryonic stem cells to DNA damage. J. Cell. Physiol 2009, 220, 586–592. [Google Scholar]
- Sokolov, M.V.; Neumann, R.D. Human embryonic stem cell responses to ionizing radiation exposures: current state of knowledge and future challenges. Stem Cell. Int 2012, 2012, 579104. [Google Scholar]
- Sokolov, M.; Neumann, R. Lessons learned about human stem cell responses to ionizing radiation exposures: a long road still ahead of us. Int. J. Mol. Sci 2013, 14, 15695–15723. [Google Scholar]
- Becker, K.A.; Ghule, P.N.; Therrien, J.A.; Lian, J.B.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S. Self-renewal of human embryonic stem cells is supported by a shortened G1 cell cycle phase. J. Cell. Physiol 2006, 209, 883–893. [Google Scholar]
- Qin, H.; Yu, T.; Qing, T.; Liu, Y.; Zhao, Y.; Cai, J.; Li, J.; Song, Z.; Qu, X.; Zhou, P.; et al. Regulation of apoptosis and differentiation by p53 in human embryonic stem cells. J. Biol. Chem 2007, 282, 5842–5852. [Google Scholar]
- Dolezalova, D.; Mraz, M.; Barta, T.; Plevova, K.; Vinarsky, V.; Holubcova, Z.; Jaros, J.; Dvorak, P.; Pospisilova, S.; Hampl, A. MicroRNAs regulate p21(Waf1/Cip1) protein expression and the DNA damage response in human embryonic stem cells. Stem Cells 2012, 30, 1362–1372. [Google Scholar]
- Bae, I.; Smith, M.L.; Sheikh, M.S.; Zhan, Q.; Scudiero, D.A.; Friend, S.H.; O’Connor, P.M.; Fornace, A.J., Jr. An abnormality in the p53 pathway following gamma-irradiation in many wild-type p53 human melanoma lines. Cancer Res 1996, 56, 840–847. [Google Scholar]
- Momcilovic, O.; Choi, S.; Varum, S.; Bakkenist, C.; Schatten, G.; Navara, C. Ionizing radiation induces ataxia telangiectasia mutated-dependent checkpoint signaling and G(2) but not G(1) cell cycle arrest in pluripotent human embryonic stem cells. Stem Cells 2009, 27, 1822–1835. [Google Scholar]
- Bahassi, el M.; Conn, C.W.; Myer, D.L.; Hennigan, R.F.; McGowan, C.H.; Sanchez, Y.; Stambrook, P.J. Mammalian Polo-like kinase 3 (Plk3) is a multifunctional protein involved in stress response pathways. Oncogene 2002, 21, 6633–6640. [Google Scholar]
- Xie, S.; Wu, H.; Wang, Q.; Cogswell, J.P.; Husain, I.; Conn, C.; Stambrook, P.; Jhanwar-Uniyal, M.; Dai, W. Plk3 functionally links DNA damage to cell cycle arrest and apoptosis at least in part via the p53 pathway. J. Biol. Chem 2001, 276, 43305–43312. [Google Scholar]
- Li, Z.; Niu, J.; Uwagawa, T.; Peng, B.; Chiao, P.J. Function of polo-like kinase 3 in NF-κB-mediated proapoptotic response. J. Biol. Chem 2005, 280, 16843–16850. [Google Scholar]
- Zimmerman, W.C.; Erikson, R.L. Polo-like kinase 3 is required for entry into S phase. Proc. Natl. Acad. Sci. USA 2007, 104, 1847–1852. [Google Scholar]
- Wang, Q.; Xie, S.; Chen, J.; Fukasawa, K.; Naik, U.; Traganos, F.; Darzynkiewicz, Z.; Jhanwar-Uniyal, M.; Dai, W. Cell cycle arrest and apoptosis induced by human Polo-like kinase 3 is mediated through perturbation of microtubule integrity. Mol. Cell. Biol 2002, 22, 3450–3459. [Google Scholar]
- Yang, H.; Filipovic, Z.; Brown, D.; Breit, S.N.; Vassilev, L.T. Macrophage inhibitory cytokine-1: A novel biornarker for p53 pathway activation. Mol. Cancer Ther 2003, 2, 1023–1029. [Google Scholar]
- Imran, M.; Lim, I.K. Regulation of Btg2 expression via reactive oxygen species-protein kinase C-NF-κB pathway under stress conditions. Cell. Signal 2013, 25, 2400–2412. [Google Scholar]
- Choi, K.S.; Kim, J.Y.; Lim, S.K.; Choi, Y.W.; Kim, Y.H.; Kang, S.Y.; Park, T.J.; Lim, I.K. TIS21(/BTG2/PC3) accelerates the repair of DNA double strand breaks by enhancing Mre11 methylation and blocking damage signal transfer to the Chk2(T68)-p53(S20) pathway. DNA Repair 2012, 11, 965–975. [Google Scholar]
- Karve, T.M.; Rosen, E.M. B-cell translocation gene 2 (BTG2) stimulates cellular antioxidant defenses through the antioxidant transcription factor NFE2L2 in human mammary epithelial cells. J. Biol. Chem 2012, 287, 31503–31514. [Google Scholar]
- Wang, P.; Yu, J.; Zhang, L. The nuclear function of p53 is required for PUMA-mediated apoptosis induced by DNA damage. Proc. Natl. Acad. Sci. USA 2007, 104, 4054–4059. [Google Scholar]
- Gomes, N.P.; Espinosa, J.M. Gene-specific repression of the p53 target gene PUMA via intragenic CTCF-Cohesin binding. Gene Develop 2010, 24, 1022–1034. [Google Scholar]
- Wang, P.; Qiu, W.; Dudgeon, C.; Liu, H.; Huang, C.; Zambetti, G.P.; Yu, J.; Zhang, L. PUMA is directly activated by NF-kappa B and contributes to TNF-alpha-induced apoptosis. Cell Death Differ 2009, 16, 1192–1202. [Google Scholar]
- Fayolle, C.; Pourchet, J.; Cohen, A.; Pedeux, R.; Puisieux, A.; Caron de Fromentel, C.; Dore, J.F.; Voeltzel, T. UVB-induced G2 arrest of human melanocytes involves Cdc2 sequestration by Gadd45a in nuclear speckles. Cell Cycle 2006, 5, 1859–1864. [Google Scholar]
- Coleman, M.A.; Yin, E.; Peterson, L.E.; Nelson, D.; Sorensen, K.; Tucker, J.D.; Wyrobek, A.J. Low-dose irradiation alters the transcript profiles of human lymphoblastoid cells including genes associated with cytogenetic radioadaptive response. Radiat. Res 2005, 164, 369–382. [Google Scholar]
- Liu, J.C.; Guan, X.; Ryan, J.A.; Rivera, A.G.; Mock, C.; Agarwal, V.; Letai, A.; Lerou, P.H.; Lahav, G. High mitochondrial priming sensitizes hESCs to DNA-damage-induced apoptosis. Cell Stem Cell 2013, 13, 483–491. [Google Scholar]
- Dumitru, R.; Gama, V.; Fagan, B. M.; Bower, J. J.; Swahari, V.; Pevny, L. H.; Deshmukh, V. Human embryonic stem cells have constitutively active Bax at the Golgi and are primed to undergo rapid apoptosis. Molecular Cell 2012, 46, 573–583. [Google Scholar]
- Wyrobek, A.J.; Manohar, C.F.; Krishnan, V.V.; Nelson, D.O.; Furtado, M.R.; Bhattacharya, M.S.; Marchetti, F.; Coleman, M.A. Low dose radiation response curves, networks and pathways in human lymphoblastoid cells exposed from 1 to 10cGy of acute gamma radiation. Mutat. Res 2011, 722, 119–130. [Google Scholar]
- Sokolov, M.V.; Neumann, R.D. Radiation-induced bystander effects in cultured human stem cells. PLoS One 2010, 5, e14195. [Google Scholar]


© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sokolov, M.; Neumann, R. Effects of Low Doses of Ionizing Radiation Exposures on Stress-Responsive Gene Expression in Human Embryonic Stem Cells. Int. J. Mol. Sci. 2014, 15, 588-604. https://doi.org/10.3390/ijms15010588
Sokolov M, Neumann R. Effects of Low Doses of Ionizing Radiation Exposures on Stress-Responsive Gene Expression in Human Embryonic Stem Cells. International Journal of Molecular Sciences. 2014; 15(1):588-604. https://doi.org/10.3390/ijms15010588
Chicago/Turabian StyleSokolov, Mykyta, and Ronald Neumann. 2014. "Effects of Low Doses of Ionizing Radiation Exposures on Stress-Responsive Gene Expression in Human Embryonic Stem Cells" International Journal of Molecular Sciences 15, no. 1: 588-604. https://doi.org/10.3390/ijms15010588



