PG-2, a Potent AMP against Pathogenic Microbial Strains, from Potato (Solanum tuberosum L cv. Gogu Valley) Tubers Not Cytotoxic against Human Cells
Abstract
:1. Introduction
2. Results and Discussion
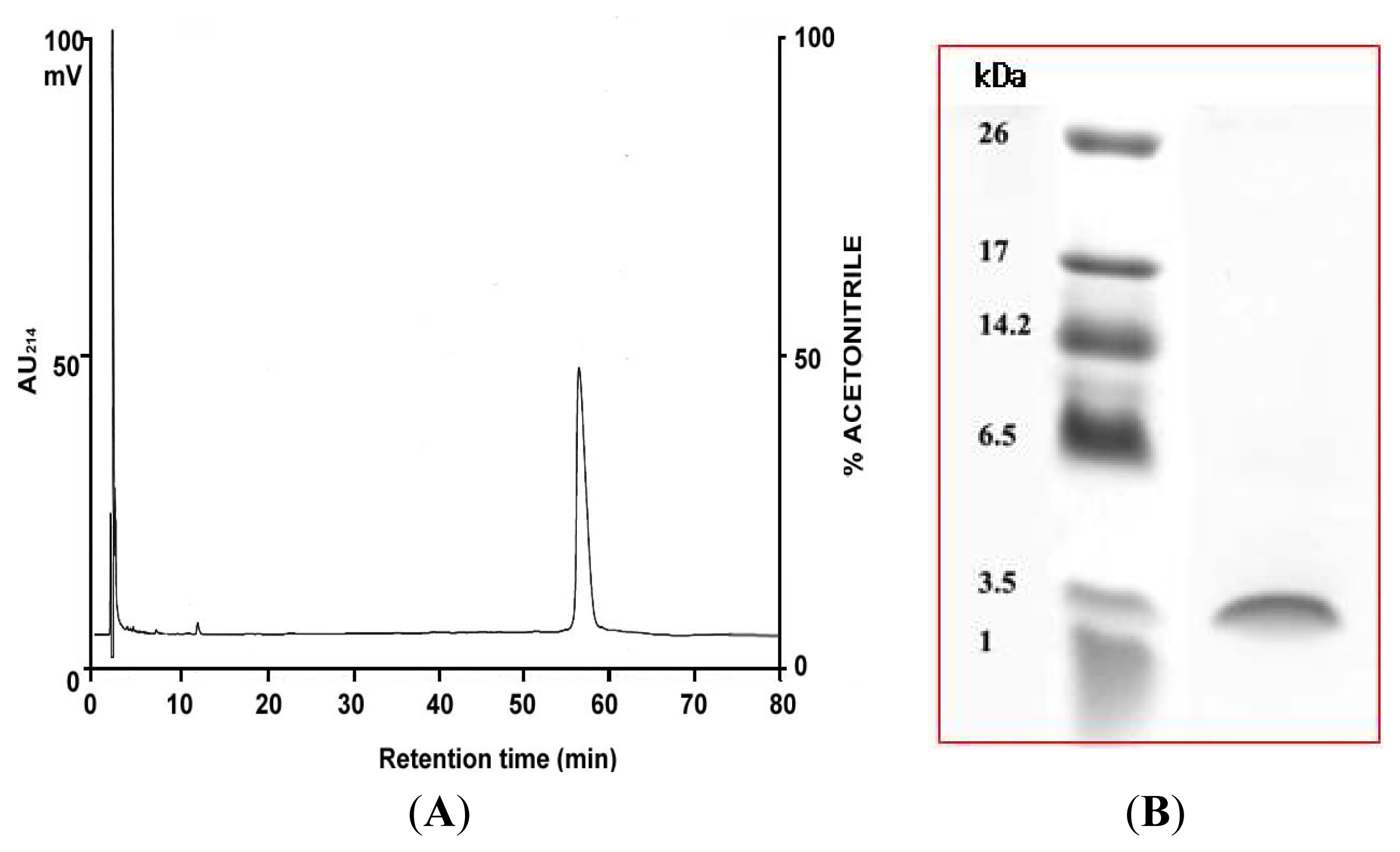
2.1. Purification of PG-2 from Potato Tubers
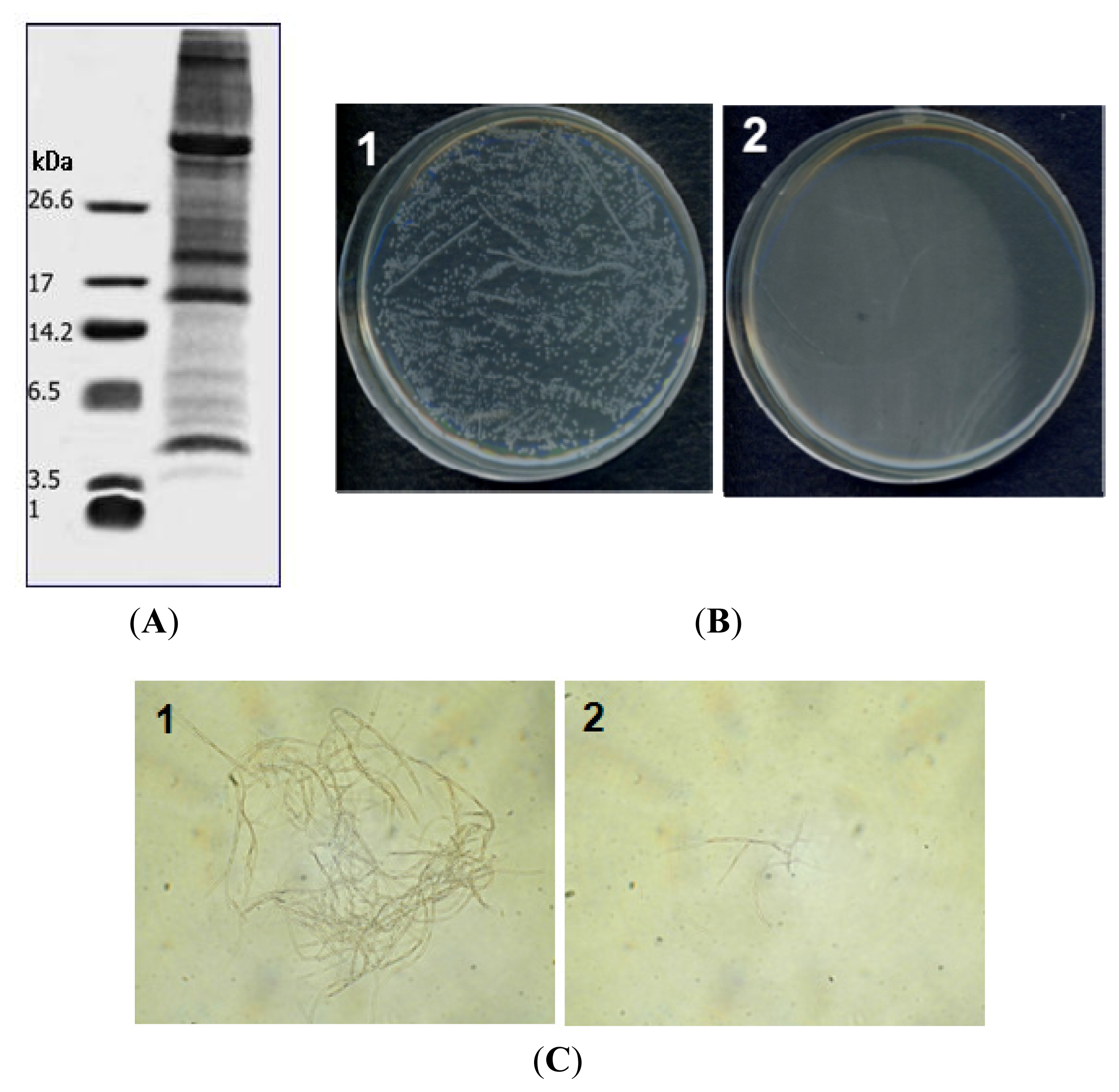
2.2. Antimicrobial Activity
2.3. Hemolytic Activity of PG-2
2.4. N-Terminal Amino Acid Sequence Analysis of PG-2
2.5. AMPs from Potatoes
3. Experimental Section
3.1. Materials
3.2. Bacterial and Fungal Strains for Antimicrobial Activity
3.3. Purification of PG-2
3.4. Tricine Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis
3.5. Antimicrobial Assay
3.6. Hemolysis of hRBCs (Human Red Blood Cells)
3.7. N-Terminal Amino Acid Sequence Analysis and Mass Spectroscopy of PG-2
4. Conclusion
Acknowledgements
References
- Osborn, W.F.; Broekaert, F.R.; Terras, B.P.; Cammue, R.W. Plant defensins: Novel antimicrobial peptides as components of the host defense system. Plant Physiol 1995, 108, 1353–1358. [Google Scholar]
- Hultmark, H.G.; Boman, D. Cell-free immunity in insects. Annu. Rev. Microbiol 1987, 41, 103–126. [Google Scholar]
- Hoffmann, J.A. Innate immunity of insects. Curr. Opin. Immunol 1995, 7, 4–10. [Google Scholar]
- Park, J.-K.; Lee, R.; Gopal, C.H.; Seo, H.; Cheong, Y. Isolation and Purification of a Novel Deca-Antifungal Peptide from Potato (Solanum tuberosum L cv. Jopung) Against Candida albicans. Int. J. Mol. Sci 2012, 13, 4021–4032. [Google Scholar]
- Simmaco, D.; Barra, M. Amphibian skin: A promising resource for antimicrbial peptides. Trends Biotechnol 1995, 13, 205–209. [Google Scholar]
- Ganz, R.I.; Lehrer, A.K.; Lichtenstein, T. Defensins: Antimicrobial and cytotoxic peptides of mammalian cells. Annu. Rev. Immunol 1993, 1, 105–128. [Google Scholar]
- Sitaram, C.; Subbalakshmi, N. Mechanism of antimicrobial action of indolicidin. FEMS Microbiol. Lett 1998, 16, 91–96. [Google Scholar]
- Glazebrook, J. Genes controlling expression of defense responses in Arabidopsis. Curr. Opin. Plant Biol 1999, 2, 280–286. [Google Scholar]
- Hahm, J.-Y.; Kim, S.Y.; Lee, S.-C.; Park, S.Y.; Shin, S.J.; Choi, Y.; Park, K.-S. Purification and antimicrobial activity studies of the N-terminal fragment of ubiquitin from human amniotic fluid. Biochim. Biophys. Acta 2007, 1774, 1221–1226. [Google Scholar]
- Garcia-Olmedo, A.; Segura, M.; Moreno, F.; Madueno, A.; Molina, F. Snakin-1, a peptide from potato that is active against plant pathogens. Mol. Plant Microb Interact 1999, 12, 16–23. [Google Scholar]
- Molina, M.; Berrocal-Lobo, A.; Segura, M.; Moreno, G.; Lopez, F.; Garcia-Olmedo, A. Snakin-2, an antimicrobial peptide from potato whose gene is locally induced by wounding and responds to pathogen infection. Plant Physiol 2002, 128, 951–961. [Google Scholar]
- Park, M.H.; Kim, S.C.; Park, J.Y.; Kim, S.Y.; Lee, H.T.; Lim, H.; Cheong, K.S.; Hahm, Y. Purification and characterization of a heat-stable serine protease inhibitor from the tubers of new potato variety “Golden Valley”. Biochem. Biophys. Res. Commun 2006, 346, 681–686. [Google Scholar]
- Hahm, J.Y.; Kim, S.C.; Park, M.H.; Kim, H.T.; Lim, Y.; Park, K.S. Antimicrobial activity studies on a trypsin-chymotrypsin protease inhibitor obtained from potato. Biochem. Biophys. Res. Commun 2005, 330, 921–927. [Google Scholar]
- Van Strien, L.C.; van Loon, E.A. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol. Mol. Plant Pathol 1999, 55, 85–97. [Google Scholar]
- Garg, V.R.; Tripathi, S.; Kumar, S.K. A study on trypsin, Aspergillus flavus and Bacillus sp. protease inhibitory activity in Cassia tora (L.) syn Senna tora (L.) Roxb. seed extract. BMC Complement Altern. Med 2011, 11, 56. [Google Scholar]
- Tian, S.B.; Liu, X.C.; Wang, M.J.; Shi, Y.Y.; Chen, Z.H.; Hu, W.M. Vegetative storage protein with trypsin inhibitor activity occurs in Sapindus mukorassi, a sapindaceae deciduous tree. J. Integr. Plant Biol 2009, 51, 352–359. [Google Scholar]
- Sano, Y.K.; Yap, Y.; Kodama, F.; Waller, K.M.; Chung, H.; Ueda, K.; Nakamura, M.; Oldsen, H.; Yoda, Y.; Yamaguchi, H. Activation of a novel transcription factor through phosphorylation by WIPK, a wound-induced mitogen-activated protein kinase in tobacco plants. Plant Physiol 2005, 139, 127–137. [Google Scholar]
- Wu, M.; Heinrich, I.T.; Baldwin, J. Two mitogen-activated protein kinase kinases, MKK1 and MEK2, are involved in wounding- and specialist lepidopteran herbivore Manduca sexta-induced responses in Nicotiana attenuata. J. Exp. Bot 2011, 62, 4355–4365. [Google Scholar]
- Domoney, A.; Clemente, M.; Carmen Marín-Manzano, E.; Jiménez, M.; Carmen Arques, C. The anti-proliferative effect of TI1B, a major Bowman-Birk isoinhibitor from pea (Pisum sativum L.), on HT29 colon cancer cells is mediated through protease inhibition. Br. J. Nutr 2012, 108, 135–144. [Google Scholar]
- Park, J.Y.; Kim, S.C.; Park, J.K.; Lee, S.J.; Choi, K.S.; Hahm, Y. Novel antibacterial activity of β(2)-microglobulin in human amniotic fluid. PLoS One 2012, 7, e47642. [Google Scholar]
- Ozols, J. Amino acid analysis. Murray, P.D., Ed.; Academic Press: London, UK, 1990; pp. 587–601. [Google Scholar]
- Park, S.C.; Park, J.Y.; Kim, J.K.; Lee, I.; Hwang, H.; Cheong, J.W.; Nah, K.S.; Hahm, Y. Antifungal mechanism of a novel antifungal protein from pumpkin rinds against various fungal pathogens. J. Agric. Food Chem 2009, 57, 9299–9304. [Google Scholar]
- Park, S.; Yoo, J.Y.; Kim, S.C.; Park, Y.; do Choi, C.H.; Seo, K.S.; Hahm, Y. Effect of acidic pH on antibacterial action of peptide isolated from Korean pen shell (Atrina pectinata). J. Pept. Sci 2011, 17, 353–357. [Google Scholar]
- Park, S.C.; Park, J.Y.; Kim, C.; Jeong, S.; Yoo, K.S.; Hahm, Y. A plausible mode of action of pseudin-2, an antimicrobial peptide from Pseudis paradoxa. Biochim. Biophys. Acta 2011, 1808, 171–182. [Google Scholar]


| Step | Amount of PG-2 crude proteins (mg/200 g powder) | Yield |
|---|---|---|
| 1. Protein Extraction Buffer | 87.18 | 43.59 |
| 2. Ultrafiltration (under 10 kDa) | 11.07 | 5.55 |
| 3. C18-HPLC | 0.12 | 0.06 |
| Microorganisms | MIC (μM) |
|---|---|
| Fungal pathogens | |
| C. albicans | 25 |
| R. solani | 25 |
| Bacterial pathogens | |
| S. aureus | 3.12 |
| C. michiganense subsp. michiganensis | 3.12 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, J.-Y.; Gopal, R.; Kim, S.Y.; Seo, C.H.; Lee, H.B.; Cheong, H.; Park, Y. PG-2, a Potent AMP against Pathogenic Microbial Strains, from Potato (Solanum tuberosum L cv. Gogu Valley) Tubers Not Cytotoxic against Human Cells. Int. J. Mol. Sci. 2013, 14, 4349-4360. https://doi.org/10.3390/ijms14024349
Kim J-Y, Gopal R, Kim SY, Seo CH, Lee HB, Cheong H, Park Y. PG-2, a Potent AMP against Pathogenic Microbial Strains, from Potato (Solanum tuberosum L cv. Gogu Valley) Tubers Not Cytotoxic against Human Cells. International Journal of Molecular Sciences. 2013; 14(2):4349-4360. https://doi.org/10.3390/ijms14024349
Chicago/Turabian StyleKim, Jin-Young, Ramamourthy Gopal, Sang Young Kim, Chang Ho Seo, Hyang Burm Lee, Hyeonsook Cheong, and Yoonkyung Park. 2013. "PG-2, a Potent AMP against Pathogenic Microbial Strains, from Potato (Solanum tuberosum L cv. Gogu Valley) Tubers Not Cytotoxic against Human Cells" International Journal of Molecular Sciences 14, no. 2: 4349-4360. https://doi.org/10.3390/ijms14024349




