2.2. Discussion
The present study was conducted to demonstrate the potential usefulness of chimeric mice with humanized livers in both
in vivo and
in vitro enzyme induction studies. In the
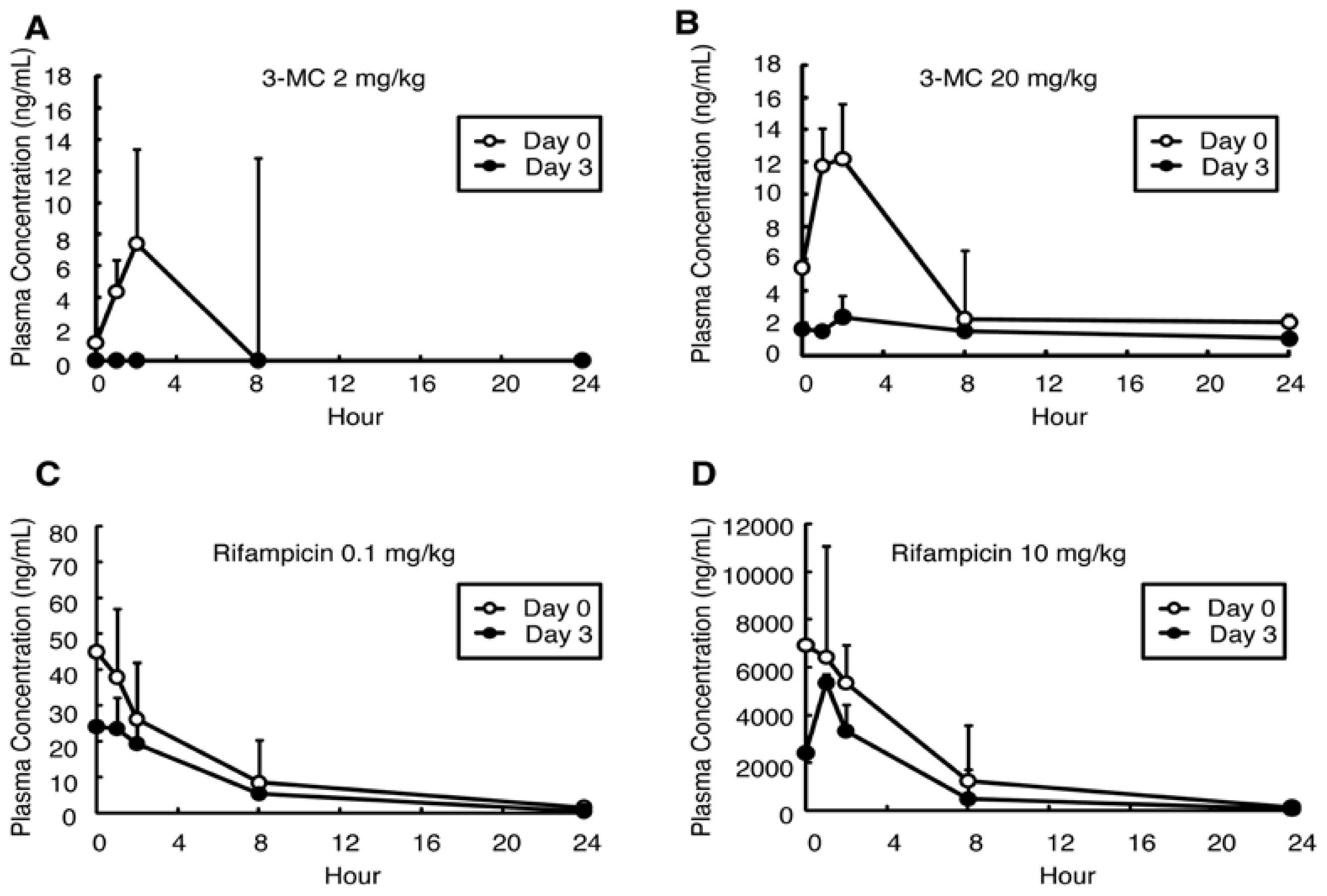
in vivo study, a significant decrease in
AUC of 3-MC was induced by four days 3-MC administration (20 mg/kg), which was associated with a significant increase in h
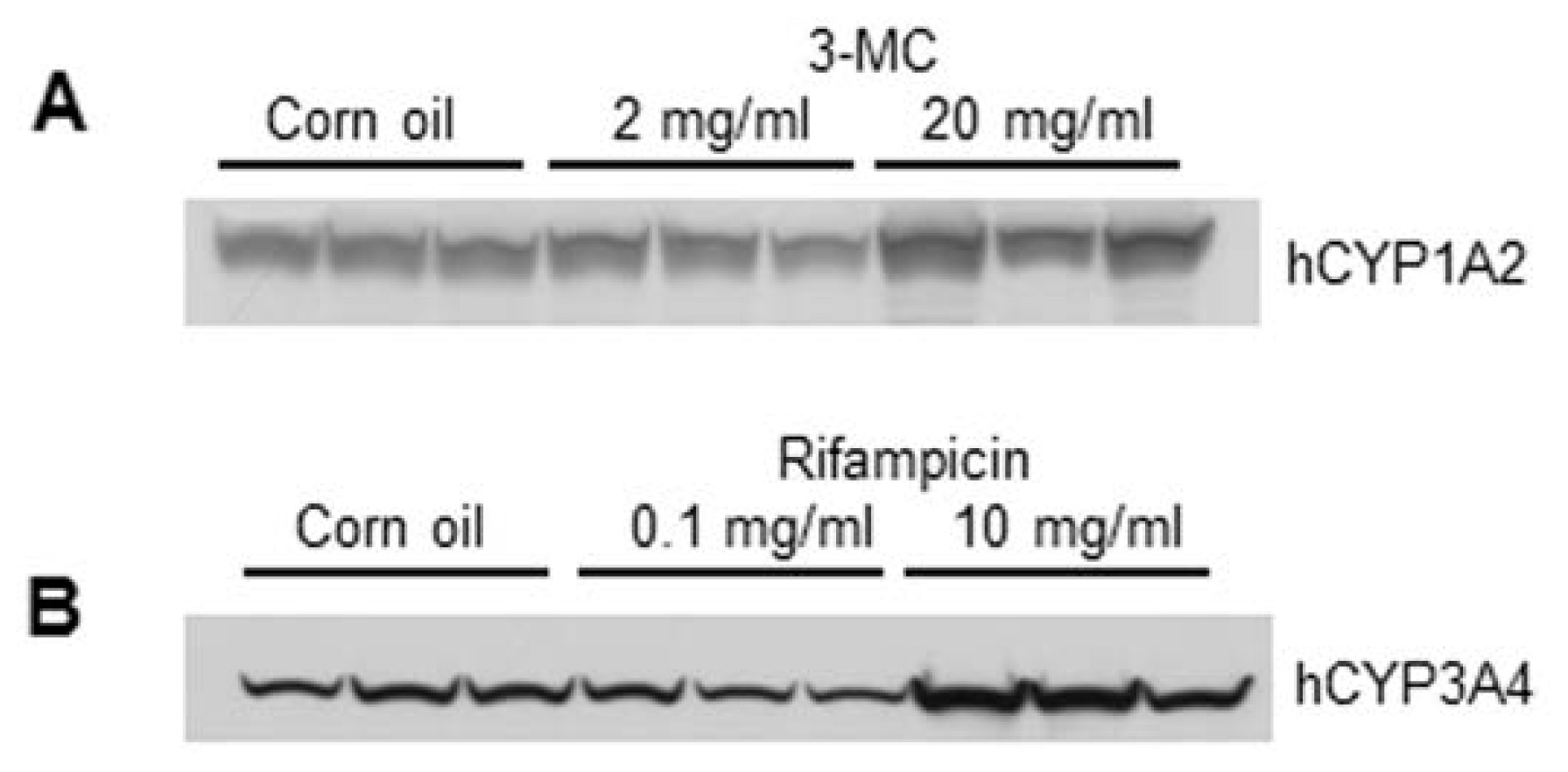
CYP1A1 mRNA expression and hCYP1A2 protein expression.
AUC decreases were observed at 2 mg/kg in the 3-MC group (but not significant) although the expression levels of mRNA (h
CYP1A1 and h
CYP1A2) and protein (hCYP1A2) did not increase significantly after dosing. These results show that although 3-MC is known to be metabolized by CYP1A [
11], we expected that up-regulation of mRNA or protein of, not only CYP1A1 and 1A2, but also other metabolic enzymes or transporters might contribute to the apparent auto-induction by 3-MC. There was no significant difference in
AUC levels between the control and rifampicin-treated groups; however, the
AUC decreased by about one-half in the 10 mg/kg rifampicin group, and this change was associated with a significant increase in h
CYP3A4 mRNA and protein expression levels. Although the exact mechanism is not known, chronic dosing of rifampicin induces its own metabolism [
12]. In addition, the
AUCs in 10 mg/kg of rifampicin-administered group were similar to the
AUC at steady state (
AUCss) of human treated with a therapeutic dose of rifampicin (22,400 to 35,300 ng·h/mL [
13]). Rifampicin is a potent and selective activator of the human nuclear pregnane X receptor (PXR). It has been reported that there are significant differences in ligand recognition by PXR between rodents and humans [
14]. Although the chimeric mice retain less than 30% mouse hepatocytes, decreases in
AUC might have occurred by PXR-related induction in human hepatocytes but not in mouse hepatocytes. Recently we reported that CYP3A4 and CYP2C subfamilies were induced by treatment of the PXB mice with 50 mg/kg rifampicin for four days. However treatment with 10 mg/kg decreased only the
AUC of the CYP3A4 substrate, and did not affect the
AUC of CYP2C substrates [
15]. These data suggest that induction of CYP3A4 might contribute to the
AUC decrease of rifampcin. On the other hand, as 3-MC induces mouse Cyp1a1 and hCYP1A [
11], the Cyp1a1 induction in less than 30% mouse hepatocytes in the chimeric liver might contribute to 3-MC auto-induction.
The protein induction of hCYP1A2 and hCYP3A4 in the chimeric mice was supported by the immunohistochemistry results. In the
in vivo situation, there is morphological, biochemical, molecular, and functional heterogeneity between periportal and pericentral hepatocytes [
16–
20]. CYP3A and CYP1A2 are expressed in pericentral, but not in periportal, hepatocytes in rodent and human liver lobules [
19,
20]. It has been reported that administering phenobarbital to rats induces the expression of CYP3a1 in periportal hepatocytes [
20]. In the present study, we demonstrated that
in vivo treatment of human hepatocytes with rifampicin and 3-MC induced hCYP3A4 and hCYP1A2 expression in periportal hepatocytes of the chimeric mice, resulting in uniform expression of these enzymes throughout the liver lobules. These data are consistent with our previous study showing induction of CYP3A4 in rifampicin-treated chimeric mice [
21]. These results demonstrated that chimeric mice with humanized liver are appropriate for evaluating enzyme induction in human hepatocytes, based on
in vivo exposure levels of the inducer and tissue levels.
Recently, we demonstrated that these chimeric mice may be useful for supplying fresh human hepatocytes on demand, thus, promising high and stable phase I enzyme and glucuronidation activities [
10]. In the present
in vitro study, we also demonstrated the feasibility of conducting 24- or 48-h enzyme induction studies in both monolayer and spheroid cultures using human hepatocytes freshly isolated from the chimeric mice. Given the scarcity of fresh human hepatocytes for conducting
in vitro studies, fresh human hepatocytes isolated from chimeric mice may be a viable alternative. Additionally, it is possible to reproducibly conduct
in vitro enzyme induction studies with fresh human hepatocytes derived from the same donor and to compare results of
in vitro and
in vivo studies using cells from the same donor.
For
in vitro CYP induction study, hepatocytes are typically cultured in monolayer conditions. However, because CYP mRNAs, proteins, and activities decline immediately during monolayer culture, induction levels have been estimated at much lower levels (less than 1%) than
in vivo levels [
8]. In the present study, we compared
CYP1A1,
1A2, and
3A4 mRNA expression levels between hepatocytes just after isolation and monolayer- or spheroid-cultured hepatocytes at 72 and 96 h.
CYP1A2 and
3A4 mRNA expression levels in hepatocytes in both culture conditions decreased; however, they were maintained at more than 10-fold higher levels in spheroid cultures than in monolayer cultures, although
CYP1A1 mRNA expression levels were similar between hepatocytes in the two conditions. Based on these results, hepatocytes in the spheroid culture apparently maintain their differentiated state better than those in monolayer culture. Thus, we consider the spheroid-cultured hepatocytes to be hepatocytes with differentiation characteristics between those
in vivo and in monolayer culture.
In vitro CYP induction studies were performed in monolayer and spheroid cultures. Induction responsiveness and induction ratios of
CYP1A1 were similar between the two conditions. On the other hand, sensitivities of
CYP1A2 were higher in spheroid culture (50 ng/mL) than monolayer culture (250 ng/mL), however, the induction ratios were quite similar between them at the highest dose (3-MC 250 ng/mL). The sensitivity of
CYP3A4 was also similar (5 μg/mL), but induction ratios were higher in spheroid than monolayer cultures. From these results, sensitivities and induction ratios were similar or somewhat higher in spheroid than monolayer cultures.
Information from these in vivo and in vitro enzyme induction studies using human hepatocytes from the same donor may be useful in advancing predictions of enzyme induction in humans. For example, in the present study, 12.2 ng/mL of 3-MC in the in vivo study (20 mg/kg, Day 0) was sufficient to induce a significant decrease in AUC of 3-MC and a significant increase in hCYP1A1 mRNA expression and hCYP1A2 protein expression. On the other hand, the in vitro study revealed that 250 ng/mL of 3-MC was needed to induce a significant increase in hCYP1A1 mRNA in both monolayer and spheroid cultures, and 250 and 50 ng/mL 3-MC was needed to induce a significant increase of hCYP1A2 mRNA in the monolayer and spheroid cultures, respectively, as shown above. The sensitivity to 3-MC was higher in spheroid than monolayer cultures for both hCYP1A1 and 1A2. The sensitivity of CYP1A1 and 1A2 mRNA expression levels to 3-MC was in the following sequence: in vivo > spheroid ≥ monolayer. However, the sensitivity of CYP3A4 for rifampicin was similar between in vivo and in vitro.