RUFY, Rab and Rap Family Proteins Involved in a Regulation of Cell Polarity and Membrane Trafficking
Abstract
:Abbreviations
| CC | Coiled Coil |
| FYVE | Fab-1, YGL023, Vps27, and EEA1 proteins |
| GAP | GTPase-activating protein |
| GDF | GDI displacement factor |
| GDI | guanine nucleotide dissociation inhibitor |
| GEF | guanine nucleotide-exchange factor Glut4: glucose transporter 4 |
| PH | plekstrin homology |
| PIP | phosphoinositidephosphate |
| PIP2 | phosphatidylinositol 4,5-bisphosphate |
| PIP3 | phosphatidylinositol 3,4,5-triphosphate |
| PI3K | phosphatidylinositol-3 kinase |
| RUFY | RUN and FYVE domain-containing proteins; RUN, RPIP8, UNC-14, and NESCA proteins |
1. Introduction
2. RUN Domain Binds Several Signaling Molecules
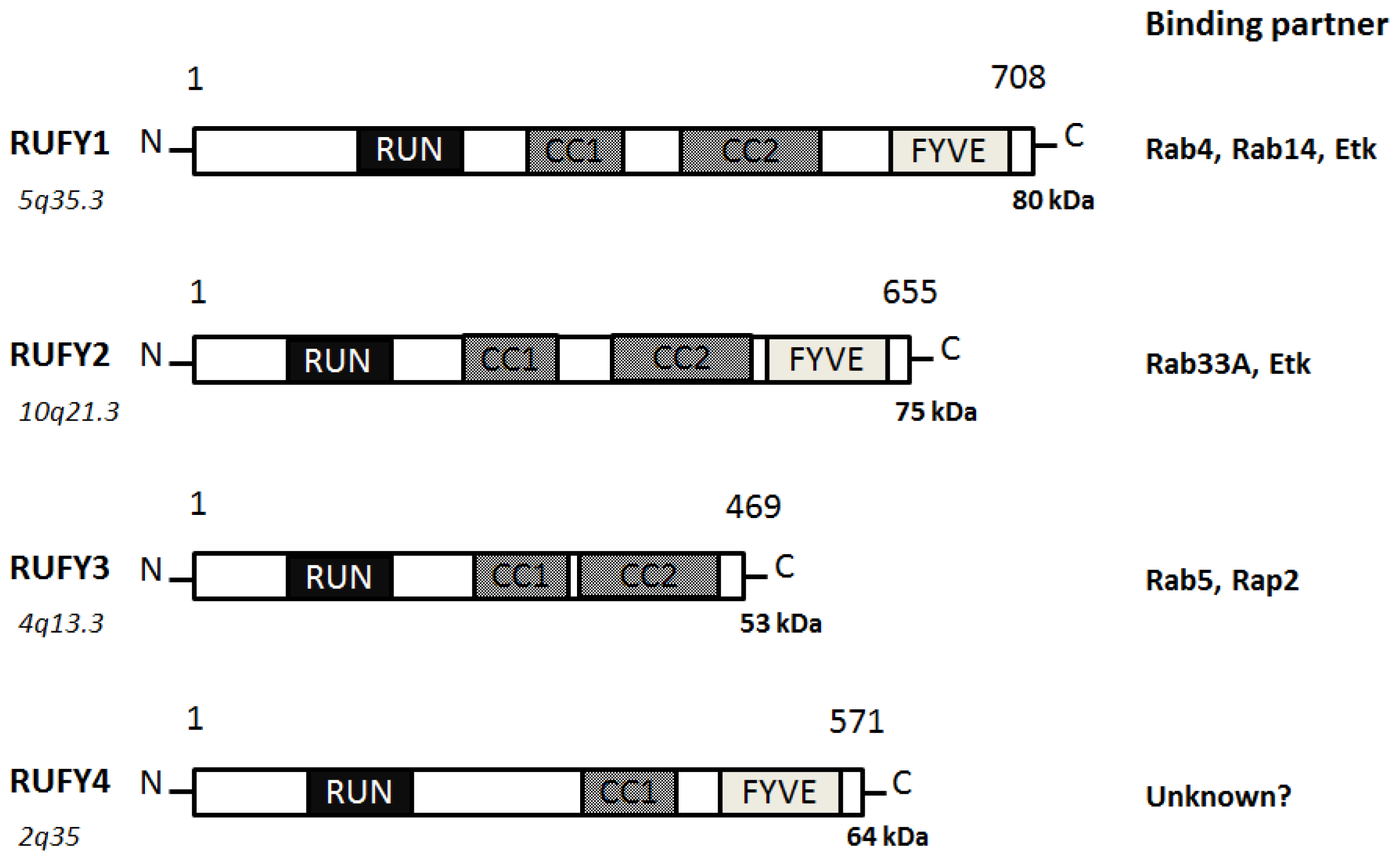
3. Function of RUFY Family Proteins with the RUN Domain
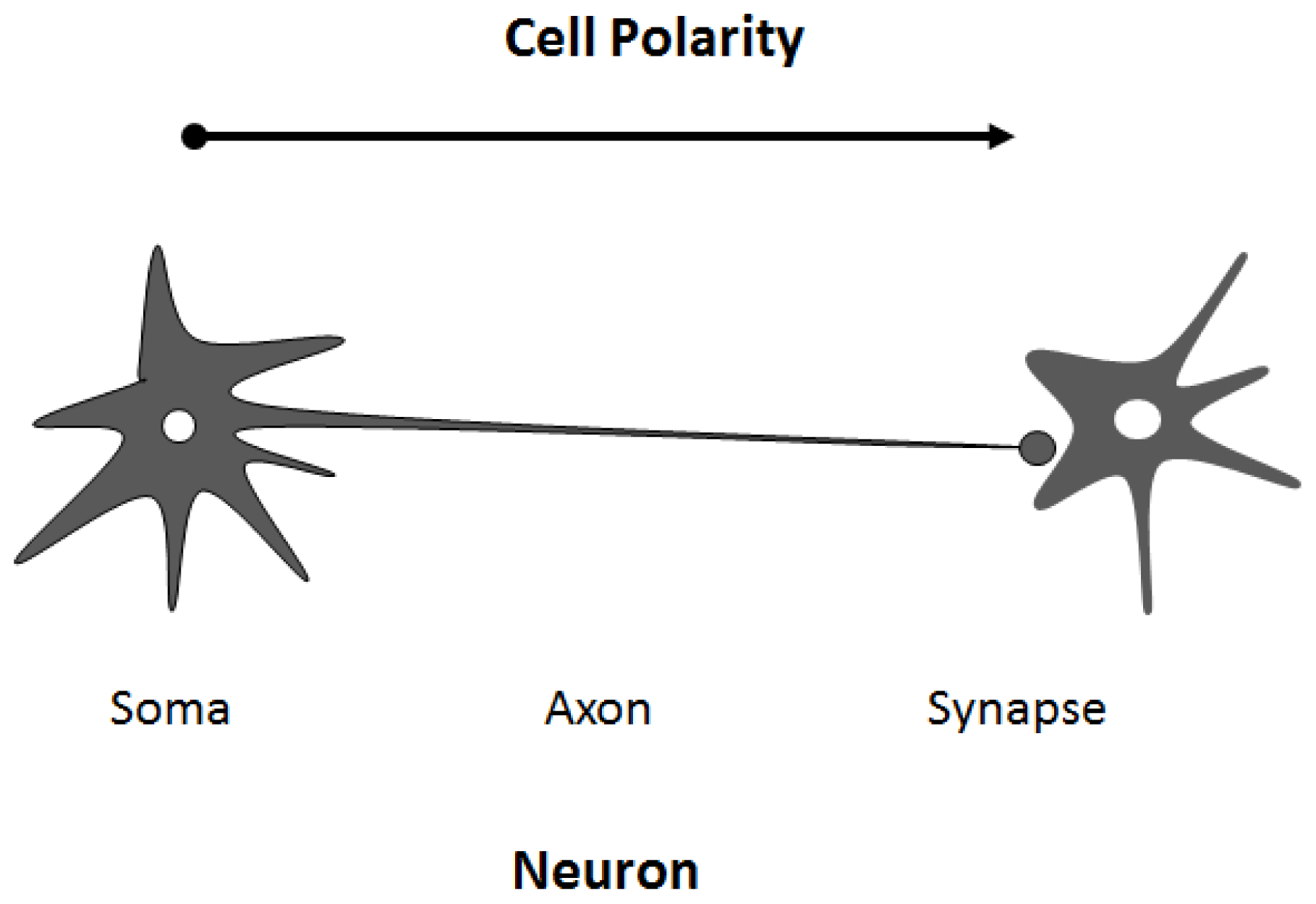
4. Regulation of Cell Polarity and Membrane Trafficking
5. Several Small GTPases Involved in the Membrane Trafficking
6. Perspective
Acknowledgments
Conflict of Interest
References
- Happé, H.; de Heer, E.; Peters, D.J. Polycystic kidney disease: The complexity of planar cell polarity and signaling during tissue regeneration and cyst formation. Biochim. Biophys. Acta 2011, 1812, 1249–1255. [Google Scholar]
- Santiago-Tirado, F.H.; Bretscher, A. Membrane-trafficking sorting hubs: Cooperation between PI4P and small GTPases at the trans-Golgi network. Trends Cell Biol 2011, 21, 515–525. [Google Scholar]
- Baum, B.; Georgiou, M. Dynamics of adherens junctions in epithelial establishment, maintenance, and remodeling. J. Cell Biol 2011, 192, 907–917. [Google Scholar]
- Wu, G.; Ge, J.; Huang, X.; Hua, Y.; Mu, D. Planar cell polarity signaling pathway in congenital heart diseases. J. Biomed. Biotechnol 2011, 2011, 589414. [Google Scholar]
- Traub, L.M. Tickets to ride: Selecting cargo for clathrin-regulated internalization. Nat. Rev. Mol. Cell Biol 2009, 10, 583–596. [Google Scholar]
- Martin-Belmonte, F.; Perez-Moreno, M. Epithelial cell polarity, stem cells and cancer. Nat. Rev. Cancer 2011, 12, 23–38. [Google Scholar]
- Layton, A.; Savage, N.S.; Howell, A.S.; Carroll, S.Y.; Drubin, D.G.; Lew, D.J. Modeling vesicle traffic reveals unexpected consequences for Cdc42p-mediated polarity establishment. Curr. Biol 2011, 21, 184–194. [Google Scholar]
- Golachowska, M.R.; Hoekstra, D.; van IJzendoorn, S.C. Recycling endosomes in apical plasma membrane domain formation and epithelial cell polarity. Trends Cell Biol 2010, 20, 618–626. [Google Scholar]
- Tóth, D.J.; Tóth, J.T.; Gulyá, S.G.; Balla, A.; Balla, T.; Hunyady, L.; Várnai, P. Acute depletion of plasma membrane phosphatidylinositol 4,5-bisphosphate impairs specific steps in endocytosis of the G-protein-coupled receptor. J. Cell Sci. 2012, 125, 2185–2197. [Google Scholar]
- Horgan, C.P.; McCaffrey, M.W. Endosomal trafficking in animal cytokinesis. Front. Biosci 2012, 4, 547–555. [Google Scholar]
- Callebaut, I.; de Gunzburg, J.; Goud, B.; Mornon, J.P. RUN domains: A new family of domains involved in Ras-like GTPase signaling. Trends Biochem. Sci 2001, 26, 79–83. [Google Scholar]
- MacDonald, J.I.; Kubu, C.J.; Meakin, S.O. Nesca, a novel adapter, translocates to the nuclear envelope and regulates neurotrophin-induced neurite outgrowth. Cell Biol 2004, 164, 851–862. [Google Scholar]
- Yoshida, H.; Kitagishi, Y.; Okumura, N.; Murakami, M.; Nishimura, Y.; Matsuda, S. How do you RUN on? FEBS Lett 2011, 585, 1707–1710. [Google Scholar]
- Kukimoto-Niino, M.; Takagi, T.; Akasaka, R.; Murayama, K.; Uchikubo-Kamo, T.; Terada, T.; Inoue, M.; Watanabe, S.; Tanaka, A.; Hayashizaki, Y.; et al. Crystal structure of the RUN domain of the RAP2-interacting protein x. J. Biol. Chem 2006, 281, 31843–31853. [Google Scholar]
- Sun, Q.; Han, C.; Liu, L.; Wang, Y.; Deng, H.; Bai, L.; Jiang, T. Crystal structure and functional implication of the RUN domain of human NESCA. Protein Cell 2012, 3, 609–617. [Google Scholar]
- Pankiv, S.; Alemu, E.A.; Brech, A.; Bruun, J.A.; Lamark, T.; Overvatn, A.; Bjørkøy, G.; Johansen, T. FYCO1 is a Rab7 effector that binds to LC3 and PI3P to mediate microtubule plus end-directed vesicle transport. J. Cell Biol 2010, 188, 253–269. [Google Scholar]
- Sakamoto, R.; Byrd, D.T.; Brown, H.M.; Hisamoto, N.; Matsumoto, K.; Jin, Y. The Caenorhabditis elegans UNC-14 RUN domain protein binds to the kinesin-1 and UNC-16 complex and regulates synaptic vesicle localization. Mol. Biol. Cell 2005, 16, 483–496. [Google Scholar]
- Ogura, K.; Goshima, Y. The autophagy-related kinase UNC-51 and its binding partner UNC-14 regulate the subcellular localization of the Netrin receptor UNC-5 in Caenorhabditis elegans. Development 2006, 133, 3441–3450. [Google Scholar]
- Yang, J.; Kim, O.; Wu, J.; Qiu, Y. Interaction between tyrosine kinase Etk and a RUN domain- and FYVE domain-containing protein RUFY1. A possible role of ETK in regulation of vesicle trafficking. J. Biol. Chem 2002, 277, 30219–30226. [Google Scholar]
- Larance, M.; Ramm, G.; Stöckli, J.; van Dam, E.M.; Winata, S.; Wasinger, V.; Simpson, F.; Graham, M.; Junutula, J.R.; Guilhaus, M.; James, D.E. Characterization of the role of the Rab GTPase-activating protein AS160 in insulin-regulated GLUT4 trafficking. J. Biol. Chem 2005, 280, 37803–37813. [Google Scholar]
- Fouraux, M.A.; Deneka, M.; Ivan, V.; van der Heijden, A.; Raymackers, J.; van Suylekom, D.; van Venrooij, W.J.; van der Sluijs, P.; Pruijn, G.J. Rabip4′ is an effector of rab5 and rab4 and regulates transport through early endosomes. Mol. Biol. Cell 2004, 15, 611–624. [Google Scholar]
- Simonsen, A.; Wurmser, A.E.; Emr, S.D.; Stenmark, H. The role of phosphoinositides in membrane transport. Curr. Opin. Cell Biol 2001, 13, 485–492. [Google Scholar]
- Mari, M.; Macia, E.; le Marchand-Brustel, Y.; Cormont, M. Role of the FYVE finger and the RUN domain for the subcellular localization of Rabip4. J. Biol. Chem 2001, 276, 42501–42508. [Google Scholar]
- Yamamoto, H.; Koga, H.; Katoh, Y.; Takahashi, S.; Nakayama, K.; Shin, H.W. Functional cross-talk between Rab14 and Rab4 through a dual effector, RUFY1/Rabip4. Mol. Biol. Cell 2010, 21, 2746–2755. [Google Scholar]
- Mari, M.; Monzo, P.; Kaddai, V.; Keslair, F.; Gonzalez, T.; le Marchand-Brustel, Y.; Cormont, M. The Rab4 effector Rabip4 plays a role in the endocytotic trafficking of Glut 4 in 3T3-L1 adipocytes. J. Cell Sci 2006, 119, 1297–1306. [Google Scholar]
- Cormont, M.; Mari, M.; Galmiche, A.; Hofman, P.; le Marchand-Brustel, Y. A FYVE-finger-containing protein, Rabip4, is a Rab4 effector involved in early endosomal traffic. Proc. Natl. Acad. Sci. USA 2001, 98, 1637–1642. [Google Scholar]
- Barbe, L.; Lundberg, E.; Oksvold, P.; Stenius, A.; Lewin, E.; Björling, E.; Asplund, A.; Pontén, F.; Brismar, H.; Uhlén, M.; Andersson-Svahn, H. Toward a confocal subcellular atlas of the human proteome. Mol. Cell Proteomics 2008, 7, 499–508. [Google Scholar]
- Fukuda, M.; Kobayashi, H.; Ishibashi, K.; Ohbayashi, N. Genome-wide investigation of the Rab binding activity of RUN domains: Development of a novel tool that specifically traps GTP-Rab35. Cell Struct. Funct 2011, 36, 155–170. [Google Scholar]
- Mori, T.; Wada, T.; Suzuki, T.; Kubota, Y.; Inagaki, N. Singar1, a novel RUN domain-containing protein, suppresses formation of surplus axons for neuronal polarity. J. Biol. Chem 2007, 282, 19884–19893. [Google Scholar]
- Yoshida, H.; Okumura, N.; Kitagishi, Y.; Shirafuji, N.; Matsuda, S. Rab5(Q79L) interacts with the carboxyl terminus of RUFY3. Int. J. Biol. Sci 2010, 6, 187–189. [Google Scholar]
- Hammad, S.M.; Twal, W.O.; Barth, J.L.; Smith, K.J.; Saad, A.F.; Virella, G.; Argraves, W.S.; Lopes-Virella, M.F. Oxidized LDL immune complexes and oxidized LDL differentially affect the expression of genes involved with inflammation and survival in human U937 monocytic cells. Atherosclerosis 2009, 202, 394–404. [Google Scholar]
- Kimura, K.; Wakamatsu, A.; Suzuki, Y.; Ota, T.; Nishikawa, T.; Yamashita, R.; Yamamoto, J.; Sekine, M.; Tsuritani, K.; Wakaguri, H.; et al. Diversification of transcriptional modulation: Large-scale identification and characterization of putative alternative promoters of human genes. Genome Res 2006, 16, 55–65. [Google Scholar]
- Pfeffer, S.R. Multiple routes of protein transport from endosomes to the trans Golgi network. FEBS Lett 2009, 583, 3811–3816. [Google Scholar]
- González-Gaitán, M. Endocytic trafficking during Drosophila development. Mech. Dev 2003, 120, 1265–1282. [Google Scholar]
- Bakhru, S.H.; Altiok, E.; Highley, C.; Delubac, D.; Suhan, J.; Hitchens, T.K.; Ho, C.; Zappe, S. Enhanced cellular uptake and long-term retention of chitosan-modified iron-oxide nanoparticles for MRI-based cell tracking. Int. J. Nanomed 2012, 7, 4613–4623. [Google Scholar]
- Monck, J.R.; Fernandez, J.M. The exocytotic fusion pore and neurotransmitter release. Neuron 1994, 12, 707–716. [Google Scholar]
- Zaid, H.; Antonescu, C.N.; Randhawa, V.K.; Klip, A. Insulin action on glucose transporters through molecular switches, tracks and tethers. Biochem. J 2008, 413, 201–215. [Google Scholar]
- Saxena, S.K.; Kaur, S. Regulation of epithelial ion channels by Rab GTPases. Biochem. Biophys. Res. Commun 2006, 351, 582–587. [Google Scholar]
- Kanamarlapudi, V. Centaurin-alpha1 and KIF13B kinesin motor protein interaction in ARF6 signalling. Biochem. Soc. Trans 2005, 33, 1279–1281. [Google Scholar]
- Saito, K.; Tautz, L.; Mustelin, T. The lipid-binding SEC14 domain. Biochim. Biophys. Acta 2007, 1771, 719–726. [Google Scholar]
- Stenmark, H.; Gillooly, D.J. Intracellular trafficking and turnover of phosphatidylinositol 3-phosphate. Semin. Cell Dev. Biol 2001, 12, 193–199. [Google Scholar]
- Shisheva, A. Phosphoinositides in insulin action on GLUT4 dynamics: Not just PtdIns(3,4,5)P3. Am. J. Physiol. Endocrinol. Metab 2008, 295, E536–E544. [Google Scholar]
- Lundquist, E.A. Small GTPases. WormBook 2006, 17, 1–18. [Google Scholar]
- Yang, H.; Sasaki, T.; Minoshima, S.; Shimizu, N. Identification of three novel proteins (SGSM1, 2, 3) which modulate small G protein (RAP and RAB)-mediated signaling pathway. Genomics 2007, 90, 249–260. [Google Scholar]
- Grosshans, B.L.; Ortiz, D.; Novick, P. Rabs and their effectors: Achieving specificity in membrane traffic. Proc. Natl. Acad. Sci. USA 2006, 103, 11821–11827. [Google Scholar]
- Mohrmann, K.; van der Sluijs, P. Regulation of membrane transport through the endocytic pathway by rabGTPases. Mol. Membr. Biol 1999, 16, 81–87. [Google Scholar]
- Jones, M.C.; Caswell, P.T.; Norman, J.C. Endocytic recycling pathways: Emerging regulators of cell migration. Curr. Opin. Cell Biol 2006, 18, 549–557. [Google Scholar]
- Nachury, M.V.; Seeley, E.S.; Jin, H. Trafficking to the ciliary membrane: How to get across the periciliary diffusion barrier? Annu. Rev. Cell Dev. Biol 2010, 26, 59–87. [Google Scholar]
- Foster, L.J.; Klip, A. Mechanism and regulation of GLUT-4 vesicle fusion in muscle and fat cells. Am. J. Physiol. Cell Physiol 2000, 279, C877–C890. [Google Scholar]
- Cavalli, V.; Corti, M.; Gruenberg, J. Endocytosis and signaling cascades: A close encounter. FEBS Lett 2001, 498, 190–196. [Google Scholar]
- Hayakawa, A.; Hayes, S.; Leonard, D.; Lambright, D.; Corvera, S. Evolutionarily conserved structural and functional roles of the FYVE domain. Biochem. Soc. Symp 2007, 74, 95–105. [Google Scholar]
- Wang, T.; Ming, Z.; Xiaochun, W.; Hong, W. Rab7: Role of its protein interaction cascades in endo-lysosomal traffic. Cell Signal 2011, 23, 516–521. [Google Scholar]
- Recacha, R.; Boulet, A.; Jollivet, F.; Monier, S.; Houdusse, A.; Goud, B.; Khan, A.R. Structural basis for recruitment of Rab6-interacting protein 1 to Golgi via a RUN domain. Structure 2009, 17, 21–30. [Google Scholar]
- Dugani, C.B.; Klip, A. Glucose transporter 4: Cycling, compartments and controversies. EMBO Rep 2005, 6, 1137–1142. [Google Scholar]
- Chibalina, M.V.; Roberts, R.C.; Arden, S.D.; Kendrick-Jones, J.; Buss, F. Rab8-optineurin-myosin VI: Analysis of interactions and functions in the secretory pathway. Methods Enzymol 2008, 438, 11–24. [Google Scholar]
- Chia, P.Z.; Gasnereau, I.; Lieu, Z.Z.; Gleeson, P.A. Rab9-dependent retrograde transport and endosomal sorting of the endopeptidase furin. J. Cell Sci 2011, 124, 2401–2413. [Google Scholar]
- Bruce, E.A.; Digard, P.; Stuart, A.D. The Rab11 pathway is required for influenza A virus budding and filament formation. J. Virol 2010, 84, 5848–5859. [Google Scholar]
- Kuroda, T.S.; Itoh, T.; Fukuda, M. Functional analysis of slac2-a/melanophilin as a linker protein between Rab27A and myosin Va in melanosome transport. Methods Enzymol 2005, 403, 419–431. [Google Scholar]
- Westbroek, W.; Lambert, J.; de Schepper, S.; Kleta, R.; van den Bossche, K.; Seabra, M.C.; Huizing, M.; Mommaas, M.; Naeyaert, J.M. Rab27b is up-regulated in human Griscelli syndrome type II melanocytes and linked to the actin cytoskeleton via exon F-Myosin Va transcripts. Pigment Cell Res 2004, 17, 498–505. [Google Scholar]
- Chua, C.E.; Lim, Y.S.; Tang, B.L. Rab35—A vesicular traffic-regulating small GTPase with actin modulating roles. FEBS Lett 2010, 584, 1–6. [Google Scholar]
- Xu, J.; Shi, S.; Matsumoto, N.; Noda, M.; Kitayama, H. Identification of Rgl3 as a potential binding partner for Rap-family small G-proteins and profilin II. Cell Signal 2007, 19, 1575–1582. [Google Scholar]
- Kinashi, T.; Katagiri, K. Regulation of lymphocyte adhesion and migration by the small GTPase Rap1 and its effector molecule, RAPL. Immunol Lett 2004, 93, 1–5. [Google Scholar]
- Kardassis, D.; Murphy, C.; Fotsis, T.; Moustakas, A.; Stournaras, C. Control of transforming growth factor beta signal transduction by small GTPases. FEBS J 2009, 276, 2947–2965. [Google Scholar]
- Fernandes, H.; Franklin, E.; Jollivet, F.; Bliedtner, K.; Khan, A.R. Mapping the interactions between a RUN domain from DENND5/Rab6IP1 and sorting nexin 1. PLoS One 2012, 7, e35637. [Google Scholar]
- Wassmer, T.; Attar, N.; Harterink, M.; van Weering, J.R.; Traer, C.J.; Oakley, J.; Goud, B.; Stephens, D.J.; Verkade, P.; Korswagen, H.C.; Cullen, P.J. The retromer coat complex coordinates endosomal sorting and dynein-mediated transport, with carrier recognition by the trans-Golgi network. Dev. Cell 2009, 17, 110–122. [Google Scholar]
- Cullen, P.J.; Korswagen, H.C. Sorting nexins provide diversity for retromer-dependent trafficking events. Nat. Cell Biol 2011, 14, 29–37. [Google Scholar]
- Fehrenbacher, K.; Huckaba, T.; Yang, H.C.; Boldogh, I.; Pon, L. Actin comet tails, endosomes and endosymbionts. J. Exp. Biol 2003, 206, 1977–1984. [Google Scholar]
- Strick, D.J.; Elferink, L.A. Rab15 effector protein: A novel protein for receptor recycling from the endocytic recycling compartment. Mol. Biol. Cell 2005, 16, 5699–5709. [Google Scholar]
- Maher-Laporte, M.; Berthiaume, F.; Moreau, M.; Julien, L.A.; Lapointe, G.; Mourez, M.; DesGroseillers, L. Molecular composition of staufen2-containing ribonucleoproteins in embryonic rat brain. PLoS One 2010, 5, e11350. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kitagishi, Y.; Matsuda, S. RUFY, Rab and Rap Family Proteins Involved in a Regulation of Cell Polarity and Membrane Trafficking. Int. J. Mol. Sci. 2013, 14, 6487-6498. https://doi.org/10.3390/ijms14036487
Kitagishi Y, Matsuda S. RUFY, Rab and Rap Family Proteins Involved in a Regulation of Cell Polarity and Membrane Trafficking. International Journal of Molecular Sciences. 2013; 14(3):6487-6498. https://doi.org/10.3390/ijms14036487
Chicago/Turabian StyleKitagishi, Yasuko, and Satoru Matsuda. 2013. "RUFY, Rab and Rap Family Proteins Involved in a Regulation of Cell Polarity and Membrane Trafficking" International Journal of Molecular Sciences 14, no. 3: 6487-6498. https://doi.org/10.3390/ijms14036487



