Optimization of Synthesis Parameters for Mesoporous Shell Formation on Magnetic Nanocores and Their Application as Nanocarriers for Docetaxel Cancer Drug
Abstract
:1. Introduction
2. Results and Discussion
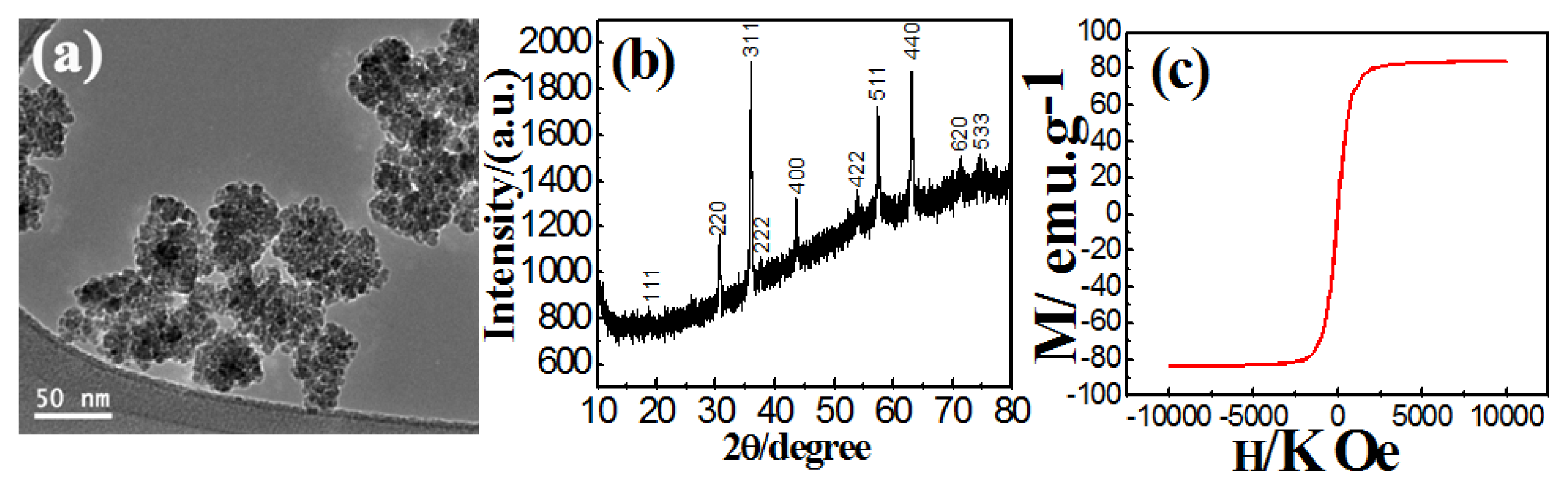
2.1. Synthesis of Fe3O4 Nanoparticles
2.2. Dense Silica Coating of Magnetic Nanoparticles
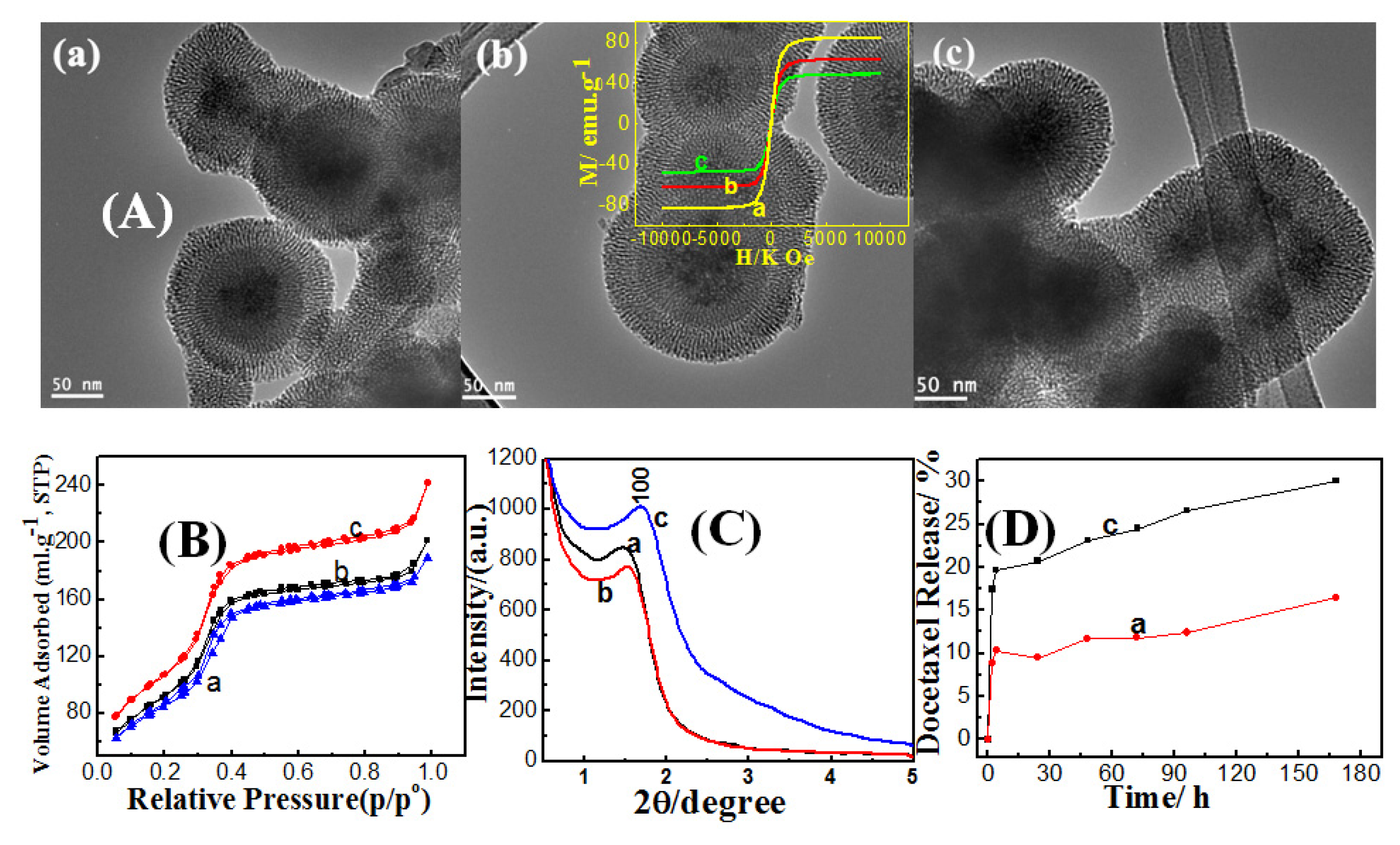
2.2.1. Effect of Ethanol Concentration
2.2.2. Effect of Ammonia Concentration
2.3. Mesoporous Shell Formation on Silica Coated Magnetic Nanoparticles
2.3.1. Dense Silica Coating Step Parameters Influencing Mesoporous Shell Formation
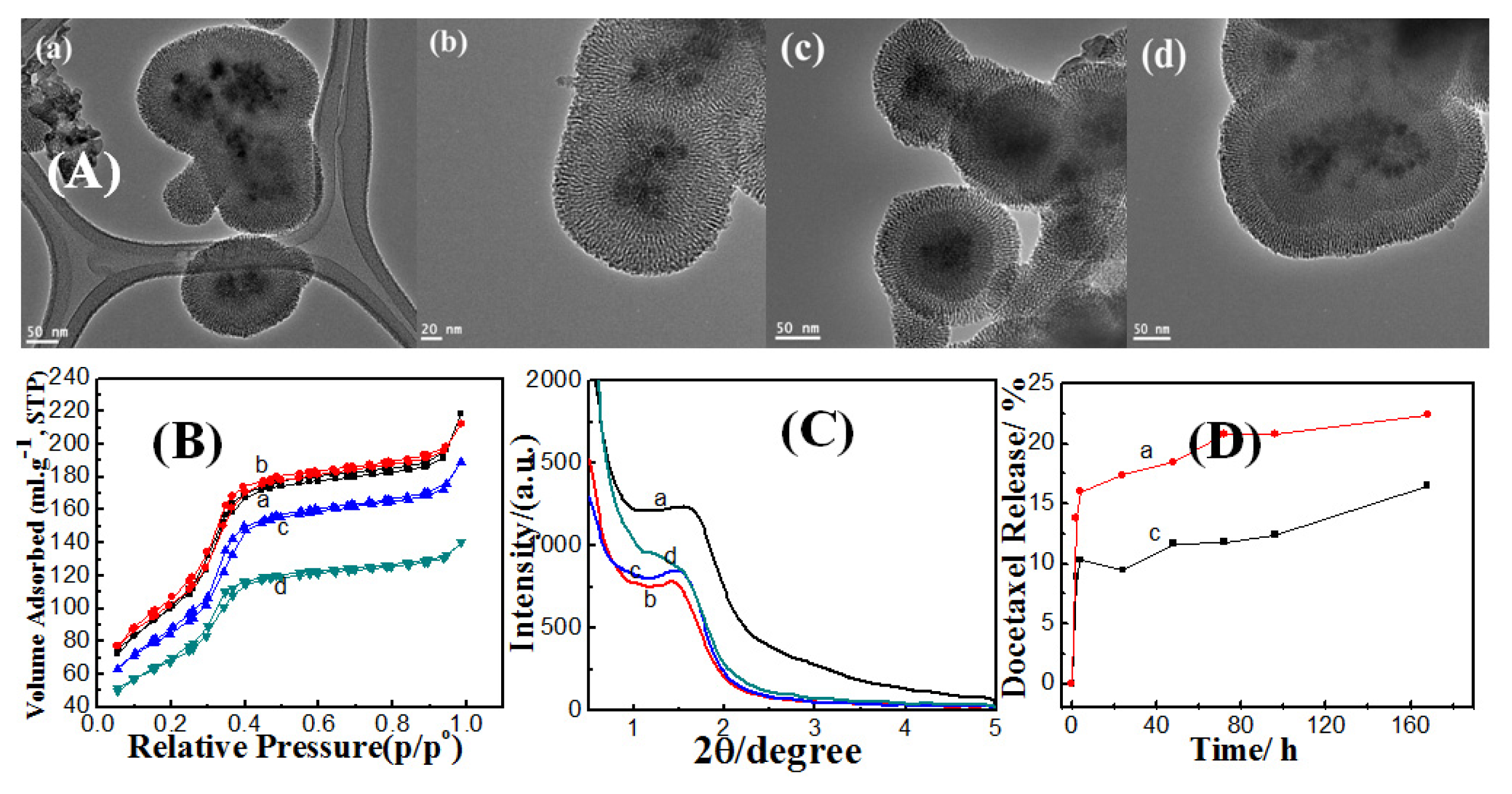
2.3.1.1. Effect of Ethanol
2.3.1.2. Effect of Ammonia
2.3.2. Mesoporous Shell Formation Parameters
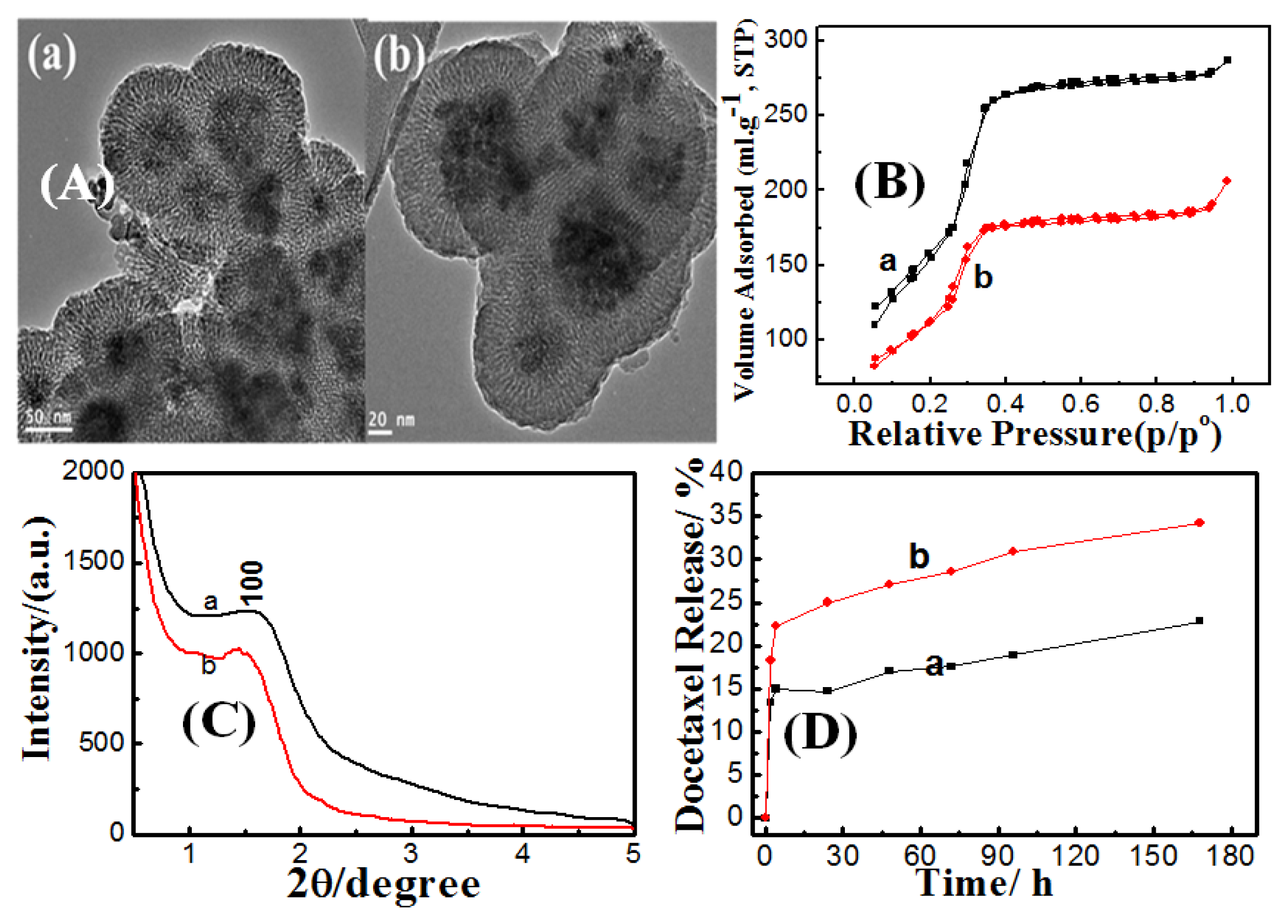
Effect of Surfactant
3. Experimental Section
3.1. Synthesis
3.1.1. Fe3O4 Synthesis
3.1.2. Dense Silica Coating
3.1.3. Construction of Mesoporous Silica Shell by Anionic Surfactant
3.2. Docetaxel Loading and Release Study
3.3. Characterization
4. Conclusions
Supplementary Information
ijms-14-11496-s001.pdfAcknowledgments
Conflict of Interest
References
- Gopin, A.; Pessah, N.; Shamis, M.; Rader, C.; Shabat, D. A chemical adaptor system designed to link a tumor-targeting device with a prodrug and an enzymatic trigge. Angew. Chem. Int. Ed. Engl 2003, 42, 327–332. [Google Scholar]
- Kataoka, K.; Hrada, A.; Nagasaki, Y. Block copolymer micelles for drug delivery: Design, characterization and biological significance. Adv. Drug Deliv. Rev 2001, 47, 113–131. [Google Scholar]
- Oussoren, C.; Storm, G. liposomes to target the lymphatics by subcutaneous administration. Adv. Drug Deliv. Rev. 2001, 50, 143–156. [Google Scholar]
- Couvreur, P.; Dubernet, C.; Puisieux, F. Controlled drug delivery with nanoparticles: Current possibilities and future trends. Eur. J. Pharm. Biopharm. 1995, 41, 2–13. [Google Scholar]
- Zhu, Y.; Shi, J.; Shen, W.; Dong, X.; Feng, J.; Ruan, M.; Li, Y. Uniform magnetic hollow spheres with a magnetic core/mesoporous silica shell structure. Angew. Chem. Int. Ed 2005, 4, 5083–5087. [Google Scholar]
- Mal, N.K.; Fujiwara, M.; Tanaka, Y. Photocontrolled reversible release of guest molecules from coumarin-modified mesoporous silica. Nature 2003, 421, 350–353. [Google Scholar]
- Li, X.; Zhang, L.; Dong, X.; Liang, J.; Shi, J. Preparation of mesoporous calcium doped silica spheres with narrow size dispersion and their drug loading and degradation behavior. Microporous Mesoporous Mater 2007, 102, 151–158. [Google Scholar]
- Gross, A.F.; Diehl, M.R.; Beverly, K.C.; Richman, E.K.; Tolbert, S.H. Controlling magnetic coupling between cobalt nanoparticles through nanoscale confinement in hexagonal mesoporous silica. J. Phys. Chem. B 2003, 107, 5475–5482. [Google Scholar]
- Lu, A.H.; Li, W.C.; Kiefer, A.; Schmidt, W.; Bill, E.; Fink, G.; Schüth, F. Fabrication of magnetically separable mesostructured silica with an open pore system. J. Am. Chem. Soc 2004, 126, 8616–8617. [Google Scholar]
- Wu, P.; Zhu, J.; Xu, Z. Template-assisted synthesis of mesoporous magnetic nanocomposite particles. Adv. Funct. Mater 2004, 14, 345–351. [Google Scholar]
- Zhao, W.; Gu, J.; Zhang, L.; Chen, H.; Shi, J. Fabrication of uniform magnetic nanocomposite spheres with a magnetic core/mesoporous silica shell structure. J. Am. Chem. Soc 2005, 127, 8916–8917. [Google Scholar]
- Buchel, G.; Grün, M.; Unger, K.K.; Matsumoto, A.; Tatsumi, K. A novel pathway for synthesis of submicrometer-size solid core/mesoporous shell silica spheres. Adv. Mater 1998, 10, 1036–1038. [Google Scholar]
- Zhao, W.; Chen, H.; Li, Y.; Li, L.; Lang, M.; Shi, J. Uniform rattle-type hollow magnetic mesoporous spheres as drug delivery carriers and their sustained-release property. Adv. Funct. Mater 2008, 18, 2780–2788. [Google Scholar]
- Yoon, S.B.; Kim, J.-Y.; Kim, J.H.; Park, Y.J.; Yoon, K.R.; Park, S.-K.; Yu, J.-S. Synthesis of monodisperse spherical silica particles with solid core and mesoporous shell: Mesopore channels perpendicular to the surface. J. Mater. Chem 2007, 17, 1758–1761. [Google Scholar]
- Deng, Y.; Qi, D.; Deng, C.; Zhang, X.; Zhao, D. Superparamagnetic high-magnetization microspheres with an Fe3O4@SiO2 core and perpendicularly aligned mesoporous SiO2 shell for removal of microcystins. J. Am. Chem. Soc 2008, 130, 28–29. [Google Scholar]
- Yang, P.; Quan, Z.; Hou, Z.; Li, C.; Kang, X.; Cheng, Z.; Lin, J. A magnetic, luminescent and mesoporous core-shell structured composite material as drug carrier. Biomaterials 2009, 30, 4786–4795. [Google Scholar]
- Che, S.; Garcia-Bennett, A.E.; Yokoi, T.; Sakamoto, K.; Kunieda, H.; Terasaki, O.; Tatsumi, T. A novel anionic surfactant templating route for synthesizing mesoporous silica with unique structure. Nat. Mater 2003, 2, 801–805. [Google Scholar]
- Garcia-Bennett, A.E.; Miyasaka, K.; Terasaki, O.; Che, S. Structural solution of mesocaged material AMS-8. Chem. Mater 2004, 16, 3597–3605. [Google Scholar]
- Gao, C.; Sakamoto, Y.; Sakamoto, K.; Terasaki, O.; Che, S. Synthesis and characterization of mesoporous silica AMS-10 with a novel bicontinuous cubic Pn3m symmetry. Angew. Chem. Int. Ed 2006, 45, 4295–4298. [Google Scholar]
- Che, S.; Liu, Z.; Ohsuna, T.; Sakamoto, K.; Terasaki, O.; Tatsumi, T. Synthesis and characterization of chiral mesoporous silica. Nature 2004, 429, 281–284. [Google Scholar]
- Jin, H.; Liu, Z.; Ohsuna, T.; Terasaki, O.; Inoue, Y.; Sakamoto, K.; Nakanishi, T.; Ariga, K.; Che, S. Control of morphology and helicity of chiral mesoporous silica. Adv. Mater 2006, 18, 593–596. [Google Scholar]
- Yu, Y.; Qiu, H.; Wu, X.; Li, H.; Li, Y.; Sakamoto, T.; Inoue, Y.; Sakamoto, K.; Terasaki, O.; Che, S. Synthesis and characterization of silica nanotubes with radially oriented mesopores. Adv. Funct. Mater 2008, 18, 541–550. [Google Scholar]
- Yokoi, T.; Yoshitake, H.; Yamada, T.; Kubota, Y.; Tatsumi, T. Amino-functionalized mesoporous silica synthesized by an anionic surfactant templating route. J. Mater. Chem 2006, 16, 1125–1135. [Google Scholar]
- Kim, K.-S.; Kim, J.-K.; Kim, W.-S. Influence of reaction conditions on sol-precipitation process producing silicon oxide particles. Ceram. Int 2002, 28, 187–194. [Google Scholar]
- Deng, Y.-H.; Wang, C.-C.; Hu, J.-H.; Yang, W.-L.; Fu, S.-K. Investigation of formation of silica-coated magnetite nanoparticles via sol-gel approach. Colloids Surf. A Physicochem. Eng. Asp 2005, 262, 87–93. [Google Scholar]
- Mine, E.; Konno, M. Secondary particle generation at low monomer concentrations in seeded growth reaction of tetraethyl orthosilicate. J. Chem. Eng. Jpn 2001, 34, 545–548. [Google Scholar]
- El-Toni, A.M.; Khan, A.; Ibrahim, M.A.; Labis, J.P.; Badr, G.; Al-Hoshan, M.; Yin, S.; Sato, T. Synthesis of double mesoporous core-shell silica spheres with tunable core porosity and their drug release and cancer cell apoptosis properties. J. Colloid Interface Sci 2012, 378, 83–92. [Google Scholar]
- Baeza, A.; Guisasola, E.; Ruiz-Hernández, E.; Vallet-Regí, M. Magnetically triggered multidrug release by hybrid mesoporous silica nanoparticles. Chem. Mater 2012, 24, 517–524. [Google Scholar]
- Wang, J.; Li, F.; Zhou, H.; Sun, P.; Ding, D.; Chen, T. Silica hollow spheres with ordered and radially oriented amino-functionalized mesochannels. Chem. Mater 2009, 21, 612–620. [Google Scholar]
- Huang, S.; Fan, Y.; Cheng, Z.; Kong, D.; Yang, P.; Quan, Z.; Zhang, C.; Lin, J. Magnetic mesoporous silica spheres for drug targeting and controlled release. J. Phys. Chem. C 2009, 113, 1775–1784. [Google Scholar]
- Lecloux, A.J.; Bronckart, J.; Noville, F.; Dodet, C.; Marchot, P.; Pirard, J.P. Study of the texture of monodisperse silica sphere samples in the nano size range. Colloids Surf. A Physicochem. Eng. Asp 1986, 19, 359–374. [Google Scholar]
- Park, S.-J.; Kim, Y.-J.; Park, S.-J. Size-dependent shape evolution of silica nanoparticles into hollow structures. Langmuir 2008, 24, 12134–12137. [Google Scholar]
- Andersson, J.; Rosenholm, J.; Lindén, M. Topics in Multifunctional Biomaterials and Devices e-book. Ashammakhi, N., Ed.; 1. Available online: http://www.oulu.fi/spareparts/ebook_topics_multifunctional/index.html (accessed on 22 May 2013).
- Jin, H.; Qiu, H.; Gao, C.; Che, S. Synthetic design towards targeted chiral anionic surfactant templated chiral mesoporous silica. Microporous Mesoporous Mater 2008, 116, 171–179. [Google Scholar]


| Sample code | NH4OH-1st a g/mL | EtOH g/mL | Surfactant mmol | HCl g/mL | Shell b 1st 2nd/nm | BET cm2/g | Vp c Cc/g | Rp d /nm | Drug EE e /% | |
|---|---|---|---|---|---|---|---|---|---|---|
| MCMSS1 | 0.03 | 0.87 | 5 | 0.16 | 10 | 43 | 411.09 | 0.373 | 3.6 | 27.54 |
| MCMSS2 | 0.03 | 0.84 | 5 | 0.16 | 21 | 36 | 352.9 | 0.311 | 3.8 | - |
| MCMSS3 | 0.03 | 0.8 | 5 | 0.16 | 25 | 27 | 319.81 | 0.292 | 3.6 | 33.81 |
| MCMSS4 | 0.06 | 0.8 | 5 | 0.16 | 37 | 26 | 259.62 | 0.216 | 3.6 | - |
| MCMSS5 | 0.016 | 0.8 | 5 | 0.16 | 14 | 44 | 389.32 | 0.328 | 3.8 | - |
| MCMSS6 | 0.008 | 0.8 | 1 | 0.16 | 8 | 37 | 621.79 | 0.443 | 3.6 | 31.03 |
| MCMSS7 | 0.008 | 0.8 | 5 | 0.16 | 8 | 50 | 386.94 | 0.338 | 3.8 | 25.67 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Toni, A.M.; Ibrahim, M.A.; Labis, J.P.; Khan, A.; Alhoshan, M. Optimization of Synthesis Parameters for Mesoporous Shell Formation on Magnetic Nanocores and Their Application as Nanocarriers for Docetaxel Cancer Drug. Int. J. Mol. Sci. 2013, 14, 11496-11509. https://doi.org/10.3390/ijms140611496
El-Toni AM, Ibrahim MA, Labis JP, Khan A, Alhoshan M. Optimization of Synthesis Parameters for Mesoporous Shell Formation on Magnetic Nanocores and Their Application as Nanocarriers for Docetaxel Cancer Drug. International Journal of Molecular Sciences. 2013; 14(6):11496-11509. https://doi.org/10.3390/ijms140611496
Chicago/Turabian StyleEl-Toni, Ahmed Mohamed, Mohamed Abbas Ibrahim, Joselito Puzon Labis, Aslam Khan, and Mansour Alhoshan. 2013. "Optimization of Synthesis Parameters for Mesoporous Shell Formation on Magnetic Nanocores and Their Application as Nanocarriers for Docetaxel Cancer Drug" International Journal of Molecular Sciences 14, no. 6: 11496-11509. https://doi.org/10.3390/ijms140611496






