Sodium Dodecyl Sulfate (SDS)-Loaded Nanoporous Polymer as Anti-Biofilm Surface Coating Material
Abstract
:1. Introduction
2. Results and Discussion
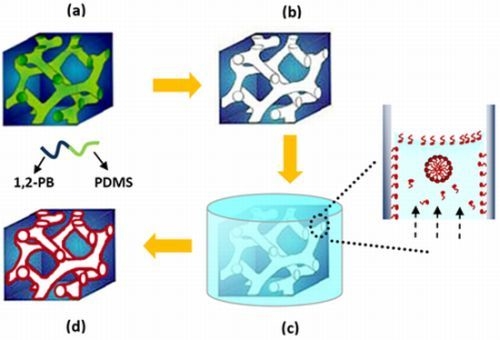
2.1. Preparation of Nanoporous Polymer
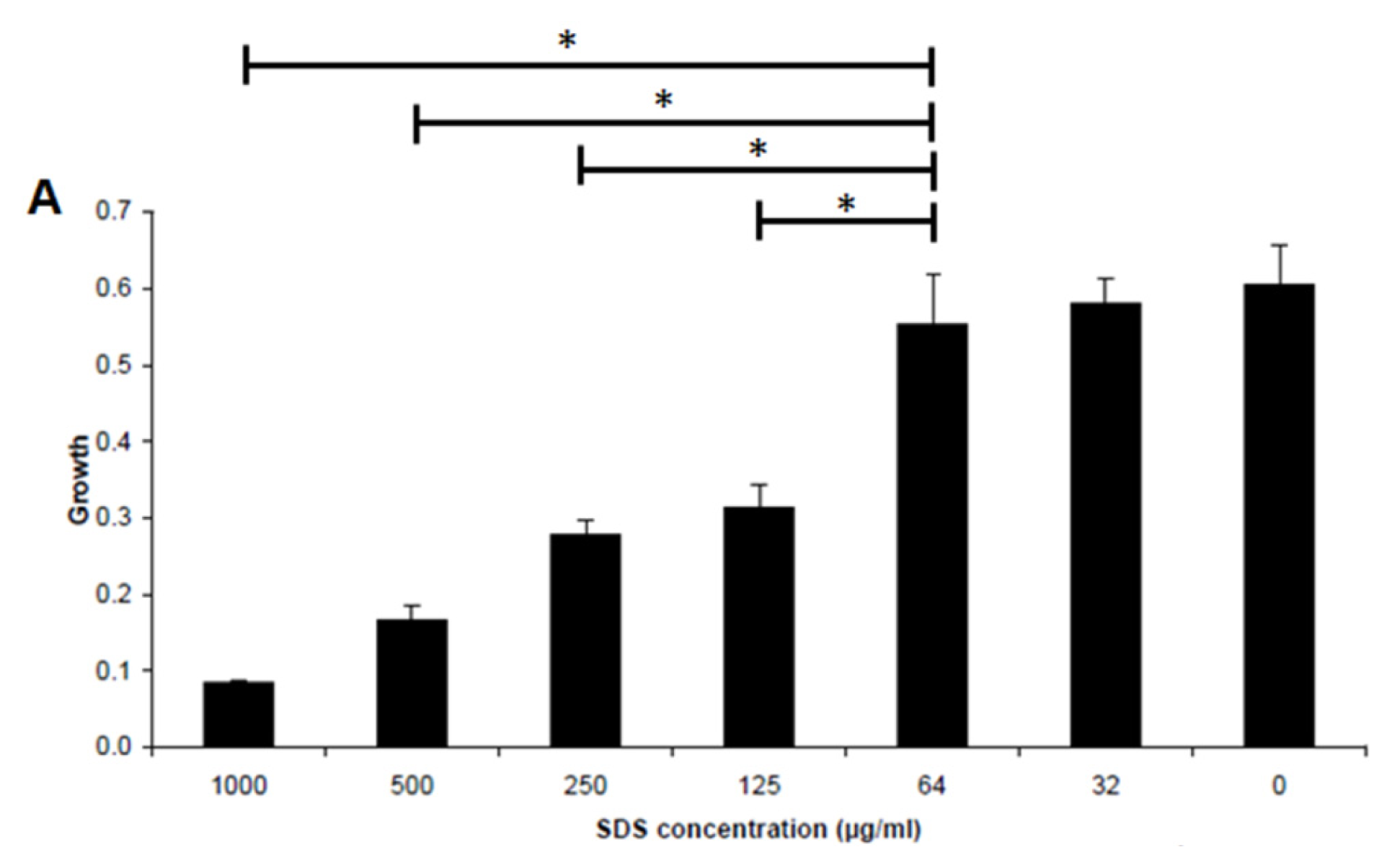
2.2. Effect of SDS on Inhibiting Biofilm Formation by E. coli Sar18
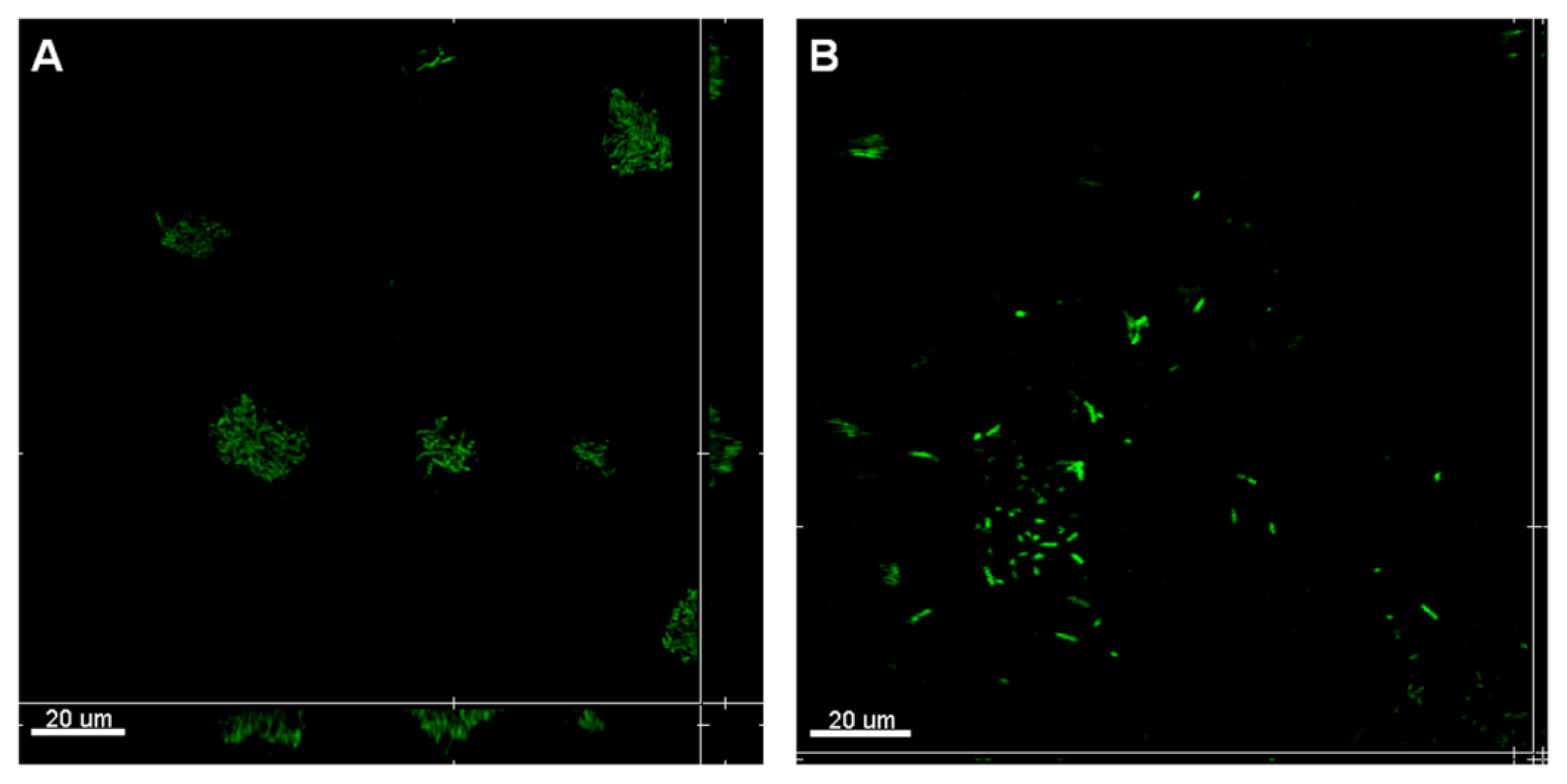
2.3. Attachment Inhibition by SDS-Loaded Nanoporous Film
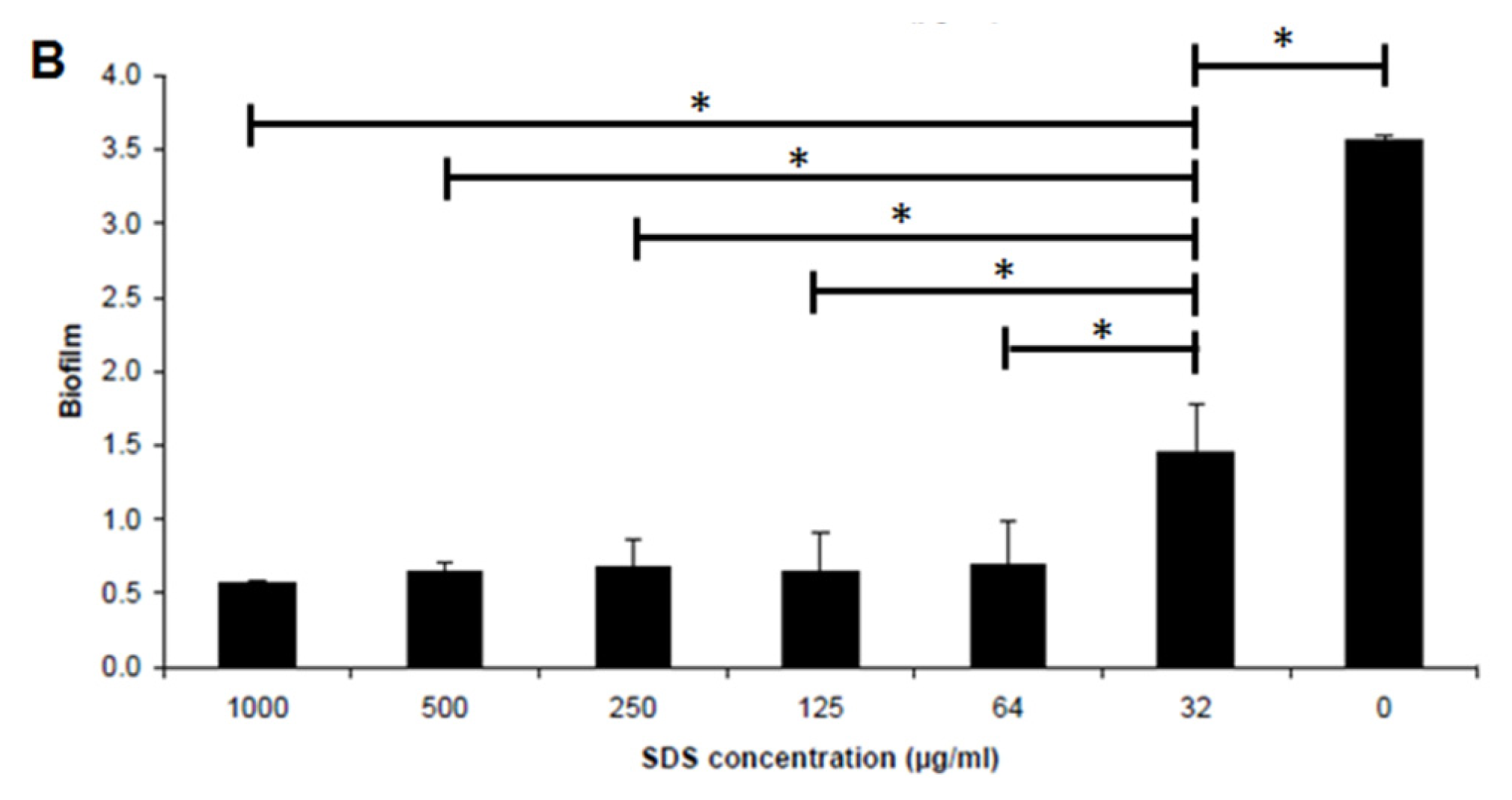
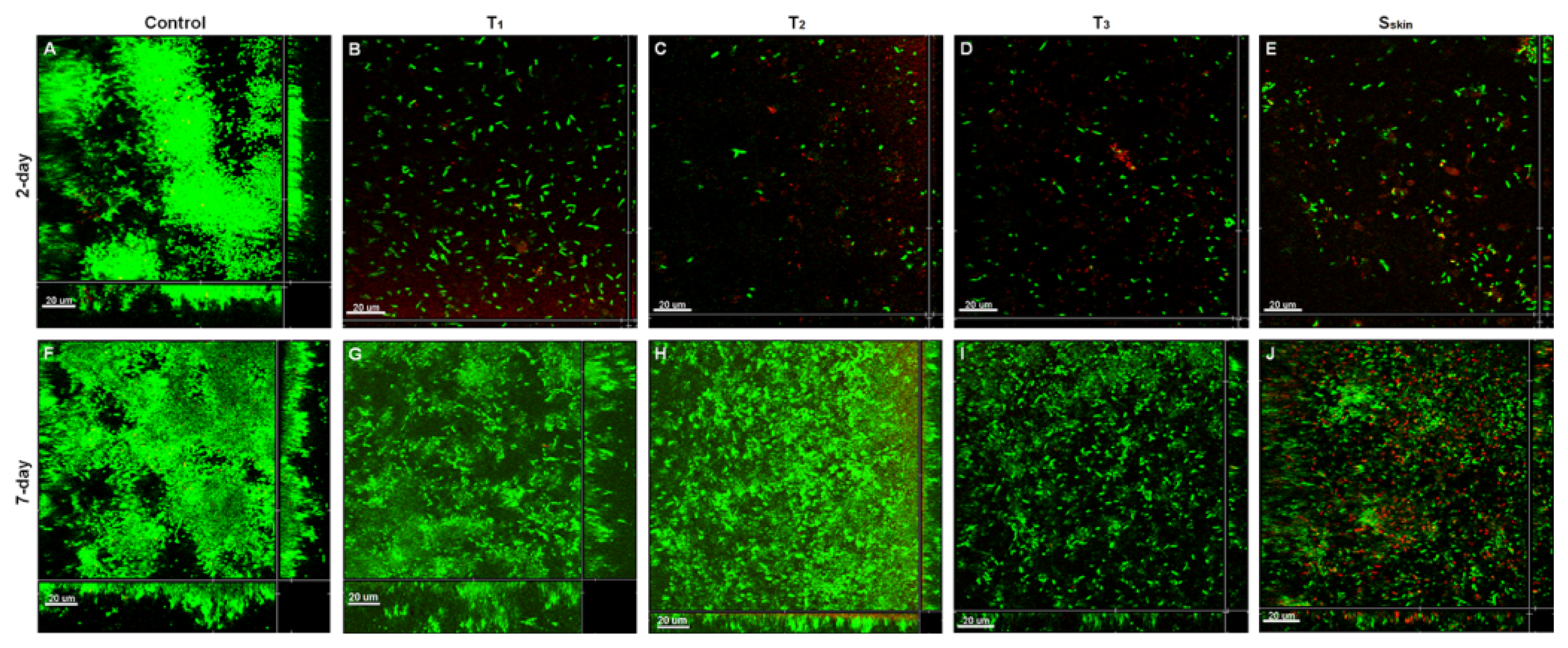
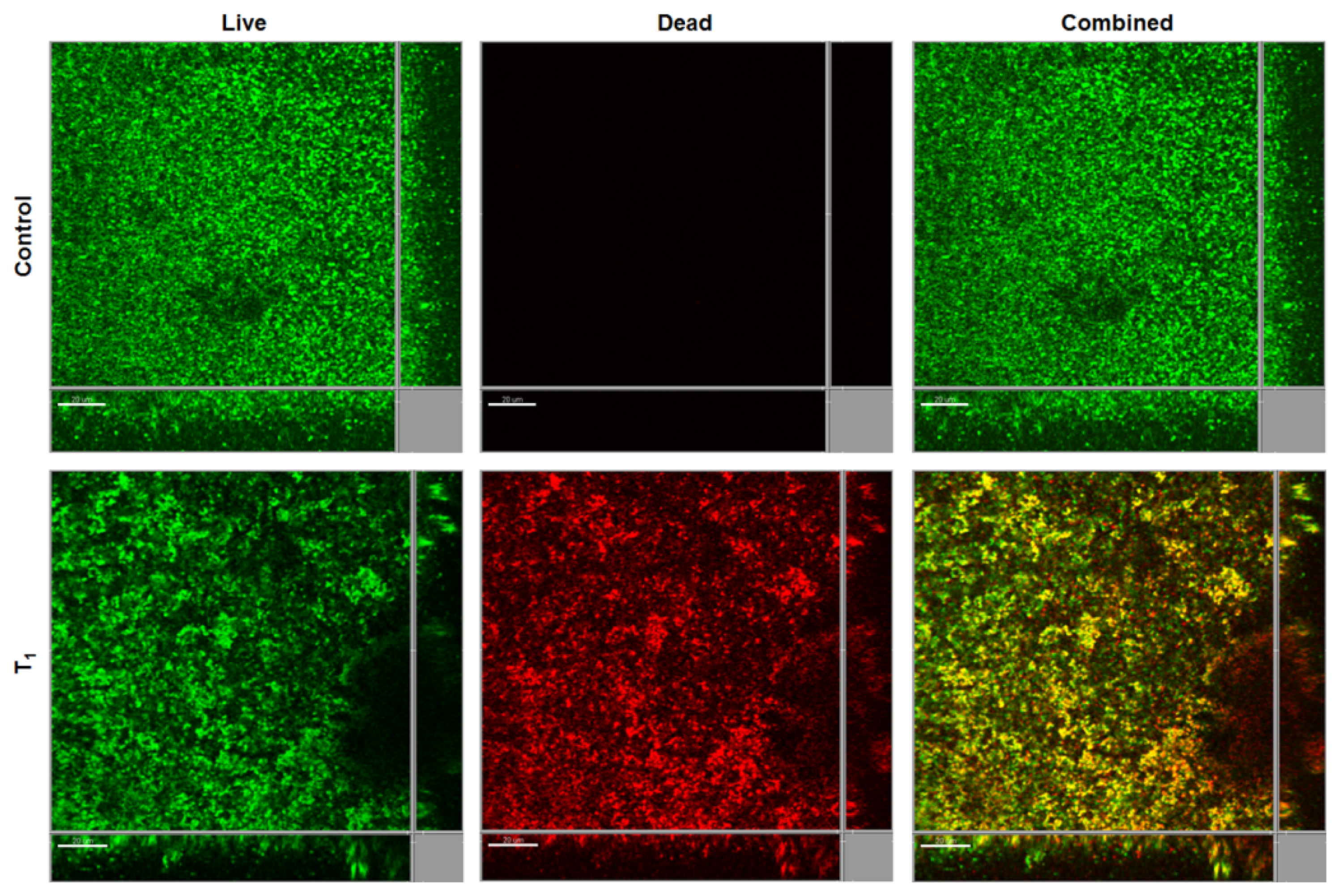
2.4. Biofilm Inhibition by SDS-Loaded Nanoporous Film
3. Experimental Section
3.1. Preparation of Nanoporous Film
3.2. Characterization of Nanoporous Films
3.3. Loading of Nanoporous Films with SDS
3.4. Minimum Biofilm Inhibitory Concentration (MBIC) Assay
3.5. Attachment Assay
3.6. Biofilm Formation Assay
3.7. CLSM Observation
3.8. COMSTAT Analysis
3.9. Estimation of the SDS Concentration Near the Surface
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Costerton, J.W.; Lewandowski, Z.; Caldwell, D.E.; Korber, D.R.; Lappin-Scott, H.M. Microbial biofilms. Annu. Rev. Microbiol 1995, 49, 711–745. [Google Scholar]
- Yang, L.; Liu, Y.; Wu, H.; Song, Z.; Hoiby, N.; Molin, S.; Givskov, M. Combating biofilms. FEMS Immunol. Med. Microbiol 2012, 65, 146–157. [Google Scholar]
- Yang, L.; Liu, Y.; Wu, H.; Hoiby, N.; Molin, S.; Song, Z.J. Current understanding of multi-species biofilms. Int. J. Oral Sci 2011, 3, 74–81. [Google Scholar]
- De Prijck, K.; Nelis, H.; Coenye, T. Efficacy of silver-releasing rubber for the prevention of Pseudomonas aeruginosa biofilm formation in water. Biofouling 2007, 23, 405–411. [Google Scholar]
- Olsen, S.M.; Pedersen, L.T.; Laursen, M.H.; Kiil, S.; Dam-Johansen, K. Enzyme-based antifouling coatings: A review. Biofouling 2007, 23, 369–383. [Google Scholar]
- Almaguer-Flores, A.; Ximenez-Fyvie, L.A.; Rodil, S.E. Oral bacterial adhesion on amorphous carbon and titanium films: Effect of surface roughness and culture media. J. Biomed. Mater. Res. B Appl. Biomater 2010, 92, 196–204. [Google Scholar]
- Kaper, H.J.; Busscher, H.J.; Norde, W. Characterization of poly(ethylene oxide) brushes on glass surfaces and adhesion of Staphylococcus epidermidis. J. Biomater. Sci. Polym. Ed 2003, 14, 313–324. [Google Scholar]
- Majumdar, P.; Lee, E.; Patel, N.; Ward, K.; Stafslien, S.J.; Daniels, J.; Chisholm, B.J.; Boudjouk, P.; Callow, M.E.; Callow, J.A.; et al. Combinatorial materials research applied to the development of new surface coatings IX: An investigation of novel antifouling/fouling-release coatings containing quaternary ammonium salt groups. Biofouling 2008, 24, 185–200. [Google Scholar]
- Nagel, J.A.; Dickinson, R.B.; Cooper, S.L. Bacterial adhesion to polyurethane surfaces in the presence of pre-adsorbed high molecular weight kininogen. J. Biomater. Sci. Polym. Ed 1996, 7, 769–780. [Google Scholar]
- Hetrick, E.M.; Schoenfisch, M.H. Reducing implant-related infections: Active release strategies. Chem. Soc. Rev 2006, 35, 780–789. [Google Scholar]
- Price, J.S.; Tencer, A.F.; Arm, D.M.; Bohach, G.A. Controlled release of antibiotics from coated orthopedic implants. J. Biomed. Mater. Res 1996, 30, 281–286. [Google Scholar]
- Schierholz, J.M.; Steinhauser, H.; Rump, A.F.; Berkels, R.; Pulverer, G. Controlled release of antibiotics from biomedical polyurethanes: Morphological and structural features. Biomaterials 1997, 18, 839–844. [Google Scholar]
- Lee, D.; Cohen, R.E.; Rubner, M.F. Antibacterial properties of Ag nanoparticle loaded multilayers and formation of magnetically directed antibacterial microparticles. Langmuir 2005, 21, 9651–9659. [Google Scholar]
- Hillmyer, M.A. Nanoporous materials from block copolymer precursors. Adv. Polym. Sci. 2005, 137–181. [Google Scholar]
- Orilall, M.C.; Wiesner, U. Block copolymer based composition and morphology control in nanostructured hybrid materials for energy conversion and storage: Solar cells, batteries, and fuel cells. Chem. Soc. Rev 2011, 40, 520–535. [Google Scholar]
- Logar, N.Z.; Kaucic, V. Nanoporous materials: From catalysis and hydrogen storage to wastewater treatment. Acta Chim. Slov 2006, 53, 117–135. [Google Scholar]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar]
- Jackson, E.A.; Hillmyer, M.A. Nanoporous membranes derived from block copolymers: From drug delivery to water filtration. ACS Nano 2010, 4, 3548–3553. [Google Scholar]
- Schulte, L.; Grydgaard, A.; Jakobsen, M.R.; Szewczykowski, P.P.; Guo, F.X.; Vigild, M.E.; Berg, R.H.; Ndoni, S. Nanoporous materials from stable and metastable structures of 1,2-PB-b-PDMS block copolymers. Polymer 2011, 52, 422–429. [Google Scholar]
- Guo, F.; Jankova, K.; Schulte, L.; Vigild, M.E.; Ndoni, S. Surface modification of nanoporous 1,2-polybutadiene by atom transfer radical polymerization or click chemistry. Langmuir 2010, 26, 2008–2013. [Google Scholar]
- Li, L.; Wang, Y.; Vigild, M.E.; Ndoni, S. Physisorption of SDS in a hydrocarbon nanoporous polymer. Langmuir 2010, 26, 13457–13465. [Google Scholar]
- Hamersky, M.W.; Hillmyer, M.A.; Tirrell, M.; Bates, F.S.; Lodge, T.P.; von Meerwall, E.D. Block copolymer self-diffusion in the gyroid and cylinder morphologies. Macromolecules 1998, 31, 5363–5370. [Google Scholar]
- Gawande, P.V.; LoVetri, K.; Yakandawala, N.; Romeo, T.; Zhanel, G.G.; Cvitkovitch, D.G.; Madhyastha, S. Antibiofilm activity of sodium bicarbonate, sodium metaperiodate and SDS combination against dental unit waterline-associated bacteria and yeast. J. Appl. Microbiol 2008, 105, 986–992. [Google Scholar]
- Allesen-Holm, M.; Barken, K.B.; Yang, L.; Klausen, M.; Webb, J.S.; Kjelleberg, S.; Molin, S.; Givskov, M.; Tolker-Nielsen, T. A characterization of DNA release in Pseudomonas aeruginosa cultures and biofilms. Mol. Microbiol 2006, 59, 1114–1128. [Google Scholar]
- Furukawa, S.; Akiyoshi, Y.; Komoriya, M.; Ogihara, H.; Morinaga, Y. Removing Staphylococcus aureus and Escherichia coli biofilms on stainless steel by cleaning-in-place (CIP) cleaning agents. Food Control 2010, 21, 669–672. [Google Scholar]
- Yang, L.; Hu, Y.; Liu, Y.; Zhang, J.; Ulstrup, J.; Molin, S. Distinct roles of extracellular polymeric substances in Pseudomonas aeruginosa biofilm development. Environ. Microbiol 2011, 13, 1705–1717. [Google Scholar]
- Yang, L.; Nilsson, M.; Gjermansen, M.; Givskov, M.; Tolker-Nielsen, T. Pyoverdine and PQS mediated subpopulation interactions involved in Pseudomonas aeruginosa biofilm formation. Mol. Microbiol 2009, 74, 1380–1392. [Google Scholar]
- Dubey, G.P.; Ben-Yehuda, S. Intercellular nanotubes mediate bacterial communication. Cell 2011, 144, 590–600. [Google Scholar]
- Achtman, M.; Morelli, G.; Schwuchow, S. Cell-cell interactions in conjugating Escherichia coli: Role of F pili and fate of mating aggregates. J. Bacteriol 1978, 135, 1053–1061. [Google Scholar]
- Ghigo, J.M. Natural conjugative plasmids induce bacterial biofilm development. Nature 2001, 412, 442–445. [Google Scholar]
- Li, L.; Schulte, L.; Clausen, L.D.; Hansen, K.M.; Jonsson, G.E.; Ndoni, S. Gyroid nanoporous membranes with tunable permeability. ACS Nano 2011, 5, 7754–7766. [Google Scholar]
- Gill, S.R.; Fouts, D.E.; Archer, G.L.; Mongodin, E.F.; Deboy, R.T.; Ravel, J.; Paulsen, I.T.; Kolonay, J.F.; Brinkac, L.; Beanan, M.; et al. Insights on evolution of virulence and resistance from the complete genome analysis of an early methicillin-resistant Staphylococcus aureus strain and a biofilm-producing methicillin-resistant Staphylococcus epidermidis strain. J. Bacteriol 2005, 187, 2426–2438. [Google Scholar]
- Yang, L.; Barken, K.B.; Skindersoe, M.E.; Christensen, A.B.; Givskov, M.; Tolker-Nielsen, T. Effects of iron on DNA release and biofilm development by Pseudomonas aeruginosa. Microbiology 2007, 153, 1318–1328. [Google Scholar]
- Reisner, A.; Haagensen, J.A.; Schembri, M.A.; Zechner, E.L.; Molin, S. Development and maturation of Escherichia coli K-12 biofilms. Mol. Microbiol 2003, 48, 933–946. [Google Scholar]
- Heydorn, A.; Nielsen, A.T.; Hentzer, M.; Sternberg, C.; Givskov, M.; Ersboll, B.K.; Molin, S. Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 2000, 146, 2395–2407. [Google Scholar]




© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, L.; Molin, S.; Yang, L.; Ndoni, S. Sodium Dodecyl Sulfate (SDS)-Loaded Nanoporous Polymer as Anti-Biofilm Surface Coating Material. Int. J. Mol. Sci. 2013, 14, 3050-3064. https://doi.org/10.3390/ijms14023050
Li L, Molin S, Yang L, Ndoni S. Sodium Dodecyl Sulfate (SDS)-Loaded Nanoporous Polymer as Anti-Biofilm Surface Coating Material. International Journal of Molecular Sciences. 2013; 14(2):3050-3064. https://doi.org/10.3390/ijms14023050
Chicago/Turabian StyleLi, Li, Soeren Molin, Liang Yang, and Sokol Ndoni. 2013. "Sodium Dodecyl Sulfate (SDS)-Loaded Nanoporous Polymer as Anti-Biofilm Surface Coating Material" International Journal of Molecular Sciences 14, no. 2: 3050-3064. https://doi.org/10.3390/ijms14023050