Abstract
The extensive application of nanomaterials in industry, medicine and consumer products has raised concerns about their potential toxicity. The female population is particularly vulnerable and deserves special attention because toxicity in this group may impact both female reproductivity and fetal development. Mouse and zebrafish models each have their own unique features and studies using these models to examine the potential toxicity of various nanoparticles are compared and summarized in this review. Several nanoparticles exhibit detrimental effects on female reproductivity as well as fetal development, and these adverse effects are related to nanoparticle composition, surface modification, dose, exposure route and animal species. Limited studies on the mechanisms of nanotoxicity are also documented and reviewed herein.
1. Introduction
Nanoparticles have unique thermal, mechanical, magnetic and optical properties that allow for their widespread application in biomedicine and many industrial sectors [1–3]. More than 1310 marketed consumer products are based on nanomaterials [4], and this number is rapidly increasing. Nanoparticles cause pulmonary injury [5], hepatotoxicity [6,7], immunotoxicity [8], neurotoxicity [9], renal toxicity [10] and reversible testis damage [11] in animals. Recently, severe pulmonary fibrosis caused by polymer nanoparticles in seven young female workers provided new evidence for nanotoxicity in humans [12]. Consequently, the increasing public and occupational exposure to nanomaterials is a call for concern with respect to nanotoxicity.
Normal female reproduction and fetal development are essential for the perpetuation of the species. However, the female reproductive system is considerably more fragile than other systems, as described below. First, compared with the reproductive male gametes, female gametes are rather limited. During a woman’s lifetime, only about 400 follicles sequentially mature and ovulate [13]. Second, the female reproductive organs, such as the ovary and uterus, exhibit periodic growth and regression, which is strictly regulated by hormones. Its dynamic activity and rigorous hormonal control make this system more sensitive to foreign bodies and physiological stress compared to other physiological processes [14,15]. Third, the disturbance of female reproduction inevitably leads to abnormal fetal development. Many environmental chemicals have already demonstrated detrimental effects on the female reproductive system and embryonic development [15,16].
The emergence of nanoparticles has added a new threat to the vulnerable female population. The toxicity of nanoparticles to female reproductive and developmental health has been studied in various models [17–19]. In this review, we focus on nanotoxicity studies that use mouse and zebrafish models. The genomic similarity between mice and humans is the primary reason for the wide use of mice in the life sciences; the mouse as a mammalian model provides analogous experimental conditions and comparable results to humans, albeit with certain limitations. For example, animals are expensive, the study cycle is rather long, the study throughput is low and several animal protection organizations are active around the world. Furthermore, the investigation of early developmental effects is challenging in mice because the process occurs in utero and is thus not easily detectable. In contrast, studies in zebrafish are fast, less expensive and can be modelled using a high-throughput format. Embryonic development in zebrafish can be studied in vitro, which allows for testing at all stages [20]. Therefore, the zebrafish has become the model of choice for molecular mechanism studies in embryonic development and one of the most valued models in developmental biology. In the following review, we summarise the main findings of these studies.
2. Effects of Nanotoxicity on Female Reproductivity and Development in Murine Models
The mouse is a commonly used animal model for toxicological evaluation. Its genomic similarity to humans as well as its short generation time and large litter size in mammals are the key elements for its extensive use. The availability of multiple species and knockout mice satisfies the needs of many specific studies.
2.1. Toxicity to the Female Reproductive System
The mammalian female reproductive system is composed of the hypothalamic-pituitary-ovarian axis and reproductive organs, including the oviducts, uterus, vagina and external genitalia. The normal operation of the reproductive system depends on the precise positive and negative feedback between various components of the axis (Figure 1) [21]. The interference of xenobiotics with the female reproductive system may impair normal gonadal processes, such as oogenesis, ovulation, hormone production by granulosa cells and the structure or function of the accessory reproductive structures [22].
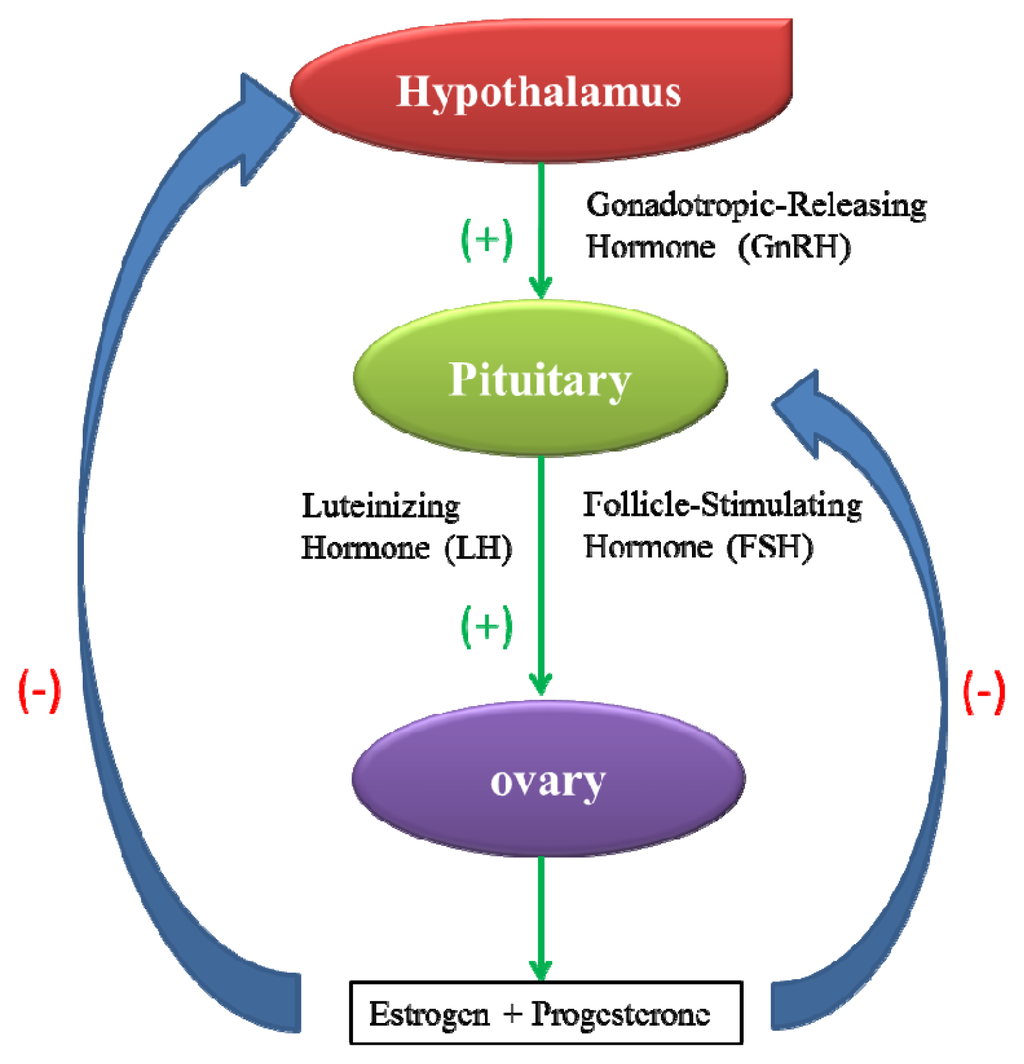
Figure 1.
The hypothalamic-pituitary-ovarian axis of the female reproductive system. (+) represents positive feedback, and (−) represents negative feedback.
In an experiment conducted in non-pregnant female mice, long-term exposure to TiO2 nanoparticles (5–6 nm, intragastric administration) at a concentration of 10 mg/kg was found to cause ovarian dysfunction and alterations in functional gene expression levels. The hormone-related gene Cyp17a1 was up-regulated, indicating the increased biosynthesis of estradiol. Additionally, three genes regulating apoptosis were down-regulated, while eighteen genes such as bmf were up-regulated. Changes in the expression of genes regulating immune and inflammatory responses, oxidative stress, ion transport, cell proliferation, transcription and oxidoreductase activity of the ovary were also observed. TiO2 nanoparticles were detected in the ovarian cells of these mice, and the resultant cellular damage led to an imbalance in sex hormones and decreased fertility [23]. In another study [24], the daily inhalation of CdO nanoparticles (230 μg/m3) increased the uterine weight and altered the placental weight of pregnant CD-1 mice. Furthermore, reduced levels of 17β-estradiol and altered expression levels of estrogen receptor α and β (ERα and ERβ) in the uterus eventually led to decreased implantation. Cd ions that released from the CdO nanoparticles may act as an endocrine disruptor to prevent implantation and perturb the implanted blastocysts. However, the mechanism of action has still not been verified. The intravenous injection of SiO2 and TiO2 nanoparticles at a dose of 0.8 mg/mouse in pregnant mice was shown to result in a decreased uterine weight and an increased fetal reabsorption rate [25]. These studies demonstrate that nanoparticles may adversely impact the female reproductive system and fertility, as has been shown for other toxic chemicals [26].
2.2. Transplacental Ability of Nanoparticles
The placenta is a hormonally regulated organ that is responsible for maternal-fetal exchange and is essential for the maintenance of gestation and embryonic growth (Figure 2) [25]. However, the placenta is not an effective barrier, as environmental pollutants and drugs are known causes of birth defects [27,28]. Many nanoparticles, such as Au nanoparticles [29], TiO2 nanoparticles [30], SiO2 nanoparticles [25,31], quantum dots (QDs) [32] and carbon nanoparticles [33], can also penetrate the placental barrier.
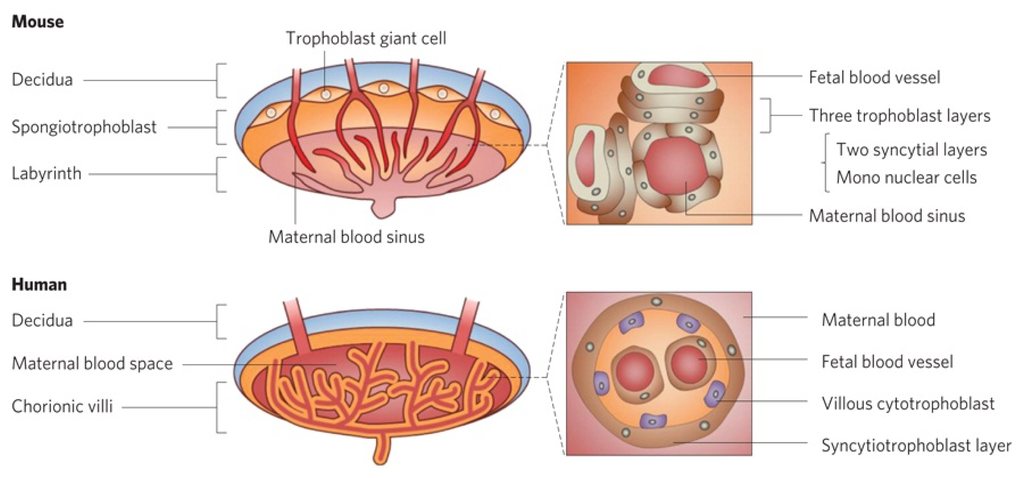
Figure 2.
Pathological examination of the mouse and human placenta (reproduced with permission from [25]©2011, Nature Publishing Group).
Smaller particles seem to have a stronger transplacental ability than larger particles with an identical chemical composition. When SiO2 nanoparticles (70, 300 and 3000 nm) were intravenously injected into pregnant BALB/c mice at a concentration of 0.8 mg/mouse, nanoparticles of all sizes were detected in the liver, while only nanoparticles with a diameter of 70 nm were detected in the placental trophoblasts, the fetal liver and the fetal brain [25]. Similarly, when Au nanoparticles of two different sizes were injected into pregnant rats, the smaller nanoparticles (1.4 nm) were detected in the placenta rather than the larger nanoparticles (18 nm) [29].
In addition to their size, surface modification also regulates the biodistribution of nanoparticles. The transplacental ability of CdTe/CdS QDs was reduced following modification with polyethylene glycol (PEG) or capping with an inorganic silica shell [32]. Similarly, the phenomenon that surface modification regulates the uptake of nanoparticles was also observed when tested with mouse embryos in vitro [34]. In this study, amine- and carboxyl-modified polystyrene beads of different sizes (200 nm for the former, and 20, 100, 500 nm for the latter) were injected into the extra-embryonic tissue of cultured embryos in vitro. The results showed that carboxylic polystyrene beads with a diameter greater than 100 nm were located solely in the extra-embryonic tissue, while 200 nm amine-modified beads crossed into the embryos.
Therefore, the transplacental ability of nanoparticles is dependent on size and surface modification [33–36]. A human placental perfusion model study confirmed that nanoparticles have the ability to cross the placenta by means of endocytosis [37]. Placental damage caused by nanoparticles may potentially lead to the deformity or developmental retardation of the fetus.
2.3. Effects of Nanoparticles on Fetal Developmental
Although nanoparticles may cause damage to embryos as a result of their transplacental ability [38], they may also affect the offspring through altered signaling pathways. Maternal exposure to carbon black nanoparticles (100 μg/mouse) by intranasal instillation of ICR mice was found to induce the overexpression of renal type VIII collagen in the offspring [39]. Inhalation of TiO2 nanoparticles (1 h/day, 42 mg/m3) by pregnant C57BL/6 mice from gestational day (GD) 8 to18 resulted in the abnormal expression of genes of the retinoic acid signaling pathway in the livers of newborn female mice [40]. Nanoparticles may also cause altered organogenesis and morphology as well as defects in the reproductive and nervous systems of the offspring (Table 1). These effects are discussed in the table below.

Table 1.
Fetal toxicity of nanoparticles in murine models.
2.3.1. Toxicity on Fetal Organogenesis and Morphology
Exposure to nanoparticles during the gestational period affects fetal organogenesis and morphology [17]. With intravenous administration, both pristine and oxidized single-walled carbon nanotubes (SWCNTs) with concentrations varying from 10 ng to 30 μg/mouse was observed to induce morphological abnormalities in the fetuses of pregnant CD-1 mice. The fetuses exhibited deformities in the abdominal wall or head, retarded development of the limbs and snout, swollen abdomens with abnormal torsion of the trunks (Figure 3). Furthermore, oxidized SWCNTs caused more abnormalities in the fetuses than did pristine SWCNTs [52]. The intragastric administration of hydroxyl-modified SWCNTs (10 mg/kg) to pregnant CD-1 mice was demonstrated to cause increased skeletal defects, such as forked cervical vertebrae, reduced ossification of sternebra and phalanges and morphological abnormalities [41]. However, toxic effects were not observed when multi-walled carbon nanotubes (MWCNTs) were administered by gavage to pregnant Sprague-Dawley rats. The offspring of the tested group showed no differences in morphological, visceral or skeletal malformations compared with the control group. The no-observed-adverse-effect level for embryonic-fetal development is considered to be 1000 mg/kg/day [53].
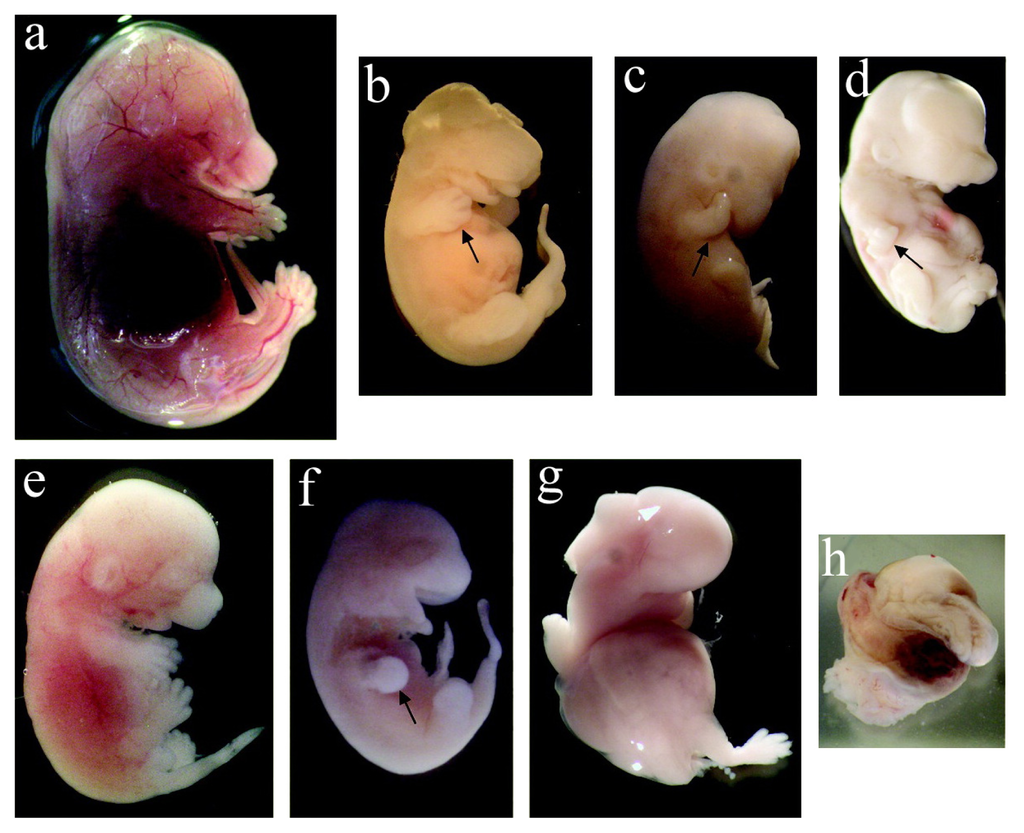
Figure 3.
Exposure of pregnant mice to SWCNTs causes malformed fetuses. (a) Normal fetus; (b–h) Malformed fetuses from SWCNT groups (reproduced with permission from [52]©2011, American Chemical Society).
To mimic the exposure of females to nanoparticles during their entire pregnancy, a study was conducted using platinum nanoparticles from pre-gestation to post-delivery. Platinum nanoparticles (0.25, 0.5 and 1 mg/kg) were orally administered to ICR mice from 14 days before mating to 4 days after delivery. Decreased growth and increased mortality of pups during the lactation period were observed, although no deformity was observed in any of the pups [54].
2.3.2. Toxicity to the Fetal Reproductive Function
Diesel exhaust (DE) is generated from the combustion of diesel fuel and is comprised of fine and ultrafine particles. DE has been shown to disrupt the reproductive development of offspring [55,56]. Pre- and postnatal exposure of ICR mice to DE at a dose of 0.17 mg/m3caused a reduced daily sperm production (DSP) and deformed the sertoli and spermatozoa cells in the offspring [57].
Because DE contains various compounds and particles, its reactive components were evaluated. Nanoparticle-rich DE (148.86 μg/m3, 5 h/day) and filtered DE (3.10 μg/m3, 5 h/day) were administered to pregnant F344 rats from GD 1 to GD 19. In both groups, the organ indices of the seminal vesicle and prostate and the concentrations of various hormones, such as testosterone, progesterone, corticosterone, LH and FSH, in the serum of the male offspring decreased. Testicular histology and real-time reverse transcription polymerase chain reaction analysis showed a loss of germ cells in the seminiferous tubules and altered expression of steroidogenic acute regulatory protein, 17β-hydroxysteroid dehydrogenase and follicle-stimulating hormone receptor mRNA [45]. The alterations observed in both groups were similar, indicating that the gaseous phase of the exhaust, rather than the particles, played a major role in disrupting the reproductive function. The hormone-like activity of the compounds adsorbed to the particles disturbed the hormone levels and thereby restricted growth [58]. Thus, the particles only served as carriers of the hazardous compounds.
The exposure of pregnant C57BL/6BomTac mice to DE (20 mg/m3, 1 h/day) from GD 7 to GD 19 caused a reduced DSP in the adult offspring. However, the testis weight, concentrations of testosterone and estradiol, gene expression of hormone-related receptors were not altered compared with the controls. These results indicate that in utero exposure to DE may not affect the endocrine activity in adult offspring [44], although another report suggested an endocrine-like activity for DE [45].
2.3.3. Effects on Fetal Neurodevelopment
Traffic-related air pollution may cause adverse effects on neurodevelopment in children [59]. Prenatal exposure of pregnant mice to nanoparticles caused neurological disorders in their offspring [50,60]. In an inhalation study, prenatal exposure to DE (0.3, 1 and 3 mg/m3) resulted in various types of damage, including caspase-3-positive cells in the cerebral cortex and hippocampus and crescent-shaped spaces in some cells. Furthermore, the granular epithelial cells and scavenger cells that constitute the blood brain barrier (BBB) underwent apoptosis [49]. Maternal exposure to TiO2 (0.1 mg/mouse) by subcutaneous injection also resulted in the apoptosis of endothelial cells, capillary stenosis and degenerative changes in the neighboring parenchyma [30,60]. The nanoparticle-induced reduction of dopamine (DA) turnover in the nucleus accumbens and striatum induced a decrease in spontaneous motor activity, thereby emphasizing the adverse effects of TiO2 nanoparticles on the central dopaminergic system [48]. Analysis of the gene expression in the brain of the offspring indicated that the alterations are related to inflammation, oxidative stress and neurotransmitters [47].
Several methods have been used to evaluate offspring behavior after the treatment of pregnant mice with nanoparticles. The open field test is used to test locomotor activity, the Morris water maze is used to test learning and memory and the acoustic startle test is used to test sensorimotor function. Using the open field test, prenatal exposure of ICR mice to DE (1.0 mg/m3, 8 h/day) from GD 2 to GD 17 decreased the spontaneous motor activity in the male offspring [48]. The Morris water maze test showed that the cognitive ability of female offspring was enhanced after maternal DE (19 mg/m3, 1 h/day) exposure from GD 9 to GD 19 [50]. Intratracheal implantation of carbon black (268 μg/mouse) in pregnant mice led to no effect on acoustic startle of their offspring [51].
2.3.4. Mechanistic Studies
Reactive oxygen species (ROS) are generated from molecular oxygen through multiple perturbations [61]. Excessive ROS overwhelms the cellular antioxidant capacity and leads to cellular injuries and malignant diseases [62,63]. According to the hierarchical oxidative stress model [64], an intermediate amount of ROS induces inflammatory responses through the MAPK and NF-κB signaling cascades [65,66]. Nanoparticle-generated ROS has been identified as a source of female reproductive and developmental toxicity [67–69]. The increased level of ROS reacts with biomacromolecules (DNA, protein, lipids), disturbs intracellular homeostasis, triggers apoptosis [70] and eventually leads to maternal and embryonic toxicity.
Maternal exposure to nanoparticles likely causes fetal dysfunction in two ways: (1) nanoparticles are transferred to the fetus through blood circulation, where they result in the production of ROS; and (2) nanoparticles generate ROS in the mother, and the resulting inflammatory cytokines affect the fetus [69]. Oxidative stress produced in the fetus is the dominating factor of nanoparticle-mediated teratogenesis. Because ROS is not stable enough to travel through the cell [64], ROS in the mother may indirectly contribute to embryonic dysfunction [38,51,54,71]. The balance between oxidation, antioxidation and damage repair determines ROS-mediated risk. Except for the indirect effects from ROS and inflammation, nanoparticles may also cause direct DNA damage following nuclear translocation [72,73].
2.4. In Vitro Models of Murine Origin
Blastocysts and granulosa cells have been used to evaluate the reproductive toxicity of nanoparticles in vitro. Ag nanoparticles (13 nm) and CdSe-core QDs (3.5 nm) were shown to inhibit cell proliferation and increase apoptosis in blastocysts [74,75]. Compared to the controls, treatment with Ag nanoparticles and QDs affected embryonic development post-implantation. Blastocysts pretreated with either the Ag nanoparticles (50 μM) or QDs (500 nmol/L) induced a high resorption rate of post-implantation embryos and a decrease in fetal weight. Granulosa cells, which are responsible for follicular growth, are the primary functional cells in the ovary. It was observed that Au nanoparticles (2.85 × 1010 particles/mL medium) can enter the ovarian granulosa cells and inhibit the synthesis of estradiol [76]. All of these in vitro studies further confirm the potential toxicity of nanoparticles on reproduction and early embryonic development.
3. Nanotoxicity to Female Reproductivity and Embryonic Development in Zebrafish
Zebrafish is a well-known model for the evaluation of reproduction and development and can be used for studying the biodistribution and potential toxicity of nanoparticles. Zebrafish possess several advantages as a model for studying vertebrate development. First, their genome shares significant homology with the human genome [77]; thus, it is possible to study various physiological functions and biological processes, such as angiogenesis, malformation (including pericardial oedema and bent notochords) and oxidative stress caused by foreign substances. Second, theirs small size and high fecundity make zebrafish a cost-effective model for research [78]. Approximately 200–300 eggs can be produced from a single mating every 5–7 days, providing a sufficient number of organisms and a reduced experimental time compared to mice. In mice, the gestational period is approximately 20 days, and their litter size is far less than zebrafish. Third, transparent embryos and a rapid embryonic development facilitate the observation of morphogenetic changes and organogenesis in real time [79,80]. Fourth, various mutant phenotypes of zebrafish are available to link gene function with the corresponding pathophysiology [20,81]. These advantages make zebrafish a cheaper and time-saving alternative to rodent models.
3.1. Translocation of Nanoparticles to the Zebrafish Embryo
The chorion is a three-layered acellular envelope surrounding the embryo [82] that protects the embryo from foreign interference during development before hatching [83]. The chorion pore canals are critical for the transport of nutrients and oxygen from the aquatic environment to the embryo and for the excretion of waste in the opposite direction. Scanning-electron microscopy has shown that the pore size is 0.5–0.7 μm in diameter; thus, small nanoparticles may enter the embryo through these canals (Figure 4) [84]. It was observed that single Ag nanoparticles (5–46 nm and 11.3 ± 2.3 nm) enter the chorionic space of the embryo by Brownian diffusion [84,85], while SWCNT agglomerates are too large to cross the canals [86]. The SWCNTs were found to aggregate in the culture medium and formed agglomerates ranging in size from several hundred nanometers to several micrometers at the outer layer of the chorion. Similar results were observed when fluorescent SiO2 nanoparticles (~60 and ~200 nm in diameter) were exposed to zebrafish embryos; the nanoparticles adhered to the surface of the chorion but did not enter the embryo [87].
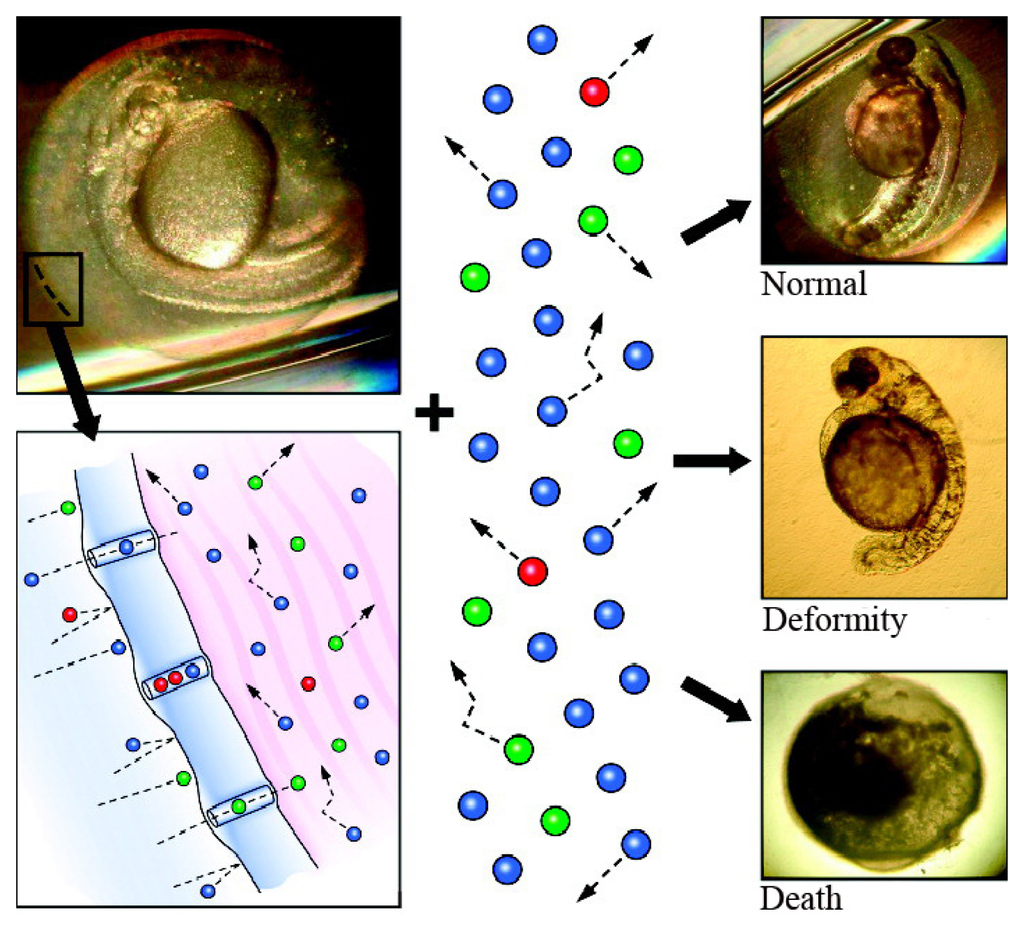
Figure 4.
Single Ag nanoparticles diffuse through the chorion pore canals and are observed in normal, deformed and dead zebrafish (reproduced with permission from [84]©2007, American Chemical Society).
3.2. Toxicity of Nanoparticles in Zebrafish Embryos
Zebrafish embryos have been used to evaluate developmental toxicity and biocompatibility in their rapid growth phase. Following treatment with nanoparticles, the embryos are placed in microtiter plates, and their survival states and phenotypic changes are examined. As a predictive model, zebrafish have been used to assess the nanotoxicity of metal and metal oxide nanoparticles, QDs and carbon nanomaterials.
3.2.1. Metal Nanoparticles
Both Ag and platinum nanoparticles were shown to cause concentration-dependent hatching delays, mortality, a drop in heart rate and other abnormalities. Furthermore, exposure to Ag nanoparticles (5–35 nm) caused defective eyes or eyeless phenotypes, pericardial oedema, pericardial effusion and circulatory defects starting at a concentration of 25 mg/mL [88]. The critical concentration of Ag nanoparticles (5–46 nm) that resulted in embryonic abnormalities and death was determined to be 1.9 nM [84]. Surface modifications, such as cysteine, decreased the toxicity of Ag nanoparticles in zebrafish [89]. It was also observed that Ag nanoparticles are more toxic than Au nanoparticles in zebrafish [88]. The teratogenicity and lethality of Au nanoparticles vary with the surface coating [90], and they show more biocompatibility than Ag nanoparticles [91,92].
The microinjection of TiO2 nanoparticles (<25 nm, 8.5 ng/g) into zebrafish embryos was observed to down-regulate the expression of genes that regulate the circadian rhythm, kinase-related activities, the immune response and vesicular transport [93]. However, embryonic incubation with a suspension of TiO2 nanoparticles (≤20 nm) displayed no toxic effects up to a concentration of 500 μg/mL [94]. In addition to the nanotoxicity of TiO2 nanoparticles, photocatalysis of TiO2 under illumination produces ROS, offering another key factor for toxicity. When zebrafish embryos were incubated with TiO2 nanoparticles (23.3 nm), the half lethal concentrations (LC50) of the TiO2 nanoparticles with/without illumination were determined to be 300 μg/mL and >1000 μg/mL, respectively, at 120 h post-fertilization (hpf) [95].
ZnO nanoparticles (20 nm) exhibit toxicity in zebrafish embryos, and the 96 hpf LC50 of their suspension was observed to be 1.793 mg/mL [94,96]. QDs have potent toxicity due to their small size and heavy metal content [97–101]. Studies from different laboratories have shown that the exposure of zebrafish to QDs causes a decreased hatch rate, malformation, a slower heart rate and delayed growth.
3.2.2. Carbon Nanomaterials
Carboxyl-functionalized MWCNTs caused mortality and alter gene expression in zebrafish embryos. Incubation with MWCNTs (30–40 nm) at a concentration of 60 μg/mL induced hatching delays and an inflammatory response. Detrimental effects on the cardiovascular system, an increased mortality rate, and apoptosis in treated embryos were also observed [102]. The length and aggregation status of CNTs are the factors that influence their toxicity [86,103].
Fullerene (C60) exposure was observed to lead to malformations and mortality in zebrafish embryos and also to induce a concentration-dependent increase in cell death (both necrosis and apoptosis) in the head and trunk. In contrast, C60 (OH)24 was shown to cause much less cell death (no apoptosis) in the head region [104]. Oxidative stress elicited by C60 was identified as a factor for malformation in zebrafish embryos [105,106].
4. Conclusions
Nanoparticles are capable of inducing detrimental effects on the reproductive systems of mice and zebrafish as well as their fetal development both in vivo and in vitro. In mice, maternal exposure to nanoparticles causes their localization in the embryo through transplacental transfer and results in fetal toxicity, such as physical defects, neurotoxicity and reproductive toxicity in the offspring. In zebrafish, incubation or microinjection of a low dose of nanoparticles induces hatching delays, mortality, axis curvatures and various morphological defects. Although the consequences of toxicity in these two models are not directly comparable, the toxicity of nanomaterials in diverse living species further highlights the universality of nanotoxicity in animals. The toxicity of nanoparticles is dose- and size-dependent [34,89]. For some nanoparticles (such as TiO2 nanoparticles), the crystal forms and illumination are also important [107]. Oxidative stress induced by nanoparticles is considered to be a main factor for female reproductive and developmental toxicity.
Numerous studies have confirmed the toxicity of nanoparticles in different species, and utilizing approaches to make nanomaterials safer will ensure their future use. The biodistribution of many nanoparticles is size-dependent; nanoparticles with a small particle size are better able to make contact with their functional units to exert their effects. However, their size may not necessarily determine their toxicity [108]. Therefore, a compromise between the size of the nanoparticles, their function (as in sewage disposal or medical applications) and their side effects could be one way to alleviate their toxicity. In addition, their shape and aggregation potential could also regulate the toxicity of nanoparticles. Plate-shaped Ag nanoparticles exhibit more severe toxicity on zebrafish embryos than nanospheres and nanowires due to the high levels of crystal defects on the surface of the plate [89]. The 10% and median lethal concentration (LD10 and LD50) of dendritic clusters of 60 nm nickel nanoparticles in zebrafish embryos were found to be much lower than well-dispersed 60 nm nanospheres, which showed a higher level of toxicity when aggregated [109]. Furthermore, surface modification is the most common method used to obtain nanoparticles with a low toxicity [90]. The toxicity induced by C60, QDs, polyamidoamine dendrimers and lead sulfide nanoparticles can also be regulated by functionalization. A change in the chemical components during manufacture could be another route to reduce toxicity; for instance, ZnO nanoparticles doped with iron showed reduced detrimental effects in mice and zebrafish.
Investigations on the effects of nanotoxicity on female reproductivity and fetal development have several issues. First, some of the reported results contradict each other, possibly due to differences in the quality of the nanoparticles used, variable experimental protocols and the different animal species selected. Unified standards, protocols and coordinated efforts are greatly needed. Second, the investigations to date have been limited to one generation. Multi-generation investigations should be launched in the near future to evaluate the long-term influence of nanoparticles. Third, because of the limited data on the reproductive and developmental toxicity of nanoparticles in humans, the use of animal models may help to speculate their effects. However, we should recognise the structural distinctions between experimental models and humans. For example, the duration of gestation in the mouse is only 20 days compared with 10 months in humans. Moreover, the reproductive structures and endocrine functions are different between mice and humans [110,111]. Therefore, conclusions derived from animal models should not be extrapolated to humans without considering their physiological differences. In short, research in this field is still quite preliminary. With more sophisticated studies, a better understanding of female reproductive and developmental toxicity will emerge in the near future.
Acknowledgments
This work was supported by the National Basic Research Program of China (973 Program 2010CB933504) and National Science Foundation of China (21137002 and 21077068).
Conflict of Interest
The authors have no conflict of interest to declare.
References
- Zhang, L.; Gu, F.; Chan, J.; Wang, A.; Langer, R.; Farokhzad, O. Nanoparticles in medicine: Therapeutic applications and developments. Clin. Pharmacol. Ther 2007, 83, 761–769. [Google Scholar]
- Lee, J.H.; Huh, Y.M.; Jun, Y.; Seo, J.; Jang, J.; Song, H.T.; Kim, S.; Cho, E.J.; Yoon, H.G.; Suh, J.S. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med 2006, 13, 95–99. [Google Scholar]
- Das, S.K.; Das, A.R.; Guha, A.K. Gold nanoparticles: Microbial synthesis and application in water hygiene management. Langmuir 2009, 25, 8192–8199. [Google Scholar]
- An inventory of nanotechnology-based consumer products currently on the market. Available online: http://www.nanotechproject.org/inventories/consumer/ (accessed on 23 April 2013).
- Chou, C.C.; Hsiao, H.Y.; Hong, Q.S.; Chen, C.H.; Peng, Y.W.; Chen, H.W.; Yang, P.C. Single-walled carbon nanotubes can induce pulmonary injury in mouse model. Nano Lett 2008, 8, 437–445. [Google Scholar]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett 2004, 4, 11–18. [Google Scholar]
- Bartneck, M.; Ritz, T.; Keul, H.A.; Wambach, M.; Bornemann, J.; Gbureck, U.; Ehling, J.; Lammers, T.; Heymann, F.; Gassler, N. Peptide-functionalized gold nanorods increase liver injury in hepatitis. ACS Nano 2012, 6, 8767–8777. [Google Scholar]
- Schipper, M.L.; Nakayama-Ratchford, N.; Davis, C.R.; Kam, N.W.S.; Chu, P.; Liu, Z.; Sun, X.; Dai, H.; Gambhir, S.S. A pilot toxicology study of single-walled carbon nanotubes in a small sample of mice. Nature Nanotech 2008, 3, 216–221. [Google Scholar]
- Wu, J.; Wang, C.; Sun, J.; Xue, Y. Neurotoxicity of silica nanoparticles: Brain localization and dopaminergic neurons damage pathways. ACS nano 2011, 5, 4476–4489. [Google Scholar]
- Lin, P.; Chen, J.W.; Chang, L.W.; Wu, J.P.; Redding, L.; Chang, H.; Yeh, T.K.; Yang, C.S.; Tsai, M.H.; Wang, H.J. Computational and ultrastructural toxicology of a nanoparticle, Quantum Dot 705, in mice. Environ. Sci. Technol 2008, 42, 6264–6270. [Google Scholar]
- Bai, Y.; Zhang, Y.; Zhang, J.; Mu, Q.; Zhang, W.; Butch, E.R.; Snyder, S.E.; Yan, B. Repeated administrations of carbon nanotubes in male mice cause reversible testis damage without affecting fertility. Nature Nanotech 2010, 5, 683–689. [Google Scholar]
- Song, Y.; Li, X.; Du, X. Exposure to nanoparticles is related to pleural effusion, pulmonary fibrosis and granuloma. Eur. Respir. J 2009, 34, 559–567. [Google Scholar]
- Hillier, S. Current concepts of the roles of follicle stimulating hormone and luteinizing hormone in folliculogenesis. Hum. Reprod 1994, 9, 188–191. [Google Scholar]
- Warren, M.; Perlroth, N. The effects of intense exercise on the female reproductive system. J. Endocrinol 2001, 170, 3–11. [Google Scholar]
- Armenti, A.E.; Zama, A.M.; Passantino, L.; Uzumcu, M. Developmental methoxychlor exposure affects multiple reproductive parameters and ovarian folliculogenesis and gene expression in adult rats. Toxicol. Appl. Pharm 2008, 233, 286–296. [Google Scholar]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar]
- Tsuchiya, T.; Oguri, I.; Yamakoshi, Y.N.; Miyata, N. Novel harmful effects of [60]fullerene on mouse embryos in vitro and in vivo. FEBS Lett 1996, 393, 139–145. [Google Scholar]
- Wang, J.; Zhu, X.; Zhang, X.; Zhao, Z.; Liu, H.; George, R.; Wilson-Rawls, J.; Chang, Y.; Chen, Y. Disruption of zebrafish (Danio rerio) reproduction upon chronic exposure to TiO2 nanoparticles. Chemosphere 2011, 83, 461–467. [Google Scholar]
- Shoults-Wilson, W.A.; Reinsch, B.C.; Tsyusko, O.V.; Bertsch, P.M.; Lowry, G.V.; Unrine, J.M. Effect of silver nanoparticle surface coating on bioaccumulation and reproductive toxicity in earthworms (Eisenia fetida). Nanotoxicology 2011, 5, 432–444. [Google Scholar]
- Dooley, K.; Zon, L.I. Zebrafish: A model system for the study of human disease. Curr. Opin. Genet. Dev 2000, 10, 252–256. [Google Scholar]
- Apter, D. Development of the hypothalamic-pituitary-ovarian axis. Ann. N. Y. Acad. Sci 1997, 816, 9–21. [Google Scholar]
- Mattison, D.; Plowchalk, D.; Meadows, M.; Al-Juburi, A.; Gandy, J.; Malek, A. Reproductive toxicity: Male and female reproductive systems as targets for chemical injury. Med. Clin. N. Am 1990, 74, 391–411. [Google Scholar]
- Gao, G.; Ze, Y.; Li, B.; Zhao, X.; Zhang, T.; Sheng, L.; Hu, R.; Gui, S.; Sang, X.; Sun, Q. Ovarian dysfunction and gene-expressed characteristics of female mice caused by long-term exposure to titanium dioxide nanoparticles. J. Hazard. Mater 2012, 243, 19–27. [Google Scholar]
- Blum, J.L.; Xiong, J.Q.; Hoffman, C.; Zelikoff, J.T. Cadmium associated with inhaled cadmium oxide nanoparticles impacts fetal and neonatal development and growth. Toxicol. Sci 2012, 126, 478–486. [Google Scholar]
- Yamashita, K.; Yoshioka, Y.; Higashisaka, K.; Mimura, K.; Morishita, Y.; Nozaki, M.; Yoshida, T.; Ogura, T.; Nabeshi, H.; Nagano, K. Silica and titanium dioxide nanoparticles cause pregnancy complications in mice. Nature Nanotech 2011, 6, 321–328. [Google Scholar]
- Martino-Andrade, A.J.; Chahoud, I. Reproductive toxicity of phthalate esters. Mol. Nutr. Food Res 2009, 54, 148–157. [Google Scholar]
- Olivero, O.A.; Yuspa, S.H.; Poirier, M.C.; Anderson, L.M.; Jones, A.B.; Wang, C.; Diwan, B.A.; Haines, D.C.; Logsdon, D.; Harbaugh, S.W. Transplacental effects of 3′-azido-2′, 3′-dideoxythymidine (AZT): Tumorigenicity in mice and genotoxicity in mice and monkeys. J. Natl. Cancer Inst 1997, 89, 1602–1608. [Google Scholar]
- Perera, F.P.; Rauh, V.; Tsai, W.Y.; Kinney, P.; Camann, D.; Barr, D.; Bernert, T.; Garfinkel, R.; Tu, Y.H.; Diaz, D. Effects of transplacental exposure to environmental pollutants on birth outcomes in a multiethnic population. Environ. Health Persp 2003, 111, 201–205. [Google Scholar]
- Semmler-Behnke, M.; Fertsch, S.; Schmid, G.; Wenk, A.; Kreyling, W.G. Uptake of 1.4 nm versus 18 nm gold nanoparticles in secondary target organs is size dependent in control and pregnant rats after intratracheal or intravenous application. Proceedings of EuroNanoForum 2007, Nanotechnology in Industrial Applications, Düsseldorf, Germany, 19–21 June 2007.
- Takeda, K.; Suzuki, K.; Ishihara, A.; Kubo-Irie, M.; Fujimoto, R.; Tabata, M.; Oshio, S.; Nihei, Y.; Ihara, T.; Sugamata, M. Nanoparticles transferred from pregnant mice to their offspring can damage the genital and cranial nerve systems. J. Health Sci 2009, 55, 95–102. [Google Scholar]
- Refuerzo, J.S.; Godin, B.; Bishop, K.; Srinivasan, S.; Shah, S.K.; Amra, S.; Ramin, S.M.; Ferrari, M. Size of the nanovectors determines the transplacental passage in pregnancy: Study in rats. Am. J. Obstet. Gynecol 2011, 204, 546, e5–546.e9.. [Google Scholar]
- Chu, M.; Wu, Q.; Yang, H.; Yuan, R.; Hou, S.; Yang, Y.; Zou, Y.; Xu, S.; Xu, K.; Ji, A.; et al. Transfer of quantum dots from pregnant mice to pups across the placental barrier. Small 2010, 6, 670–678. [Google Scholar]
- Sumner, S.C.J.; Fennell, T.R.; Snyder, R.W.; Taylor, G.F.; Lewin, A.H. Distribution of carbon-14 labeled C60 ([14C] C60) in the pregnant and in the lactating dam and the effect of C60 exposure on the biochemical profile of urine. J. Appl. Toxicol 2010, 30, 354–360. [Google Scholar]
- Tian, F.; Razansky, D.; Estrada, G.G.; Semmler-Behnke, M.; Beyerle, A.; Kreyling, W.; Ntziachristos, V.; Stoeger, T. Surface modification and size dependence in particle translocation during early embryonic development. Inhal. Toxicol 2009, 21, 92–96. [Google Scholar]
- Morgan, K. Development of a preliminary framework for informing the risk analysis and risk management of nanoparticles. Risk Anal 2005, 25, 1621–1635. [Google Scholar]
- Zhang, L.; Fischer, W.; Pippel, E.; Hause, G.; Brandsch, M.; Knez, M. Receptor-mediated cellular uptake of nanoparticles: A switchable delivery system. Small 2011, 7, 1538–1541. [Google Scholar]
- Wick, P.; Malek, A.; Manser, P.; Meili, D.; Maeder-Althaus, X.; Diener, L.; Diener, P.-A.; Zisch, A.; Krug, H.F.; von Mandach, U. Barrier capacity of human placenta for nanosized materials. Environ. Health Persp 2009, 118, 432–436. [Google Scholar]
- Fujimoto, A.; Tsukue, N.; Watanabe, M.; Sugawara, I.; Yanagisawa, R.; Takano, H.; Yoshida, S.; Takeda, K. Diesel exhaust affects immunological action in the placentas of mice. Environ. Toxicol 2005, 20, 431–440. [Google Scholar]
- Umezawa, M.; Kudo, S.; Yanagita, S.; Shinkai, Y.; Niki, R.; Oyabu, T.; Takeda, K.; Ihara, T.; Sugamata, M. Maternal exposure to carbon black nanoparticle increases collagen type VIII expression in the kidney of offspring. J. Toxicol. Sci 2011, 36, 461–468. [Google Scholar]
- Jackson, P.; Halappanavar, S.; Hougaard, K.S.; Williams, A.; Madsen, A.M.; Lamson, J.S.; Andersen, O.; Yauk, C.; Wallin, H.; Vogel, U. Maternal inhalation of surface-coated nanosized titanium dioxide (UV-Titan) in C57BL/6 mice: Effects in prenatally exposed offspring on hepatic DNA damage and gene expression. Nanotoxicology 2012. [Google Scholar] [CrossRef]
- Philbrook, N.A.; Walker, V.K.; Afrooz, A.R.M.N.; Saleh, N.B.; Winn, L.M. Investigating the effects of functionalized carbon nanotubes on reproduction and development in Drosophila melanogaster and CD-1 mice. Reprod. Toxicol 2011, 32, 442–448. [Google Scholar]
- Hougaard, K.S.; Jackson, P.; Jensen, K.A.; Sloth, J.J.; Löschner, K.; Larsen, E.H.; Birkedal, R.K.; Vibenholt, A.; Boisen, A.-M.Z.; Wallin, H.; et al. Effects of prenatal exposure to surface-coated nanosized titanium dioxide (UV-Titan). A study in mice. Part. Fiber Toxicol. 2010, 7. [Google Scholar] [CrossRef]
- Yoshida, S.; Hiyoshi, K.; Oshio, S.; Takano, H.; Takeda, K.; Ichinose, T. Effects of fetal exposure to carbon nanoparticles on reproductive function in male offspring. Fertil. Steril 2010, 93, 1695–1699. [Google Scholar]
- Hemmingsen, J.G.; Hougaard, K.S.; Talsness, C.; Wellejus, A.; Loft, S.; Wallin, H.; Møller, P. Prenatal exposure to diesel exhaust particles and effect on the male reproductive system in mice. Toxicology 2009, 264, 61–68. [Google Scholar]
- Li, C.M.; Taneda, S.; Taya, K.; Watanabe, G.; Li, X.; Fujitani, Y.; Nakajima, T.; Suzuki, A.K. Effects of in utero exposure to nanoparticle-rich diesel exhaust on testicular function in immature male rats. Toxicol. Lett 2009, 185, 1–8. [Google Scholar]
- Takahashi, Y.; Mizuo, K.; Shinkai, Y.; Oshio, S.; Takeda, K. Prenatal exposure to titanium dioxide nanoparticles increases dopamine levels in the prefrontal cortex and neostriatum of mice. J. Toxicol. Sci 2010, 35, 749–756. [Google Scholar]
- Shimizu, M.; Tainaka, H.; Oba, T.; Mizuo, K.; Umezawa, M.; Takeda, K. Maternal exposure to nanoparticulate titanium dioxide during the prenatal period alters gene expression related to brain development in the mouse. Part. Fiber Toxicol. 2009, 6. [Google Scholar] [CrossRef]
- Yokota, S.; Mizuo, K.; Moriya, N.; Oshio, S.; Sugawara, I.; Takeda, K. Effect of prenatal exposure to diesel exhaust on dopaminergic system in mice. Neurosci. Lett 2009, 449, 38–41. [Google Scholar]
- Sugamata, M.; Ihara, T.; Takano, H.; Oshio, S.; Takeda, K. Maternal diesel exhaust exposure damages newborn murine brains. J. Health Sci 2006, 52, 82–84. [Google Scholar]
- Hougaard, K.S.; Jensen, K.A.; Nordly, P.; Taxvig, C.; Vogel, U.; Saber, A.T.; Wallin, H. Effects of prenatal exposure to diesel exhaust particles on postnatal development, behavior, genotoxicity and inflammation in mice. Part. Fibre Toxicol. 2008, 5. [Google Scholar] [CrossRef]
- Jackson, P.; Vogel, U.; Wallin, H.; Hougaard, K.S. Prenatal exposure to carbon black (Printex 90): Effects on sexual development and neurofunction. Basic Clin. Pharmacol 2011, 109, 434–437. [Google Scholar]
- Pietroiusti, A.; Massimiani, M.; Fenoglio, I.; Colonna, M.; Valentini, F.; Palleschi, G.; Camaioni, A.; Magrini, A.; Siracusa, G.; Bergamaschi, A. Low doses of pristine and oxidized single wall carbon nanotubes affect mammalian embryonic development. ACS Nano 2011, 5, 4624–4633. [Google Scholar]
- Lim, J.-H.; Kim, S.-H.; Shin, I.-S.; Park, N.-H.; Moon, C.; Kang, S.-S.; Kim, S.-H.; Park, S.-C.; Kim, J.-C. Maternal exposure to multi-wall carbon nanotubes does not induce embryo-fetal developmental toxicity in rats. Birth Defects Res. B 2011, 92, 69–76. [Google Scholar]
- Park, E.J.; Kim, H.; Kim, Y.; Park, K. Effects of platinum nanoparticles on the postnatal development of mouse pups by maternal exposure. Evn. Heal.Toxicol 2010, 25, 279–286. [Google Scholar]
- Tsukue, N.; Tsubone, H.; Suzuki, A.K. Diesel exhaust affects the abnormal delivery in pregnant mice and the growth of their young. Inhal. Toxicol 2002, 14, 635–651. [Google Scholar]
- Ono, N.; Oshio, S.; Niwata, Y.; Yoshida, S.; Tsukue, N.; Sugawara, I.; Takano, H.; Takeda, K. Prenatal exposure to diesel exhaust impairs mouse spermatogenesis. Inhal. Toxicol 2007, 19, 275–281. [Google Scholar]
- Kubo-Irie, M.; Oshio, S.; Niwata, Y.; Ishihara, A.; Sugawara, I.; Takeda, K. Pre-and postnatal exposure to low-dose diesel exhaust impairs murine spermatogenesis. Inhal. Toxicol 2011, 23, 805–813. [Google Scholar]
- Watanabe, N. Decreased number of sperms and Sertoli cells in mature rats exposed to diesel exhaust as fetuses. Toxicol. Lett 2005, 155, 51–58. [Google Scholar]
- Freire, C.; Ramos, R.; Puertas, R.; Lopez-Espinosa, M.J.; Julvez, J.; Aguilera, I.; Cruz, F.; Fernandez, M.F.; Sunyer, J.; Olea, N. Association of traffic-related air pollution with cognitive development in children. J. Epidemiol. Comm. H 2010, 64, 223–228. [Google Scholar]
- Sugamata, M.; Ihara, T.; Umezawa, M.; Takeda, K. P-999-Maternal exposure to nanoparticles enhances the risk of mental neurological disorders in offspring. Eur. Psychiat 2012, 27, 1. [Google Scholar]
- Gloire, G.; Legrand-Poels, S.; Piette, J. NF-κB activation by reactive oxygen species: Fifteen years later. Biochem. Pharmacol 2006, 72, 1493–1505. [Google Scholar]
- Jomova, K.; Jenisova, Z.; Feszterova, M.; Baros, S.; Liska, J.; Hudecova, D.; Rhodes, C.; Valko, M. Arsenic: Toxicity, oxidative stress and human disease. J. Appl. Toxicol 2011, 31, 95–107. [Google Scholar]
- Peden, D.B. The role of oxidative stress and innate immunity in O3 and endotoxin-induced human allergic airway disease. Immunol. Rev 2011, 242, 91–105. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 1999; Volume 3, pp. 1–543. [Google Scholar]
- Schoonbroodt, S.; Piette, J. Oxidative stress interference with the nuclear factor-kappa B activation pathways. Biochem. Pharmacol 2000, 60, 1075–1083. [Google Scholar]
- Wang, X.; Martindale, J.L.; Liu, Y.; Holbrook, N.J. The cellular response to oxidative stress: Influences of mitogen-activated protein kinase signalling pathways on cell survival. Biochem. J 1998, 333, 291–300. [Google Scholar]
- Ornoy, A. Embryonic oxidative stress as a mechanism of teratogenesis with special emphasis on diabetic embryopathy. Reprod. Toxicol 2007, 24, 31–41. [Google Scholar]
- Wells, P.G.; Winn, L.M. Biochemical toxicology of chemical teratogenesis. Crit. Rev. Biochem. Mol 1996, 31, 1–40. [Google Scholar]
- Wells, P.G.; Bhuller, Y.; Chen, C.S.; Jeng, W.; Kasapinovic, S.; Kennedy, J.C.; Kim, P.M.; Laposa, R.R.; McCallum, G.P.; Nicol, C.J. Molecular and biochemical mechanisms in teratogenesis involving reactive oxygen species. Toxicol. Appl. Pharm 2005, 207, 354–366. [Google Scholar]
- Li, Y.; Liu, Y.; Fu, Y.; Wei, T.; Le Guyader, L.; Gao, G.; Liu, R.S.; Chang, Y.Z.; Chen, C. The triggering of apoptosis in macrophages by pristine graphene through the MAPK and TGF-beta signaling pathways. Biomaterials 2011, 33, 402–411. [Google Scholar]
- Meyer, U.; Feldon, J.; Fatemi, S.H. In-vivo rodent models for the experimental investigation of prenatal immune activation effects in neurodevelopmental brain disorders. Neurosci. Biobehav. Rev 2009, 33, 1061–1079. [Google Scholar]
- Tkachenko, A.G.; Xie, H.; Coleman, D.; Glomm, W.; Ryan, J.; Anderson, M.F.; Franzen, S.; Feldheim, D.L. Multifunctional gold nanoparticle-peptide complexes for nuclear targeting. J. Am. Chem. Soc 2003, 125, 4700–4701. [Google Scholar]
- Zhu, L.; Chang, D.W.; Dai, L.; Hong, Y. DNA damage induced by multiwalled carbon nanotubes in mouse embryonic stem cells. Nano Lett 2007, 7, 3592–3597. [Google Scholar]
- Li, P.-W.; Kuo, T.-H.; Chang, J.-H.; Yeh, J.-M.; Chan, W.-H. Induction of cytotoxicity and apoptosis in mouse blastocysts by silver nanoparticles. Toxicol. Lett 2010, 197, 82–87. [Google Scholar]
- Chan, W.; Shiao, N. Cytotoxic effect of CdSe quantum dots on mouse embryonic development. Acta Pharmacol. Sin 2008, 29, 259–266. [Google Scholar]
- Stelzer, R.; Hutz, R.J. Gold nanoparticles enter rat ovarian granulosa cells and subcellular organelles, and alter in vitro estrogen accumulation. J. Reprod. Develop 2009, 55, 685–690. [Google Scholar]
- Amatruda, J.F.; Shepard, J.L.; Stern, H.M.; Zon, L.I. Zebrafish as a cancer model system. Cancer Cell 2002, 1, 229–231. [Google Scholar]
- Hill, A.J.; Teraoka, H.; Heideman, W.; Peterson, R.E. Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol. Sci 2005, 86, 6–19. [Google Scholar]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dynam 1995, 203, 253–310. [Google Scholar]
- Fadool, J.M.; Dowling, J.E. Zebrafish: A model system for the study of eye genetics. Prog. Retin. Eye Res 2008, 27, 89–110. [Google Scholar]
- Zon, L.I.; Peterson, R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov 2005, 4, 35–44. [Google Scholar]
- Selman, K.; Wallace, R.A.; Sarka, A.; Qi, X. Stages of oocyte development in the zebrafish, Brachydanio rerio. J. Morphol 1993, 218, 203–224. [Google Scholar]
- Bonsignorio, D.; Perego, L.; Giacco, L.D.; Cotelli, F. Structure and macromolecular composition of the zebrafish egg chorion. Zygote 1996, 4, 101–108. [Google Scholar]
- Lee, K.J.; Nallathamby, P.D.; Browning, L.M.; Osgood, C.J.; Xu, X.H.N. In vivo imaging of transport and biocompatibility of single silver nanoparticles in early development of zebrafish embryos. ACS Nano 2007, 1, 133–143. [Google Scholar]
- Nallathamby, P.D.; Lee, K.J.; Xu, X.H.N. Design of stable and uniform single nanoparticle photonics for in vivo dynamics imaging of nanoenvironments of zebrafish embryonic fluids. ACS Nano 2008, 2, 1371–1380. [Google Scholar]
- Cheng, J.; Flahaut, E.; Cheng, S.H. Effect of carbon nanotubes on developing zebrafish (Danio rerio) embryos. Environ. Toxicol. Chem 2007, 26, 708–716. [Google Scholar]
- Fent, K.; Weisbrod, C.J.; Wirth-Heller, A.; Pieles, U. Assessment of uptake and toxicity of fluorescent silica nanoparticles in zebrafish (Danio rerio) early life stages. Aquat. Toxicol 2010, 100, 218–228. [Google Scholar]
- Asharani, P.V.; lianwu, Y.; Gong, Z.; Valiyaveettil, S. Comparison of the toxicity of silver, gold and platinum nanoparticles in developing zebrafish embryos. Nanotoxicology 2011, 5, 43–54. [Google Scholar]
- George, S.; Lin, S.; Ji, Z.; Thomas, C.R.; Li, L.J.; Mecklenburg, M.; Meng, H.; Wang, X.; Zhang, H.; Xia, T. Surface defects on plate-shaped silver nanoparticles contribute to its hazard potential in a fish gill cell line and zebrafish embryos. ACS Nano 2012, 6, 3745–3759. [Google Scholar]
- Truong, L.; Tilton, S.C.; Zaikova, T.; Richman, E.; Waters, K.M.; Hutchison, J.E.; Tanguay, R.L. Surface functionalities of gold nanoparticles impact embryonic gene expression responses. Nanotoxicology 2013, 7, 192–201. [Google Scholar]
- Bar-Ilan, O.; Albrecht, R.M.; Fako, V.E.; Furgeson, D.Y. Toxicity assessments of multisized gold and silver nanoparticles in zebrafish embryos. Small 2009, 5, 1897–1910. [Google Scholar]
- Browning, L.M.; Lee, K.J.; Huang, T.; Nallathamby, P.D.; Lowman, J.E.; Xu, X.H.N. Random walk of single gold nanoparticles in zebrafish embryos leading to stochastic toxic effects on embryonic developments. Nanoscale 2009, 1, 138–152. [Google Scholar]
- Jovanović, B.; Ji, T.; Palić, D. Gene expression of zebrafish embryos exposed to titanium dioxide nanoparticles and hydroxylated fullerenes. Ecotoxicol. Environ. Saf 2011, 74, 1518–1525. [Google Scholar]
- Zhu, X.; Zhu, L.; Duan, Z.; Qi, R.; Li, Y.; Lang, Y. Comparative toxicity of several metal oxide nanoparticle aqueous suspensions to Zebrafish (Danio rerio) early developmental stage. J. Environ. Sci. Health A 2008, 43, 278–284. [Google Scholar]
- Bar-Ilan, O.; Louis, K.M.; Yang, S.P.; Pedersen, J.A.; Hamers, R.J.; Peterson, R.E.; Heideman, W. Titanium dioxide nanoparticles produce phototoxicity in the developing zebrafish. Nanotoxicology 2012, 6, 670–679. [Google Scholar]
- Truong, L.; Moody, I.S.; Stankus, D.P.; Nason, J.A.; Lonergan, M.C.; Tanguay, R.L. Differential stability of lead sulfide nanoparticles influences biological responses in embryonic zebrafish. Arch. Toxicol 2010, 85, 787–798. [Google Scholar]
- Lei, Y.; Xiao, Q.; Huang, S.; Xu, W.; Zhang, Z.; He, Z.; Liu, Y.; Deng, F. Impact of CdSe/ZnS quantum dots on the development of zebrafish embryos. J. Nanopart. Res 2011, 13, 6895–6906. [Google Scholar]
- Zhang, W.; Sun, X.; Chen, L.; Lin, K.F.; Dong, Q.X.; Huang, C.J.; Fu, R.B.; Zhu, J. Toxicological effect of joint cadmium selenium quantum dots and copper ion exposure on zebrafish. Environ. Toxicol. Chem 2012, 31, 2117–2123. [Google Scholar]
- Zhang, W.; Lin, K.; Miao, Y.; Dong, Q.; Huang, C.; Wang, H.; Guo, M.; Cui, X. Toxicity assessment of zebrafish following exposure to CdTe QDs. J. Hazard. Mater. 2012, 213–214, 413–420. [Google Scholar]
- Zhang, W.; Lin, K.; Sun, X.; Dong, Q.; Huang, C.; Wang, H.; Guo, M.; Cui, X. Toxicological effect of MPA–CdSe QDs exposure on zebrafish embryo and larvae. Chemosphere 2012, 89, 52–59. [Google Scholar]
- King-Heiden, T.C.; Wiecinski, P.N.; Mangham, A.N.; Metz, K.M.; Nesbit, D.; Pedersen, J.A.; Hamers, R.J.; Heideman, W.; Peterson, R.E. Quantum dot nanotoxicity assessment using the zebrafish embryo. Environ. Sci. Technol 2009, 43, 1605–1611. [Google Scholar]
- Asharani, P.; Serina, N.; Nurmawati, M.; Wu, Y.; Gong, Z.; Valiyaveettil, S. Impact of multi-walled carbon nanotubes on aquatic species. J. Nanosci. Nanotechnol 2008, 8, 3603–3609. [Google Scholar]
- Cheng, J.; Cheng, S.H. Influence of carbon nanotube length on toxicity to zebrafish embryos. Int. J. Nanomed 2012, 7, 3731–3739. [Google Scholar]
- Usenko, C.Y.; Harper, S.L.; Tanguay, R.L. In vivo evaluation of carbon fullerene toxicity using embryonic zebrafish. Carbon 2007, 45, 1891–1898. [Google Scholar]
- Yamakoshi, Y.; Umezawa, N.; Ryu, A.; Arakane, K.; Miyata, N.; Goda, Y.; Masumizu, T.; Nagano, T. Active oxygen species generated from photoexcited fullerene (C60) as potential medicines: O2 −.versus 1O2. J. Am. Chem. Soc 2003, 125, 12803–12809. [Google Scholar]
- Usenko, C.Y.; Harper, S.L.; Tanguay, R.L. Fullerene C60 exposure elicits an oxidative stress response in embryonic zebrafish. Toxicol. Appl. Pharm 2008, 229, 44–55. [Google Scholar]
- Zhu, R.R.; Wang, S.L.; Chao, J.; Shi, D.L.; Zhang, R.; Sun, X.Y.; Yao, S.D. Bio-effects of Nano-TiO2 on DNA and cellular ultrastructure with different polymorph and size. Mater. Sci. Eng. C 2009, 29, 691–696. [Google Scholar]
- Gorth, D.J.; Rand, D.M.; Webster, T.J. Silver nanoparticle toxicity in Drosophila: Size does matter. Int. J. Nanomed 2011, 6, 343–350. [Google Scholar]
- Ispas, C.; Andreescu, D.; Patel, A.; Goia, D.V.; Andreescu, S.; Wallace, K.N. Toxicity and developmental defects of different sizes and shape nickel nanoparticles in zebrafish. Environ. Sci. Technol 2009, 43, 6349–6356. [Google Scholar]
- Carter, A. Animal models of human placentation–a review. Placenta 2007, 28, S41–S47. [Google Scholar]
- Malassine, A.; Frendo, J.L.; Evain-Brion, D. A comparison of placental development and endocrine functions between the human and mouse model. Hum. Reprod. Update 2003, 9, 531–539. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).