Naringin Enhances CaMKII Activity and Improves Long-Term Memory in a Mouse Model of Alzheimer’s Disease
Abstract
:1. Introduction
2. Results and Discussion
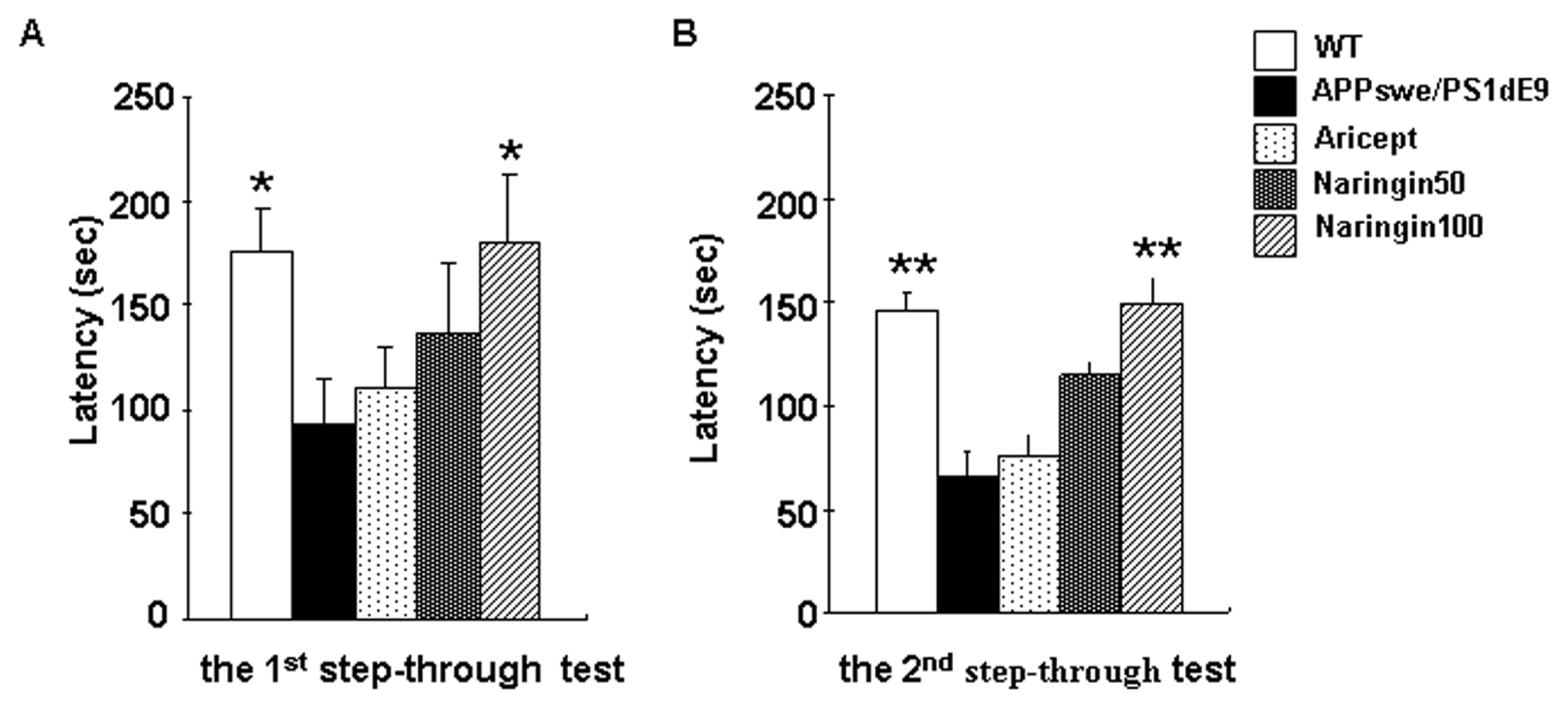
2.1. Naringin Delays the Passive-Avoidance Decline of APPswe/PS1dE9 Mice in a Step-Through Test
2.2. Naringin Improves Long-Term Learning and Memory of APPswe/PS1dE9 Mice in the Morris Water Maze
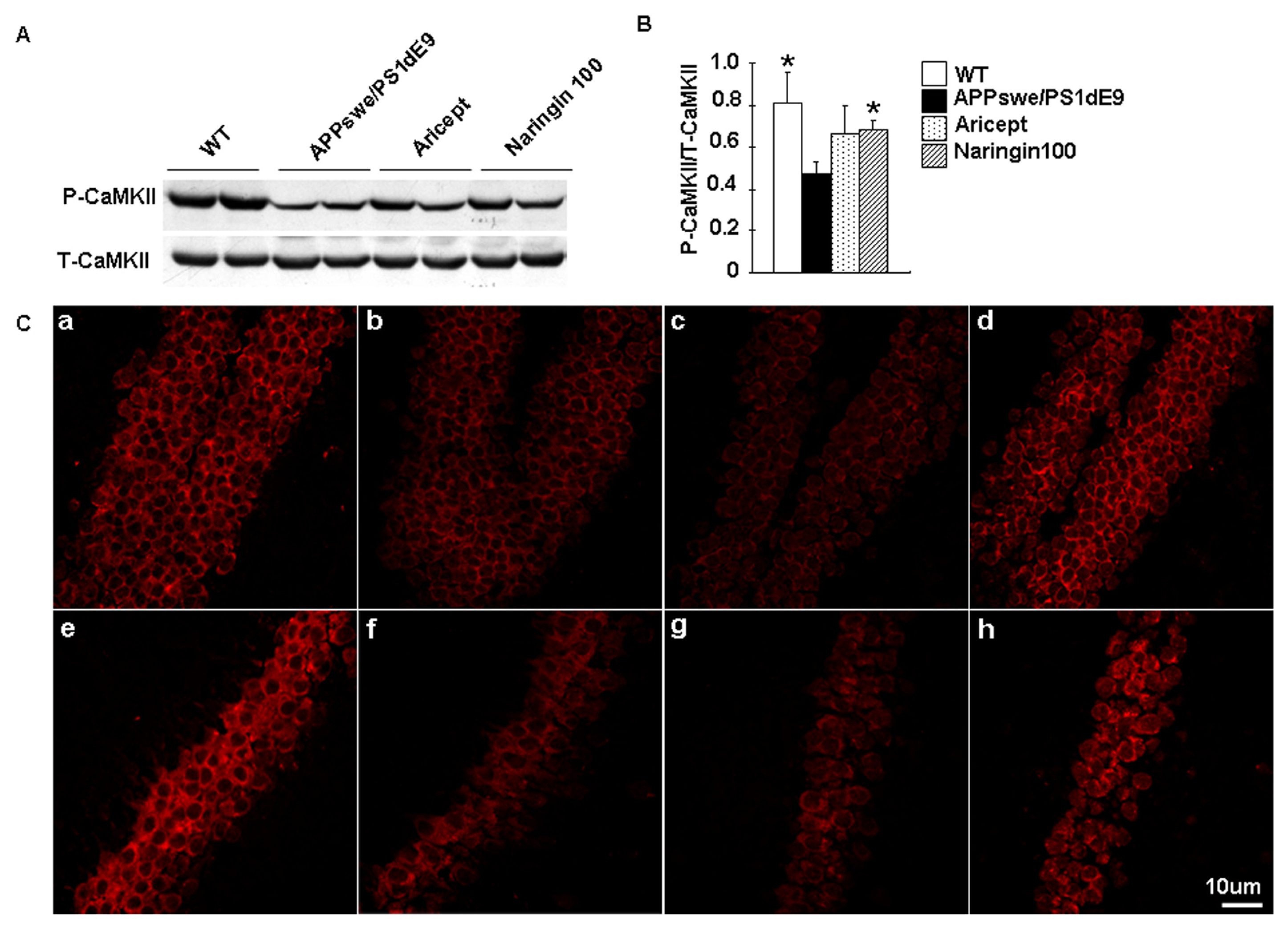
2.3. Naringin Enhances CaMKII Activity in an AD Model
3. Experimental Section
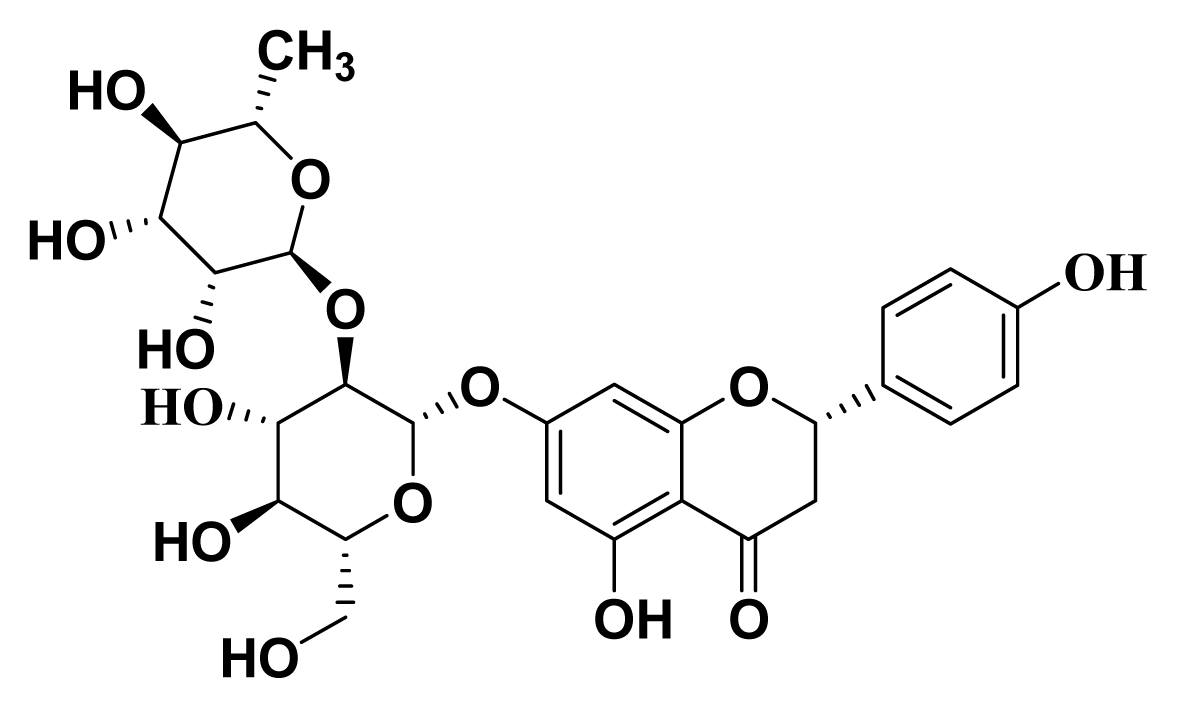
3.1. Isolation of Naringin
3.2. APPswe/PS1dE9 Mice and Naringin Treatment
3.3. Passive-Avoidance Test
3.4. Morris Water Maze
3.5. Tissue Preparation
3.6. Western Blot Analysis
3.7. Immunofluorescence Staining
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Stokin, G.B.; Lillo, C.; Falzone, T.L.; Brusch, R.G.; Rockenstein, E.; Mount, S.L.; Raman, R.; Davies, P.; Masliah, E.; Williams, D.S.; et al. Axonopathy and transport deficits early in the pathogenesis of Alzheimer’s disease. Science 2005, 307, 1282–1288. [Google Scholar]
- Ballatore, C.; Lee, V.M.; Trojanowski, J.Q. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat. Rev. Neurosci 2007, 8, 663–672. [Google Scholar]
- Auld, D.S.; Kornecook, T.J.; Bastianetto, S.; Quirion, R. Alzheimer’s disease and the basal forebrain cholinergic system: Relations to beta-amyloid peptides, cognition, and treatment strategies. Prog. Neurobiol 2002, 68, 209–245. [Google Scholar]
- Martinez-Coria, H.; Green, K.N.; Billings, L.M.; Kitazawa, M.; Albrecht, M.; Rammes, G.; Parsons, C.G.; Gupta, S.; Banerjee, P.; LaFerla, F.M. Memantine improves cognition and reduces Alzheimer’s-like neuropathology in transgenic mice. Am. J. Pathol 2010, 176, 870–880. [Google Scholar]
- Selkoe, D.J. Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav. Brain Res 2008, 192, 106–113. [Google Scholar]
- Shankar, G.M.; Walsh, D.M. Alzheimer’s disease: Synaptic dysfunction and Abeta. Mol. Neurodegener 2009, 4, 48. [Google Scholar]
- Chakroborty, S.; Kim, J.; Schneider, C.; Jacobson, C.; Molgo, J.; Stutzmann, G.E. Early presynaptic and postsynaptic calcium signaling abnormalities mask underlying synaptic depression in presymptomatic Alzheimer’s disease mice. J. Neurosci 2012, 32, 8341–8353. [Google Scholar]
- Zeng, Y.; Zhao, D.; Xie, C.W. Neurotrophins enhance CaMKII activity and rescue amyloid-beta-induced deficits in hippocampal synaptic plasticity. J. Alzheimer’s Dis 2010, 21, 823–831. [Google Scholar]
- Schroeter, H.; Boyd, C.; Spencer, J.P.; Williams, R.J.; Cadenas, E.; Rice-Evans, C. MAPK signaling in neurodegeneration: Influences of flavonoids and of nitric oxide. Neurobiol. Aging 2002, 23, 861–880. [Google Scholar]
- Pereira, R.M.S.; Andrades, N.E.D.; Paulino, N.; Sawaya, A.C.H.F.; Eberlin, M.N.; Marcucci, M.C.; Favero, G.M.; Novak, E.M.; Bydlowski, S.P. Synthesis and characterization of a metal complex containing naringin and Cu, and its antioxidant, antimicrobial, antiinflammatory and tumor cell cytotoxicity. Molecules 2007, 12, 1352–1366. [Google Scholar]
- Cui, L.; Zhang, Z.H.; Sun, E.; Jia, X.B. Effect of β-cyclodextrin complexation on solubility and enzymatic conversion of naringin. Int. J. Mol. Sci 2012, 13, 14251–14261. [Google Scholar]
- Golechha, M.; Chaudhry, U.; Bhatia, J.; Saluja, D.; Arya, D.S. Naringin protects against kainic acid-induced status epilepticus in rats: Evidence for an antioxidant, anti-inflammatory and neuroprotective intervention. Biol. Pharm. Bull 2011, 34, 360–365. [Google Scholar]
- Kumar, P.; Kumar, A. Protective effect of hesperidin and naringin against 3-nitropropionic acid induced Huntington’s like symptoms in rats: Possible role of nitric oxide. Behav. Brain Res 2010, 206, 38–46. [Google Scholar]
- Kumar, A.; Dogra, S.; Prakash, A. Protective effect of naringin, a Citrus flavonoid, against colchicine-induced cognitive dysfunction and oxidative damage in rats. J. Med. Food 2010, 13, 976–984. [Google Scholar]
- Kumar, A.; Prakash, A.; Dogra, S. Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem. Toxicol 2010, 48, 626–632. [Google Scholar]
- Wang, D.M.; Gao, K.; Li, X.Y.; Shen, X.H.; Zhang, X.; Ma, C.M.; Qin, C.; Zhang, L.F. Long-term naringin consumption reverses a glucose uptake defect and improves cognitive deficits in a mouse model of Alzheimer’s disease. Pharm. Biochem. Behav 2012, 102, 13–20. [Google Scholar]
- Insausti, R.; Tunon, T.; Sobreviela, T.; Insausti, A.M.; Gonzalo, L.M. The human entorhinal cortex: A cytoarchitectonic analysis. J. Comp. Neurol 1995, 355, 171–198. [Google Scholar]
- Shankar, G.M.; Li, S.; Mehta, T.H.; Garcia-Munoz, A.; Shepardson, N.E.; Smith, I.; Brett, F.M.; Farrell, M.A.; Rowan, M.J.; Lemere, C.A.; et al. Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat. Med 2008, 14, 837–842. [Google Scholar]
- Amada, N.; Aihara, K.; Ravid, R.; Horie, M. Reduction of NR1 and phosphorylated Ca2+/calmodulin-dependent protein kinase II levels in Alzheimer’s disease. Neuroreport 2005, 16, 1809–1813. [Google Scholar]
- Lisman, J.; Yasuda, R.; Raghavachari, S. Mechanisms of CaMKII action in long-term potentiation. Nat. Rev. Neurosci 2012, 13, 169–182. [Google Scholar]
- Strack, S.; Choi, S.; Lovinger, D.M.; Colbran, R.J. Translocation of autophosphorylated calcium/calmodulin-dependent protein kinase II to the postsynaptic density. J. Biol. Chem 1997, 272, 13467–13470. [Google Scholar]
- Benfenati, F.; Valtorta, F.; Rubenstein, J.L.; Gorelick, F.S.; Greengard, P.; Czernik, A.J. Synaptic vesicle-associated Ca2+/calmodulin-dependent protein kinase II is a binding protein for synapsin I. Nature 1992, 359, 417–420. [Google Scholar]
- Derkach, V.; Barria, A.; Soderling, T.R. Ca2+/calmodulin-kinase II enhances channel conductance of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionate type glutamate receptors. Proc. Natl. Acad. Sci. USA 1999, 96, 3269–3274. [Google Scholar]
- Liu, S.J.; Gasperini, R.; Foa, L.; Small, D.H. Amyloid-beta decreases cell-surface AMPA receptors by increasing intracellular calcium and phosphorylation of GluR2. J. Alzheimer’s Dis 2010, 21, 655–666. [Google Scholar]
- Minano-Molina, A.J.; Espana, J.; Martin, E.; Barneda-Zahonero, B.; Fado, R.; Sole, M.; Trullás, R.; Saura, C.A.; Rodríguez-Alvarez, J. Soluble oligomers of amyloid-beta peptide disrupt membrane trafficking of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor contributing to early synapse dysfunction. J. Biol. Chem 2011, 286, 27311–27321. [Google Scholar]
- Lanz, T.A.; Carter, D.B.; Merchant, K.M. Dendritic spine loss in the hippocampus of young PDAPP and Tg2576 mice and its prevention by the ApoE2 genotype. Neurobiol. Dis 2003, 13, 246–253. [Google Scholar]
- Hayashi, Y.; Shi, S.H.; Esteban, J.A.; Piccini, A.; Poncer, J.C.; Malinow, R. Driving AMPA receptors into synapses by LTP andCaMKII: Requirement for GluR1 and PDZ domain interaction. Science 2000, 287, 2262–2267. [Google Scholar]
- Jourdain, P.; Fukunaga, K.; Muller, D. Calcium/calmodulin-dependent protein kinase II contributes to activity-dependent filopodia growth and spine formation. J. Neurosci 2003, 23, 10645–10649. [Google Scholar]
- Wayman, G.A.; Lee, Y.S.; Tokumitsu, H.; Silva, A.J.; Soderling, T.R. Calmodulin-kinases: Modulators of neuronal development and plasticity. Neuron 2008, 59, 914–931. [Google Scholar]
- Zhu, L.Q.; Wang, S.H.; Liu, D.; Yin, Y.Y.; Tian, Q.; Wang, X.C.; Wang, Q.; Chen, J.G.; Wang, J.Z. Activation of glycogen synthase kinase-3 inhibits long-term potentiation with synapse-associated impairments. J. Neurosci 2007, 27, 12211–12220. [Google Scholar]
- Song, B.; Lai, B.; Zheng, Z.; Zhang, Y.; Luo, J.; Wang, C.; Chen, Y.; Woodgett, J.R.; Li, M. Inhibitory phosphorylation of GSK-3 by CaMKII couples depolarization to neuronal survival. J. Biol. Chem. 2010, 285, 41122–41134. [Google Scholar]
- Sudto, K.; Pornpakakul, S.; Wanichwecharungruang, S. An efficient method for the large scale isolation of naringin from pomelo (Citrus grandis) peel. Int. J. Food Sci. Tech 2009, 44, 1737–1742. [Google Scholar]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J 2008, 22, 659–661. [Google Scholar]
- Laczo, J.; Vlcek, K.; Vyhnalek, M.; Vajnerova, O.; Ort, M.; Holmerova, I.; Tolar, M.; Andel, R.; Bojar, M.; Hort, J. Spatial navigation testing discriminates two types of amnestic mild cognitive impairment. Behav. Brain Res 2009, 202, 252–259. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, D.-M.; Yang, Y.-J.; Zhang, L.; Zhang, X.; Guan, F.-F.; Zhang, L.-F. Naringin Enhances CaMKII Activity and Improves Long-Term Memory in a Mouse Model of Alzheimer’s Disease. Int. J. Mol. Sci. 2013, 14, 5576-5586. https://doi.org/10.3390/ijms14035576
Wang D-M, Yang Y-J, Zhang L, Zhang X, Guan F-F, Zhang L-F. Naringin Enhances CaMKII Activity and Improves Long-Term Memory in a Mouse Model of Alzheimer’s Disease. International Journal of Molecular Sciences. 2013; 14(3):5576-5586. https://doi.org/10.3390/ijms14035576
Chicago/Turabian StyleWang, Dong-Mei, Ya-Jun Yang, Li Zhang, Xu Zhang, Fei-Fei Guan, and Lian-Feng Zhang. 2013. "Naringin Enhances CaMKII Activity and Improves Long-Term Memory in a Mouse Model of Alzheimer’s Disease" International Journal of Molecular Sciences 14, no. 3: 5576-5586. https://doi.org/10.3390/ijms14035576



