The Influence of Metal Stress on the Availability and Redox State of Ascorbate, and Possible Interference with Its Cellular Functions
Abstract
:1. Introduction
2. Ascorbate Biosynthesis and the Influence of Metal Exposure
2.1. Biosynthesis
2.2. Influence of Metal Exposure on AsA Biosynthesis
3. Interference of Metals with Physiological Functions of AsA
3.1. Growth and Development
3.1.1. Cell Division
3.1.2. Cell Wall Metabolism and Cell Expansion
3.1.3. Senescence
3.2. Photosynthesis
4. Metabolism of AsA
5. Localization of AsA and its Biochemical Reactions
5.1. Subcellular Localization of AsA
5.2. Role of AsA as a Cellular Antioxidant
5.2.1. Protection of AsA against Excess of Non-Essential Metals
5.2.2. Protection of AsA against Excess of Essential Metals
5.3. The Interaction of AsA with the Antioxidant α-Tocopherol
5.4. AsA Oxidation by APx or AO Serves Differential Functional Goals in Plant Cells
5.5. Other Biochemical Reactions
6. Conclusions
Acknowledgments
Conflict of Interest
References
- Islam, E.; Yang, X.; He, Z.; Mahmood, Q. Assessing potential dietary toxicity of heavy metals in selected vegetables and food crops. J. Zhejiang Univ 2007, 8, 1–13. [Google Scholar]
- Sanita, T.L.; Gabbrielli, R. Response to cadmium in higher plants. Environ. Exp. Bot 1999, 41, 105–130. [Google Scholar]
- Nawrot, T.S.; Van Hecke, E.; Thijs, L.; Richart, T.; Kuznetsova, T.; Jin, Y.; Vangronsveld, J.; Roels, H.A.; Staessen, J.A. Cadmium-related mortality and long-term secular trends in the cadmium body burden of an environmentally exposed population. Environ. Health Perspect 2008, 116, 1620–1628. [Google Scholar]
- Lauwerys, R.; Amery, A.; Bernard, A.; Bruaux, P.; Buchet, J.P.; Claeys, F.; De Plaen, P.; Ducoffre, G.; Fagard, R.; Lijnen, P.; et al. Health effects of environmental exposure to cadmium: Objectives, design and organization of the Cadmibel Study: A cross-sectional morbidity study carried out in Belgium from 1985 to 1989. Environ. Health Perspect. 1990, 87, 283–289. [Google Scholar]
- Bernard, A. Cadmium & its adverse effects on human health. Indian J. Med. Res 2008, 128, 557–564. [Google Scholar]
- Cockell, K.A.; Bertinato, J.; L’Abbe, M.R. Regulatory frameworks for copper considering chronic exposures of the population. Am. J. Clin. Nutr 2008, 88, 863S–866S. [Google Scholar]
- Marschner, H. Mineral Nutrition of Higher Plants. In Functions of Mineral Nutrients: Micronutrients, 2nd ed; Marschner, H., Ed.; Academic Press: London, UK, 1995. [Google Scholar]
- Leonard, S.S.; Bower, J.J.; Shi, X. Metal-induced toxicity, carcinogenesis, mechanisms and cellular responses. Mol. Cell. Biochem 2004, 255, 3–10. [Google Scholar]
- Verbruggen, N.; Hermans, C.; Schat, H. Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 2009, 181, 759–776. [Google Scholar]
- Benavides, M.P.; Gallego, S.M.; Tomaro, M.L. Cadmium toxicity in plants. Br. J. Plant Physiol 2005, 17, 21–34. [Google Scholar]
- Cuypers, A.; Smeets, K.; Vangronsveld, J. Heavy Metal Stress in Plants. In Plant stress Biology: From Genomics to Systems Biology; Hirt, H., Ed.; Wiley-VCH: Verlag: Weinheim, Germany, 2009; pp. 161–178. [Google Scholar]
- Gallegoa, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Botany 2012, 83, 33–46. [Google Scholar]
- Cuypers, A.; Remans, T.; Vangronsveld, J.; Smeets, K. Cadmium and Pxidative Stress. In Encyclopedia of Metalloproteins; Kretsinger, R.H., Permyakov, E.A., Uversky, V.M., Eds.; Springer: Berlin, Germany, 2012. [Google Scholar]
- Stoyanova, Z.; Doncheva, S. The effect of zinc supply and succinate treatment on plant growth and mineral uptake in pea plant. Braz. J. Plant Physiol 2002, 14, 111–116. [Google Scholar]
- Yruela, I. Copper in plants. Braz. J. Plant Physiol 2005, 17, 145–156. [Google Scholar]
- Fargasova, A. Phytotoxic effects of Cd, Zn, Pb, Cu and Fe on Sinapis alba L. seedlings and their accumulation in roots and shoots. Biol. Plant 2001, 44, 471–473. [Google Scholar]
- Smeets, K.; Opdenakker, K.; Remans, T.; Van Sanden, S.; Van Belleghem, F.; Semane, B.; Horemans, N.; Guisez, Y.; Vangronsveld, J.; Cuypers, A. Oxidative stress-related responses at transcriptional and enzymatic levels after exposure to Cd or Cu in a multipollution context. J. Plant Physiol 2009, 166, 1982–1992. [Google Scholar]
- Cuypers, A.; Keunen, E.; Bohler, S.; Jozefczak, M.; Opdenakker, K.; Gielen, H.; Vercampt, H.; Bielen, A.; Schellingen, K.; Vangronsveld, J.; et al. Cadmium and Copper Stress Induce a Cellular Oxidative Challenge Leading to Damage Versus Signalling. In Metal toxicity in Plants: Perception, Signaling and Remediation; Gupta, D.K.G., Sandalios, L.M., Eds.; Springer-Verslag GmbH: Berlin, Heidelberg, Germany, 2011; pp. 65–90. [Google Scholar]
- Smeets, K.; Ruytinx, J.; van Belleghem, F.; Semane, B.; Lin, D.; Vangronsveld, J.; Cuypers, A. Critical evaluation and statistical validation of a hydroponic culture system for Arabidopsis thaliana. Plant Physiol. Biochem 2008, 46, 212–218. [Google Scholar]
- Guo, W.J.; Meetam, M.; Goldsbrough, P.B. Examining the specific contributions of individual Arabidopsis metallothioneins to copper distribution and metal tolerance. Plant Physiol 2008, 146, 1697–1706. [Google Scholar]
- Hall, J.L. Cellular mechanisms for heavy metal detoxification and tolerance. J. Exp. Botany 2002, 53, 1–11. [Google Scholar]
- Arora, A.; Sairim, R.K.; Srivastava, G.C. Oxidative stress and antioxidative system in plants. Curr. Sci 2002, 82, 1227–1238. [Google Scholar]
- Halliwell, B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 2006, 141, 312–322. [Google Scholar]
- Maksymiec, W. Signaling responses in plants to heavy metal stress. Acta Physiol. Plant 2007, 29, 177–187. [Google Scholar]
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 2010, 33, 453–467. [Google Scholar]
- Cuypers, A.; Smeets, K.; Ruytinx, J.; Opdenakker, K.; Keunen, E.; Remans, T.; Horemans, N.; Vanhoudt, N.; Van Sanden, S.; Van Belleghem, F.; et al. The cellular redox state as a modulator in cadmium and copper responses in Arabidopsis thaliana seedlings. J. Plant Physiol. 2011, 168, 309–316. [Google Scholar]
- Rizhsky, L.; Hallak-Herr, E.; van Breusegem, F.; Rachmilevitch, S.; Barr, J.E.; Rodermel, S.; Inze, D.; Mittler, R. Double antisense plants lacking ascorbate peroxidase and catalase are less sensitive tot oxidative stress than single antisense plants lacking ascorbate peroxidase or catalase. Plant J 2002, 32, 329–342. [Google Scholar]
- Conklin, P.L.; Barth, C. Ascorbic acid, a familiar small molecule intertwined in the response of plants to ozone, pathogens, and the onset of senescence. Plant Cell Environ 2004, 27, 959–970. [Google Scholar]
- Smirnoff, N.; Wheeler, G.L. Ascorbic acid in plants: Biosynthesis and function. Crit. Rev. Biochem. Mol. Biol 2000, 35, 291–314. [Google Scholar]
- Zhang, Y. Biological Role of Ascorbate in Plants. In Ascorbic Acid in Plant—Biosynthesis, Regulation and Enhancement; Zhang, Y., Ed.; Springer: Wuhan, China, 2013; pp. 7–33. [Google Scholar]
- Ishikawa, T.; Shigeoka, S. Recent advances in ascorbate biosynthesis and the physiological significance of ascorbate peroxidase in photosynthesizing organisms. Biosci. Biotechnol. Biochem 2008, 72, 1143–1154. [Google Scholar]
- Linster, C.L.; Clarke, S.G. l-Ascorbate biosynthesis in higher plants: the role of VTC2. Trends Plant Sci 2008, 13, 567–573. [Google Scholar]
- Valpuesta, V.; Botella, M.A. Biosynthesis of l-ascorbic acid in plants: New pathways for an old antioxidant. Trends Plant Sci 2004, 9, 573–577. [Google Scholar]
- Linster, C.L.; Gomez, T.A.; Christensen, K.C.; Adler, L.N.; Young, B.D.; Brenner, C.; Clarke, S.G. Arabidopsis VTC2 encodes a GDP-l-galactose phosphorylase, the last unknown enzyme in the Smirnoff-Wheeler pathway to ascorbic acid in plants. J. Biol. Chem 2007, 282, 18879–18885. [Google Scholar]
- Wheeler, G.L.; Jones, M.A.; Smirnoff, N. The biosynthetic pathway of vitamin C in higher plants. Nature 1998, 393, 365–369. [Google Scholar]
- Smirnoff, N. Ascorbate biosynthesis and function in photoprotection. Philoso. Trans. Royal Soc. London Series B Biol. Sci 2000, 355, 1455–1464. [Google Scholar]
- Ishikawa, T.; Dowdle, J.; Smirnoff, N. Progress in manipulating ascorbic acid biosynthesis and accumulation in plants. Physiol. Plant 2006, 126, 343–355. [Google Scholar]
- Conklin, P.L.; Norris, S.R.; Wheeler, G.L.; Williams, E.H.; Smirnoff, N.; Last, R.L. Genetic evidence for the role of GDP-mannose in plant ascorbic acid (vitamin C) biosynthesis. Proc. Natl. Acad. Sci. USA 1999, 96, 4198–4203. [Google Scholar]
- Smirnoff, N. Ascorbic acid: Metabolism and functions of a multi-facetted molecule. Curr. Opin. Plant Biol 2000, 3, 229–235. [Google Scholar]
- Dowdle, J.; Ishikawa, T.; Gatzek, S.; Rolinski, S.; Smirnoff, N. Two genes in Arabidopsis thaliana encoding GDP-l-galactose phosphorylase are required for ascorbate biosynthesis and seedling viability. Plant J 2007, 52, 673–689. [Google Scholar]
- Laing, W.A.; Wright, M.A.; Cooney, J.; Bulley, S.M. The missing step of the l-galactose pathway of ascorbate biosynthesis in plants, an l-galactose guanyltransferase, increases leaf ascorbate content. Proc. Natl. Acad. Sci. USA 2007, 104, 9534–9539. [Google Scholar]
- Conklin, P.L.; Gatzek, S.; Wheeler, G.L.; Dowdle, J.; Raymond, M.J.; Rolinski, S.; Isupov, M.; Littlechild, J.A.; Smirnoff, N. Arabidopsis thaliana VTC4 encodes l-galactose-1-P phosphatase, a plant ascorbic acid biosynthetic enzyme. J. Biol. Chem 2006, 281, 15662–15670. [Google Scholar]
- Loscos, J.; Matamoros, M.A.; Becana, M. Ascorbate and homoglutathione metabolism in common bean nodules under stress conditions and during natural senescence. Plant Physiol 2008, 146, 1282–1292. [Google Scholar]
- Tamaoki, M.; Mukai, F.; Asai, N.; Nakajima, N.; Kubo, A.; Aono, M.; Saji, H. Light-controlled expression of a gene encoding l-galactono-g-lactone dehydrogenase which affects ascorbate pool size in Arabidopsis thaliana. Plant Sci 2003, 164, 1111–1117. [Google Scholar]
- Keunen, E.; Remans, T.; Opdenakker, K.; Jozefczak, M.; Gielen, H.; Guisez, Y.; Vangronsveld, J.; Cuypers, A. A mutant of the Arabidopsis thaliana LIPOXYGENASE1 gene shows altered signalling and oxidative stress related responses after cadmium exposure. Plant Physiol. Biochem 2013, 63, 272–280. [Google Scholar]
- Smeets, K.; Cuypers, A.; Lambrechts, A.; Semane, B.; Hoet, P.; Van Laere, A.; Vangronsveld, J. Induction of oxidative stress and antioxidative mechanisms in Phaseolus vulgaris after Cd application. Plant Physiol. Biochem 2005, 43, 437–444. [Google Scholar]
- Schutzendubel, A.; Schwanz, P.; Teichmann, T.; Gross, K.; Langenfeld-Heyser, R.; Godbold, D.L.; Polle, A. Cadmium-induced changes in antioxidative systems, hydrogen peroxide content, and differentiation in Scots pine roots. Plant Physiol 2001, 127, 887–898. [Google Scholar]
- Aravind, P.; Prasad, M.N. Modulation of cadmium-induced oxidative stress in Ceratophyllum demersum by zinc involves ascorbate-glutathione cycle and glutathione metabolism. Plant Physiol. Biochem 2005, 43, 107–116. [Google Scholar]
- Mohamed, A.A.; Castagna, A.; Ranieri, A.; Sanità di Toppi, L. Cadmium tolerance in Brassica juncea roots and shoots is affected by antioxidant status and phytochelatin biosynthesis. Plant Physiol. Biochem 2012, 57, 15–22. [Google Scholar]
- Liu, Y.; Wang, X.; Zeng, G.; Qu, D.; Gu, J.; Zhou, M.; Chai, L. Cadmium-induced oxidative stress and response of the ascorbate-glutathione cycle in Bechmeria nivea (L.) Gaud. Chemosphere 2007, 69, 99–107. [Google Scholar]
- Pant, P.P.; Tripathi, A.K.; Dwivedi, V. Effect of heavy metals on some biochemical parameters of sal (Shorea robusta) seedling at nursery level, Doon Valley, India. J. Agr. Sci 2011, 2, 45–51. [Google Scholar]
- Sharma, P.; Dubey, R.S. Involvement of oxidative stress and role of antioxidative defense system in growing rice seedlings exposed to toxic concentrations of aluminum. Plant Cell Rep 2007, 26, 2027–2038. [Google Scholar]
- Yin, L.; Wang, S.; Eltayeb, A.E.; Uddin, M.I.; Yamamoto, Y.; Tsuji, W.; Takeuchi, Y.; Tanaka, K. Overexpression of dehydroascorbate reductase, but not monodehydroascorbate reductase, confers tolerance to aluminum stress in transgenic tobacco. Planta 2010, 231, 609–621. [Google Scholar]
- Drazkiewicz, M.; Skótzynska-Polit, E.; Krupa, Z. Response of the ascorbate-glutathione cycle to excess copper in Arabidopsis thaliana (L.). Plant Sci 2003, 164, 195–202. [Google Scholar]
- Gupta, M.; Cuypers, A.; Vangronsveld, J.; Clijsters, H. Copper affects the enzymes of the ascorbate-glutathione cycle and its related metabolites in the roots of Phaseolus vulgaris. Physiol. Plant 1999, 106, 262–267. [Google Scholar]
- Cuypers, A.; Vangronsveld, J.; Clijsters, H. Biphasic effect of copper on the ascorbate-glutathione pathway in primary leaves of Phaseolus vulgaris seedlings during the early stages of metal assimilation. Physiol. Plant 2000, 110, 512–517. [Google Scholar]
- Thounaojam, T.C.; Panda, P.; Mazumdar, P.; Kumar, D.; Sharma, G.D.; Sahoo, L.; Panda, S.K. Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol. Biochem 2012, 53, 33–39. [Google Scholar]
- Tewari, R.K.; Kumar, P.; Sharma, P.N. Antioxidant responses to enhanced generation of superoxide anion radical and hydrogen peroxide in the copper-stressed mulberry plants. Planta 2006, 223, 1145–1153. [Google Scholar]
- Demirevska-Kepova, K.; Simova-Stoilova, L.; Stoyanova, Z.; Hölzer, R.; Feller, U. Biochemical changes in barley plants after excessive supply of copper and manganese. Environ. Exp. Bot 2004, 52, 253–266. [Google Scholar]
- Cuypers, A.; Vangronsveld, J.; Clijsters, H. The redox status of plant cells (AsA and GSH) is sensitive to zinc imposed oxidative stress in roots and primary leaves of Phaseolus vulgaris. Plant Physiol. Biochem 2001, 39, 657–664. [Google Scholar]
- Rao, K.V.M.; Sresty, T.V.S. Antioxidative parameters in the seedlings of pigeonpea (Cajanus cajan (L.) Millspaugh) in response to Zn and Ni stresses. Plant Sci 2000, 157, 113–128. [Google Scholar]
- Prasad, K.V.S.K.; Saradhi, P.P.; Sharmila, P. Concerted action of antioxidant enzymes and curtailed growth under zinc toxicity in Brassica juncea. Environ. Exp. Bot 1999, 42, 1–10. [Google Scholar]
- Maheshwari, R.; Dubey, R.S. Nickel-induced oxidative stress and the role of antioxidant defence in rice seedlings. Plant Growth Regul 2009, 59, 37–49. [Google Scholar]
- Fecht-Christoffers, M.M.; Maier, P.; Horst, W.J. Apoplastic peroxidases and ascorbate are involved in manganese toxicity and tolerance of Vigna unguiculata. Physiol. Plant 2003, 117, 237–244. [Google Scholar]
- Srivastava, S.; Dubey, R.S. Manganese-excess induces oxidative stress, lowers the pool of antioxidants and elevates activities of key antioxidative enzymes in rice seedlings. Plant Growth Regul 2011, 64, 1–16. [Google Scholar]
- Wolucka, B.A.; Van Montagu, M. GDP-mannose 3′,5′-epimerase forms GDP-l-gulose, a putative intermediate for the de novo biosynthesis of vitamin C in plants. J. Biol. Chem 2003, 278, 47483–47490. [Google Scholar]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci 2004, 9, 490–498. [Google Scholar]
- Veljovic-Jovanovic, S.D.; Pignocchi, C.; Noctor, G.; Foyer, C.H. Low ascorbic acid in the vtc-1 mutant of Arabidopsis is associated with decreased growth and intracellular redistribution of the antioxidant system. Plant Physiol 2001, 127, 426–435. [Google Scholar]
- Kerk, N.M.; Feldman, L.J. A biochemical-model for the initiation and maintenance of the quiescent center—Implications for organization of root-meristems. Development 1995, 121, 2825–2833. [Google Scholar]
- Potters, G.; Horemans, N.; Caubergs, R.J.; Asard, H. Ascorbate and dehydroascorbate influence cell cycle progression in a tobacco cell suspension. Plant Physiol 2000, 124, 17–20. [Google Scholar]
- Tabata, K.; Oba, K.; Suzuki, K.; Esaka, M. Generation and properties of ascorbic aciddeficient transgenic tobacco cells expressing antisense RNA for l-galactono-1,4-lactone dehydrogenase. Plant J 2001, 27, 139–148. [Google Scholar]
- Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Mendez, A.A.; Gallego, S.M. Oxidative post translational modifications of proteins related to cell cycle are involved in cadmium toxicity in wheat seedlings. Plant Sci 2012, 196, 1–7. [Google Scholar]
- Kozhevnikova, A.D.; Seregin, I.V.; Bystrova, E.I.; Belyaeva, A.I.; Kataeva, M.N.; Ivanov, V.B. The effects of lead, nickel, and strontium nitrates on cell division and elongation in maize roots. Russ. J. Plant Physl 2009, 56, 242–250. [Google Scholar]
- Radha, J.; Srivastava, S.; Solomon, S.; Shrivastava, A.K.; Chandra, A. Impact of excess zinc on growth parameters, cell division, nutrient accumulation, photosynthetic pigments and oxidative stress of sugarcane (Saccharum spp.). Acta Physiol. Plant 2010, 32, 979–986. [Google Scholar]
- Davey, M.W.; Van Montag, M.; Inzé, D.; Sanmartin, M.; Kanellis, A.; Smirnoff, N.; Benzie, I.J.J.; Strain, J.J.; Favell, D.; Fletcher, J. Plant l-ascorbic acid: Chemistry, function, metabolism, bioavailability and effects of processing. J. Sci. Food Agr 2000, 80, 825–860. [Google Scholar]
- Smirnoff, N. The function and metabolism of ascorbic acid in plants. Ann. Bot 1996, 78, 661–669. [Google Scholar]
- Torabinejad, J.; Donahue, J.L.; Gunesekera, B.N.; Allen-Daniels, M.J.; Gillaspy, G.E. VTC4 is a bifunctional enzyme that affects myoinositol and ascorbate biosynthesis in plants. Plant Physiol 2009, 150, 951–961. [Google Scholar]
- Alhagdow, M.; Mounet, F.; Gilbert, L.; Nunes-Nesi, A.; Garcia, V.; Just, D.; Petit, J.; Beauvoit, B.; Fernie, A.R.; Rothan, C.; Baldet, P. Silencing of the mitochondrial ascorbate synthesizing enzyme l-galactono-1,4-lactone dehydrogenase affects plant and fruit development in tomato. Plant Physiol 2007, 145, 1408–1422. [Google Scholar]
- Cuypers, A.; Vangronsveld, J.; Clijsters, H. Peroxidases in roots and primary leaves of Phaseolus vulgaris. Copper and zinc phytotoxicity: A comparison. J. Plant Physiol 2002, 159, 869–876. [Google Scholar]
- Chaoui, A.; Jarrar, B.; Ferjani, E.E. Effects of cadmium and copper on peroxidase, NADH oxidase and IAA oxidase activities in cell wall, soluble and microsomal membrane fractions of pea roots. J. Plant Physiol 2004, 161, 1225–1234. [Google Scholar]
- Lin, C.C.; Chen, L.M.; Liu, Z.H. Rapid effect of copper on lignin biosynthesis in soybean roots. Plant Sci 2005, 168, 855–861. [Google Scholar]
- Yang, Y.J.; Cheng, L.M.; Liu, Z.H. Rapid effect of cadmium on lignin biosynthesis in soybean roots. Plant Sci 2007, 172, 632–639. [Google Scholar]
- Kovácik, J.; Backor, M.; Kaduková, J. Physiological responses of Matricaria chamomilla to cadmium and copper excess. Environ. Toxicol 2008, 23, 123–130. [Google Scholar]
- Finger-Teixeira, A.; de Lourdes Lucio Ferrarese, M.; Soares, A.R.; da Silva, D.; Ferrarese-Filho, O. Cadmium-induced lignifications restricts soybean root growth. Ecotox. Environ. Safe 2010, 73, 1959–1964. [Google Scholar]
- Tamás, L.; Mistrík, I.; Huttová, J.; Halusková, L.; Valentovicová, K.; Zelinova, V. Role of reactive oxygen species-generating enzymes and hydrogen peroxide during cadmium, mercury and osmotic stresses in barley root tip. Planta 2010, 231, 221–231. [Google Scholar]
- Zimmermann, P.; Zentgraf, U. The correlation between oxidative stress and leaf senescence during plant development. Cell. Mol. Biol. Lett 2005, 10, 515–534. [Google Scholar]
- Barth, C.; Moeder, W.; Klessig, D.F.; Conklin, P.L. The timing of senescence and response to pathogens is altered in the ascorbate-deficient Arabidopsis mutant vitamin c-1. Plant Physiol 2004, 134, 1784–1792. [Google Scholar]
- Barth, C.; De Tullio, M.; Conklin, P.L. The role of ascorbic acid in the control of flowering time and the onset of senescence. J. Exp. Bot 2006, 57, 1657–1665. [Google Scholar]
- Distefano, S.; Palma, J.M.; McCarthy, I.; del Río, L.A. Proteolytic cleavage of plant proteins by peroxisomal endoproteases from senescent pea leaves. Planta 1999, 209, 308–313. [Google Scholar]
- McCarthy, I.; Romero-Puertas, M.C.; Palma, J.M.; Sandalio, L.M.; Corpas, F.J.; Gómez, M.; del Rio, L.A. Cadmium induces senescence symptoms in leaf peroxisomes of pea plants. Plant Cell Environ 2001, 24, 1065–1073. [Google Scholar]
- Chaffei, C.; Pageau, K.; Suzuki, A.; Gouia, H.; Ghorbel, H.M.; Mascalaux-Daubresse, C. Cadmium toxicity induced changes in nitrogen management in Lycopersicon esculentum leading to a metabolic safeguard through an amino acid storage strategy. Plant Cell Physiol 2004, 45, 1681–1693. [Google Scholar]
- Mazid, M.; Khan, T.A.; Khan, Z.H.; Quddusi, S.; Mohammad, F. Occurence, biosynthesis, and potentialities of ascorbic acid in plants. Int. J. Plant Anim. Environ. Sci 2011, 1, 167–184. [Google Scholar]
- Arvidsson, P.O.; Bratt, C.E.; Carlsson, M.; Akerlund, H.E. Purification and identification of the violaxanthin deepoxidase as a 43 kDa protein. Photosynth. Res 1996, 49, 119–129. [Google Scholar]
- Liu, Y.H.; Yu, L.; Wang, R.Z. Level of ascorbic acid in transgenic rice for l-galactono-1,4-lactone dehydrogenase overexpressing or suppressed is associated with plant growth and seed set. Acta Physiol. Plant 2011, 33, 1353–1363. [Google Scholar]
- Prasad, M.N.; Strzalka, K. Heavy Metal Influence on the Light Phase of Photosynthesis. In Physiology and Biochemistry of Metal Toxicity and Tolerance in Plants; Mysliwa-Kurdziel, B., Prasad, M.N., Strzalka, K., Eds.; Kluwer Academic Publishers: Dordrecht, The Nederlanths, 2002; pp. 229–250. [Google Scholar]
- Kucera, T.; Horakova, H.; Sonska, A. Toxic metal ions in photoautotrophic organisms. Photosynthetica 2008, 46, 481–489. [Google Scholar]
- Ding, B.; Shi, G.; Xu, Y.; Hu, J.; Xu, Q. Physiological responses of Alternanthera philoxeroides (Mart.) Griseb leaves to cadmium stress. Environ. Pollut 2007, 147, 800–803. [Google Scholar]
- Anjum, N.A.; Umar, S.; Ahmad, A.; Iqbal, M.; Khan, N.A. Sulphur protects mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul 2008, 54, 271–279. [Google Scholar]
- Ekmekci, Y.; Tanyolac, D.; Ayhan, B. Effects of cadmium on antioxidant enzyme and photosynthetic activities in leaves of two maize cultivars. J. Plant Physiol 2008, 165, 600–611. [Google Scholar]
- He, J.Y.; Ren, Y.F.; Zhu, C.; Yan, Y.P.; Jiang, D.A. Effect of Cd on growth, photosynthetic gas exchange, and chlorophyll fluorescence of wild and Cd-sensitive mutant rice. Photosynthetica 2008, 46, 466–470. [Google Scholar]
- Debolt, S.; Melino, V.; Ford, C.M. Ascorbate as a biosynthetic precursor in plants. Ann. Bot 2007, 99, 3–8. [Google Scholar]
- Debolt, S.; Hardie, W.J.; Tyerman, S.; Ford, C.M. Composition and synthesis of raphide crystals and druse crystals in berries of Vitis vinifera L. cv. Cabernet Sauvignon: ascorbic acid as precursor for both oxalic and tartaric acids as revealed by radiolabelling studies. Aust. J. Grape Wine R 2004, 10, 134–142. [Google Scholar]
- Loewus, F.A. Biosynthesis and metabolism of ascorbic acid in plants and of analogs of ascorbic acid in fungi. Phytochemistry 1999, 52, 193–210. [Google Scholar]
- El-Jaoual, T.; Cox, D.A. Manganese toxicity in plants. J. Plant Nutr 1998, 21, 353–386. [Google Scholar]
- González, A.; Lynch, J. Subcellular and tissue Mn compartmentation in bean leaves under Mn toxicity stress. Aust. J. Plant Physiol 1999, 26, 811–822. [Google Scholar]
- González, A.; Steffen, K.L.; Lynch, J. Light and excess manganese. Implications for oxidative stress in common bean. Plant Physiol 1998, 118, 493–504. [Google Scholar]
- Zeng, F.; Zhou, W.; Qiu, B.; Ali, S.; Wu, F.; Zhang, G. Subcellular distribution and chemical forms of chromium in rice plants suffering from different levels of chromium toxicity. J. Plant Nutr. Soil Sci 2011, 174, 249–256. [Google Scholar]
- Zhang, X.H.; Liu, J.; Huang, H.T.; Chen, J.; Zhu, Y.N.; Wang, D.Q. Chromium accumulation by the hyperaccumulator plant Leersia hexandra Swartz. Chemosphere 2007, 67, 1138–1143. [Google Scholar]
- Lytle, C.M.; Qian, J.H.; Hansen, D.; Zayed, A.; Terry, N.; Lytle, F.W.; Yang, N. Reduction of Cr(VI) to Cr(III) by wetland plants: Potential for in situ heavy metal detoxification. Envir. Sci. Tech 1998, 32, 3087–3093. [Google Scholar]
- Zechmann, B.; Stumpe, M.; Mauch, F. Immunocytochemical determination of the subcellular distribution of ascorbate in plants. Planta 2011, 233, 1–12. [Google Scholar]
- Takahama, U. Oxidation of vacuolar and apoplastic phenolic substrates by peroxidase: Physiological significance of the oxidation reactions. Phytochem. Rev 2004, 3, 207–219. [Google Scholar]
- Green, R.M.; Graham, M.; O’Donovan, M.R.; Chipman, J.K.; Hodges, N.J. Subcellular compartmentalization of glutathione: Correlations with parameters of oxidative stress related to genotoxicity. Mutagenesis 2006, 21, 383–390. [Google Scholar]
- Pignocchi, C.; Foyer, C.H. Apoplastic ascorbate metabolism and its role in the regulation of cell signalling. Curr. Opin. Plant Biol 2003, 6, 379–389. [Google Scholar]
- Hong-Bo, S.; Li-ye, C.; Zhao-Hua, L.; Cong-Min, K. Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. Int. J. Biol. Sci 2008, 4, 8–14. [Google Scholar]
- Suza, W.P.; Avila, C.A.; Carruthers, K.; Kulkarni, S.; Goggin, F.L.; Lorence, A. Exploring the impact of wounding and jasmonates on ascorbate metabolism. Plant Physiol. Biochem 2010, 48, 337–350. [Google Scholar]
- Foyer, C.H.; Noctor, G. Redox homeostasis and antioxidant signaling: A metabolic interface between stress perception and physiological responses. The Plant Cell 2005, 17, 1866–1875. [Google Scholar]
- Foyer, C.H.; Noctor, G. Redox sensing and signaling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Physiol. Plant 2003, 119, 355–364. [Google Scholar]
- Foyer, C.H.; Noctor, G. Oxidant and antioxidant signaling in plants: A re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ 2005, 28, 1056–1071. [Google Scholar]
- Asada, K. The water-water cycle in Chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu Rev. Plant Phys 1999, 50, 601–639. [Google Scholar]
- Koussevitzky, S.; Suzuki, N.; Huntington, S.; Armijo, L.; Sha, W.; Cortes, D.; Shulaev, V.; Mittler, R. Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. J. Biol. Chem 2008, 283, 34197–34203. [Google Scholar]
- Dehghan, G.; Rezazadeh, L.; Habibi, G. Exogenous ascorbate improves antioxidant defense system and induces salinity tolerance in soybean seedlings. Acta Biol. Szeg 2011, 55, 261–264. [Google Scholar]
- Khan, T.A.; Mazid, M.; Mohammad, F. Role of ascorbic acid against pathogenesis in plants. J. Stress Physiol. Biochem 2011, 7, 222–234. [Google Scholar]
- Conklin, P.L.; Williams, E.H.; Last, R.L. Environmental stress sensitivity of an ascorbic acid-deficient Arabidopsis mutant. Proc. Natl. Acad. Sci. USA 1996, 93, 9970–9974. [Google Scholar]
- Karpinski, S.; Reynolds, H.; Karpinska, B.; Wingsle, G.; Creissen, G.; Mullineaux, P. Systemic signaling and acclimation in response to excess excitation energy in Arabidopsis. Science 1999, 284, 654–657. [Google Scholar]
- Prasad, T.K.; Anderson, M.D.; Martin, B.A.; Steward, C.R. Evidence for chilling-induced oxidative stress in maize seedlings and a regulatory role for hydrogen peroxide. Plant Cell 1994, 6, 65–74. [Google Scholar]
- Sun, X.; Zhang, J.; Zhang, H.; Ni, Y.; Zhang, Q.; Chen, J.; Guan, Y. The responses of Arabidopsis thaliana to cadmium exposure explored via metabolite profiling. Chemosphere 2010, 78, 840–845. [Google Scholar]
- Munné-Bosch, S.; Alegre, L. Drought-induced changes in the redox state of alfa-tocopherol, ascorbate, and the diterpene carnosic acid in chloroplasts of labiatae species differing in carnosic acid contents. Plant Physiol 2003, 131, 1816–1825. [Google Scholar]
- Simontacchi, M.; Caro, A.; Fraga, C.G.; Puntarulo, S. Oxidative stress affects a-tocopherol content in soybean embryonic axes upon imbibition and following germination. Plant Physiol 1993, 103, 949–953. [Google Scholar]
- Yusuf, M.A.; Kumar, D.; Rajwanshi, R.; Strasser, R.J.; Tsimilli-Michael, M.; Govindjee Sarin, N.B. Overexpression of γ-tocopherol methyl transferase gene in transgenic Brassica juncea plants alleviates abiotic stress: Physiological and chlorophyll a fluorescence measurements. Biochim. Biophys. Acta 2010, 1797, 1428–1438. [Google Scholar]
- Karpinski, S.; Escobar, C.; Karpinska, B.; Creissen, G.; Mullineaux, P.M. Photosynthetic electron transport regulates the expression of cytosolic ascorbate peroxidase genes in Arabidopsis during excess light stress. Plant Cell 1997, 9, 627–640. [Google Scholar]
- Keunen, E.; Remans, T.; Bohler, S.; Vangronsveld, J.; Cuypers, A. Metal-induced oxidative stress and plant mitochondria. Int. J. Mol. Sci 2011, 12, 6894–6918. [Google Scholar]
- Kato, N.; Esaka, M. Changes in ascorbate oxidase gene expression and ascorbate levels in cell division and cell elongation in tobacco cells. Physiol. Plant 1999, 105, 321–329. [Google Scholar]
- Pignocchi, C.; Fletcher, J.M.; Wilkinson, J.E.; Barnes, J.D.; Foyer, C.H. The function of ascorbate oxidase in tobacco. Plant Physiol 2003, 132, 1631–1641. [Google Scholar]
- Pignocchi, C.; Kiddle, G.; Hernandez, I.; Foster, S.J.; Asensi, A.; Taybi, T.; Barnes, J.; Foyer, C.H. Ascorbate oxidase-dependent changes in the redox state of the apoplast modulate gene transcript accumulation leading to modified hormone signaling and orchestration of defense processes in tobacco. Plant Physiol 2006, 141, 423–435. [Google Scholar]
- Fotopoulos, V.; Sanmartin, M.; Kanellis, A.K. Effect of ascorbate oxidase over-expression on ascorbate recycling gene expression in response to agents imposing oxidative stress. J. Exp. Bot 2006, 57, 3933–3943. [Google Scholar]
 ) or through direct binding to ROS and produce MDHA. The other secondary manner is to regenerate antioxidants such as α-tocopherol and zeaxanthin. Abbrevations: d-gluc, d-glucose; d-gluc-6P, d-glucose-6-P; d-fruc-6P, d-fructose-6-P; d-man-6P, d-mannose-6-P; d-man-1P, d-mannose-1-P; GDP-man, GDP-d-mannose; GDP-gal, GDP-l-galactose; l-gal-1P, l-galactose-1-P; l-gal, l-galactose; l-gal-1,4-lac, l-galactono-1,4-lactone; PGI, phosphoglucose isomerase; PMI, phosphomannose isomeras; PMM, phosphomannomutase; GMP, GDP-mannose-pyrophosphorylase, GME, GDP-mannose-3′,5′-epimerase; GLGalPP, GDP-l-galactose phosphorylase; GalPP, l-galactose-1-P-phosphatase; GalDH, l-galactose dehydrogenase; GalLDH, l-galactone-1,4-lactone dehydrogenase; AsA, ascorbate; DHA, dehydroascorbate; MDHA, monodehydroascorbate; APx, ascorbate peroxidase; MDHAR, monodehydroascorbate reductase; DHAR, dehydroascorbate reductase; GSH, glutathione; GSSG, oxidized glutathione; GR, glutathione reductase; α-toc, α-tocopherol; VDE, violaxanthin de-epoxidase; Fd, ferredoxin; Cyt b, cytochrome b.
) or through direct binding to ROS and produce MDHA. The other secondary manner is to regenerate antioxidants such as α-tocopherol and zeaxanthin. Abbrevations: d-gluc, d-glucose; d-gluc-6P, d-glucose-6-P; d-fruc-6P, d-fructose-6-P; d-man-6P, d-mannose-6-P; d-man-1P, d-mannose-1-P; GDP-man, GDP-d-mannose; GDP-gal, GDP-l-galactose; l-gal-1P, l-galactose-1-P; l-gal, l-galactose; l-gal-1,4-lac, l-galactono-1,4-lactone; PGI, phosphoglucose isomerase; PMI, phosphomannose isomeras; PMM, phosphomannomutase; GMP, GDP-mannose-pyrophosphorylase, GME, GDP-mannose-3′,5′-epimerase; GLGalPP, GDP-l-galactose phosphorylase; GalPP, l-galactose-1-P-phosphatase; GalDH, l-galactose dehydrogenase; GalLDH, l-galactone-1,4-lactone dehydrogenase; AsA, ascorbate; DHA, dehydroascorbate; MDHA, monodehydroascorbate; APx, ascorbate peroxidase; MDHAR, monodehydroascorbate reductase; DHAR, dehydroascorbate reductase; GSH, glutathione; GSSG, oxidized glutathione; GR, glutathione reductase; α-toc, α-tocopherol; VDE, violaxanthin de-epoxidase; Fd, ferredoxin; Cyt b, cytochrome b.
 ) or through direct binding to ROS and produce MDHA. The other secondary manner is to regenerate antioxidants such as α-tocopherol and zeaxanthin. Abbrevations: d-gluc, d-glucose; d-gluc-6P, d-glucose-6-P; d-fruc-6P, d-fructose-6-P; d-man-6P, d-mannose-6-P; d-man-1P, d-mannose-1-P; GDP-man, GDP-d-mannose; GDP-gal, GDP-l-galactose; l-gal-1P, l-galactose-1-P; l-gal, l-galactose; l-gal-1,4-lac, l-galactono-1,4-lactone; PGI, phosphoglucose isomerase; PMI, phosphomannose isomeras; PMM, phosphomannomutase; GMP, GDP-mannose-pyrophosphorylase, GME, GDP-mannose-3′,5′-epimerase; GLGalPP, GDP-l-galactose phosphorylase; GalPP, l-galactose-1-P-phosphatase; GalDH, l-galactose dehydrogenase; GalLDH, l-galactone-1,4-lactone dehydrogenase; AsA, ascorbate; DHA, dehydroascorbate; MDHA, monodehydroascorbate; APx, ascorbate peroxidase; MDHAR, monodehydroascorbate reductase; DHAR, dehydroascorbate reductase; GSH, glutathione; GSSG, oxidized glutathione; GR, glutathione reductase; α-toc, α-tocopherol; VDE, violaxanthin de-epoxidase; Fd, ferredoxin; Cyt b, cytochrome b.
) or through direct binding to ROS and produce MDHA. The other secondary manner is to regenerate antioxidants such as α-tocopherol and zeaxanthin. Abbrevations: d-gluc, d-glucose; d-gluc-6P, d-glucose-6-P; d-fruc-6P, d-fructose-6-P; d-man-6P, d-mannose-6-P; d-man-1P, d-mannose-1-P; GDP-man, GDP-d-mannose; GDP-gal, GDP-l-galactose; l-gal-1P, l-galactose-1-P; l-gal, l-galactose; l-gal-1,4-lac, l-galactono-1,4-lactone; PGI, phosphoglucose isomerase; PMI, phosphomannose isomeras; PMM, phosphomannomutase; GMP, GDP-mannose-pyrophosphorylase, GME, GDP-mannose-3′,5′-epimerase; GLGalPP, GDP-l-galactose phosphorylase; GalPP, l-galactose-1-P-phosphatase; GalDH, l-galactose dehydrogenase; GalLDH, l-galactone-1,4-lactone dehydrogenase; AsA, ascorbate; DHA, dehydroascorbate; MDHA, monodehydroascorbate; APx, ascorbate peroxidase; MDHAR, monodehydroascorbate reductase; DHAR, dehydroascorbate reductase; GSH, glutathione; GSSG, oxidized glutathione; GR, glutathione reductase; α-toc, α-tocopherol; VDE, violaxanthin de-epoxidase; Fd, ferredoxin; Cyt b, cytochrome b.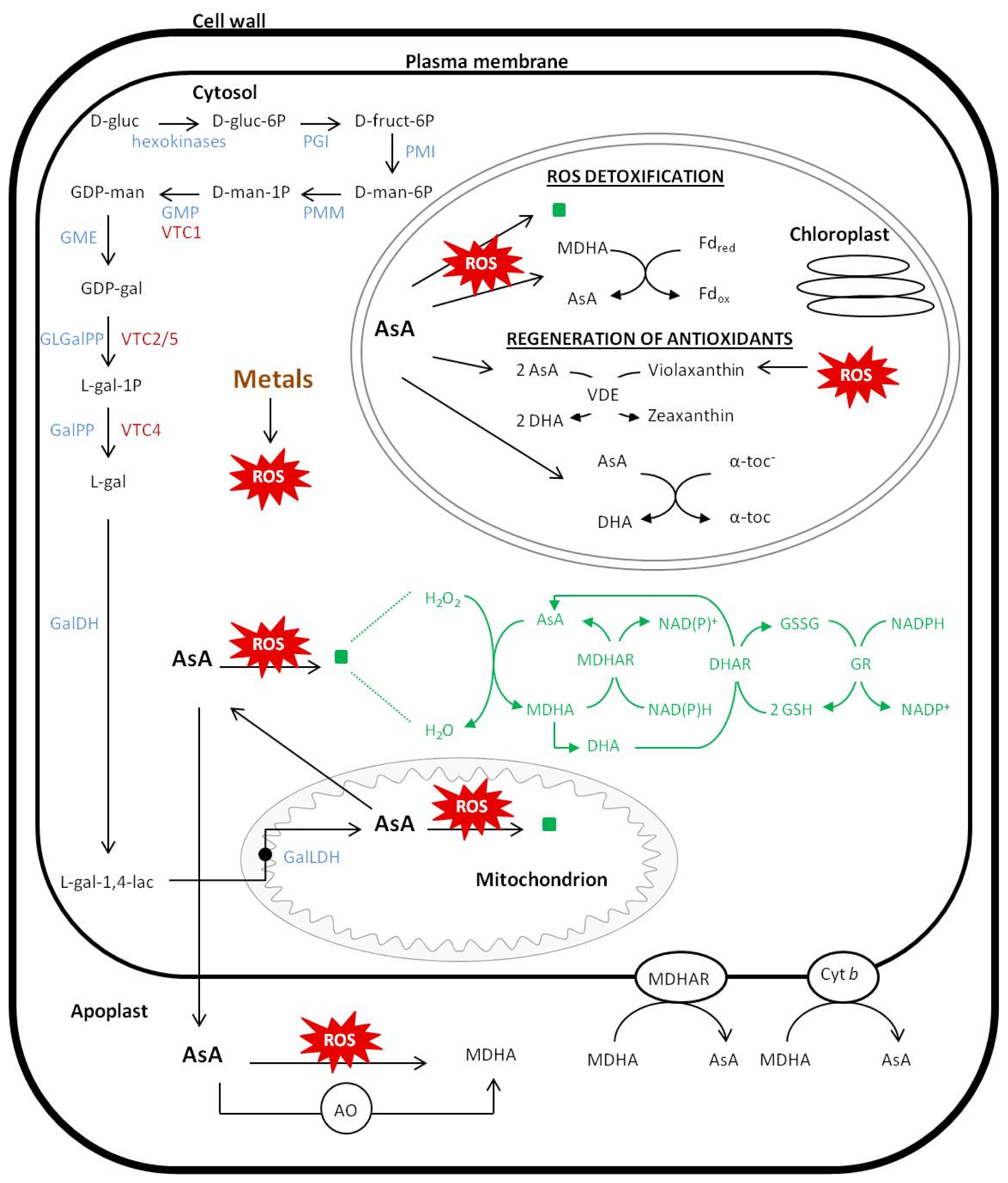
| Plant | Condition | Biosynthesis/content of total AsA | Antioxidant | Ref. | ||
|---|---|---|---|---|---|---|
| Ratios | AsA-GSH cycle | |||||
| NON-ESSENTIAL ELEMENTS | ||||||
| Cadmium (Cd) | Arabidopsis thaliana | 0, 5, 10 μM CdSO4 24 h | ↑ Total AsA (roots) | ↑ APX, GR (leaves) | [26] | |
| Arabidopsis thaliana | 0, 5, 10 μM CdSO4 24 h | ↑ Total AsA (leaves) | ↑ Reduced AsA (leaves) ↓ DHA/AsA (leaves) | [45] | ||
| Arabidopsis thaliana | 10 μM CdSO4 24 h | ↑ Total AsA (roots) | ↑ Reduced AsA (roots) ↓ DHA/AsA (n.s.) | ↑ APX, GR (leaves) | [17] | |
| Phaseolus vulgaris (leaves) | 2 μM CdSO4 0, 24, 48, 72 h | ↑ Total AsA | Long term (>48 h) ↑ DHA Long term (>48 h) ↑ DHA/AsA | ↑ APX, GR | [46] | |
| Phaseolus vulgaris | 0, 100 μM CdCl2 4 days | ↑ mRNA levels of GMP, GME, GalDH, GalLDH | [43] | |||
| Pinus sylvestris | 0, 5, 50 μM CdSO4 6, 12, 24, 48, 96 h | Transient ↑ total AsA followed by ↓ (50 μM Cd) | Transient ↑of reduced AsA followed by ↓ (>24 h) | ↑ MDHAR Ttransient ↓ APX followed by ↑ APX (>24 h) | [47] | |
| Ceratophyllum demersum | 10 μM CdCl2 1 week | ↓ Total AsA | ↓ Reduced AsA DHA > reduced AsA ↑ DHA/AsA | ↑ APX (2x) ↓ MDHAR, DHAR | [48] | |
| Brassica juncea (shoots) | 0, 50, 200 μM Cd(NO3)2 7 days | ↓ Total AsA ( <controls) (200 μM Cd) | ↑ Reduced AsA (50 μM Cd) ↓ Reduced AsA ( <controls) (200 μM Cd) | [49] | ||
| Cadmium (Cd) | Brassica juncea (roots) | 0, 50, 200 μM Cd(NO3)2 7 days | ↑ Total AsA (50 μM Cd) ↓ Total AsA ( >controls) (200 μM Cd) | ↑ Reduced AsA (50 μM Cd) ↓ Reduced AsA ( >controls) (200 μM Cd) | [49] | |
| Bechmeria nivea Gaud | 0, 1, 3, 7 mg/L Cd 1, 2, 3, 7, 10 days | ↑ Reduced AsA followed by ↓ at high Cd concentration ↑ DHA ↑ DHA/AsA | ↑ APX, GR | [50] | ||
| Shorea robusta | 1 mg/L, 5mg/L, 10 mg/L CdCl2 4 months | ↑ AsA | [51] | |||
| Aluminum (Al) | Oryza Sativa | 0, 80, 160 μM Al2(SO4)3 0, 5, 10, 15, 20 days | ↓ Total AsA | ↓ Reduced AsA ↑ DHA/AsA | ↑ APX, MDHAR, DHAR, GR | [52] |
| Nicotiana tabacum SR-1 wildtype | 0, 400 μM AlCl3 24 h | ↑ Total AsA | ↑ DHA ↑ DHA/AsA | ↑ APX | [53] | |
| Nicotiana tabacum overexpressing Arabidopsis thaliana cytosolic DHAR (DHAR-OX) | 0, 400 μM AlCl3 24 h | ↑ Total AsA | ↑ Reduced AsA ↓ DHA ↓ DHA/AsA | ↑ APX | [53] | |
| Arsenic (As) | Shorea robusta | 1 mg/L, 5mg/L, 10 mg/L As2O3 4 months | ↑ AsA | [51] | ||
| Lead (Pb) | Shorea robusta | 1 mg/L, 5mg/L, 10 mg/L Pb(C2H3O2)2·3H2O 4 months | ↑ AsA | [51] | ||
| ESSENTIAL MICRONUTRIENTS | ||||||
| Copper (Cu) | Arabidopsis thaliana | 2, 5 μM CuSO4 24 h | ↓ Total AsA (roots) ↑ Total AsA (leaves) | ↑ DHA/AsA (roots) | ↑ APX (roots) ↑ GR (leaves) | [26] |
| Arabidopsis thaliana | 10 μM CuSO4 24 h | ↓ Total AsA (roots) ↑ Total AsA (leaves) | ↓ Reduced AsA, DHA (roots) ↑ Reduced AsA (leaves) | ↓ APX (roots) | [17] | |
| Arabidopsis thaliana | 0, 5, 25, 50, 100 μM CuSO4 1, 3, 7 days | Short term ↑ reduced AsA (1, 3 days) Short term ↑ DHA (3 days) | Short term ↑ DHAR Long term ↑ GR, MDHAR | [54] | ||
| Phaseolus vulgaris (roots) | 15 μM CuSO4 1, 5, 24, 48, 72, 96, 120, 168 h | ↑ Total AsA | ↑ Reduced AsA ↑ DHA ↑ DHA/AsA | Short term ↑ DHAR Long term slightly ↓ DHAR ↑ GR Long term ↑ MDHAR, APX | [55] | |
| Phaseolus vulgaris (leaves) | 50 μM CuSO4 0, 24, 48, 72, 96, 120, 144, 168 h | ↑ Total AsA | ↑ Reduced AsA Short term ↓ DHA followed by transient ↑ DHA Long term ↓ DHA ↓ DHA/AsA | ↑ MDHAR, GR Long term ↑ APX, DHAR | [56] | |
| Oryza Sativa (MSE-9) | 0, 10, 50, 100 μM CuSO4 1, 5 days | ↑ Total AsA | ↑ APX Long term ↑ GR (>50 μM Cu) | [57] | ||
| Morus alba L. cv. Kanva-2 | 0.0, 0.1, 1.0, 100 μM CuSO4 25, 50 days | ↑ Total AsA | ↑ DHA (Cu-deficient,-excess) ↑ DHA/AsA (Cu-deficient,-excess) | ↑ APX, GR | [58] | |
| Hordeum vulgare L. cv. Obzor | 15, 150, 1500 μM CuSO4 5 days | ↑ Total AsA | ↓ % reduced AsA | [59] | ||
| Znic (Zn) | Phaseolus vulgaris (roots) | 50 μM ZnSO4 1, 5, 24, 48, 72, 96, 120, 144, 168 h | 0–120 h ↓ Total AsA >120 h ↑ Total AsA | 1–96 h ↓ reduced AsA >96 h ↑ reduced AsA >72 h ↑ DHA ↑ DHA/AsA | 1–96 h ↑ APX | [60] |
| Phaseolus vulgaris (leaves) | 50 μM ZnSO4 1, 5, 24, 48, 72, 96, 120, 144, 168 h | ↑ Total AsA | ↑ Reduced AsA 48 h, >96 h ↑ DHA >96 h ↑ DHA/AsA | ↑ DHAR Long term ↑ MDHAR, APX, GR | [60] | |
| Cajanus cajan LRG30 (long duration type) | 2.5, 5.0, 7.5 mM ZnSO4·7H2O germinate for 6 days | ↓ AsA | ↑ GR | [61] | ||
| Cajanus cajan ICPL87 (short duration type) | 2.5, 5.0, 7.5 mM ZnSO4·7H2O germinate for 6 days | ↓ AsA | ↑ GR | [61] | ||
| Brassica juncea | 0.007, 0.05, 5.0, 10 mM ZnSO4 10 days | ↑ Total AsA | ↑ MDHAR, DHAR, APX, GR | [62] | ||
| Nickel (Ni) | Oryza sativa | 200, 400 μM NiSO4 0, 5, 10, 15, 20 days | ↑ Reduced AsA ↑ DHA ↑ DHA/AsA | ↑ MDHAR, APX, GR | [63] | |
| Cajanus cajan LRG30 (long duration type) | 0.5, 1.0, 1.5 mM NiSO4·6H2O germinate for 6 days | ↓ AsA | ↑ GR | [61] | ||
| Cajanus cajan ICPL87 (short duration type) | 0.5, 1.0, 1.5 mM NiSO4·6H2O germinate for 6 days | ↓ AsA | ↑ GR | [61] | ||
| Manganese (Mn) | Vigna unguiculata cv. TVu 91 | 0.2, 50, 100 μM MnSO4 6 days | ↓ Reduced AsA ↓ AsA/total AsA | [64] | ||
| Vigna unguiculata cv. TVu1987 | 0.2, 50, 100 μM MnSO4 6 days | ↑ MDHAR, GR | [64] | |||
| Hordeum vulgare L. cv. Obzor | 183, 1830, 18300 μM MnCl2 5 days | Transient ↑ total AsA followed by ↓ | ↓ APX | [59] | ||
| Oryza sativa L. cv. Pant-12 | 3, 6 mM MnCl2 0, 10, 20 days | ↓ Reduced AsA ↓ DHA ↑ DHA/AsA | ↑ MDHAR, DHAR, GR, APX | [65] | ||
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bielen, A.; Remans, T.; Vangronsveld, J.; Cuypers, A. The Influence of Metal Stress on the Availability and Redox State of Ascorbate, and Possible Interference with Its Cellular Functions. Int. J. Mol. Sci. 2013, 14, 6382-6413. https://doi.org/10.3390/ijms14036382
Bielen A, Remans T, Vangronsveld J, Cuypers A. The Influence of Metal Stress on the Availability and Redox State of Ascorbate, and Possible Interference with Its Cellular Functions. International Journal of Molecular Sciences. 2013; 14(3):6382-6413. https://doi.org/10.3390/ijms14036382
Chicago/Turabian StyleBielen, An, Tony Remans, Jaco Vangronsveld, and Ann Cuypers. 2013. "The Influence of Metal Stress on the Availability and Redox State of Ascorbate, and Possible Interference with Its Cellular Functions" International Journal of Molecular Sciences 14, no. 3: 6382-6413. https://doi.org/10.3390/ijms14036382






