A Novel Thylakoid Ascorbate Peroxidase from Jatrophacurcas Enhances Salt Tolerance in Transgenic Tobacco
Abstract
:1. Introduction
2. Results
2.1. Cloning and Characterization of the JctAPX Gene
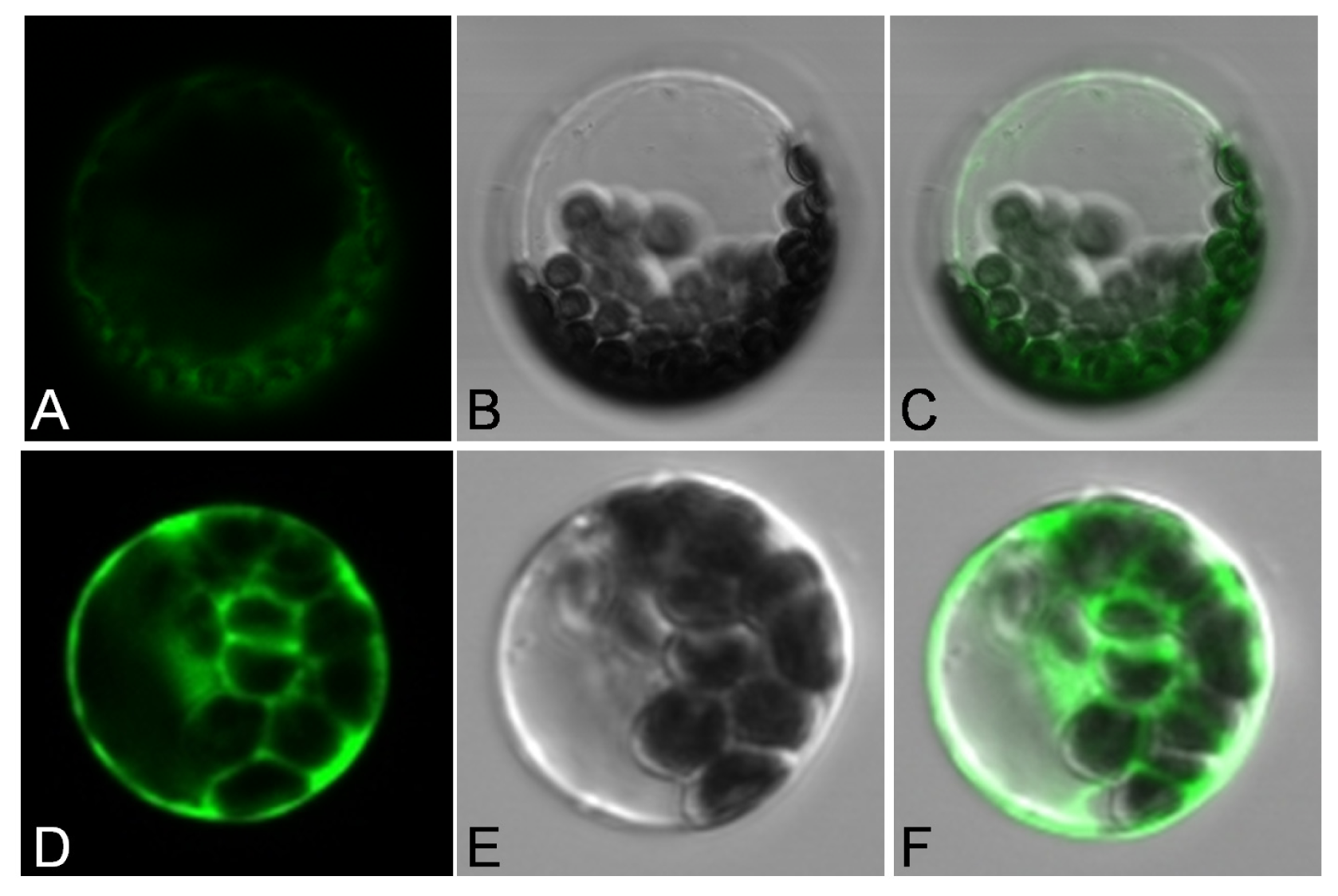
2.2. Subcellular Localization of JctAPX
2.3. Comparison of Expression Levels of JctAPX
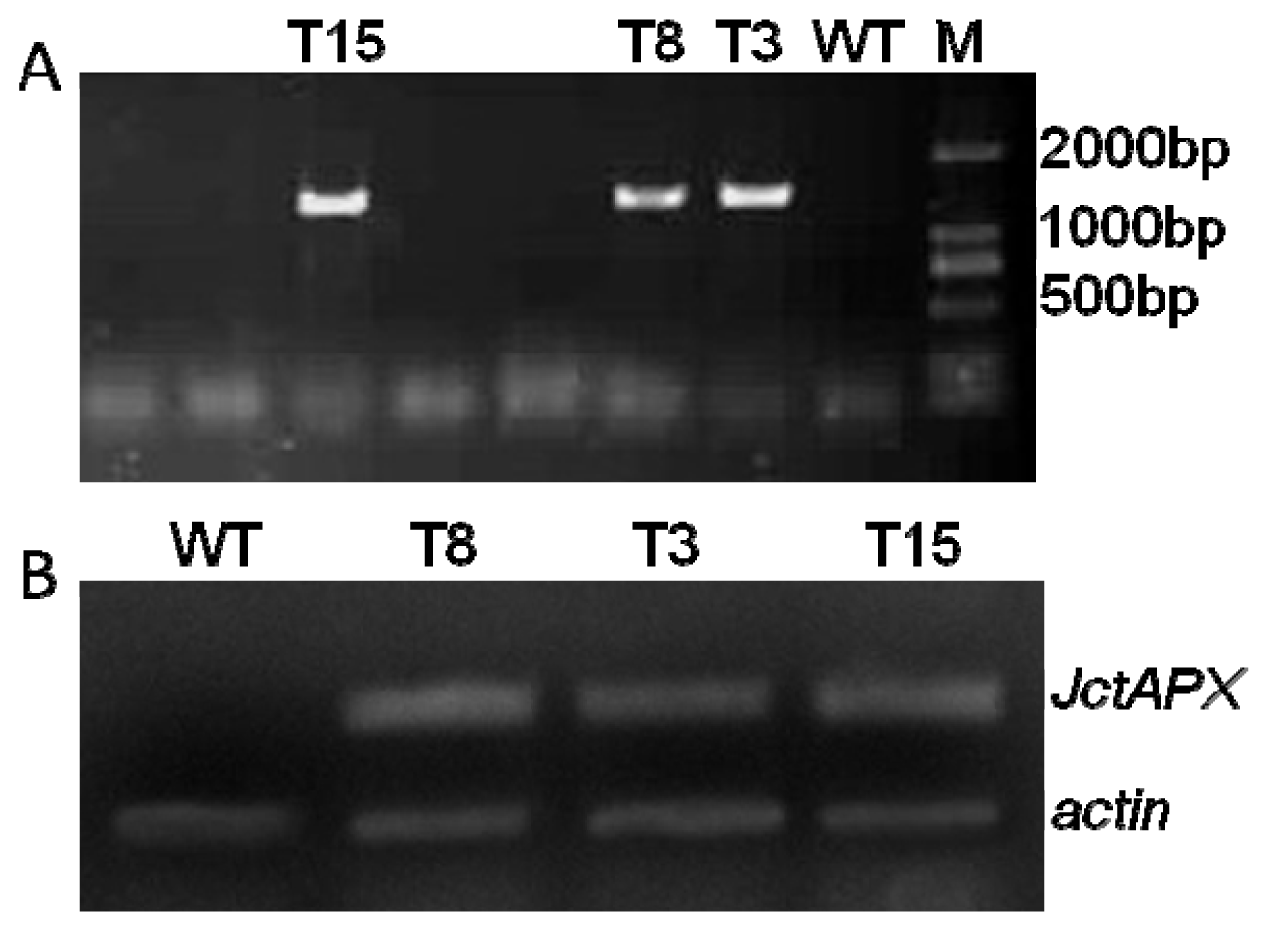
2.4. Molecular Characterization of Transgenic Tobacco
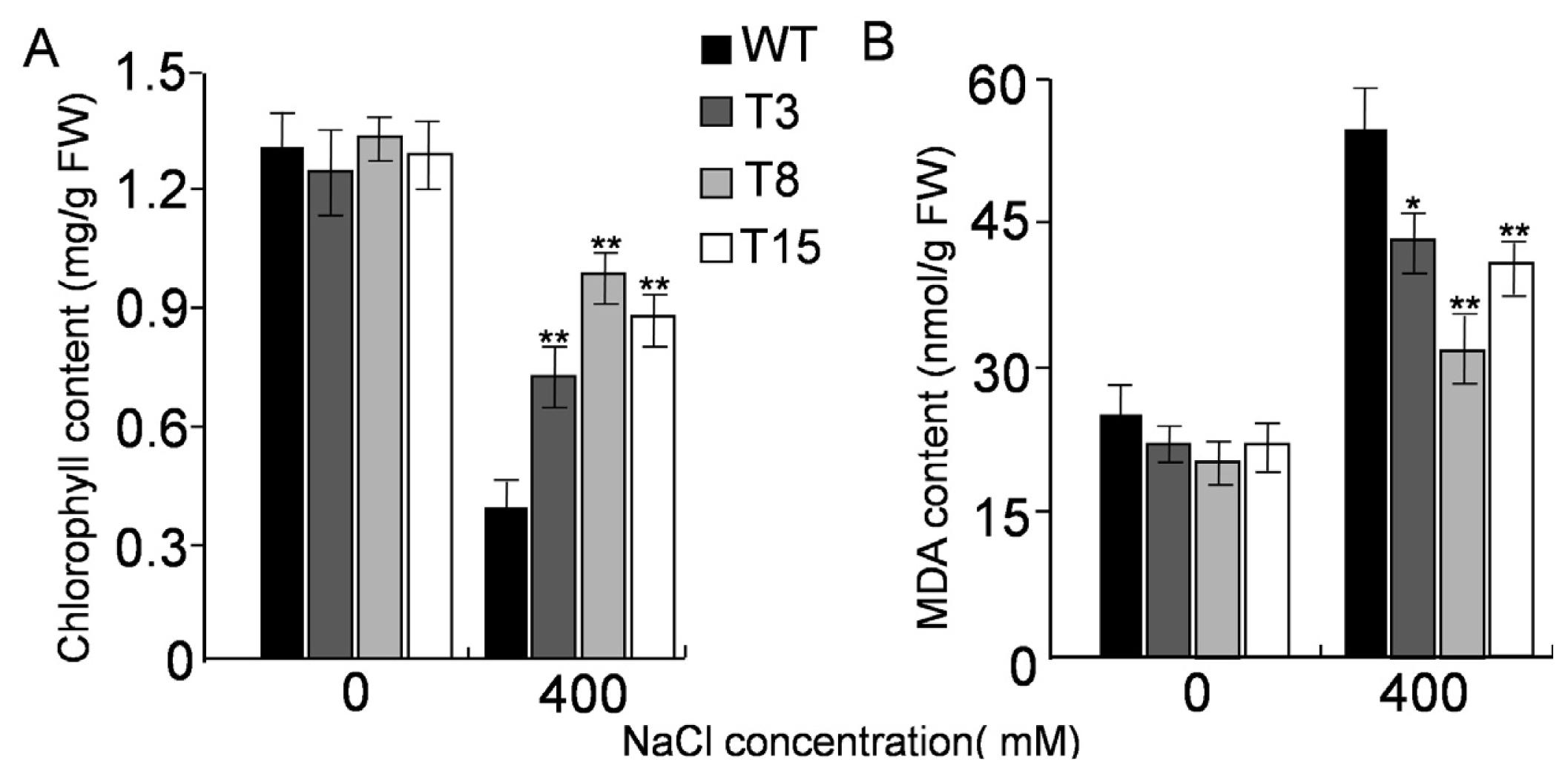
2.5. Salt Tolerance in Transgenic Tobacco Overexpressing JctAPX
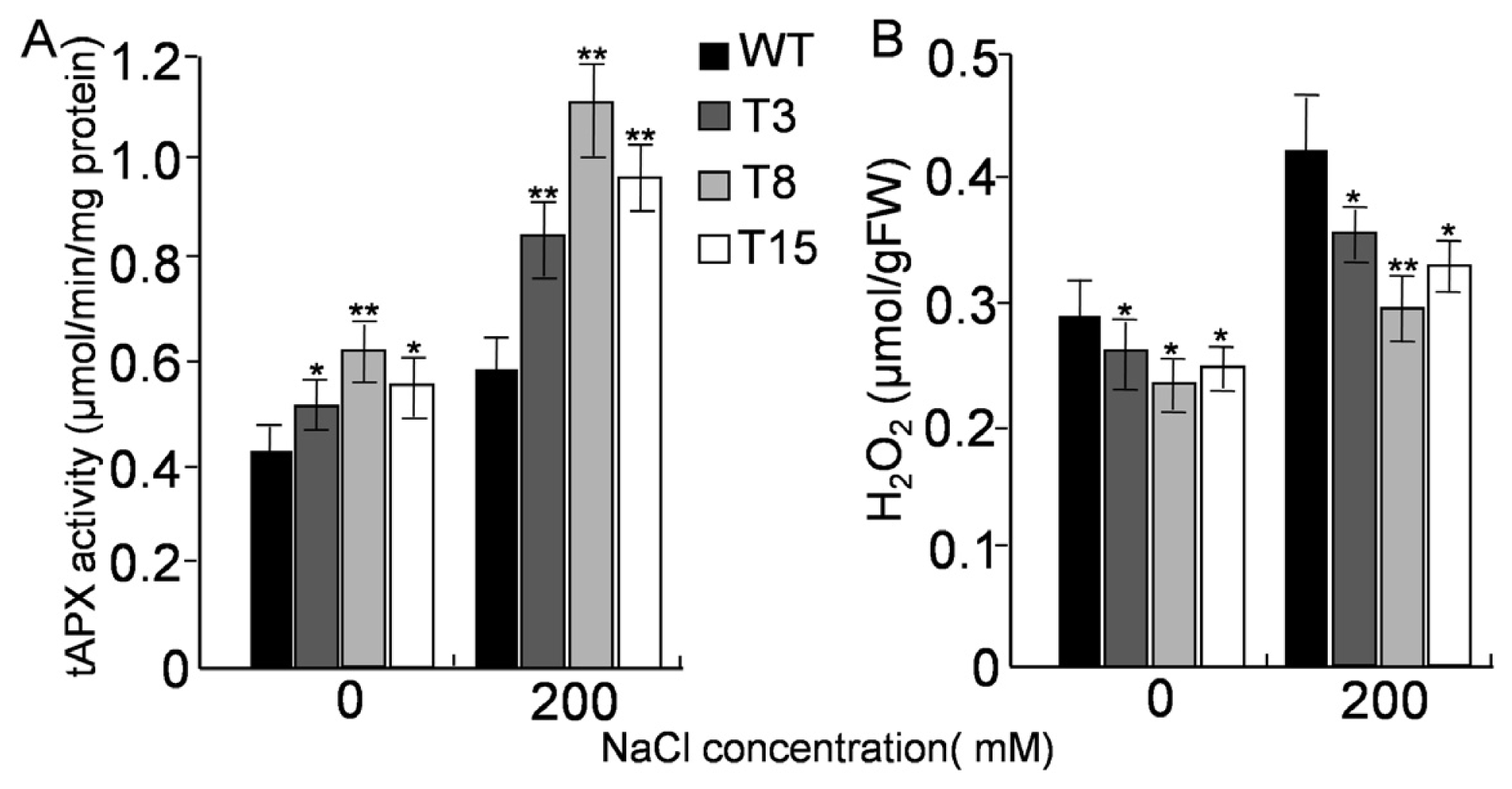
2.6. Activities of tAPX and H2O2 Level under Salt Stress
3. Discussion
4. Experimental Section
4.1. Plant Materials and Treatments
4.2. Cloning and Sequencing of the JctAPX Gene
4.3. Real-Time PCR
4.4. Subcellular Localization of JctAPX
4.5. Plasmid Construction and N. tabacum Transformation
4.6. Salt Tolerance Assay
4.7. Measurement of Chlorophyll Content
4.8. Determination of Lipid Peroxide
4.9. APX Activity Assay and Quantitative Analyses of H2O2
4.10. Data Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Walia, H.; Wilson, C.; Condamine, P.; Liu, X.; Ismail, A.M.; Zeng, L.; Wanamaker, S.I.; Manda, J.; Xu, J.; Cui, X.; et al. Comparative transcriptional profiling of two contrasting rice genotypes under salinity stress during the vegetative growth stage. Plant Physiol 2005, 139, 822–835. [Google Scholar]
- Niu, X.; Bressan, R.A.; Hasegawa, P.M.; Pardo, J.M. Ion homeostasis in NaCl stress environments. Plant Physiol 1995, 109, 735–742. [Google Scholar]
- Mittova, V.; Guy, M.; Tal, M.; Volokita, M. Response of the cultivated tomato and its salt-tolerant relative Lycopersicon pennellii to salt dependant oxidative stress: Increased activities of antioxidant enzymes in root plastids. Free Radic. Res 2002, 36, 195–202. [Google Scholar]
- Zhu, J.K. Plant salt tolerance. Trends Plant Sci 2001, 6, 66–72. [Google Scholar]
- Cavalcanti, F.R.; Oliveira, J.T.A.; Martins-Miranda, A.S.; Viégas, R.A.; Silveira, J.A.G. Superoxide dismutase, catalase and peroxidase activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. New Phytol 2004, 163, 563–571. [Google Scholar]
- Rubio, M.C.; Bustos-Sanmamed, P.; Clemente, M.R.; Becana, M. Effects of salt stress on the expression of antioxidant genes and proteins in the model legume Lotus japonicas. New Phytol 2009, 181, 851–859. [Google Scholar]
- Stepien, P.; Klobus, G. Antioxidant defense in the leaves of C3 and C4 plants under salinity stress. Physiol. Plant 2005, 125, 31–40. [Google Scholar]
- Asada, K. Ascorbate peroxidase—A hydrogen peroxide scavenging enzyme in plants. Physiol. Plant 1992, 85, 235–241. [Google Scholar]
- Shigeoka, S.; Ishikawa, T.; Tamoi, M.; Miyagawa, Y.; Takeda, T.; Yabuta, Y. Regulation and function of ascorbate peroxidase isoenzymes. J. Exp. Bot 2002, 53, 1305–1319. [Google Scholar]
- Jiménez, A.; Hernández, J.A.; del Río, L.A.; Sevilla, F. Evidence for the presence of the ascorbate-glutathione cycle in mitochondria and peroxisomes of pea (Pisumsativum L.) leaves. Plant Physiol 1997, 114, 275–284. [Google Scholar]
- Takeda, Y. Development study on Jatrophacurcas (sabudum) oil as a substitute for diesel engine oil in Thailand. J. Agric. Assoc 1982, 120, 1–8. [Google Scholar]
- Divakara, B.N.; Upadhyaya, H.D.; Wani, S.P.; Gowda, C.L. Biology and genetic improvement of Jatrophacurcas L.: A review. Appl. Energy 2010, 87, 732–742. [Google Scholar]
- Openshaw, K. A review of Jatrophacurcas: An oil plant of unfulfilled promise. Biomass Bioenergy 2000, 19, 1–15. [Google Scholar]
- Achten, W.M.; Mathijs, E.; Verchot, L.; Singh, V.P.; Aerts, R.; Muys, B. Jatropha biodiesel fueling sustainability? Biofuels Bioprod. Bioref 2007, 1, 283–291. [Google Scholar]
- Gao, S.; Ou-yang, C.; Tang, L.; Zhu, J.; Xu, Y.; Wang, S.; Chen, F. Growth and antioxidant responses in Jatrophacurcas seedling exposed to mercury toxicity. J. Hazard. Mater 2010, 182, 591–597. [Google Scholar]
- Sun, J.; Li, L.; Liu, M.; Wang, M.; Ding, M.; Deng, S.; Chen, S. Hydrogen peroxide and nitric oxide mediate K+/Na+ homeostasis and antioxidant defense in NaCl-stressed callus cells of two contrasting poplars. Plant Cell Tissue Organ Cult 2010, 103, 205–215. [Google Scholar]
- Foyer, C.H.; Shigeoka, S. Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol 2011, 155, 93–100. [Google Scholar]
- D’Arcy-Lameta, A.; Ferrari-Iliou, R.; Contour-Ansel, D.; Pham-Thi, A.T.; Zuily-Fodil, Y. Isolation and characterization of four ascorbate peroxidase cDNAs responsive to water deficit in cowpea leaves. Ann. Bot 2006, 97, 133–140. [Google Scholar]
- Sun, W.H.; Duan, M.; Shu, D.F.; Yang, S.; Meng, Q.W. Over-expression of StAPX in tobacco improves seed germination and increases early seedling tolerance to salinity and osmotic stresses. Plant Cell Rep 2010, 29, 917–926. [Google Scholar]
- Yoshimura, K.; Yabuta, Y.; Ishikawa, T.; Shigeoka, S. Expression of spinach ascorbate peroxidase isoenzymes in response to oxidative stresses. Plant Physiol 2000, 123, 223–234. [Google Scholar]
- Song, X.S.; Hu, W.H.; Mao, W.H.; Ogweno, J.O.; Zhou, Y.H.; Yu, J.Q. Response of ascorbate peroxidase isoenzymes and ascorbate regeneration system to abiotic stresses in Cucumissativus L. Plant Physiol. Biochem 2005, 43, 1082–1088. [Google Scholar]
- De Azevedo Neto, A.D.; Prisco, J.T.; Eneas-Filho, J.; Abreu, C.E.B.; Gomes-Filho, E. Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ. Exp. Bot 2006, 56, 87–94. [Google Scholar]
- Badawi, G.H.; Kawano, N.; Yamauchi, Y.; Shimada, E.; Sasaki, R.; Kubo, A.; Tanaka, K. Over-expression of ascorbate peroxidase in tobacco chloroplasts enhances the tolerance to salt stress and water deficit. Physiol. Plantarum 2004, 121, 231–238. [Google Scholar]
- Badawi, G.H.; Yamauchi, Y.; Shimada, E.; Sasaki, R.; Kawano, N.; Tanaka, K.; Tanaka, K. Enhanced tolerance to salt stress and water deficit by overexpressing superoxide dismutase in tobacco (Nicotianatabacum) chloroplasts. Plant Sci 2004, 166, 919–928. [Google Scholar]
- Faize, M.; Burgos, L.; Faize, L.; Piqueras, A.; Nicolas, E.; Barba-Espin, G.; Hernandez, J.A. Involvement of cytosolic ascorbate peroxidase and Cu/Zn-superoxide dismutase for improved tolerance against drought stress. J. Exp. Bot 2011, 62, 2599–2613. [Google Scholar]
- Diaz-Vivancos, P.; Faize, M.; Barba-Espin, G.; Faize, L.; Petri, C.; Hernández, J.A.; Burgos, L. Ectopic expression of cytosolic superoxide dismutase and ascorbate peroxidase leads to salt stress tolerance in transgenic plums. Plant Biotechnol. J 2013, 11, 976–985. [Google Scholar]
- Li, Y.J.; Hai, R.L.; Du, X.H.; Jiang, X.N.; Lu, H. Overexpression of a Populus peroxisomal ascorbate peroxidase (PpAPX) gene in tobacco plants enhances stress tolerance. Plant Breed 2009, 128, 404–410. [Google Scholar]
- Yabuta, Y.; Motoki, T.; Yoshimura, K.; Takeda, T.; Ishikawa, T.; Shigeoka, S. Thylakoid membrane-bound ascorbate peroxidase is a limiting factor of antioxidative systems under photo-oxidative stress. Plant J 2002, 32, 915–925. [Google Scholar]
- Kuo, M.C.; Kao, C.H. Aluminum effects on lipid peroxidation and antioxidative enzyme activities in rice leaves. Biol. Plant 2003, 46, 149–152. [Google Scholar]
- Li, K.; Pang, C.H.; Ding, F.; Sui, N.; Feng, Z.T.; Wang, B.S. Overexpression of Suaeda salsa stroma ascorbate peroxidase in Arabidopsis chloroplasts enhances salt tolerance of plants. South Afr. J. Bot 2012, 78, 235–245. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 1962, 15, 473–497. [Google Scholar]
- Jefferson, A.R. Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol. Biol. Reporter 1987, 5, 387–405. [Google Scholar]
- Yoo, S.D.; Cho, Y.H.; Sheen, J. Arabidopsis mesophyll protoplasts: A versatile cell system for transient gene expression analysis. Nat. Protoc 2007, 2, 1565–1572. [Google Scholar]
- Lu, H.; Zeng, Q.Y.; Zhao, Y.L.; Jiang, X.N. Xylem-specific expression of a GRP1.8 promoter: 4CL gene construct in transgenic tobacco. Plant Growth Regul 2003, 41, 279–286. [Google Scholar]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol 1987, 148, 350–382. [Google Scholar]
- Cao, Y.J.; Wei, Q.; Liao, Y.; Song, H.L.; Li, X.; Xiang, C.B.; Kuai, B.K. Ectopic overexpression of AtHDG11 in tall fescue resulted in enhanced tolerance to drought and salt stress. Plant Cell Rep 2009, 28, 579–588. [Google Scholar]
- Robinson, S.P.; Downton, W.J.S.; Millhouse, J.A. Photosynthesis and ion content of leaves and isolated chloroplasts of salt-stressed spinach. Plant Physiol 1983, 73, 238–242. [Google Scholar]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 1981, 22, 867–880. [Google Scholar]
- Lin, C.C.; Kao, C.H. Abscisic acid induced changes in cell wall peroxidase activity and hydrogen peroxide level in roots of rice seedlings. Plant Sci 2001, 160, 323–329. [Google Scholar]





| Primer | Sequence(5′-3′) |
|---|---|
| JctAPX1 | AG(A/T)AGGAT(G/C)AA(G/C)ATC(T/A)ACTTG(T/C)ATT |
| JctAPX2 | AT(T/C)G(C/T)CAAC(A/G)ACC(T/C)AC(A/G)AGC(T/C)A |
| JctAPX3 | CTTTCCAGTTGAGTATTTGGCTGCT |
| JctAPX4 | CTGCAACACCATCTAGCACAATACC |
| JctAPX5 | TTCTTTTTGATGATCCTTCGTTCAG |
| JctAPX6 | ATACTCAACTGGAAAGAGAGAATTG |
| JctAPX7 | GAAACTCCTTCAGCCAATCAA |
| JctAPX8 | TGAAACATCCACCCTTCCATAC |
| Actin-F | ATGAGCTTCGAGTTGCACCA |
| Actin-R | AGCATCAGTGAGATCACGAC |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, Z.; Bao, H.; Cai, J.; Han, J.; Zhou, L. A Novel Thylakoid Ascorbate Peroxidase from Jatrophacurcas Enhances Salt Tolerance in Transgenic Tobacco. Int. J. Mol. Sci. 2014, 15, 171-185. https://doi.org/10.3390/ijms15010171
Liu Z, Bao H, Cai J, Han J, Zhou L. A Novel Thylakoid Ascorbate Peroxidase from Jatrophacurcas Enhances Salt Tolerance in Transgenic Tobacco. International Journal of Molecular Sciences. 2014; 15(1):171-185. https://doi.org/10.3390/ijms15010171
Chicago/Turabian StyleLiu, Zhibin, Han Bao, Jin Cai, Jun Han, and Lirong Zhou. 2014. "A Novel Thylakoid Ascorbate Peroxidase from Jatrophacurcas Enhances Salt Tolerance in Transgenic Tobacco" International Journal of Molecular Sciences 15, no. 1: 171-185. https://doi.org/10.3390/ijms15010171




