Phospholipid Membrane Protection by Sugar Molecules during Dehydration—Insights into Molecular Mechanisms Using Scattering Techniques
Abstract
:1. Introduction
1.1. Membrane Protection by Small Solutes
1.2. Hydration Forces Explanation
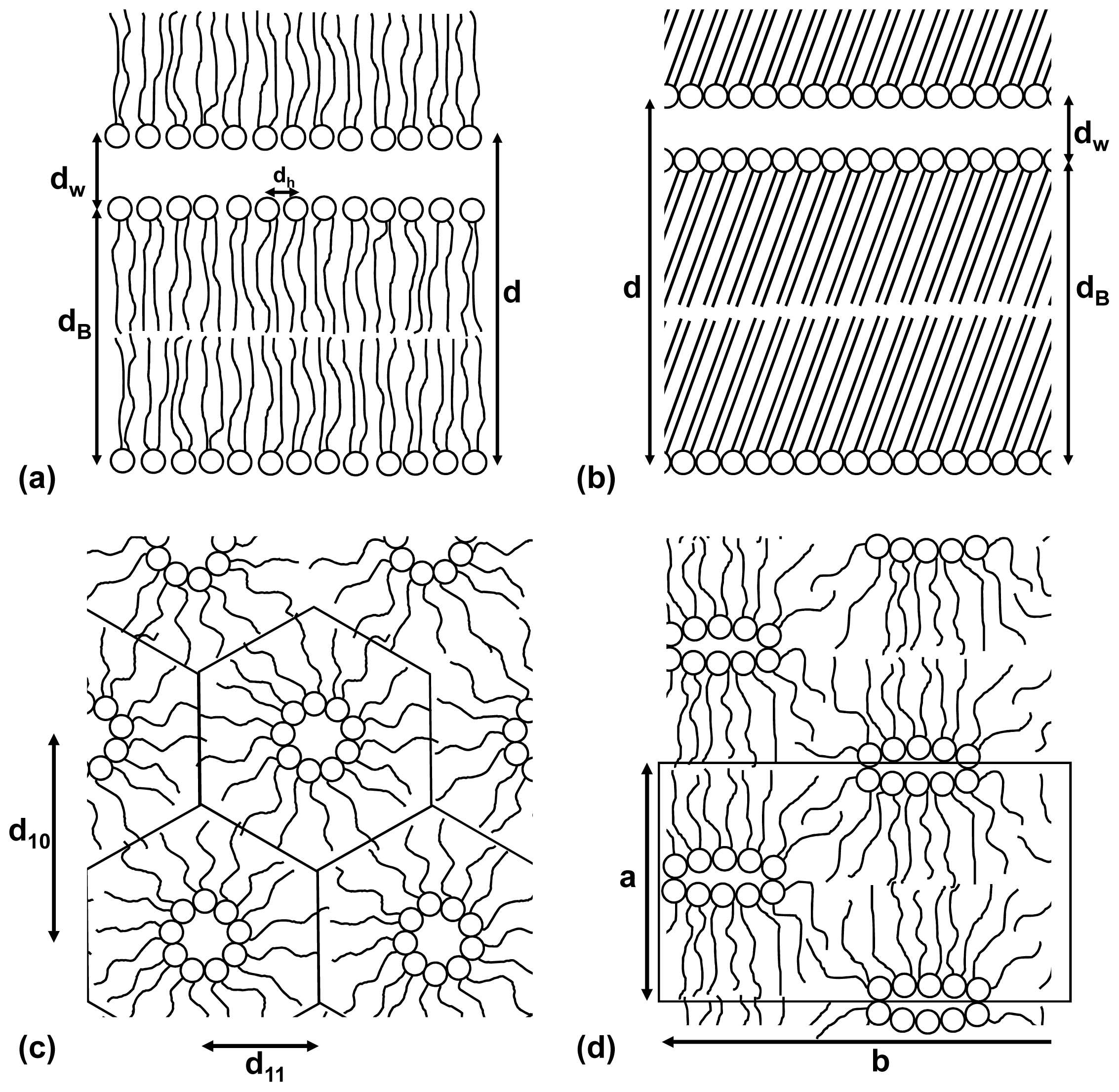
1.3. Phase Behavior of Phospholipids at Low Hydration
2. Scattering Techniques
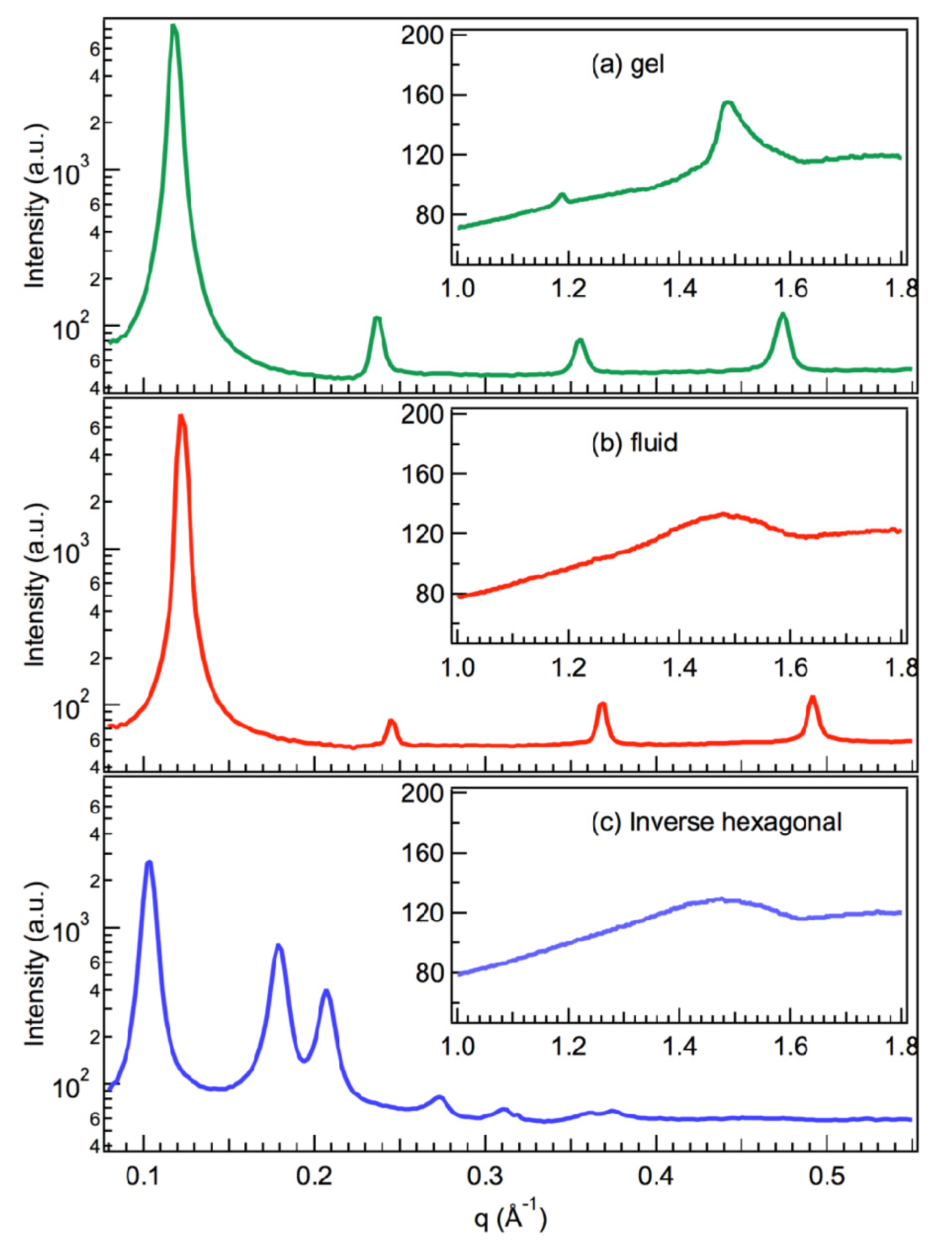
2.1. X-Ray Scattering
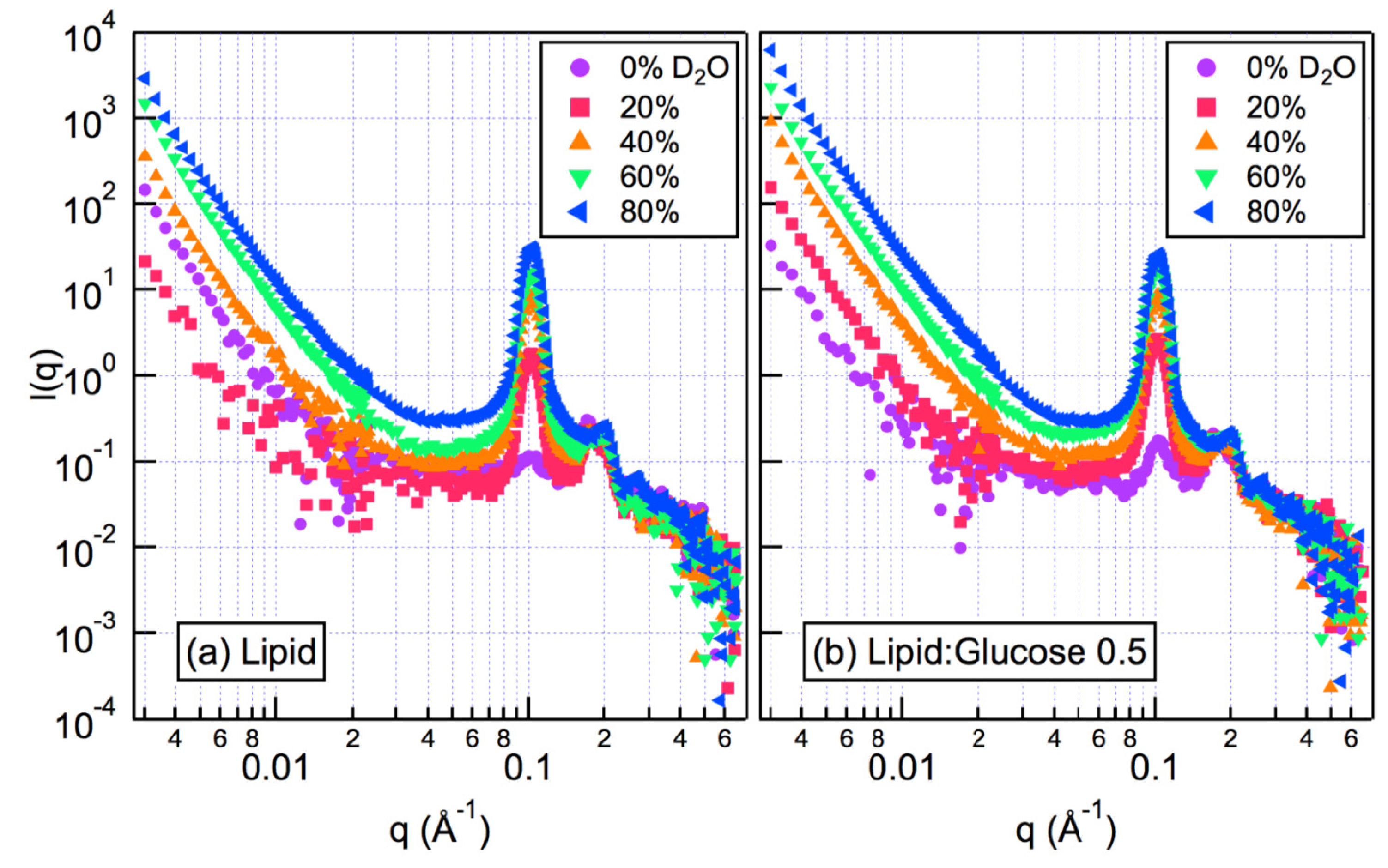
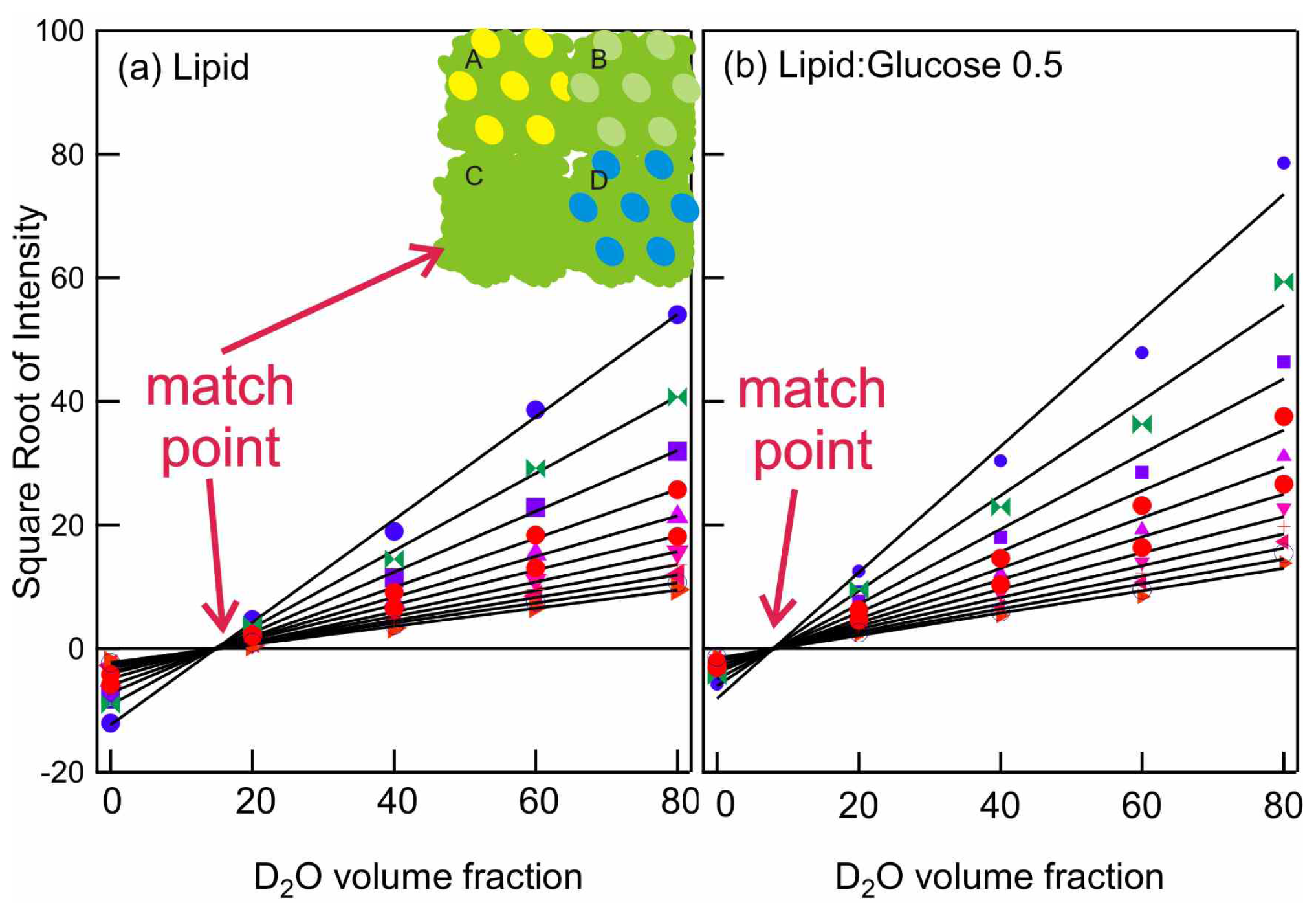
2.2. Small Angle Neutron Scattering
3. Discussion and Conclusions
4. Materials and Methods
4.1. Small Angle X-Ray Scattering
4.2. Small Angle Neutron Scattering
4.3. Sample Preparation
Acknowledgments
References
- Hoffmann, E.K.; Simonsen, L.O. Membrane mechanisms in volume and pH regulation in vertebrate cells. Physiol. Rev 1989, 69, 315–382. [Google Scholar]
- Yeo, A.R.; Flowers, T.J. Plant Solute Transport; Blackwell Publishing: Oxford, UK, 2008; p. 415. [Google Scholar]
- Maurel, C. Aquaporins and water permeability of plant membranes. Annu. Rev. Plant Physiol. Plant Molec. Biol 1997, 48, 399–429. [Google Scholar]
- Wolfe, J.; Bryant, G. Freezing, drying, and/or vitrification of membrane-solute-water systems. Cryobiology 1999, 39, 103–129. [Google Scholar]
- Benga, G. Water channel proteins (Later Called Aquaporins) and relatives: Past, present, and future. IUBMB Life 2009, 61, 112–133. [Google Scholar]
- Koster, K.L.; Bryant, G. Dehydration in Model Membranes and Protoplasts: Contrasting Effects at Low, Intermediate and High Hydrations. In Cold Hardiness in Plants: Molecular Genetics, Cell Biology and Physiology; Chen, T.H.H., Uemura, M., Fujikawa, S., Eds.; CAB International: Oxon, UK, 2005; pp. 219–234. [Google Scholar]
- Steponkus, P.L. Role of the plasma membrane in freezing injury and cold acclimation. Annu. Rev. Plant Physiol. Plant Molec. Biol 1984, 35, 543–584. [Google Scholar]
- Andersen, H.D.; Wang, C.; Arleth, L.; Peters, G.H.; Westh, P. Reconciliation of opposing views on membrane-sugar interactions. Proc. Natl. Acad. Sci. USA 2011, 108, 1874–1878. [Google Scholar]
- Westh, P. Glucose, sucrose and trehalose are partially excluded from the interface of hydrated DMPC bilayers. Phys. Chem. Chem. Phys 2008, 10, 4110–4112. [Google Scholar]
- Billi, D.; Potts, M. Life and death of dried prokaryotes. Res. Microbiol 2002, 153, 7–12. [Google Scholar]
- Koster, K.L.; Leopold, A.C. Sugars and desiccation tolerance in seeds. Plant Physiology 1988, 88, 829–832. [Google Scholar]
- Storey, K.B.; Storey, J.M. Natural freezing survival in animals. Annu. Rev. Ecol. Syst 1996, 27, 365–386. [Google Scholar]
- Uemura, M.; Warren, G.; Steponkus, P.L. Freezing sensitivity in the sfr4 mutant of Arabidopsis is due to low sugar content and is manifested by loss of osmotic responsiveness. Plant Physiol 2003, 131, 1800–1807. [Google Scholar]
- Crowe, L.M.; Crowe, J.H.; Rudolph, A.; Womersley, C.; Appel, L. Preservation of freeze-dried liposomes by trehalose. Arch. Biochem. Biophys 1985, 242, 240–247. [Google Scholar]
- Strauss, G.; Schurtenberger, P.; Hauser, H. The interaction of saccharides with lipid bilayer vesicles—Stabilization during freeze-thawing and freeze-drying. Biochim. Biophys. Acta 1986, 858, 169–180. [Google Scholar]
- Crowe, J.H.; Crowe, L.M.; Chapman, D. Preservation of membranes in anhydrobiotic organisms—The role of trehalose. Science 1984, 223, 701–703. [Google Scholar]
- Crowe, J.H.; Crowe, L.M.; Oliver, A.E.; Tsvetkova, N.; Wolkers, W.; Tablin, F. The trehalose myth revisited: Introduction to a symposium on stabilization of cells in the dry state. Cryobiology 2001, 43, 89–105. [Google Scholar]
- Potts, M. Desiccation tolerance of prokaryotes. Microbiol. Rev 1994, 58, 755–805. [Google Scholar]
- Bryant, G.; Koster, K.L.; Wolfe, J. Membrane behaviour in seeds and other systems at low water content: The various effects of solutes. Seed Sci. Res 2001, 11, 17–25. [Google Scholar]
- Koster, K.L.; Webb, M.S.; Bryant, G.; Lynch, D.V. Interactions between soluble sugars and POPC (1-palmitoyl-2-oleoylphosphatidylcholine) during dehydration-vitrification of sugars alters the phase-behavior of the phospholipid. BBA-Biomembranes 1994, 1193, 143–150. [Google Scholar]
- Sun, W.Q.; Leopold, A.C.; Crowe, L.M.; Crowe, J.H. Stability of dry liposomes in sugar glasses. Biophys. J 1996, 70, 1769–1776. [Google Scholar]
- Koster, K.L.; Lei, Y.P.; Anderson, M.; Martin, S.; Bryant, G. Effects of vitrified and nonvitrified sugars on phosphatidylcholine fluid-to-gel phase transitions. Biophys. J 2000, 78, 1932–1946. [Google Scholar]
- Wolfe, J. Lateral stresses in membranes at low water potential. Aust. J. Plant Physiol 1987, 14, 311–318. [Google Scholar]
- Bryant, G.; Wolfe, J. Lateral phase separations in lipid lamellar phases at low hydration. Cryobiology 1988, 25, 541–541. [Google Scholar]
- Bryant, G.; Wolfe, J. Can hydration forces induce lateral phase separations in lamellar phases. Euro. Biophys. J 1989, 16, 369–374. [Google Scholar]
- Bryant, G.; Wolfe, J. Interfacial forces in cryobiology and anhydrobiology. Cryo-Letters 1992, 13, 23–36. [Google Scholar]
- Horn, R.G. Direct measurement of the force between 2 lipid bilayers and observation of their fusion. Biochim. Biophys. Acta 1984, 778, 224–228. [Google Scholar]
- Lis, L.J.; McAlister, M.; Fuller, N.; Rand, R.P.; Parsegian, V.A. Interactions between neutral phospholipid bilayer membranes. Biophys. J 1982, 37, 657–665. [Google Scholar]
- Rand, R.P.; Parsegian, V.A. Hydration forces between phospholipid bilayers. Biochim. Biophys. Acta 1989, 988, 351–376. [Google Scholar]
- Lis, L.J.; McAlister, M.; Fuller, N.; Rand, R.P.; Parsegian, V.A. Measurement of the lateral compressibility of several phospholipid-bilayers. Biophys. J 1982, 37, 667–672. [Google Scholar]
- Pincet, F.; Perez, E.; Wolfe, J. Do trehalose and dimethyl sulfoxide affect intermembrane forces? Cryobiology 1994, 31, 531–539. [Google Scholar]
- Bryant, G.; Pope, J.M.; Wolfe, J. Low hydration phase properties of phospholipid mixtures—Evidence for dehydration-induced fluid-fluid separations. Euro. Biophys. J 1992, 21, 223–232. [Google Scholar]
- Tardieu, A.; Luzzati, V.; Reman, F.C. Structure and polymorphism of the hydrocarbon chains of lipids: A study of lecithin-water phases. J. Mol. Biol 1973, 75, 711–718. [Google Scholar]
- Luzzati, V. X-ray Diffraction Studies of Lipid-Water Systems. In Biological Membranes: Physical Fact and Function; Chapman,, D., Ed.; Academic Press: London, UK, 1968; Volume 1, pp. 71–123. [Google Scholar]
- Lenné, T.; Bryant, G.; Garvey, C.J.; Keiderling, U.; Koster, K.L. Location of sugars in multilamellar membranes at low hydration. Physica B 2006, 385–386, 862–864. [Google Scholar]
- Lenné, T.; Bryant, G.; Holcomb, R.; Koster, K.L. How much solute is needed to inhibit the fluid to gel membrane phase transition at low hydration? BBA-Biomembranes 2007, 1768, 1019–1022. [Google Scholar]
- Lenné, T.; Garvey, C.J.; Koster, K.L.; Bryant, G. Effects of sugars on lipid bilayers during dehydration: SAXS/WAXS measurements and quantitative model. J. Phys. Chem 2009, 113, 2486–2491. [Google Scholar]
- Lenné, T.; Garvey, C.J.; Koster, K.L.; Bryant, G. Kinetics of the lamellar gel-fluid transition in phosphatidylcholine membranes in the presence of sugars. Chem. Phys. Lipids 2010, 163, 236–242. [Google Scholar]
- Kent, B.; Garvey, C.J.; Cookson, D.; Bryant, G. The inverse hexagonal—Inverse ribbon—Lamellar gel phase transition sequence in low hydration DOPC:DOPE phospholipid mixtures. Chem. Phys. Lipids 2009, 157, 56–60. [Google Scholar]
- Kent, B.; Garvey, C.J.; Lenne, T.; Porcar, L.; Garamus, V.M.; Bryant, G. Measurement of glucose exclusion from the fully hydrated DOPE inverse hexagonal phase. Soft Matter 2010, 6, 1197–1202. [Google Scholar]
- Pohle, W.; Selle, C. Fourier-transform infrared spectroscopic evidence for a novel lyotropic phase transition occurring in dioleoylphosphatidylethanolamine. Chem. Phys. Lipids 1996, 82, 191–198. [Google Scholar]
- Pohle, W.; Selle, C.; Gauger, D.R.; Brandenburg, K. Lyotropic phase transitions in phospholipids as evidenced by small-angle synchrotron X-ray scattering. J. Biomol. Struct. Dyn 2001, 19, 351–364. [Google Scholar]
- Gruner, S.M. Stability of lyotropic phases with curved interfaces. J. Phys. Chem 1989, 93, 7562–7570. [Google Scholar]
- Tate, M.W.; Gruner, S.M. Lipid polymorphism of mixtures of dioleoylphosphatidylethanolamine and saturated and monounsaturated phosphatidylcholines of various chain lengths. Biochemistry 1987, 26, 231–236. [Google Scholar]
- Seddon, J.M. Structure of the inverted hexagonal (HII) phase, and non-lamellar phase transitions of lipids. BBA-Rev. Biomebranes 1990, 1031, 1–69. [Google Scholar]
- Epand, R.M. Functional roles of non-lamellar forming lipids. Chem. Phys. Lipids 1996, 81, 101–104. [Google Scholar]
- Pearce, R.S. A freeze-fracture study of the cell-membranes of wheat adapted to extracellular freezing and to growth at low-temperatures. J. Exp. Bot 1985, 36, 369–381. [Google Scholar]
- Pearce, R.S. The membranes of slowly drought-stressed wheat seedlings—A freeze-fracture study. Planta 1985, 166, 1–14. [Google Scholar]
- Platt-Aloia, K.A. Freeze-Fractue Evidence of Stressed-Induced Phase Separations in Plant Cell Membranes. In Physiological Regulation of Membrane Fluidity, Advances in Membrane Fluidity; Aloia,, R.C., Curtin,, C.C., Gordon,, L.M., Eds.; Alan R. Liss: New York, NY, USA, 1988; Volume 3, pp. 259–292. [Google Scholar]
- Uemura, M.; Joseph, R.A.; Steponkus, P.L. Cold-acclimation of arabidopsis-thaliana—Effect on plasma-membrane lipid-composition and freeze-induced lesions. Plant Physiol 1995, 109, 15–30. [Google Scholar]
- Caffrey, M. X-Ray Diffraction as a Technique for Studying the Mesomorphic Phase Properties of Lipids. In Membranes, Metabolism, and Dry Organisms; Conference on Anhydrous Biology, Bellagio, Italy; LEOPOLD, A.C., Ed.; Cornell University Press: Ithaca, NY, USA, 1986; pp. 350–357. [Google Scholar]
- Caffrey, M. Kinetics and mechanism of the lamellar gel lamellar liquid-crystal and lamellar inverted hexagonal phase-transition in phosphatidylethanolamine—A real-time X-ray-diffraction study using synchrotron radiation. Biochemistry 1985, 24, 4826–4844. [Google Scholar]
- Kucerka, N.; Nagle, J.F.; Sachs, J.N.; Feller, S.E.; Pencer, J.; Jackson, A.; Katsaras, J. Lipid bilayer structure determined by the simultaneous analysis of neutron and X-ray scattering data. Biophys. J 2008, 95, 2356–2367. [Google Scholar]
- Nagle, J.F.; Tristram-Nagle, S. Structure of lipid bilayers. BBA-Rev. Biomebranes 2000, 1469, 159–195. [Google Scholar]
- Ding, L.; Liu, W.; Wang, W.; Glinka, C.J.; Worcester, D.L.; Yang, L.; Huang, H.W. Diffraction techniques for nonlamellar phases of phospholipids. Langmuir 2004, 20, 9262–9269. [Google Scholar]
- Warren, B.E. X-Ray Diffraction; Addison-Wesley Publishing Company: Cambridge, Massachusetts, MA, USA, 1969; p. 381. [Google Scholar]
- Deme, B.; Zemb, T. Measurement of sugar depletion from uncharged lamellar phases by SANS contrast variation. J. Appl. Crsytallogr 2000, 33, 569–573. [Google Scholar]
- Cookson, D.; Kirby, N.; Knott, R.; Lee, M.; Schultz, D. Strategies for data collection and calibration with a pinhole-geometry SAXS instrument on a synchrotron beamline. J. Synchrotron Radiat 2006, 13, 440–444. [Google Scholar]
- Svergun, D.I.; Koch, M.H.J. Small-angle scattering studies of biological macromolecules in solution. Rep. Prog. Phys 2003, 66, 1735–1782. [Google Scholar]
- Pan, J.J.; Heberle, F.A.; Tristram-Nagle, S.; Szymanski, M.; Koepfinger, M.; Katsaras, J.; Kucerka, N. Molecular structures of fluid phase phosphatidylglycerol bilayers as determined by small angle neutron and X-ray scattering. Biochim. Biophys. Acta-Biomembr 2012, 1818, 2135–2148. [Google Scholar]
- Gilbert, E.P.; Schulz, J.C.; Noakes, T.J. Physica B 2006, 385–386, 1180–1182.
- Koster, K.L.; Maddocks, K.J.; Bryant, G. Exclusion of maltodextrins from phosphatidylcholine multilayers during dehydration: Effects on membrane phase behaviour. Eur. Biophys. J 2003, 32, 96–105. [Google Scholar]
- Gordon-Kamm, W.J.; Steponkus, P.L. Lamellar-to-hexagonal II phase-transitions in the plasma-membrane of isolated protoplasts after freeze-induced dehydration. Proc. Natl. Acad. Sci.Biol 1984, 81, 6373–6377. [Google Scholar]
- Jacrot, B. Study of biological structures by neutron-scattering from solution. Rep. Prog. Phys 1976, 39, 911–953. [Google Scholar]
- Institute Laue Langevin. Available online: http://www.ill.eu/sites/deuteration/index.htm (accessed on 11 April 2013).
- Australian Nuclear Science and Technology, organization. Available online: http://www.ansto.gov.au/ResearchHub/Bragg/Facilities/NationalDeuterationFacility/index.htm (accessed on 11 April 2013).
- Center for Structural Molecular Biology, Oak Ridge National Laboratory. Available online: http://www.csmb.ornl.gov/bdl/ (accessed on 11 April 2013).
- Wiener, M.C.; White, S.H. Fluid bilayer structure determination by the combined use of X-ray and neutron-diffraction. 1. Fluid bilayer models and the limits of resolution. Biophys. J 1991, 59, 162–173. [Google Scholar]
- Quokka—Small-Angle Neutron Scattering. Available online: http://www.ansto.gov.au/ResearchHub/Bragg/Facilities/Instruments/Quokka/index.htm (accessed on 11 April 2013).

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Garvey, C.J.; Lenné, T.; Koster, K.L.; Kent, B.; Bryant, G. Phospholipid Membrane Protection by Sugar Molecules during Dehydration—Insights into Molecular Mechanisms Using Scattering Techniques. Int. J. Mol. Sci. 2013, 14, 8148-8163. https://doi.org/10.3390/ijms14048148
Garvey CJ, Lenné T, Koster KL, Kent B, Bryant G. Phospholipid Membrane Protection by Sugar Molecules during Dehydration—Insights into Molecular Mechanisms Using Scattering Techniques. International Journal of Molecular Sciences. 2013; 14(4):8148-8163. https://doi.org/10.3390/ijms14048148
Chicago/Turabian StyleGarvey, Christopher J., Thomas Lenné, Karen L. Koster, Ben Kent, and Gary Bryant. 2013. "Phospholipid Membrane Protection by Sugar Molecules during Dehydration—Insights into Molecular Mechanisms Using Scattering Techniques" International Journal of Molecular Sciences 14, no. 4: 8148-8163. https://doi.org/10.3390/ijms14048148




