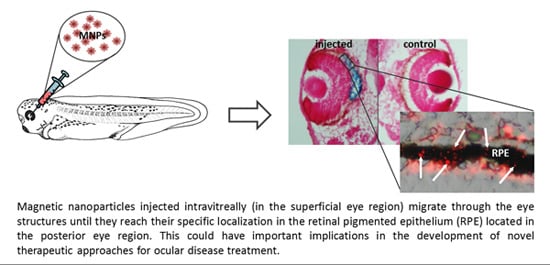
Magnetic Nanoparticles as Intraocular Drug Delivery System to Target Retinal Pigmented Epithelium (RPE)
Abstract
:1. Introduction
2. Results and Discussion
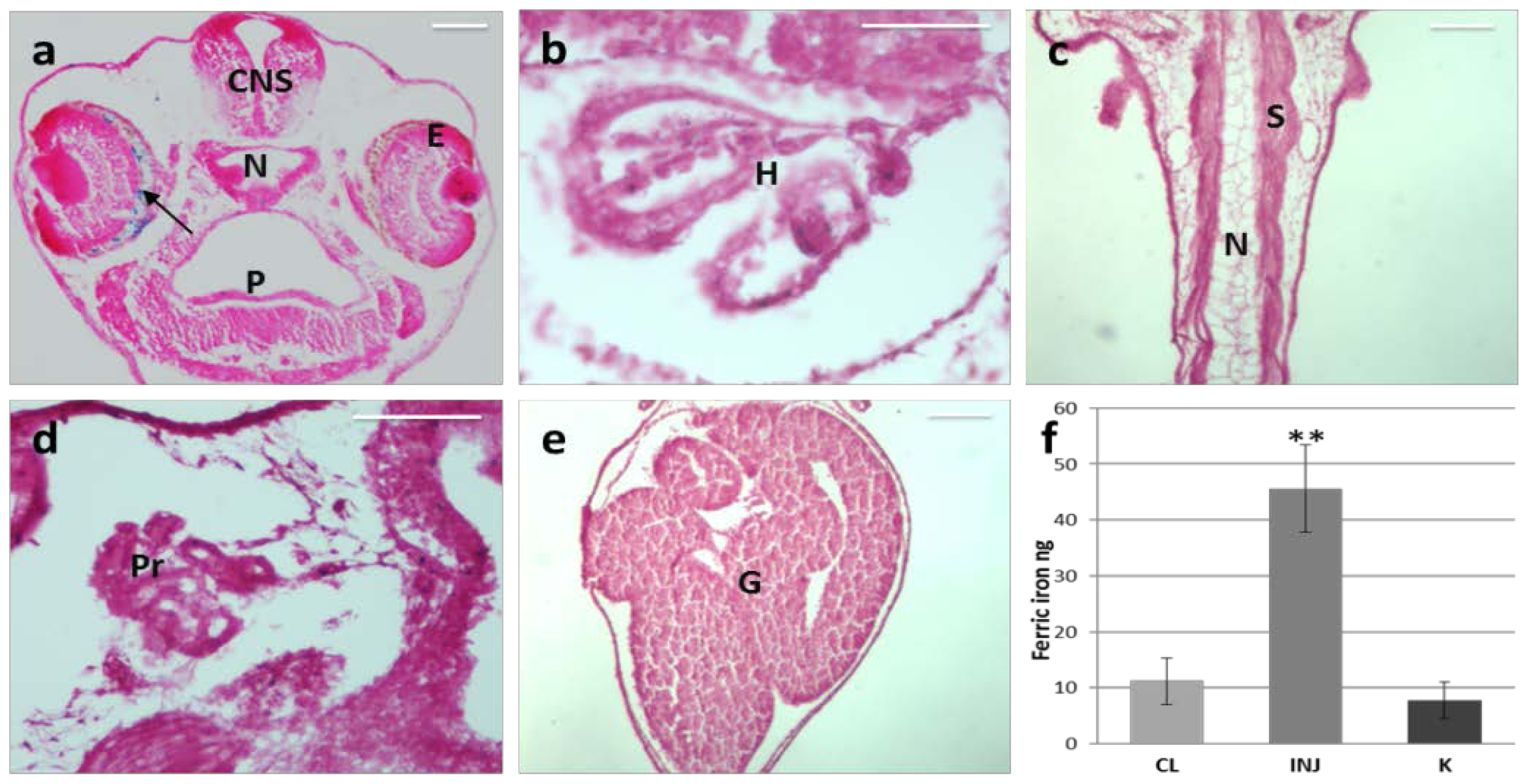
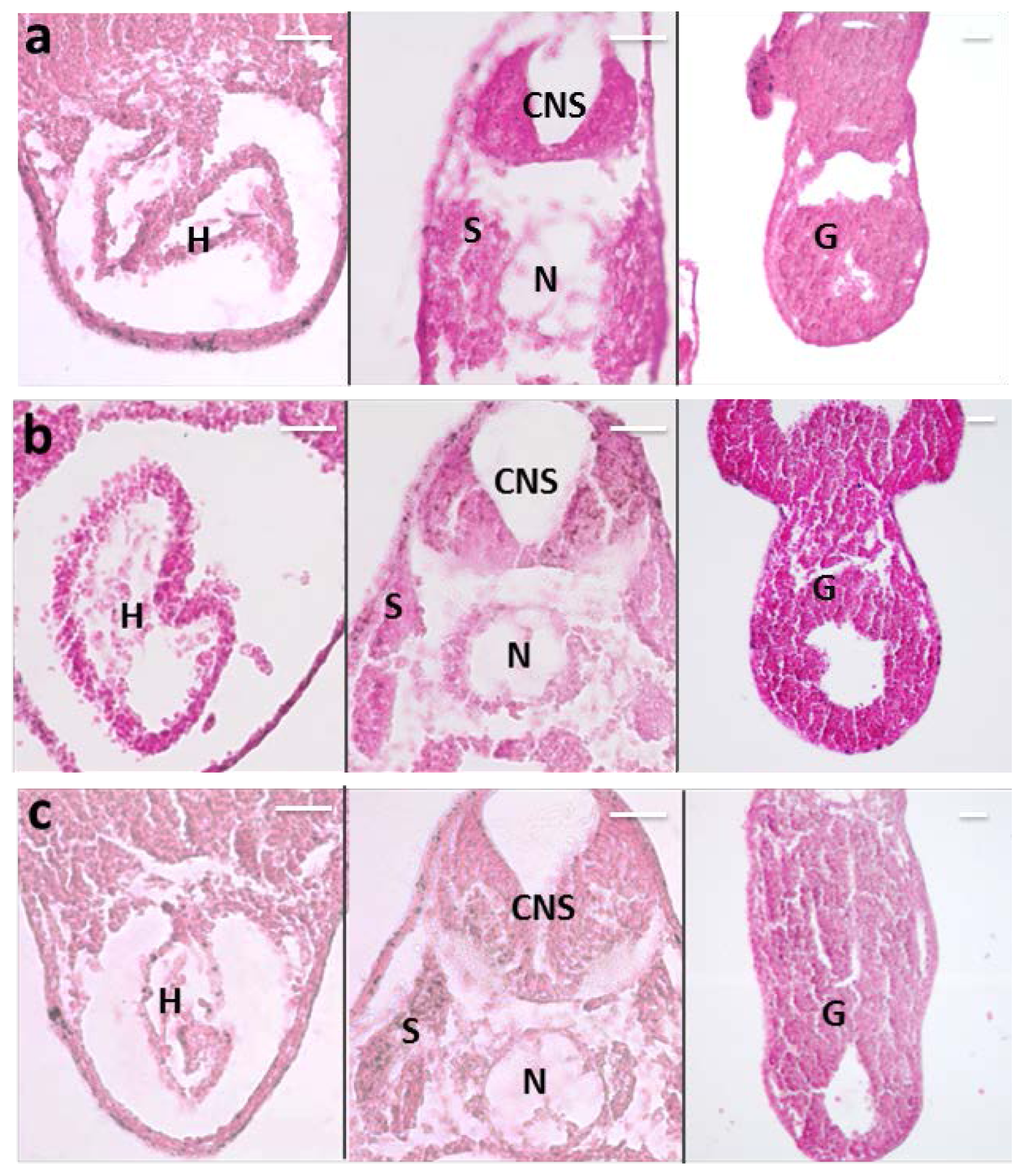
2.1. MNP Biodistribution
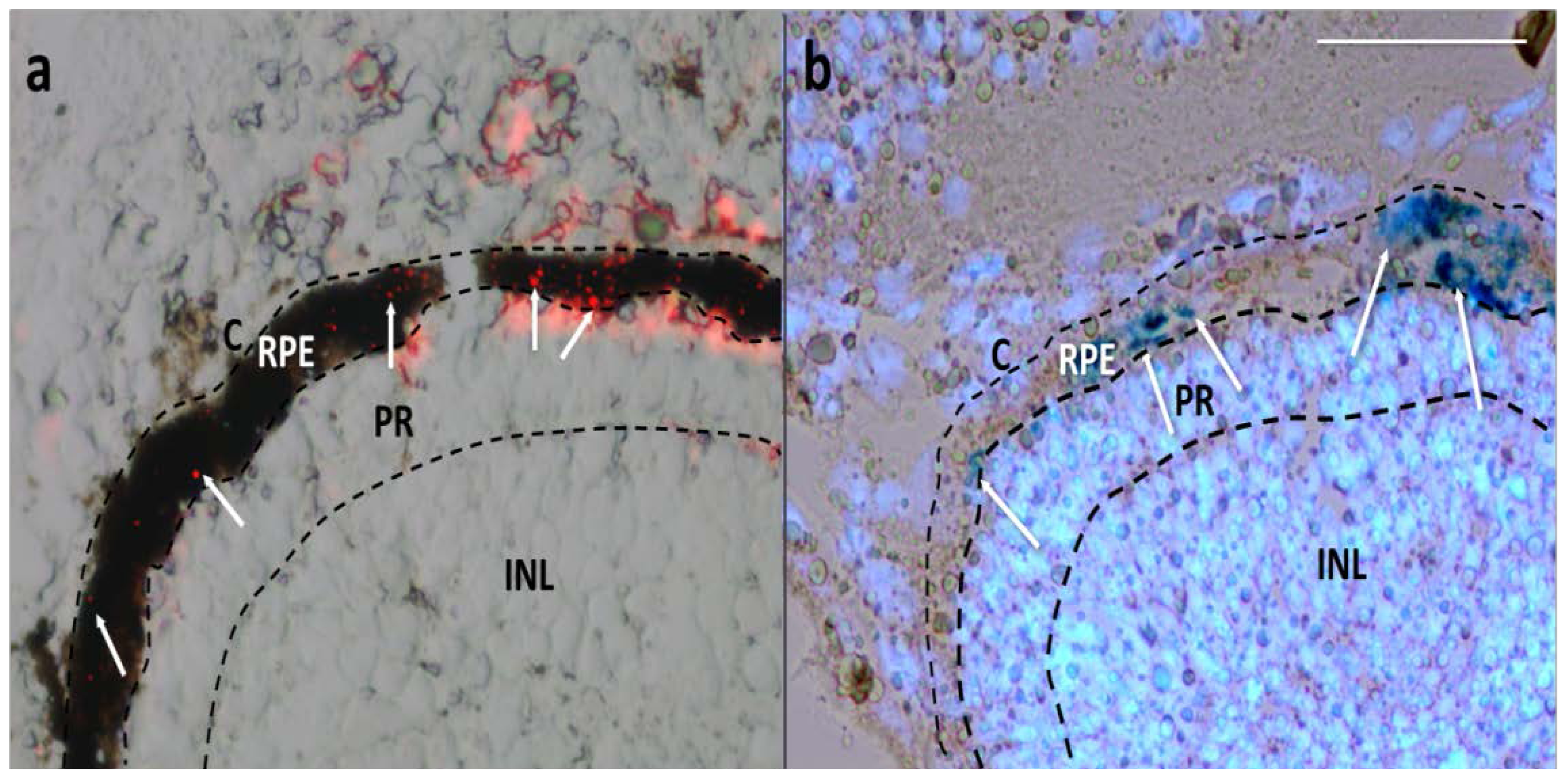
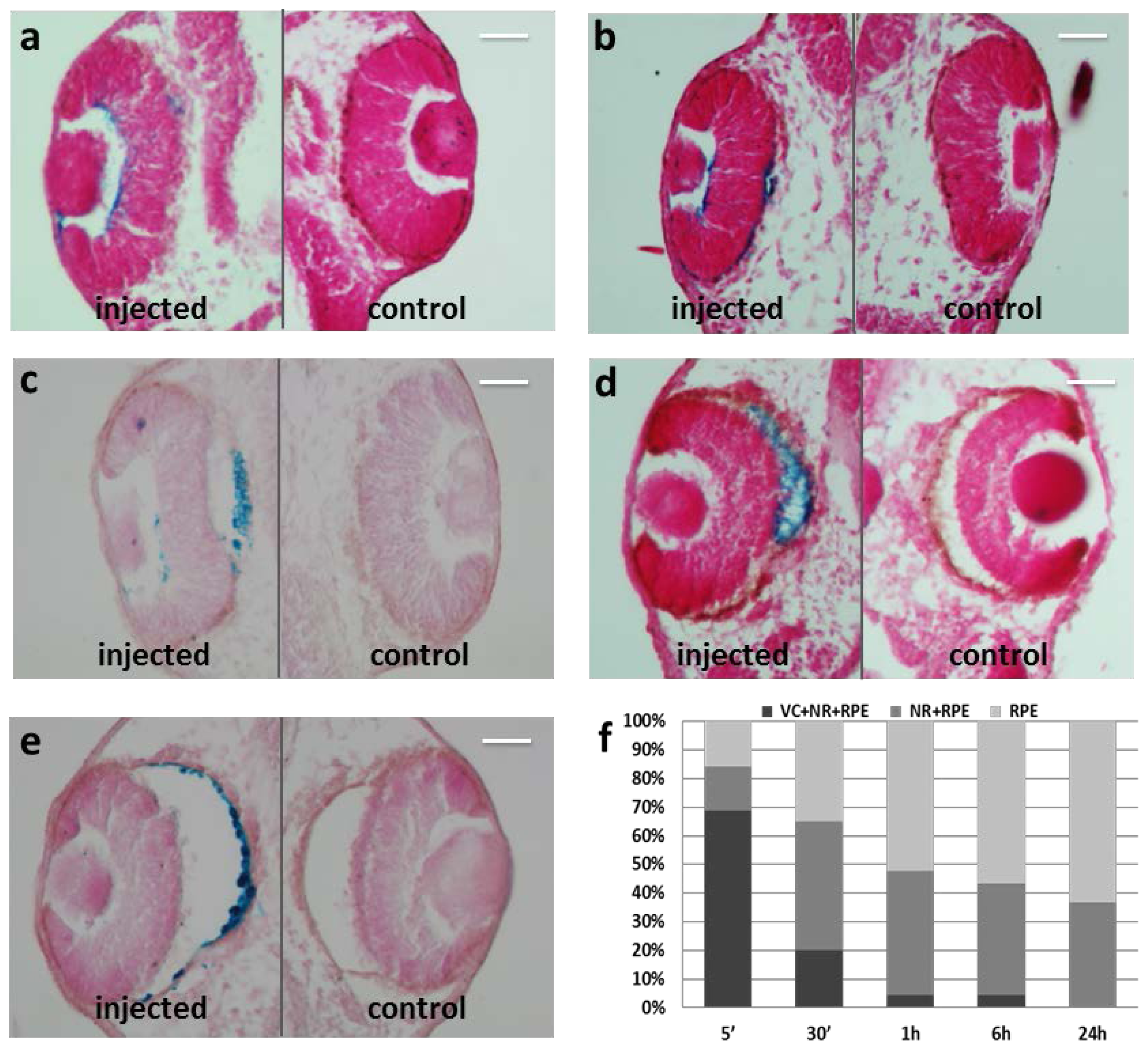
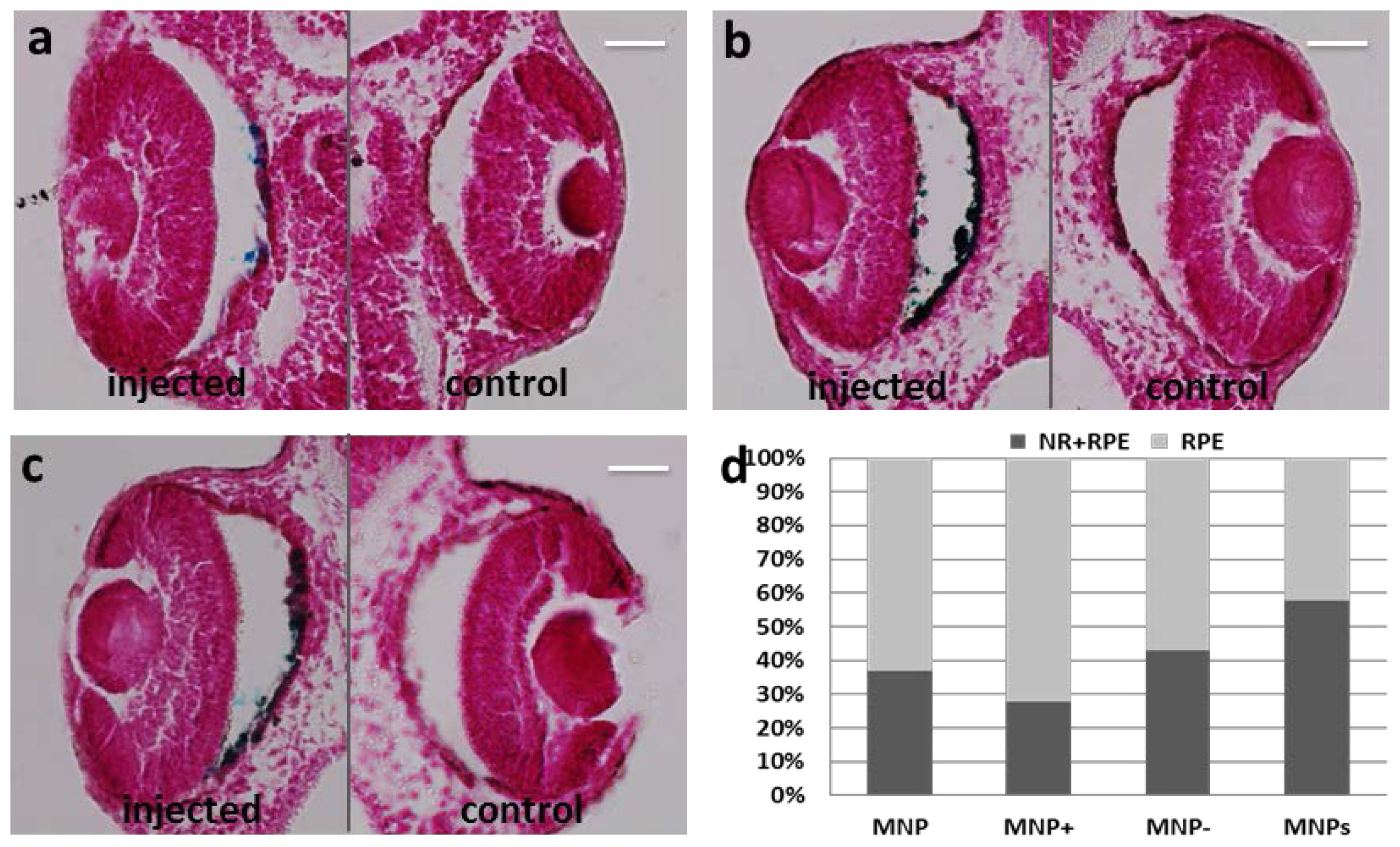
2.2. Particles Injected Intraocularly Localize behind the Neural Retina
2.3. MNP Localization Is not Driven by Superficial Charge and Size
2.4. MNP RPE/Choroid Localization Is not Species-Specific
3. Experimental Section
3.1. Nanoparticles
3.1.1. Transmission Electron Microscopy (TEM)
3.1.2. Dynamic Light Scattering (DLS) and Zeta Potential
3.2. Embryo Preparation
3.3. Embryo Microinjections
3.4. Histochemical Analysis
3.5. Iron Content Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lebrun-Julien, F.; di Polo, A. Molecular and cell-based approaches for neuroprotection in glaucoma. Optom. Vis. Sci 2008, 85, 417–424. [Google Scholar]
- Sultana, Y.; Jain, R.; Aqil, M.; Ali, A. Review of ocular drug delivery. Curr. Drug Deliv 2006, 3, 207–217. [Google Scholar]
- Raghava, S.; Hammond, M.; Kompella, U.B. Periocular routes for retinal drug delivery. Expert Opin. Drug Deliv 2004, 1, 99–114. [Google Scholar]
- Peyman, G.A.; Lad, E.M.; Moshfeghi, D.M. Intravitreal injection of therapeutic agents. Retina-J. Ret. Vit. Dis 2009, 29, 875–912. [Google Scholar]
- Gaudana, R.; Jwala, J.; Boddu, S.H.S.; Mitra, A.K. Recent perspectives in ocular drug delivery. Pharm. Res 2009, 26, 1197–1216. [Google Scholar]
- Sahoo, S.K.; Dilnawaz, F.; Krishnakumar, S. Nanotechnology in ocular drug delivery. Drug Dis. Today 2008, 13, 144–151. [Google Scholar]
- Sakurai, E.; Ozeki, H.; Kunou, N.; Ogura, Y. Effect of particle size of polymeric nanospheres on intravitreal kinetics. Ophthalmic Res 2001, 33, 31–36. [Google Scholar]
- Bourges, J.L.; Gautier, S.E.; Delie, F.; Bejjani, R.A.; Jeanny, J.C.; Gurny, R.; BenEzra, D.; Behar-Cohen, F.F. Ocular drug delivery targeting the retina and retinal pigment epithelium using polylactide nanoparticles. Investig. Ophthalmol. Vis. Sci 2003, 44, 3562–3569. [Google Scholar]
- Kim, H.; Robinson, S.B.; Csaky, K.G. Investigating the movement of intravitreal human serum albumin nanoparticles in the vitreous and retina. Pharm. Res 2009, 26, 329–337. [Google Scholar]
- Sun, C.; Lee, J.S.; Zhang, M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev 2008, 60, 1252–1265. [Google Scholar]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar]
- Gupta, A.K.; Naregalkar, R.R.; Vaidya, V.D.; Gupta, M. Recent advances on surface engineering of magnetic iron oxide nanoparticles and their biomedical applications. Nanomedicine 2007, 2, 23–39. [Google Scholar]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev 2008, 108, 2064–2110. [Google Scholar]
- Misra, R.D.K. Magnetic nanoparticle carrier for targeted drug delivery: Perspective, outlook and design. Mater. Sci. Technol 2008, 24, 1011–1019. [Google Scholar]
- McCarthy, J.R.; Kelly, K.A.; Sun, E.Y.; Weissleder, R. Targeted delivery of multifunctional magnetic nanoparticles. Nanomedicine 2007, 2, 153–167. [Google Scholar]
- Edelman, E.R.; Langer, R. Optimization of release from magnetically controlled polymeric drug-release devices. Biomaterials 1993, 14, 621–626. [Google Scholar]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D 2003, 36, R167–R181. [Google Scholar]
- Pankhurst, Q.A.; Thanh, N.T.K.; Jones, S.K.; Dobson, J. Progress in applications of magnetic nanoparticles in biomedicine. J. Phys. D 2009, 42. [Google Scholar] [CrossRef]
- Goya, G.F.; Grazu, V.; Ibarra, M.R. Magnetic nanoparticles for cancer therapy. Curr. Nanosci 2008, 4, 1–16. [Google Scholar]
- Van Landeghem, F.K.H.; Maier-Hauff, K.; Jordan, A.; Hoffmann, K.T.; Gneveckow, U.; Scholz, R.; Thiesen, B.; Brück, W.; von Deimling, V. Post-mortem studies in glioblastoma patients treated with thermotherapy using magnetic nanoparticles. Biomaterials 2009, 30, 52–57. [Google Scholar]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Brück, W.; von Deimling, V. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neuro-Oncol 2011, 103, 317–324. [Google Scholar]
- Zysler, R.; Berra, A.; Gurman, P.; Auciello, O.; Saravia, M.J. Material for Medical Use Comprising Nanoparticles with Superparamagnetic Properties and Its Utilization in Surgery. U.S. Patent 20130225906, 29 August 2013. [Google Scholar]
- Raju, H.B.; Hu, Y.; Vedula, A.; Dubovy, S.R.; Goldberg, J.L. Evaluation of magnetic micro- and nanoparticle toxicity to ocular Tissues. PLoS One 2011, 6, e17452. [Google Scholar]
- Giannaccini, M.; Cuschieri, A.; Dente, L.; Raffa, V. Non-mammalian vertebrate embryos as models in nanomedicine. Nanomedicine 2013. [Google Scholar] [CrossRef]
- Lévy, M.; Gazeau, F.; Bacri, J.; Wilhelm, C.; Devaud, M. Modeling magnetic nanoparticle dipole-dipole interactions inside living cells. Phys. Rev. B 2011, 84, 075480. [Google Scholar]
- Bulte, J.W.M. In Vivo MRI cell tracking: Clinical studies. Am. J. Roentgenol 2009, 193, 314–325. [Google Scholar]
- Kim, H.S.; Oh, S.Y.; Joo, H.J.; Son, K.R.; Song, I.C.; Moon, W.K. The effects of clinically used MRI contrast agents on the biological properties of human mesenchymal stem cells. NMR Biomed 2010, 23, 514–522. [Google Scholar]
- Strauss, O. The retinal pigment epithelium in visual function. Physiol. Rev 2005, 85, 845–881. [Google Scholar]
- Peeters, L.; Sanders, N.N.; Braeckmans, K.; Boussery, K.; van de Voorde, J.; de Smedt, S.C.; Demeester, J. Vitreous: A barrier to nonviral ocular gene therapy. Investig. Ophthalmol. Vis. Sci 2005, 46, 3553–3561. [Google Scholar]
- Calatayud, M.P.; Riggio, C.; Raffa, V.; Sanz, B.; Torres, T.E.; Ibarra, M.R.; Hoskins, C.; Cuschieri, A.; Wang, L.; Pinkernelle, J.; et al. Neuronal cells loaded with PEI-coated Fe3O4 nanoparticles for magnetically guided nerve regeneration. J. Mater. Chem. B 2013, 1, 3607–3616. [Google Scholar]
- Nguyen-Legros, J.; Hicks, D. Renewal of photoreceptor outer segments and their phagocytosis by the retinal pigment epithelium. Int. Rev. Cytol 2000, 196, 245–313. [Google Scholar]
- Kimura, H.; Ogura, Y.; Moritera, T.; Honda, Y.; Tabata, Y.; Ikada, Y. In-Vitro phagocytosis of polylactide microspheres by retinal-pigment epithelial-cells and intracellular drug-release. Curr. Eye Res 1994, 13, 353–360. [Google Scholar]
- Ogura, Y.; Kimura, H. Biodegradable polymer microspheres for targeted drug delivery to the retinal pigment epithelium. Surv. Ophthalmol 1995, 39, S17–S24. [Google Scholar]
- Nieuwkoop, P.D.; Faber, J. Normal Table of Xenopus laevis (Daudin); Elsevier: Amsterdam, The Netherlands, 1967. [Google Scholar]
- Newport, J.; Kirschner, M. A major developmental transition in early Xenopus embryos: II. Control of the onset of transcription. Cell 1982, 30, 687–696. [Google Scholar]
- Sive, H.L.; Grainger, R.M.; Harland, R.M. Early development of Xenopus laevis: A laboratory manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2000. [Google Scholar]
- Westerfield, M. The Zebrafish Book, 5th ed; A Guide for the Laboratory Use of Zebrafish (Danio Rerio); University of Oregon Press: Eugene, OR, USA, 2007. [Google Scholar]
- Arraes, J.C.; Tedesco, R.C.; Arraes, T.A.; Silva, E.B.; Ventura, A.; Avila, M.P. Intravitreal bevacizumab in pigmented rabbit eyes: Histological analysis 90 days after injection. Arq. Bras. Oftalmol 2009, 72, 656–660. [Google Scholar]

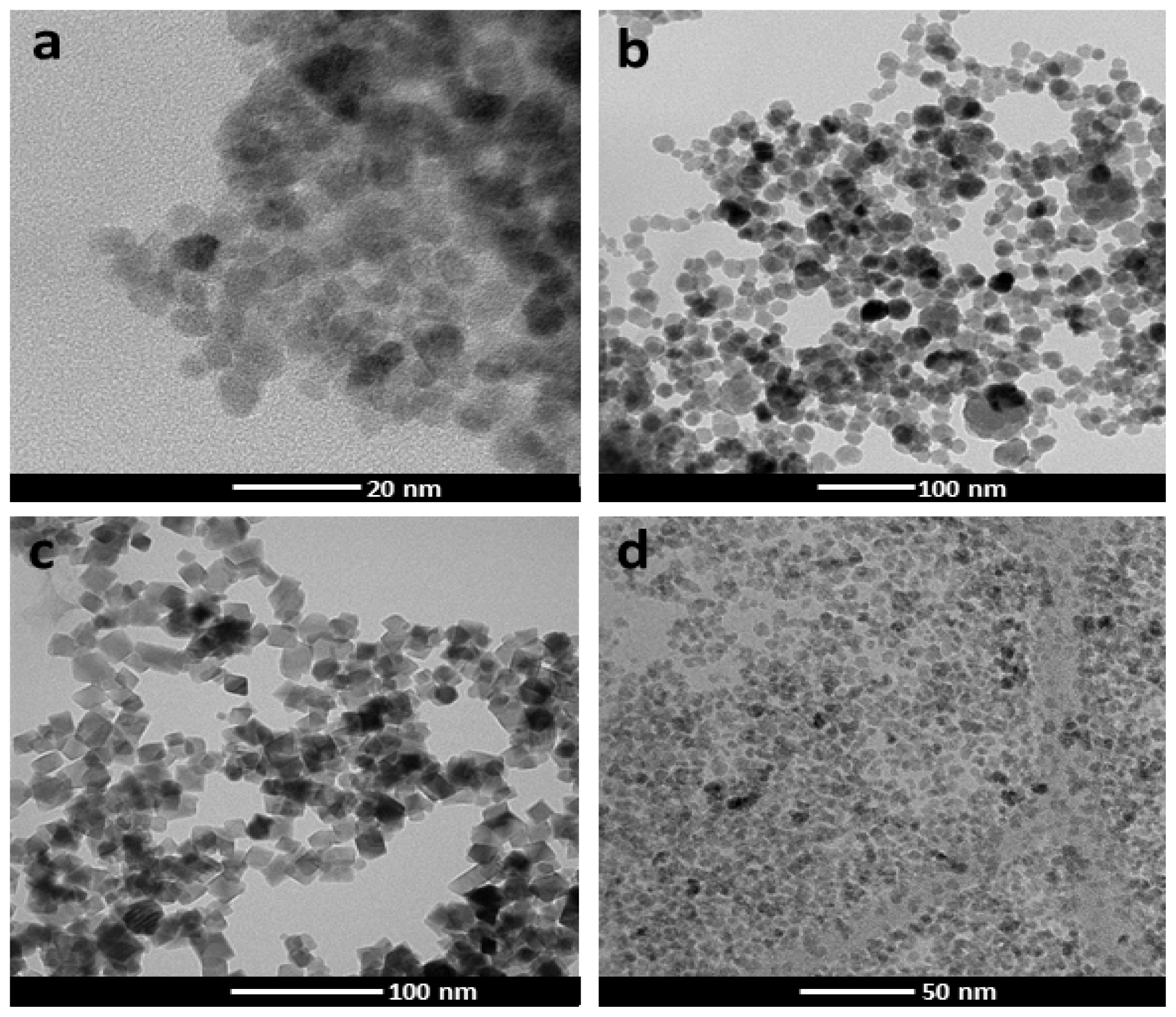

| MNP | MNP− | MNP+ | MNPs | |
|---|---|---|---|---|
| Hydrodynamic Radius | 252 nm | 158 nm | 95 nm | 17 nm |
| Zp (@water) | −17.22 mV | −28 mV | +30 mV | 0.4 mV |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Giannaccini, M.; Giannini, M.; Calatayud, M.P.; Goya, G.F.; Cuschieri, A.; Dente, L.; Raffa, V. Magnetic Nanoparticles as Intraocular Drug Delivery System to Target Retinal Pigmented Epithelium (RPE). Int. J. Mol. Sci. 2014, 15, 1590-1605. https://doi.org/10.3390/ijms15011590
Giannaccini M, Giannini M, Calatayud MP, Goya GF, Cuschieri A, Dente L, Raffa V. Magnetic Nanoparticles as Intraocular Drug Delivery System to Target Retinal Pigmented Epithelium (RPE). International Journal of Molecular Sciences. 2014; 15(1):1590-1605. https://doi.org/10.3390/ijms15011590
Chicago/Turabian StyleGiannaccini, Martina, Marianna Giannini, M. Pilar Calatayud, Gerardo F. Goya, Alfred Cuschieri, Luciana Dente, and Vittoria Raffa. 2014. "Magnetic Nanoparticles as Intraocular Drug Delivery System to Target Retinal Pigmented Epithelium (RPE)" International Journal of Molecular Sciences 15, no. 1: 1590-1605. https://doi.org/10.3390/ijms15011590