The Expression of Millettia pinnata Chalcone Isomerase in Saccharomyces cerevisiae Salt-Sensitive Mutants Enhances Salt-Tolerance
Abstract
:1. Introduction
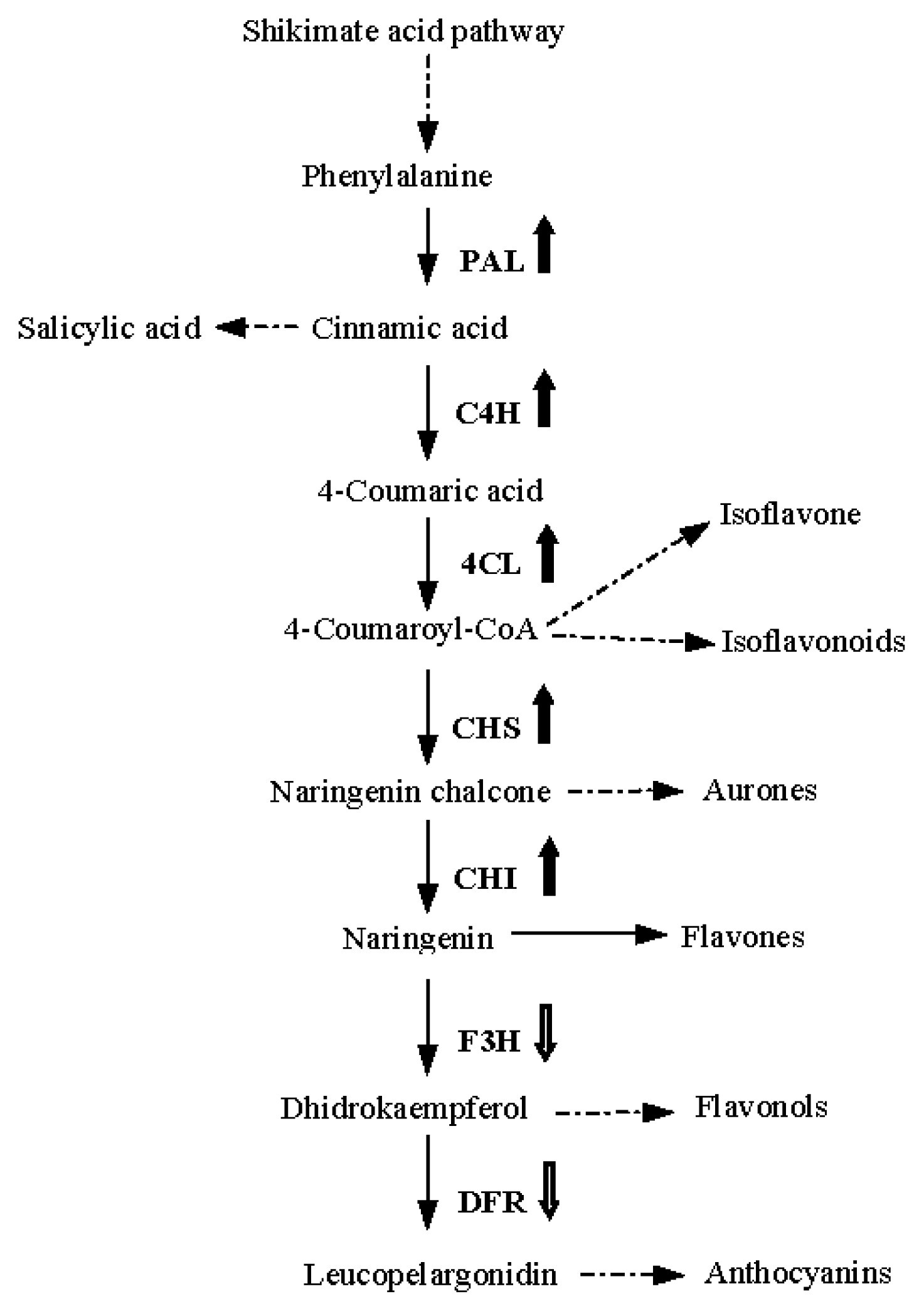
2. Results and Discussion
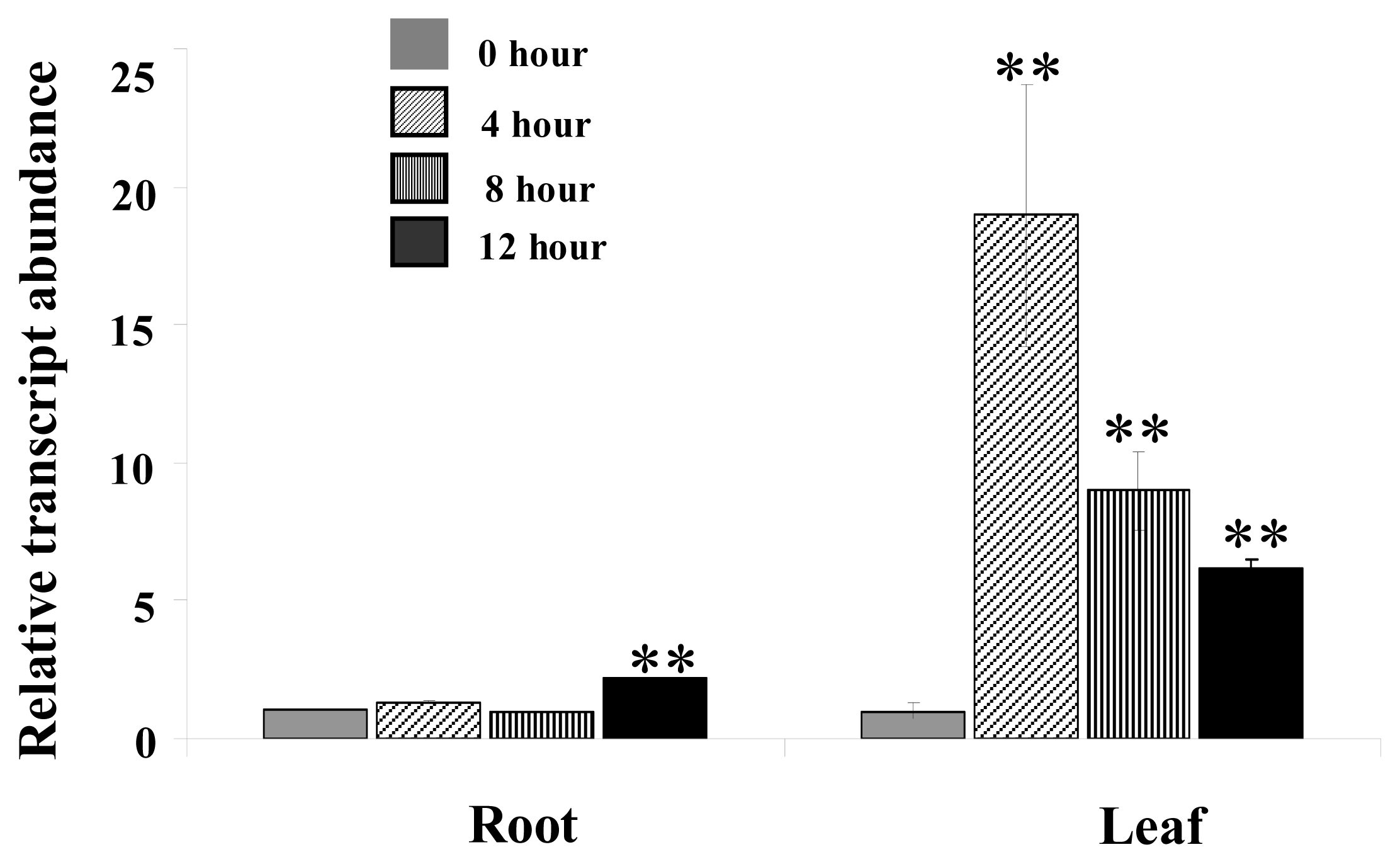
2.1. The Transcription of MpCHI Is Up Regulated by 500 mM NaCl
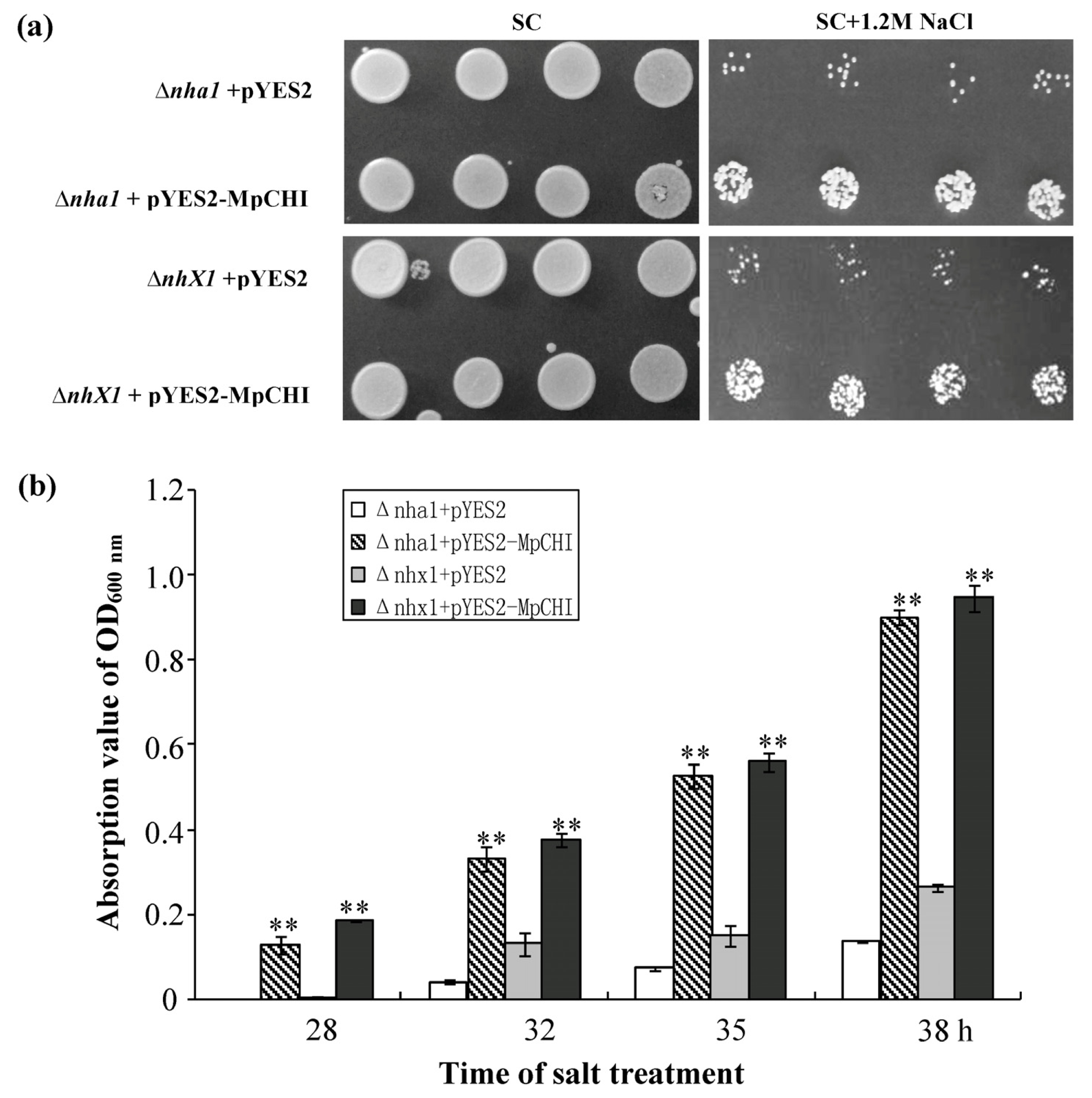
2.3. The pYES2-MpCHI Transformed Yeast Mutants Showed Improved Salt-Tolerance
3. Experimental Section
3.1. Salt-Treatment on M. pinnata Young Plants
3.2. Real-Time Quantitative Reverse Transcription PCR (QRT-PCR)
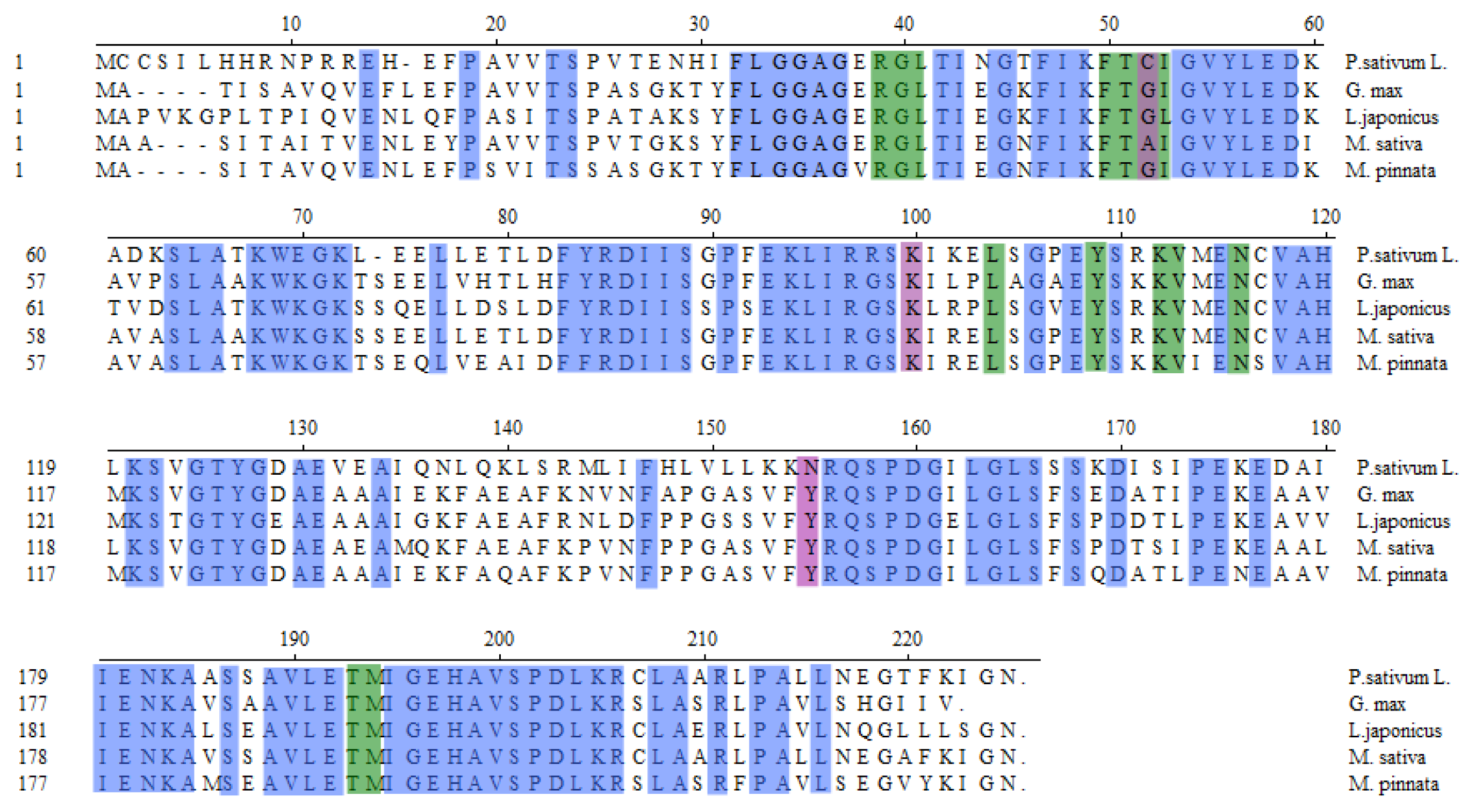
3.3. MpCHI Full Length cDNA Clone
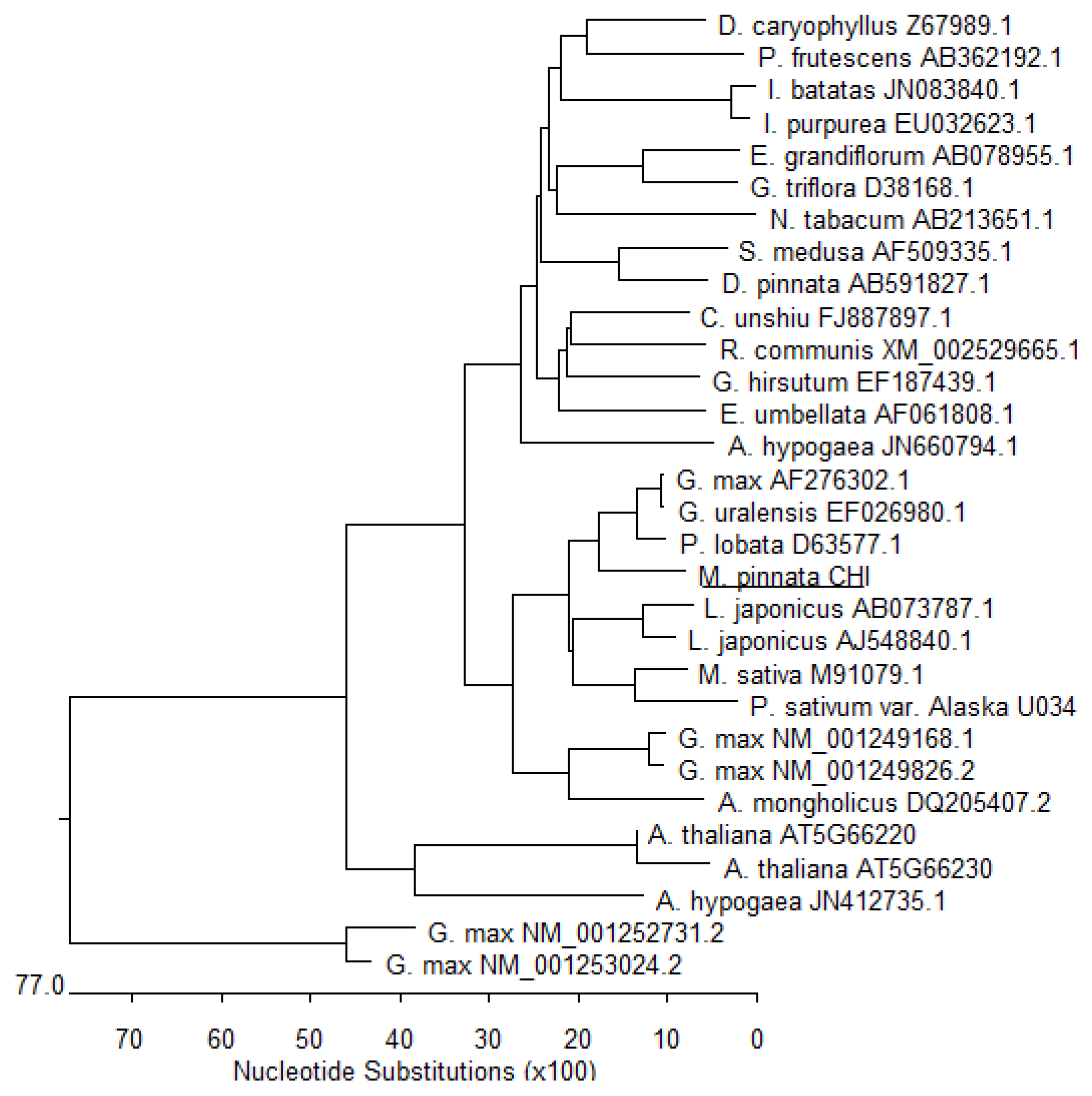
3.4. Phylogenic Analysis
3.5. Plasmid pYES2-MpCHI Construction and Yeast Transformation
3.6. Salt-Tolerance Assay on the pYES2-MpCHI Transformed Δnha1 and Δnhx1 Yeast Mutants
4. Conclusions
Supplementary Information
ijms-14-08775-s001.pdfAcknowledgments
Conflict of Interest
References
- Mukta, N.; Sreevalli, Y. Propagation techniques, evaluation and improvement of the biodiesel plant, Pongamia pinnata (L.) Pierre—A review. Ind. Crops Prod 2010, 31, 1–12. [Google Scholar]
- Huang, J.Z.; Lu, X.; Yan, H.; Chen, S.I.; Zhang, W.K.; Huang, R.F.; Zheng, Y.Z. Transcriptome characterization and sequencing-based identification of salt-responsive genes in Millettia pinnata, a semi-mangrove plant. DNA Res 2012, 19, 195–207. [Google Scholar]
- Shadle, L.; Wesley, S.V.; Korth, K.L.; Chen, F.; Lamb, C.; Dixon, R.A. Phenylpropanoid compounds and disease resistance in transgenic tobacco with altered expression of l-phenylalanine ammonia-lyase. Phytochemistry 2003, 64, 153–161. [Google Scholar]
- Mauch-Mani, B.; Slusarenko, A.J. American society of plant physiologists production of salicylic acid precursors is a major function of phenylalanine ammonia-lyase in the resistance of Arabidopsis to Peronospora parasitica. Plant Cell 1996, 8, 203–212. [Google Scholar]
- Hahlbrock, K.; Scheel, D. Physiology and molecular biology of phenylpropanoid metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol 1989, 40, 347–369. [Google Scholar]
- Winkel-Shirley, B. Biosynthesis of flavonoids and effects of stress. Curr. Opin. Plant Biol 2002, 5, 218–223. [Google Scholar]
- Christensen, A.B.; Gregersen, P.L.; Schröder, J.; Collinge, D.B. A chalcone synthase with an unusual substrate preference is expressed in barley leaves in response to UV light and pathogen attack. Plant Mol. Biol 1998, 37, 849–857. [Google Scholar]
- Winkel-Shirley, B. Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 2001, 126, 485–493. [Google Scholar]
- Tattini, M.; Galardi, C.; Pinelli, P.; Massai, R.; Remorini, D.; Agati, G. Differential accumulation of flavonoids and hydroxycinnamates in leaves of Ligustrum vulgare under excess light and drought stress. New Phytol 2004, 163, 547–561. [Google Scholar]
- Tolrà, R.P.; Poschenrieder, C.; Luppi, B.; Barceló, J. Aluminium-induced changes in the profiles of both organic acids and phenolic substances underlie Al tolerance in Rumex acetosa L. Environ. Exp. Bot 2005, 54, 231–238. [Google Scholar]
- Casati, P.; Walbot, V. Gene expression profiling in response to ultraviolet radiation in maize genotypes with varying flavonoid content. Plant Physiol 2003, 132, 1739–1754. [Google Scholar]
- Kinclova-Zimmermannova, O.; Sychrova, H. Functional study of the Nha1p C-terminus: Involvement in cell response to changes in external osmolarity. Curr. Genet 2006, 49, 229–236. [Google Scholar]
- Yoshikawa, K.; Tanaka, T.; Furusawa, C.; Nagahisa, K.; Hirasawa, T.; Shimizu, H. Comprehensive phenotypic analysis for identification of genes affecting growth under ethanol stress in Saccharomyces cerevisiae. FEMS Yeast Res 2009, 9, 32–44. [Google Scholar]
- Jez, J.M.; Bowman, M.E.; Dixon, R.A.; Noel, J.P. Structure and mechanism of the evolutionarily unique plant enzyme chalcone isomerase. Nat. Struct. Biol 2000, 7, 786–791. [Google Scholar]
- Ralston, L.; Subramanian, S.; Matsuno, M.; Yu, O. Partial reconstruction of flavonoid and isoflavonoid biosynthesis in yeast using soybean type I and type II chalcone isomerases. Plant Physiol 2005, 137, 1375–1388. [Google Scholar]
- Rodriguez-Porrata, B.; Carmona-Gutierrez, D.; Reisenbichler, A.; Bauer, M.; Lopez1, G.; Escoté1, X.; Mas1, A.; Madeo, F.; Cordero-Otero, R. Sip18 hydrophilin prevents yeast cell death during desiccation stress. J. Appl. Microbiol 2012, 112, 512–525. [Google Scholar]
- Chen, Y.; Feldman, D.E.; Deng, C.; Brown, J.A.; de Giacomo, A.F.; Gaw, A.F.; Shi, G.; Le, Q.T.; Brown, J.M.; Koong, A.C. Identification of mitogen-activated protein kinase signaling pathways that confer resistance to endoplasmic reticulum stress in Saccharomyces cerevisiae. Mol. Cancer Res 2005, 3, 669–677. [Google Scholar]
- Prick, T.; Thumm, M.; Häussinger, D.; Dahl, S. Deletion of HOG1 leads to Osmosensitivity in starvation-induced, but not rapamycin-dependent Atg8 degradation and proteolysis: Further evidence for different regulatory mechanisms in yeast autophagy. Autophagy 2006, 2, 241–243. [Google Scholar]
- Maayan, I.; Engelberg, D. The yeast MAPK Hog1 is not essential for immediate survival under osmostress. FEBS Lett 2009, 583, 2015–2020. [Google Scholar]
- Ngaki, M.N.; Louie, G.V.; Philippe, R.N.; Manning, G.; Pojer, F.; Bowman, M.E.; Li, L.; Larsen, E.; Wurtele, E.S.; Noel, J.P. Evolution of the chalcone-isomerase fold from fatty-acid binding to stereospecific catalysis. Nature 2012, 485, 530–533. [Google Scholar]
- West, R.W.J.; Yocum, R.R.; Ptashne, M. Saccharomyces cerevisiae GAL1-GAL10 divergent promoter region: Location and function of the upstream activator sequence UASG. Mol. Cell Biol 1984, 4, 2467–2478. [Google Scholar]
- Giniger, E.; Barnum, S.M.; Ptashne, M. Specific DNA Binding of GAL4, a Positive regulatory protein of yeast. Cell 1985, 40, 767–774. [Google Scholar]
- Gietz, R.D.; Schiestl, R.H. High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat. Protoc 2007, 2, 31–34. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, H.; Hu, T.; Huang, J.; Lu, X.; Huang, B.; Zheng, Y. The Expression of Millettia pinnata Chalcone Isomerase in Saccharomyces cerevisiae Salt-Sensitive Mutants Enhances Salt-Tolerance. Int. J. Mol. Sci. 2013, 14, 8775-8786. https://doi.org/10.3390/ijms14058775
Wang H, Hu T, Huang J, Lu X, Huang B, Zheng Y. The Expression of Millettia pinnata Chalcone Isomerase in Saccharomyces cerevisiae Salt-Sensitive Mutants Enhances Salt-Tolerance. International Journal of Molecular Sciences. 2013; 14(5):8775-8786. https://doi.org/10.3390/ijms14058775
Chicago/Turabian StyleWang, Hui, Tangjin Hu, Jianzi Huang, Xiang Lu, Baiqu Huang, and Yizhi Zheng. 2013. "The Expression of Millettia pinnata Chalcone Isomerase in Saccharomyces cerevisiae Salt-Sensitive Mutants Enhances Salt-Tolerance" International Journal of Molecular Sciences 14, no. 5: 8775-8786. https://doi.org/10.3390/ijms14058775



