Ubiquitin-Conjugating Enzyme 9 Promotes Epithelial Ovarian Cancer Cell Proliferation in Vitro
Abstract
:1. Introduction
2. Results and Discussion
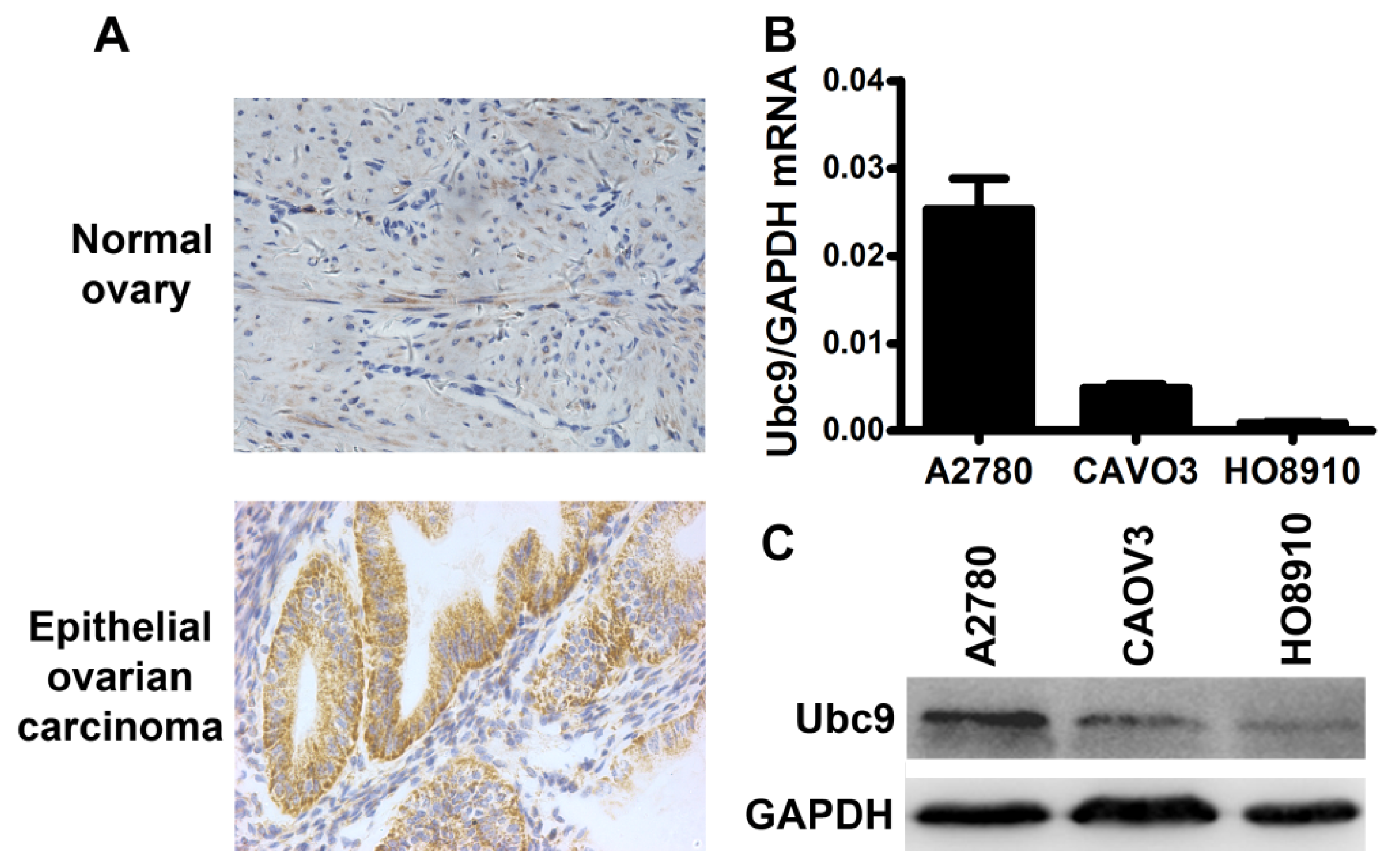
2.1. Ubc9 mRNA and Protein Expression in Ovarian Cancer Cells
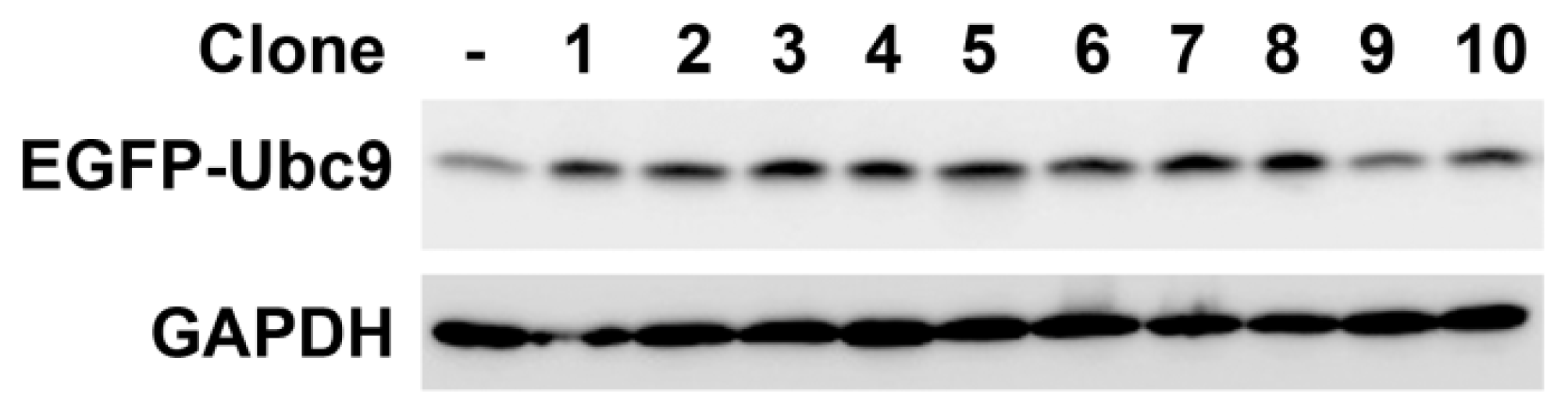
2.2. Construction of Ubc9 Recombinant Plasmid and Selection of Stable Transfected Cell Clones
2.3. Specific Silencing of the Ubc9 Gene
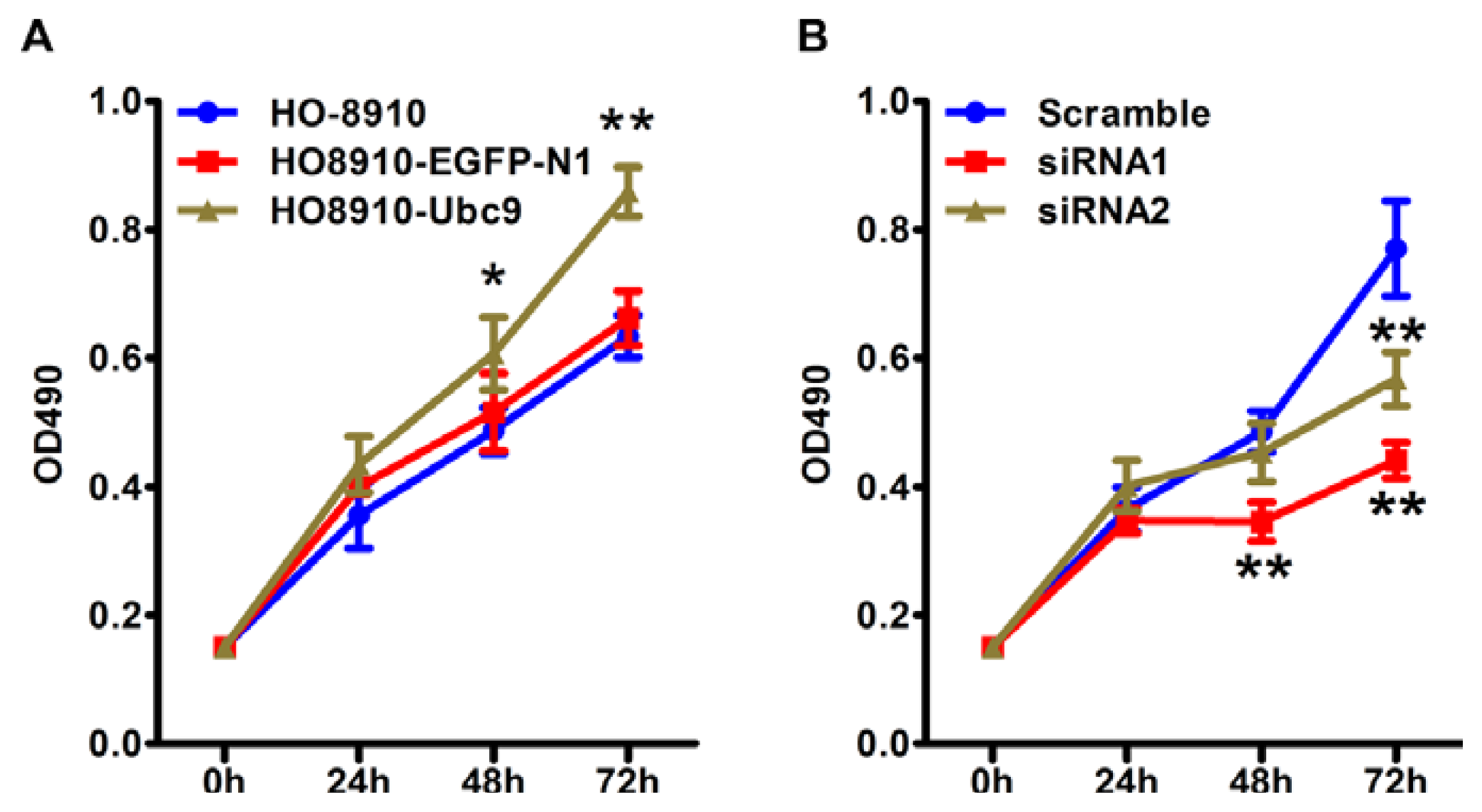
2.4. Ubc9 Overexpression Promotes Cell Proliferation, whereas siRNA Targeting Ubc9 Inhibited Epithelial Ovarian Cancer Cell Proliferation
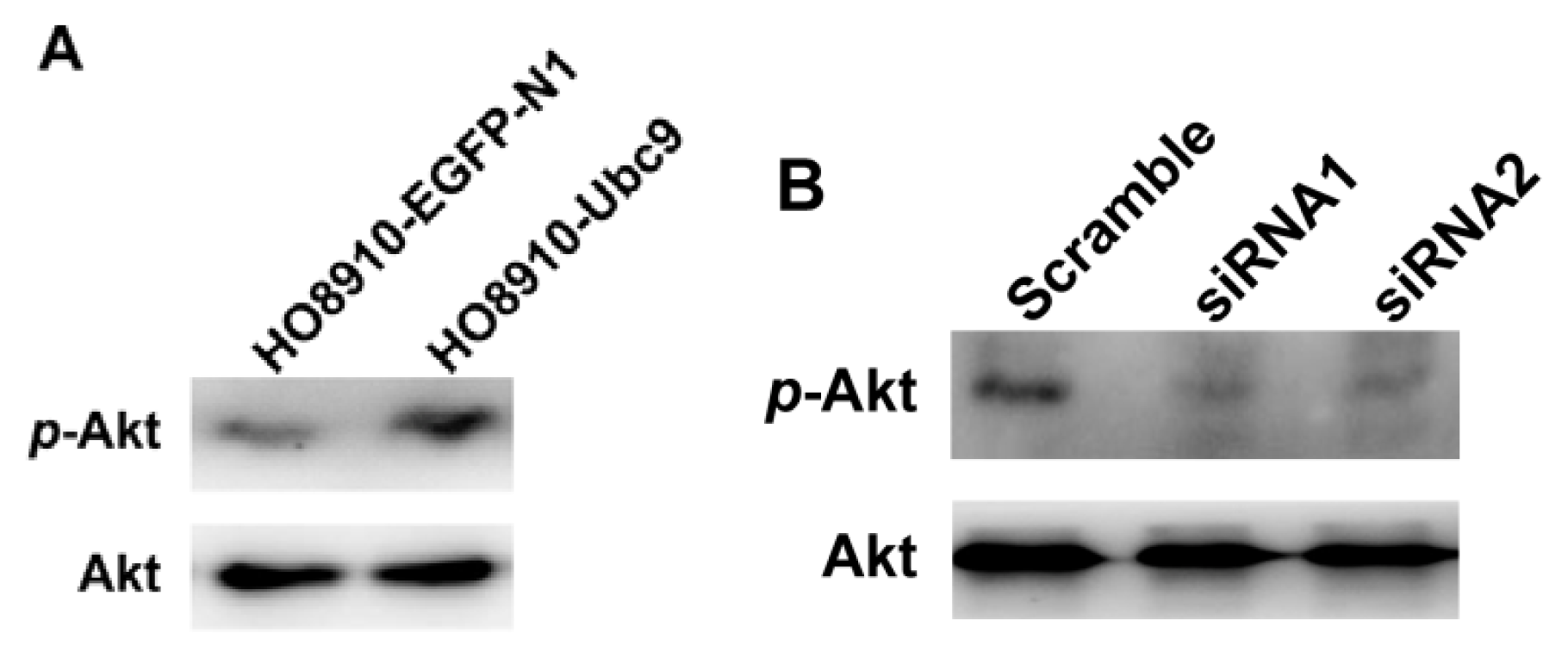
2.5. PI3K/Akt Signaling is Required for Ubc9 in Accelerating HO8910 Cell Proliferation
2.6. Discussion
3. Experimental Section
3.1. Reagents
3.2. Cell Culture
3.3. Immunohistochemical Analysis
3.4. Quantitative Real-Time PCR
3.5. Western Blot
3.6. Construction of Plasmid and Generation of Stably Transfected Cell Lines
3.7. siRNA Treatment
3.8. Cell Proliferation Assay
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Johnson, E.S.; Blobel, G. Ubc9p is the conjugating enzyme for the ubiquitin-like protein Smt3p. J. Biol. Chem 1997, 272, 26799–26802. [Google Scholar]
- Tashiro, K.; Pando, M.P.; Kanegae, Y.; Wamsley, P.M.; Inoue, S.; Verma, I. Direct involvement of the ubiquitin-conjugating enzyme Ubc9/Hus5 in the degradation of InBa. Proc. Natl. Acad. Sci. USA 1997, 94, 7862–7867. [Google Scholar]
- Desterro, J.M.; Thomson, J.; Hay, R.T. Ubch9 conjugates SUMO but not ubiquitin. FEBS Lett 1997, 417, 297–300. [Google Scholar]
- Geiss-Friedlander, R.; Melchior, F. Concepts in sumoylation: A decade on. Nat. Rev. Mol. Cell Biol 2007, 8, 947–956. [Google Scholar]
- Kurtzman, A.L.; Schechter, N. Ubc9 interacts with a nuclear localization signal and mediates nuclear localizationof the paired-like homeobox protein Vsx-1 independent of SUMO-1modification. Proc. Natl. Acad. Sci. USA 2001, 98, 5602–5607. [Google Scholar]
- Kaul, S.; Blackford, J.A., Jr; Cho, S.; Simons, S.S., Jr. Ubc9 is a novel modulator of the induction properties of glucocorticoid receptors. J. Biol. Chem. 2002, 277, 12541–12549. [Google Scholar]
- Zhu, S.; Sachdeva, M.; Wu, F.; Lu, Z.; Mo, Y. Ubc9 promotes breast cell invasion and metastasis in a sumoylation-independent manner. Oncogene 2010, 29, 1763–1772. [Google Scholar]
- Wu, F.; Zhu, S.; Ding, Y.; Beck, W.T.; Mo, Y. MicroRNA-mediated regulation of Ubc9 expression in cancer cells. Clin. Cancer Res 2009, 15, 1550–1557. [Google Scholar]
- Moschos, S.J.; Smith, A.P.; Mandic, M.; Athanassiou, C.; Watson-Hurst, K.; Jukic, D.M.; Edington, H.D.; Kirkwood, J.M.; Becker, D. SAGE and antibody array analysis of melanoma-infiltrated lymph nodes: identification of Ubc9 as an important molecule in advanced-stage melanoma. Oncogene 2007, 26, 4216–4225. [Google Scholar]
- Moschos, S.J.; Jukic, D.M.; Athanassiou, C.; Bhargava, R.; Dacic, S.; Wang, X.; Kuan, S.F.; Fayewicz, S.L.; Galambos, C.; Acquafondata, M.; et al. Expression analysis of Ubc9, the single small ubiquitin-like modifier (SUMO) E2 conjugating enzyme,in normal and malignant tissues. Hum. Pathol 2010, 41, 1286–1298. [Google Scholar]
- Chen, S.F.; Gong, C.; Luo, M.; Yao, H.R.; Zeng, Y.J.; Su, F.X. Ubc9 expression predicts chemoresistance in breast cancer. Chin. J. Cancer 2011, 30, 638–644. [Google Scholar]
- Mo, Y.; Yu, Y.; Theodosiou, E.; Ee, P.L.R.; Beck, W.T. A role for Ubc9 in tumorigenesis. Oncogene 2005, 24, 2677–2683. [Google Scholar]
- Melchior, F. SUMO-nonclassical ubiquitin. Annu. Rev. Cell Dev. Biol 2000, 16, 591–626. [Google Scholar]
- Muller, S.; Hoege, C.; Pyrowolakis, G.; Jentsch, S. SUMO, ubiquitin’s mysterious cousin. Nat. Rev. Mol. Cell Biol 2001, 2, 202–210. [Google Scholar]
- Mo, Y.Y.; Moschos, S.J. Targeting Ubc9 for cancer therapy. Expert Opin. Ther. Targets 2005, 9, 1203–1216. [Google Scholar]
- Woenckhaus, J.; Steger, K.; Sturm, K.; Münstedt, K.; Franke, F.E.; Fenic, I. Prognostic value of PIK3CA and phosphorylated AKT expression in ovarian cancer. Virchows. Archiv 2007, 450, 387–395. [Google Scholar]
- Qin, Y.; Xu, J.; Aysola, K.; Oprea, G.; Reddy, A.; Matthews, R.; Okoli, J.; Cantor, A.; Grizzle, W.E.; Partridge, E.E.; et al. BRCA1 proteins regulate growth of ovarian cancer cells by tethering Ubc9. Am. J. Cancer Res 2012, 2, 540–548. [Google Scholar]
- Do, T.V.; Kubba, L.A.; Antenos, M.; Rademaker, A.W.; Sturgis, C.D.; Woodruff, T.K. The role of activin A and Akt/GSK signaling in ovarian tumor biology. Endocrinology 2008, 149, 3809–3816. [Google Scholar]
- Shtilbans, V.; Wu, M.; Burstein, D.E. Current overview of the role of Akt in cancer studies via applied immunohistochemistry. Annu. Diagn. Pathol 2008, 12, 153–160. [Google Scholar]


© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dong, M.; Pang, X.; Xu, Y.; Wen, F.; Zhang, Y. Ubiquitin-Conjugating Enzyme 9 Promotes Epithelial Ovarian Cancer Cell Proliferation in Vitro. Int. J. Mol. Sci. 2013, 14, 11061-11071. https://doi.org/10.3390/ijms140611061
Dong M, Pang X, Xu Y, Wen F, Zhang Y. Ubiquitin-Conjugating Enzyme 9 Promotes Epithelial Ovarian Cancer Cell Proliferation in Vitro. International Journal of Molecular Sciences. 2013; 14(6):11061-11071. https://doi.org/10.3390/ijms140611061
Chicago/Turabian StyleDong, Mei, Xiaoyan Pang, Yang Xu, Fang Wen, and Yi Zhang. 2013. "Ubiquitin-Conjugating Enzyme 9 Promotes Epithelial Ovarian Cancer Cell Proliferation in Vitro" International Journal of Molecular Sciences 14, no. 6: 11061-11071. https://doi.org/10.3390/ijms140611061



