The Use of Chitosan to Enhance Photodynamic Inactivation against Candida albicans and Its Drug-Resistant Clinical Isolates
Abstract
:1. Introduction
2. Results and Discussion
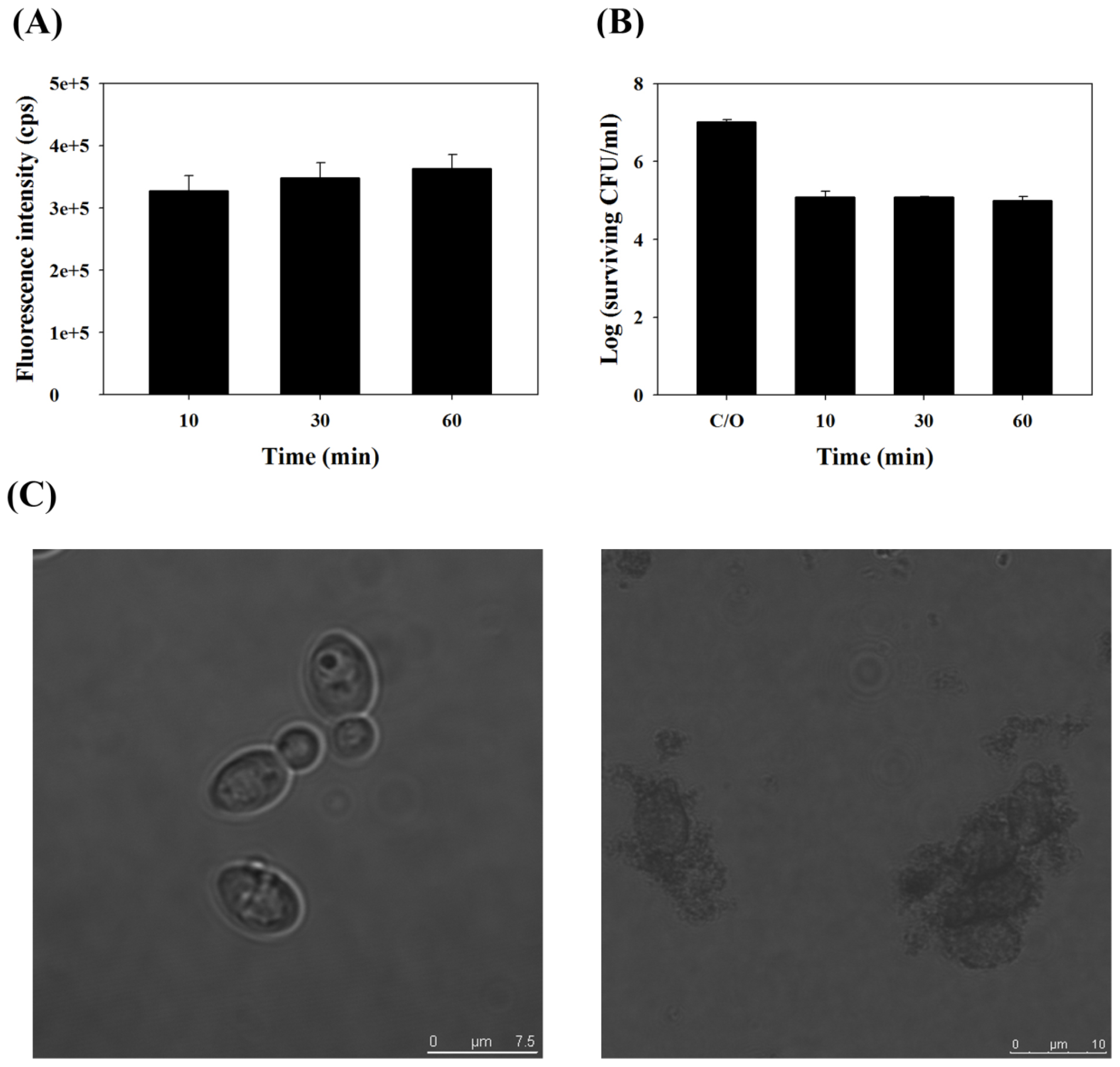
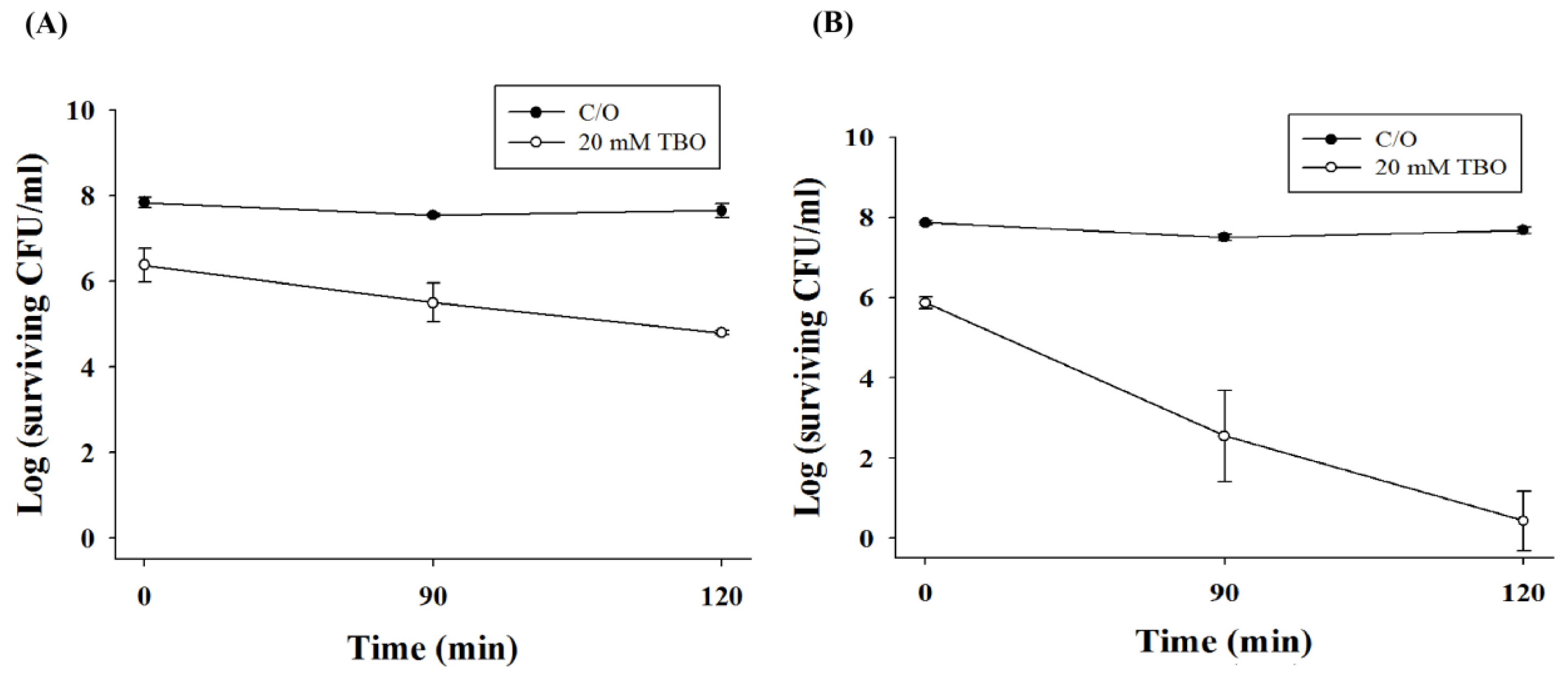
2.1. TBO Binding Assay and Survival Fraction as Related to Incubation Time and Dose
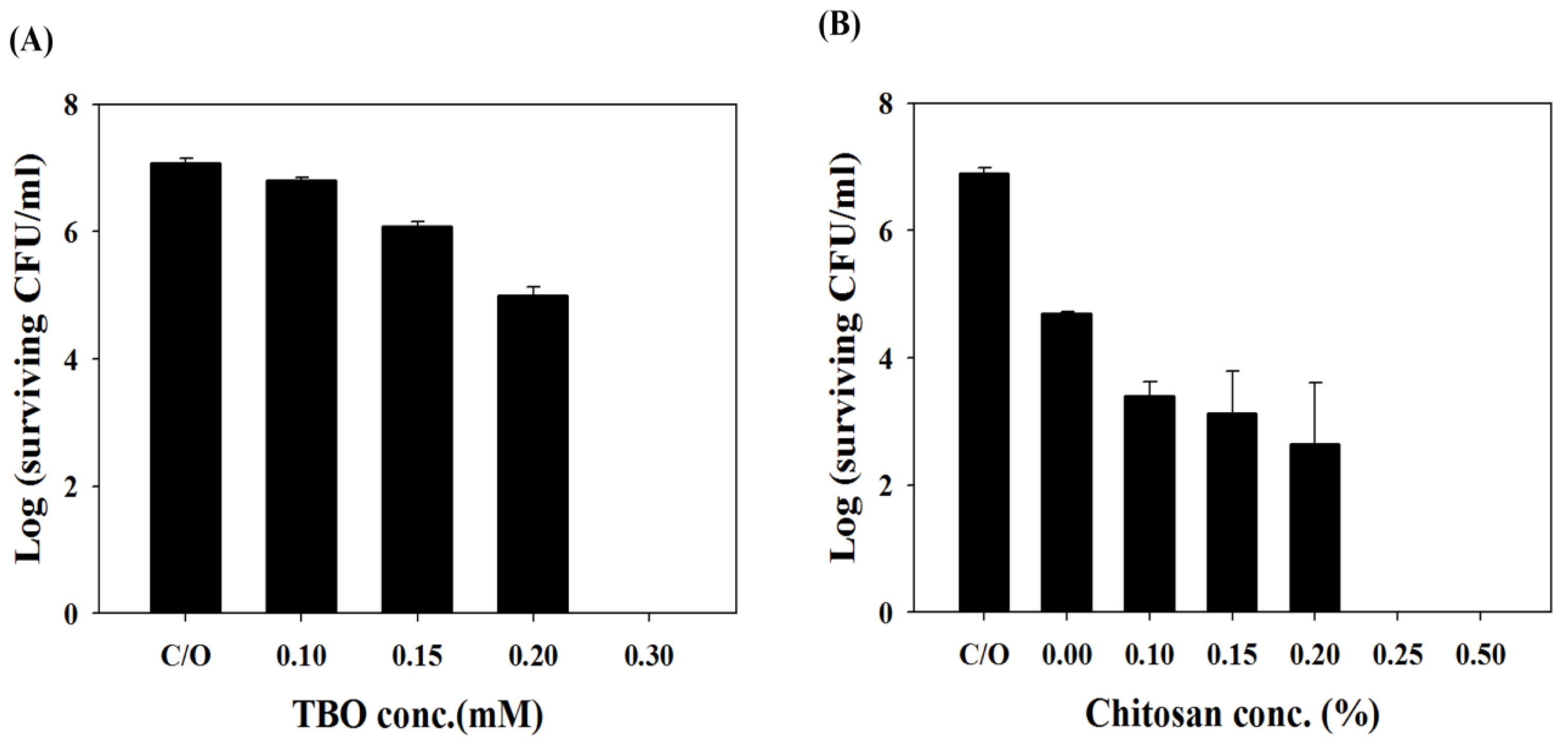
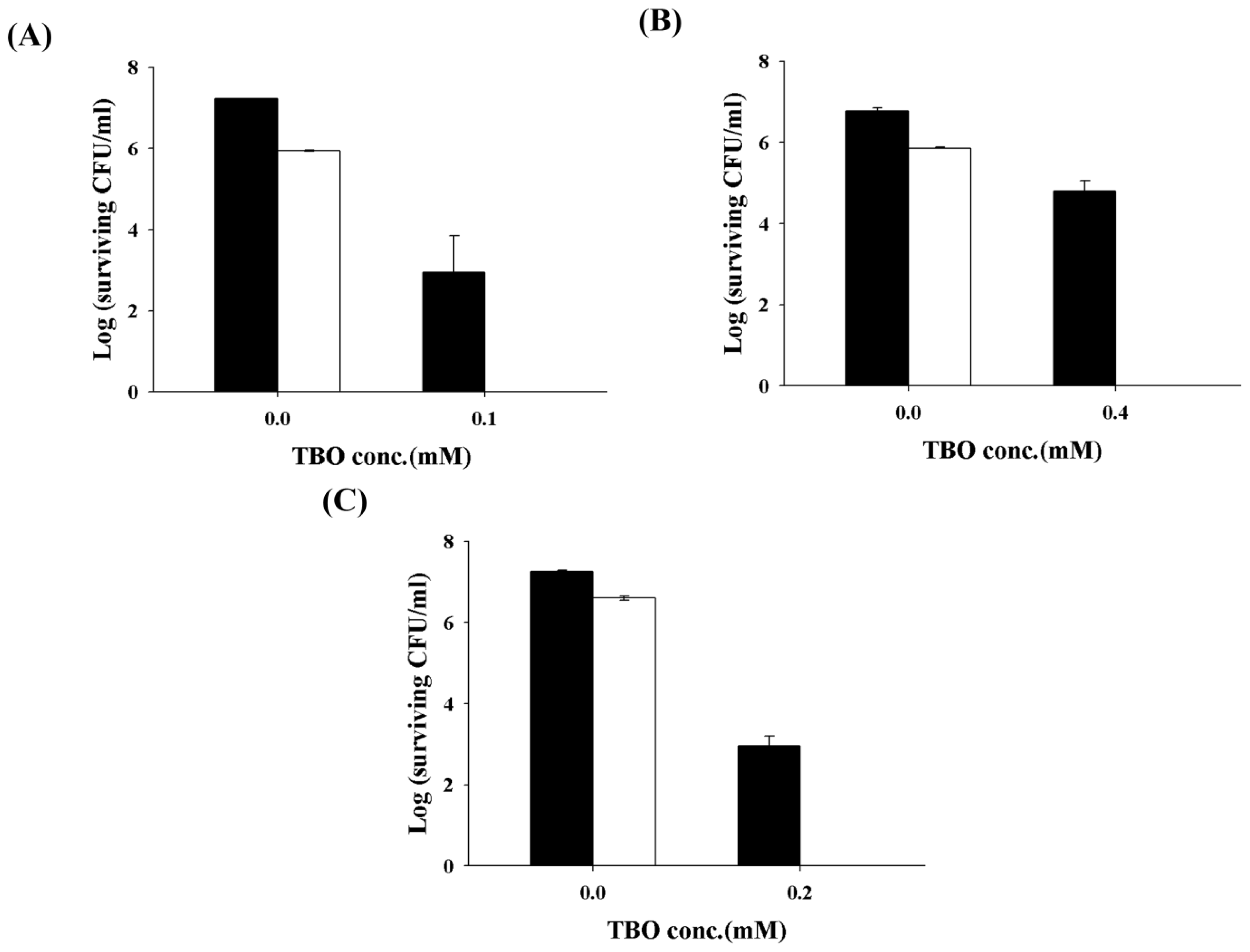
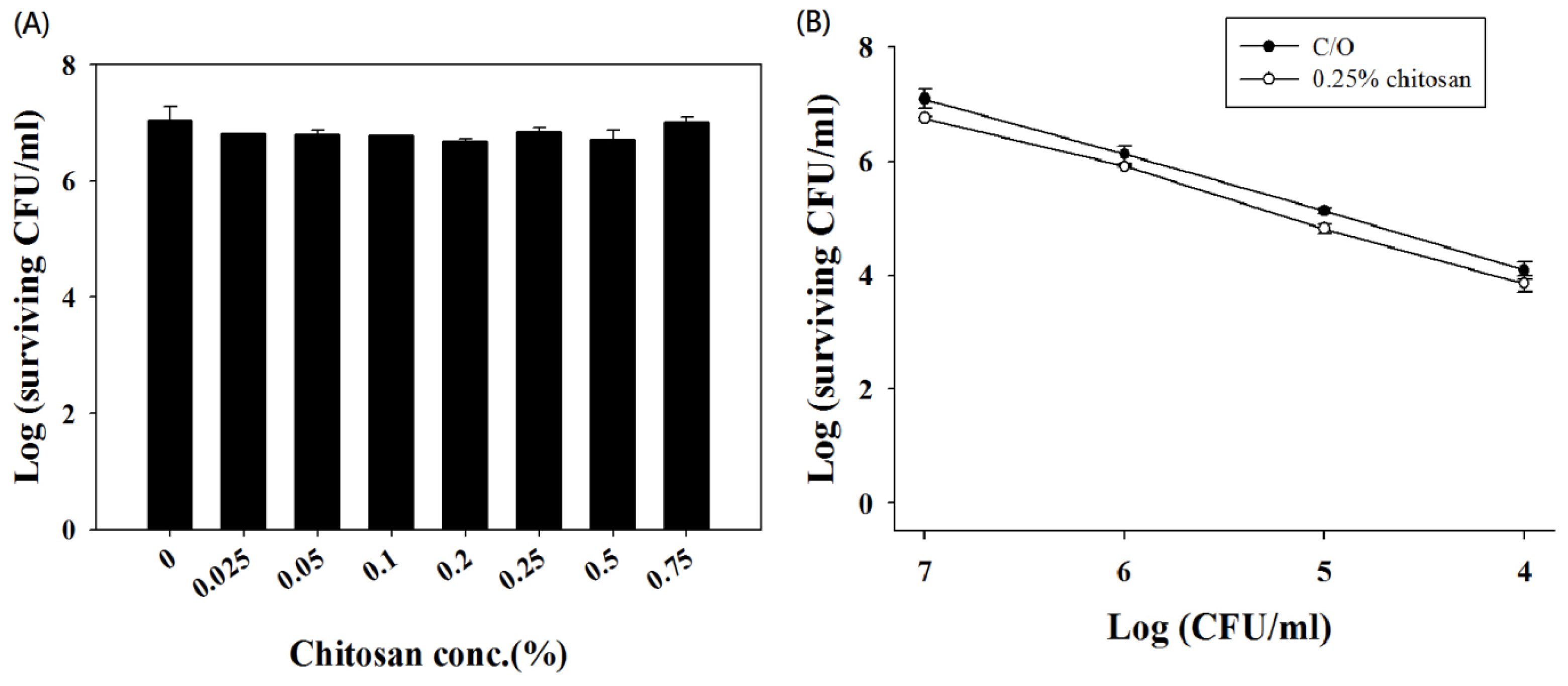
2.2. Chitosan Augments TBO or Ce6 Mediated PDI against Planktonic C. albicans
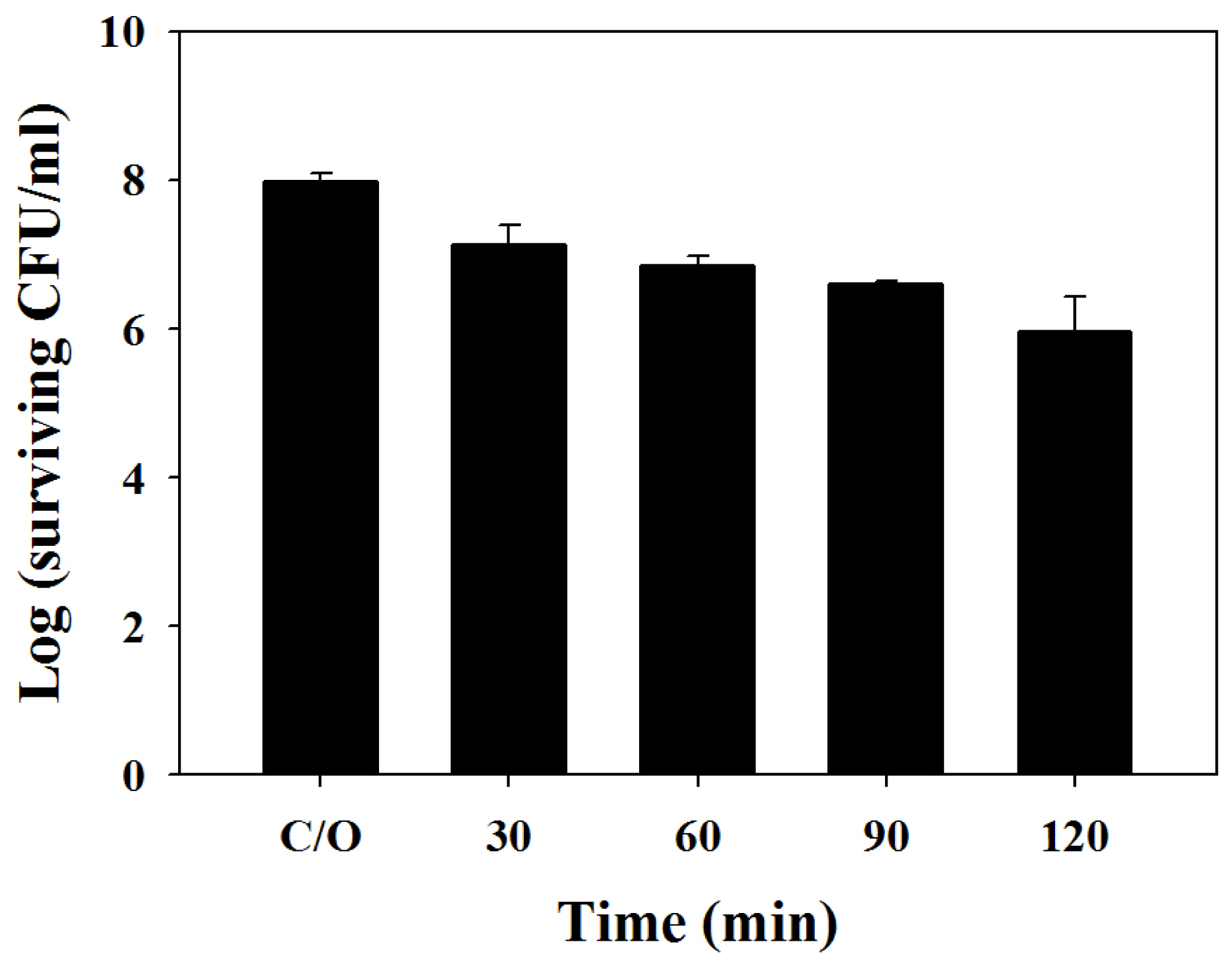
2.3. Chitosan Augments TBO Mediated PDI against the Biofilm of C. albicans
3. Experimental Section
3.1. Materials
3.2. Candida Strains and Growth Conditions
3.3. Biofilm Preparation
3.4. TBO Incubation
3.5. PDI in Planktonic Cells of C. albicans
3.6. TBO Mediated PDI in Biofilm Cells
3.7. Effect of Chitosan on PDI
3.8. C. albicans Cell Survival Assay
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Helander, I.M.; Nurmiaho-Lassila, E.L.; Ahvenainen, R.; Rhoades, J.; Roller, S. Chitosan disrupts the barrier properties of the outer membrane of gram-negative bacteria. Int. J. Food Microbiol 2001, 71, 235–244. [Google Scholar]
- Muzzarelli, R.; Tarsi, R.; Filippini, O.; Giovanetti, E.; Biagini, G.; Varaldo, P.E. Antimicrobial properties of N-carboxybutyl chitosan. Antimicrob. Agents Chemother 1990, 34, 2019–2023. [Google Scholar]
- Rabea, E.I.; Badawy, M.E.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar]
- Alonso, M.J.; Sanchez, A. The potential of chitosan in ocular drug delivery. J. Pharm. Pharmacol 2010, 55, 1451–1463. [Google Scholar]
- Ueno, H.; Yamada, H.; Tanaka, I.; Kaba, N.; Matsuura, M.; Okumura, M.; Kadosawa, T.; Fujinaga, T. Accelerating effects of chitosan for healing at early phase of experimental open wound in dogs. Biomaterials 1999, 20, 1407–1414. [Google Scholar]
- Fontana, C.R.; dos Santos, D.S.J.; Bosco, J.M.; Spolidorio, D.M.; Marcantonio, R.A.C. Evaluation of chitosan gel as antibiotic and photosensitizer delivery. Drug Deliv 2008, 15, 417–422. [Google Scholar]
- Wang, X.H.; Li, D.P.; Wang, W.J.; Feng, Q.L.; Cui, F.Z.; Xu, Y.X.; Song, X.H.; van der Werf, M. Crosslinked collagen/chitosan matrix for artificial livers. Biomaterials 2003, 24, 3213–3220. [Google Scholar]
- Hung, C.C.; Chen, Y.C.; Chang, S.C.; Luh, K.T.; Hsieh, W.C. Nosocomial candidemia in a university hospital in Taiwan. J. Formos. Med. Assoc 1996, 95, 19–28. [Google Scholar]
- Liu, C.Y.; Liao, C.H.; Chen, Y.C.; Chang, S.C. Changing epidemiology of nosocomial bloodstream infections in 11 teaching hospitals in Taiwan between 1993 and 2006. J. Microbiol. Immunol. Infect 2010, 43, 416–429. [Google Scholar]
- Phelan, J.A.; Saltzman, B.R.; Friedland, G.H.; Klein, R.S. Oral findings in patients with acquired immunodeficiency syndrome. Oral Surg. Oral Med. Oral Pathol 1987, 64, 50–56. [Google Scholar]
- Ballard, J.; Edelman, L.; Saffle, J.; Sheridan, R.; Kagan, R.; Bracco, D.; Cancio, L.; Cairns, B.; Baker, R.; Fillari, P.; et al. Positive fungal cultures in burn patients: A multicenter review. J. Burn Care Res 2008, 29, 213–221. [Google Scholar]
- Ballal, N.V.; Kundabala, M.; Bhat, K.S.; Acharya, S.; Ballal, M.; Kumar, R.; Prakash, P.Y. Susceptibility of Candida albicans and Enterococcus faecalis to Chitosan, Chlorhexidine gluconate and their combination in vitro. Aust. Endod. J 2009, 35, 29–33. [Google Scholar]
- Hamblin, M.R.; Hasan, T. Photodynamic therapy: A new antimicrobial approach to infectious disease? Photochem. Photobiol. Sci 2004, 3, 436–450. [Google Scholar]
- Jori, G.; Fabris, C.; Soncin, M.; Ferro, S.; Coppellotti, O.; Dei, D.; Fantetti, L.; Chiti, G.; Roncucci, G. Photodynamic therapy in the treatment of microbial infections: Basic principles and perspective applications. Lasers Surg. Med 2006, 38, 468–481. [Google Scholar]
- Winckler, K.D. Special section: Focus on anti-microbial photodynamic therapy (PDT). J. Photochem. Photobiol. B 2007, 86, 43–44. [Google Scholar]
- Wainwright, M.; Crossley, K.B. Photosensitising agents—Circumventing resistance and breaking down biofilms: A review. Int. Biodeter Biodeg 2004, 53, 119–126. [Google Scholar]
- Francesco, G.; Manuele, M.; Annalisa, C.; Debora, A.; Lia, F.; Gabrio, R. In vitro resistance selection studies of RLP068/Cl, a new Zn(II) phthalocyanine suitable for antimicrobial photodynamic therapy. Antimicrob. Agents Chemother 2010, 54, 637–642. [Google Scholar]
- Zeina, B.; Greenman, J.; Purcell, W.M.; Das, B. Killing of cutaneous microbial species by photodynamic therapy. Br. J. Dermatol 2001, 144, 274–278. [Google Scholar]
- Souza, R.C.; Junqueira, J.C.; Rossoni, R.D.; Pereira, C.A.; Munin, E.; Jorge, A.O. Comparison of the photodynamic fungicidal efficacy of methylene blue, toluidine blue, malachite green and low-power laser irradiation alone against Candida albicans. Lasers Med. Sci 2010, 25, 385–389. [Google Scholar]
- Zeina, B.; Greenman, J.; Corry, D.; Purcell, W.M. Cytotoxic effects of antimicrobial photodynamic therapy on keratinocytes in vitro. Br. J. Dermatol 2002, 146, 568–573. [Google Scholar]
- Soukos, N.S.; Wilson, M.; Burns, T.; Speight, P.M. Photodynamic effects of toluidine blue on human oral keratinocytes and fibroblasts and Streptococcus sanguis evaluated in vitro. Lasers Surg. Med 1996, 18, 253–259. [Google Scholar]
- Zeina, B.; Greenman, J.; Corry, D.; Purcell, W.M. Antimicrobial photodynamic therapy: Assessment of genotoxic effects on keratinocytes in vitro. Br. J. Dermatol 2003, 148, 229–232. [Google Scholar]
- Tsai, T.; Chien, H.F.; Wang, T.H.; Huang, C.T.; Ker, Y.B.; Chen, C.T. Chitosan augments photodynamic inactivation of gram-positive and gram-negative bacteria. Antimicrob. Agents Chemother 2011, 55, 1883–1890. [Google Scholar]
- Tsai, T.; Yang, Y.T.; Wang, T.H.; Chien, H.F.; Chen, C.T. Improved photodynamic inactivation of gram-positive bacteria using hematoporphyrin encapsulated in liposomes and micelles. Lasers Surg. Med 2009, 41, 316–322. [Google Scholar]
- Nett, J.E.; Guite, K.M.; Ringeisen, A.; Holoyda, K.A.; Andes, D.R. Reduced biocide susceptibility in Candida albicans biofilms. Antimicrob. Agents Chemother 2008, 52, 3411–3413. [Google Scholar]
- Chandra, J.; Kuhn, D.M.; Mukherjee, P.K.; Hoyer, L.L.; McCormick, T.; Ghannoum, M.A. Biofilm formation by the fungal pathogen Candida albicans: Development, architecture, and drug resistance. J. Bacteriol 2001, 183, 5385–5394. [Google Scholar]
- Ganan, M.; Carrascosa, A.V.; Martinez-Rodriguez, A.J. Antimicrobial activity of chitosan against Campylobacter spp. and other microorganisms and its mechanism of action. J. Food Prot 2009, 72, 1735–1738. [Google Scholar]
- Park, Y.; Kim, M.H.; Park, S.C.; Cheong, H.; Jang, M.K.; Nah, J.W.; Hahm, K.S. Investigation of the antifungal activity and mechanism of action of LMWS-Chitosan. J. Microbiol. Biotechnol 2008, 18, 1729–1734. [Google Scholar]
- Jin, Y.; Yip, H.K.; Samaranayake, Y.H.; Yau, J.Y.; Samaranayake, L.P. Biofilm-forming a ability of Candida albicans is unlikely to contribute to high levels of oral yeast carriage in cases of human immunodeficiency virus infection. J. Clin. Microbiol 2003, 41, 2961–2967. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chien, H.-F.; Chen, C.-P.; Chen, Y.-C.; Chang, P.-H.; Tsai, T.; Chen, C.-T. The Use of Chitosan to Enhance Photodynamic Inactivation against Candida albicans and Its Drug-Resistant Clinical Isolates. Int. J. Mol. Sci. 2013, 14, 7445-7456. https://doi.org/10.3390/ijms14047445
Chien H-F, Chen C-P, Chen Y-C, Chang P-H, Tsai T, Chen C-T. The Use of Chitosan to Enhance Photodynamic Inactivation against Candida albicans and Its Drug-Resistant Clinical Isolates. International Journal of Molecular Sciences. 2013; 14(4):7445-7456. https://doi.org/10.3390/ijms14047445
Chicago/Turabian StyleChien, Hsiung-Fei, Chueh-Pin Chen, Yee-Chun Chen, Po-Han Chang, Tsuimin Tsai, and Chin-Tin Chen. 2013. "The Use of Chitosan to Enhance Photodynamic Inactivation against Candida albicans and Its Drug-Resistant Clinical Isolates" International Journal of Molecular Sciences 14, no. 4: 7445-7456. https://doi.org/10.3390/ijms14047445



