Regulation of Erythropoietin Receptor Activity in Endothelial Cells by Different Erythropoietin (EPO) Derivatives: An in Vitro Study
Abstract
:1. Introduction
2. Results
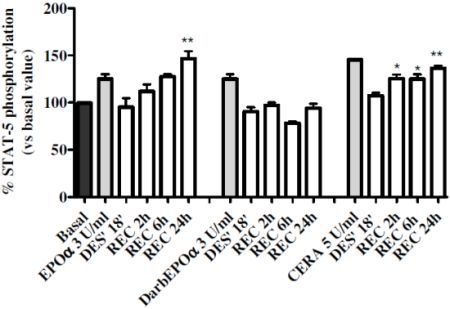
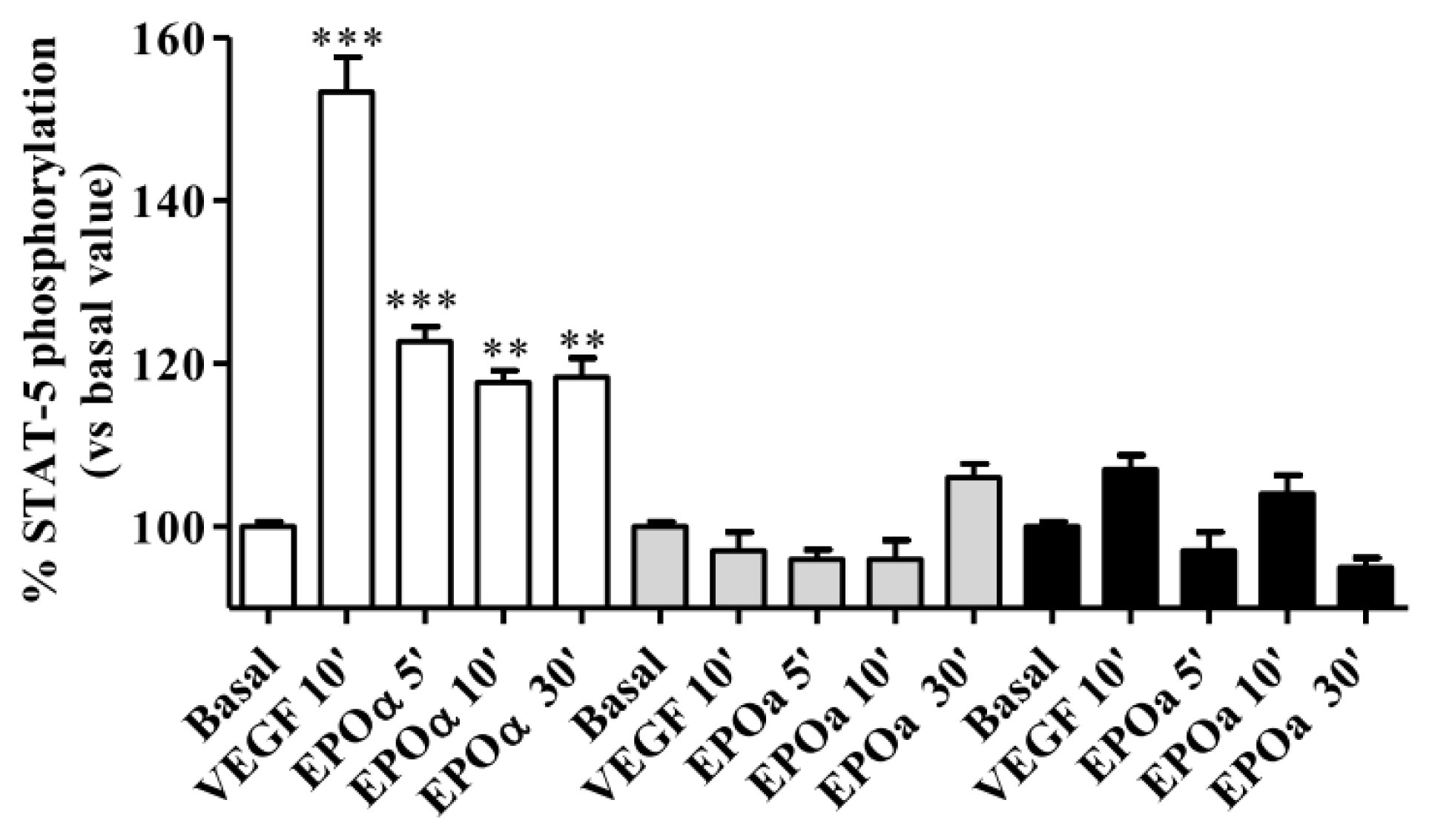
2.1. Optimization of Experimental Conditions
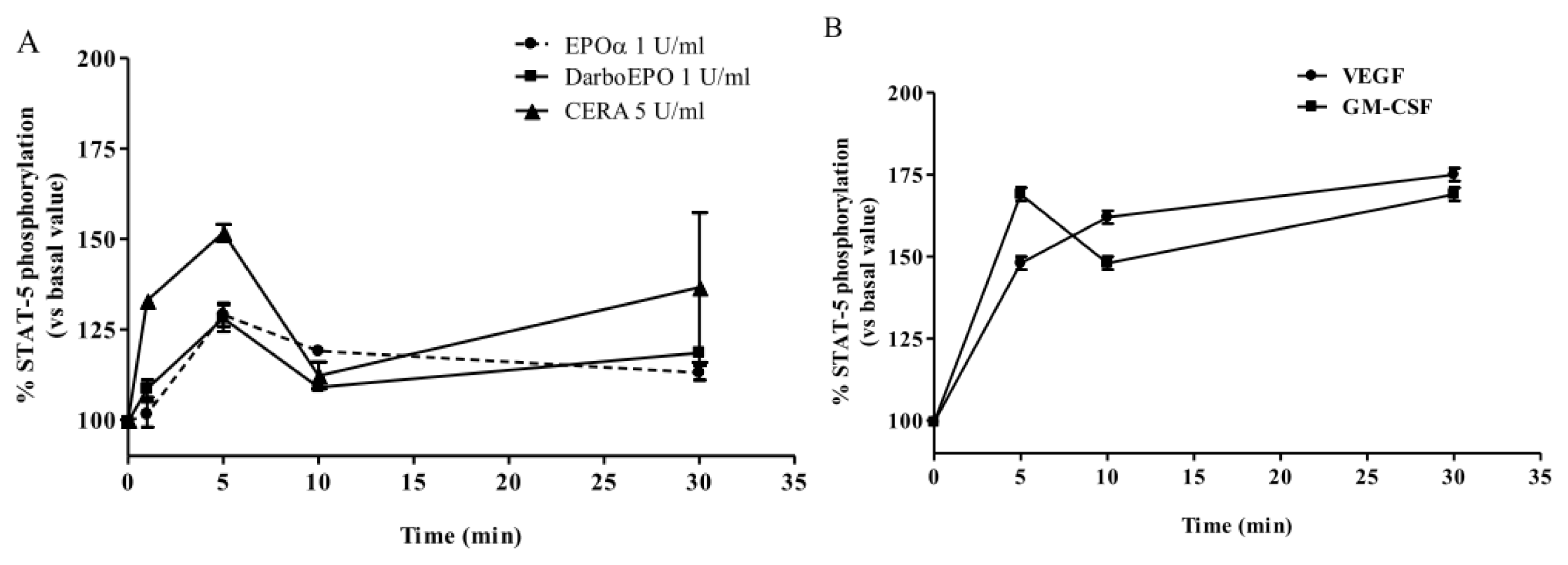
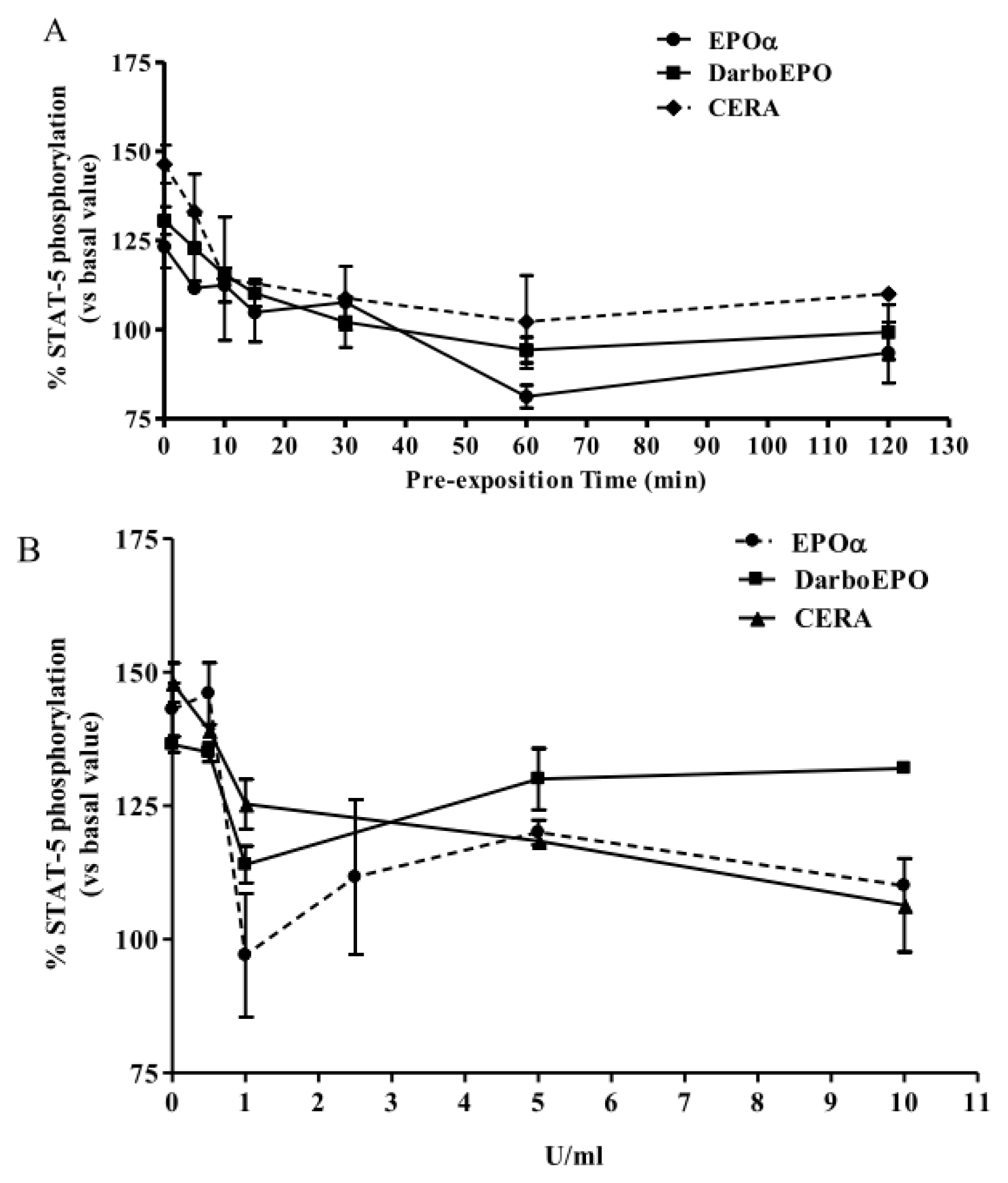
2.2. EPOα-, DarbEPO- and CERA-Mediated STAT-5 Phosphorylation: Concentration- and Time-Dependence
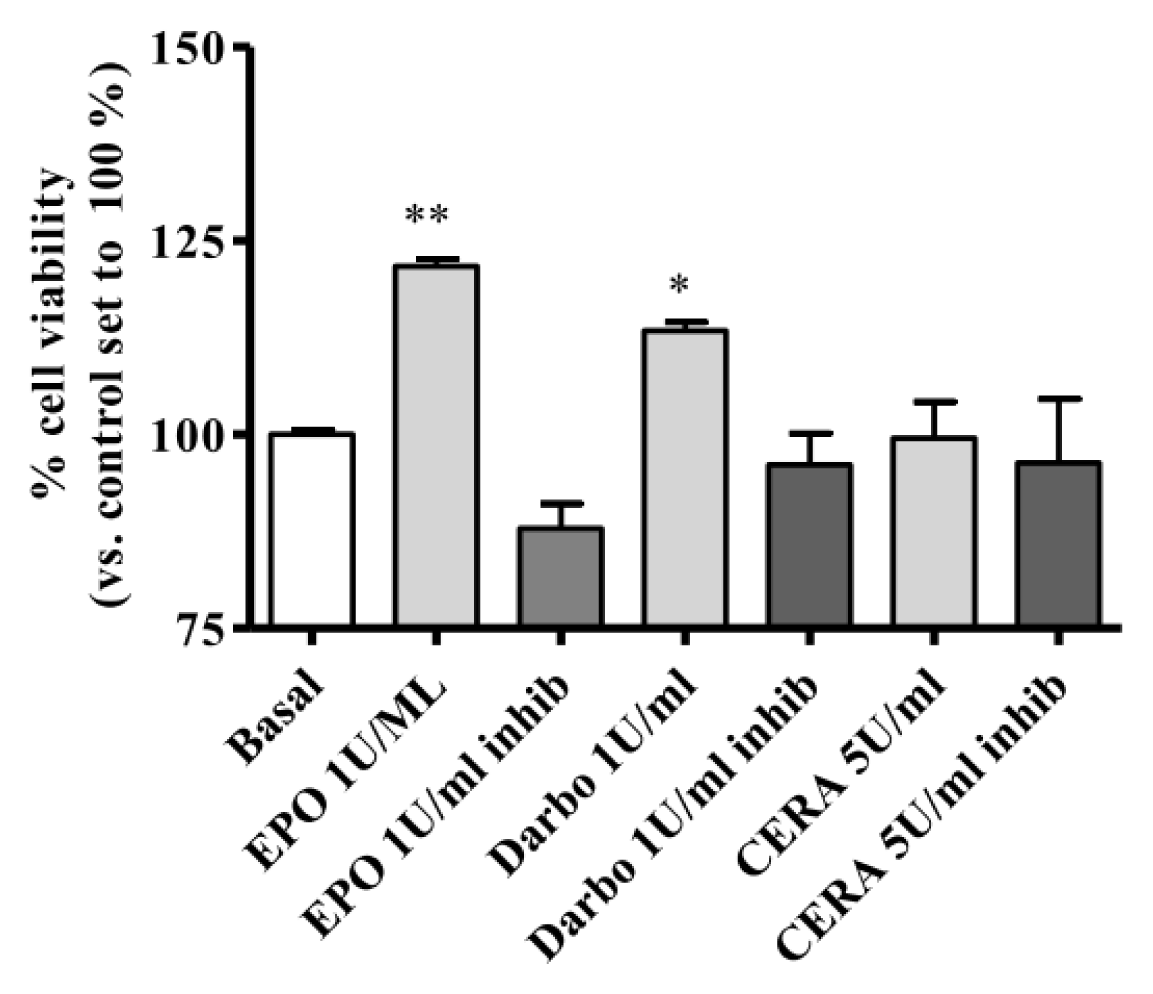
2.3. Effect of EPOα, DarbEPO and CERA on HUVEC Viability
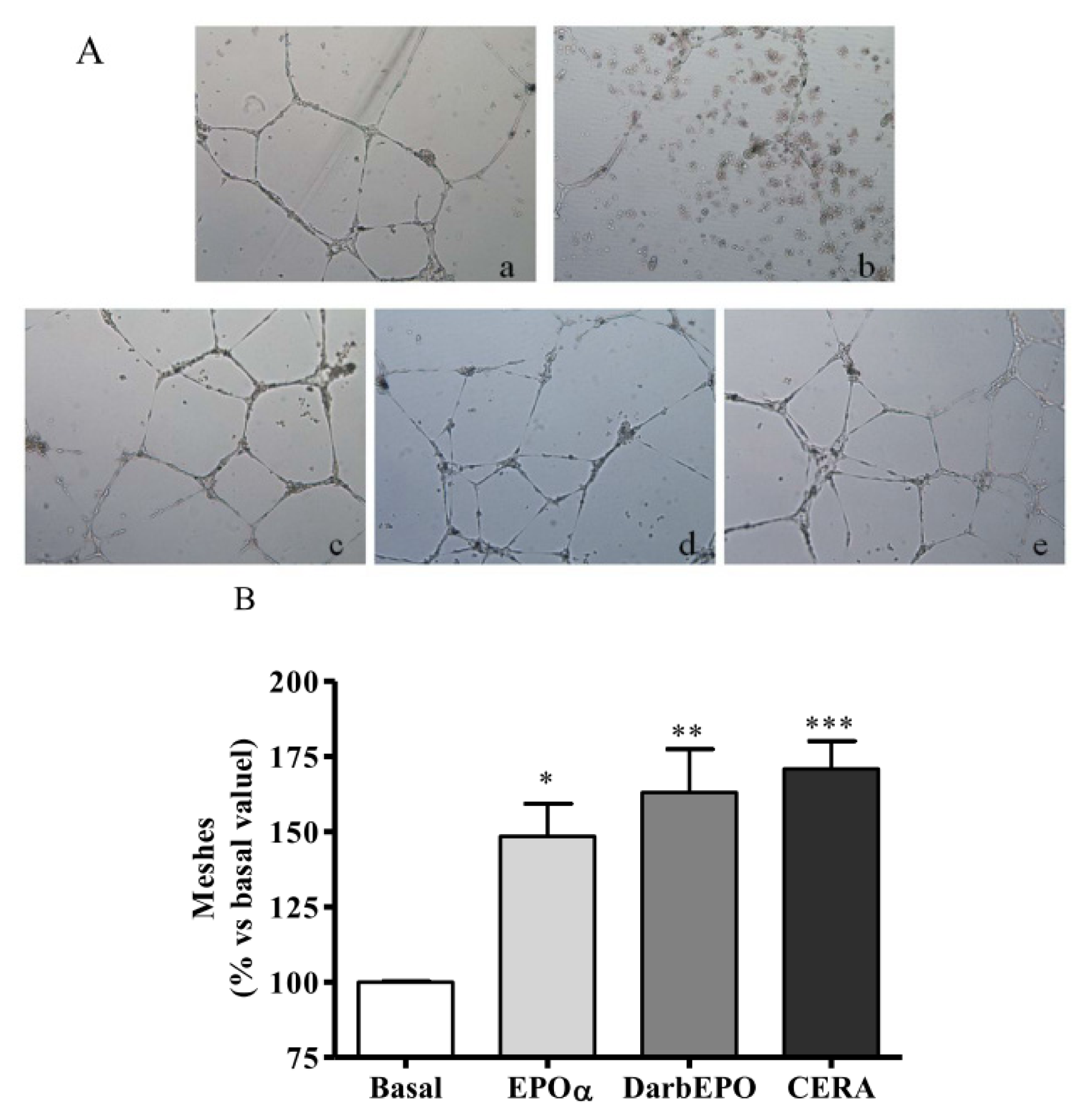
2.4. EPOα-, DarbEPO- and CERA-Mediated Angiogenesis of HUVECs In Vitro
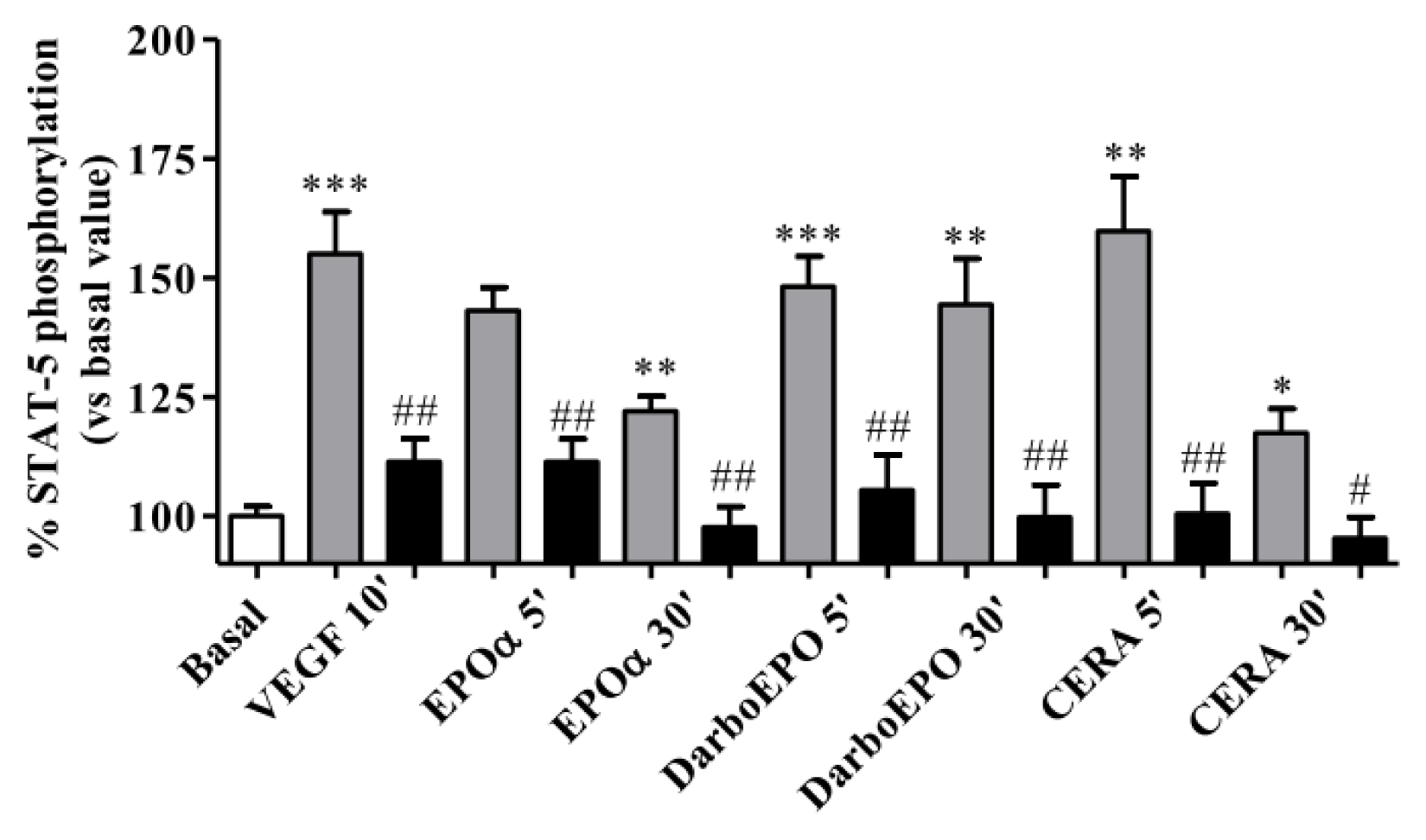
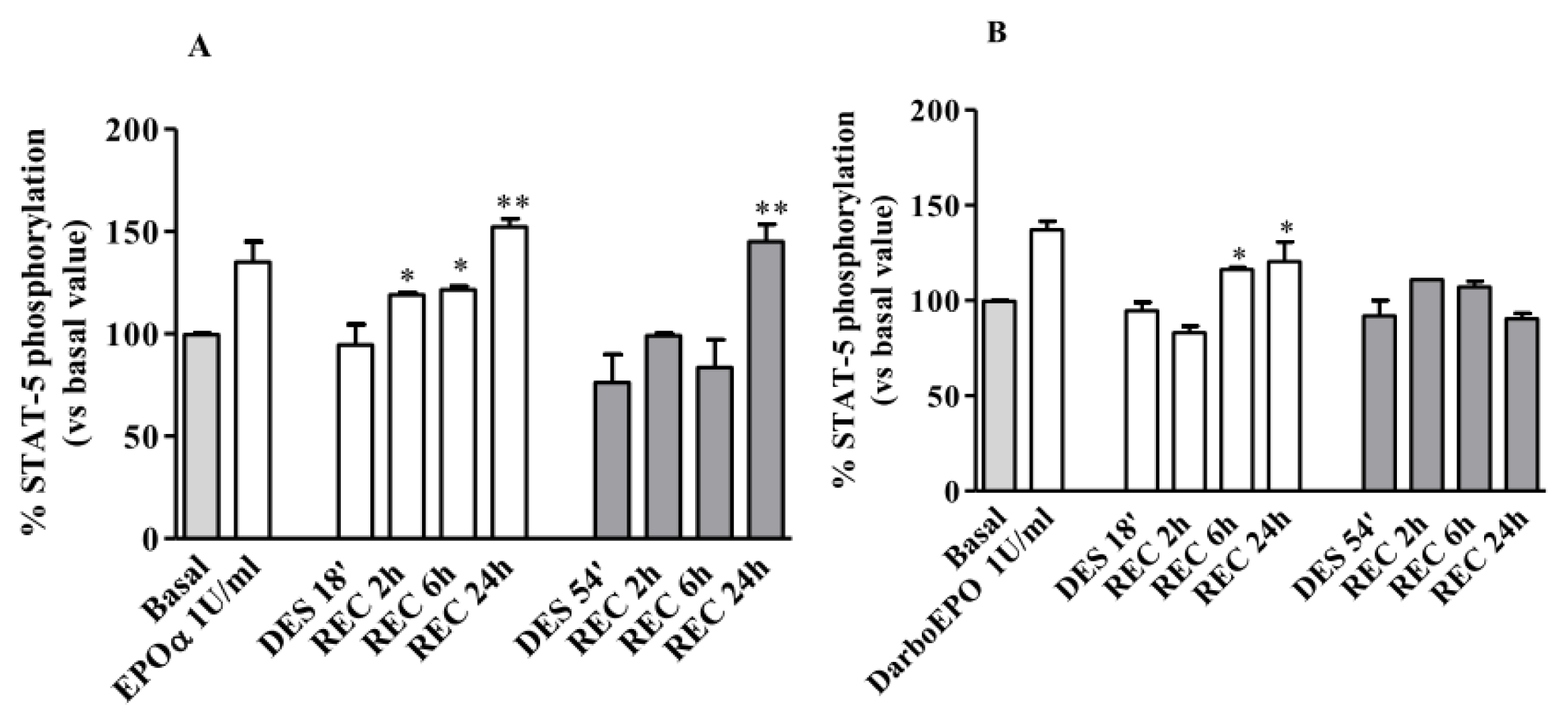
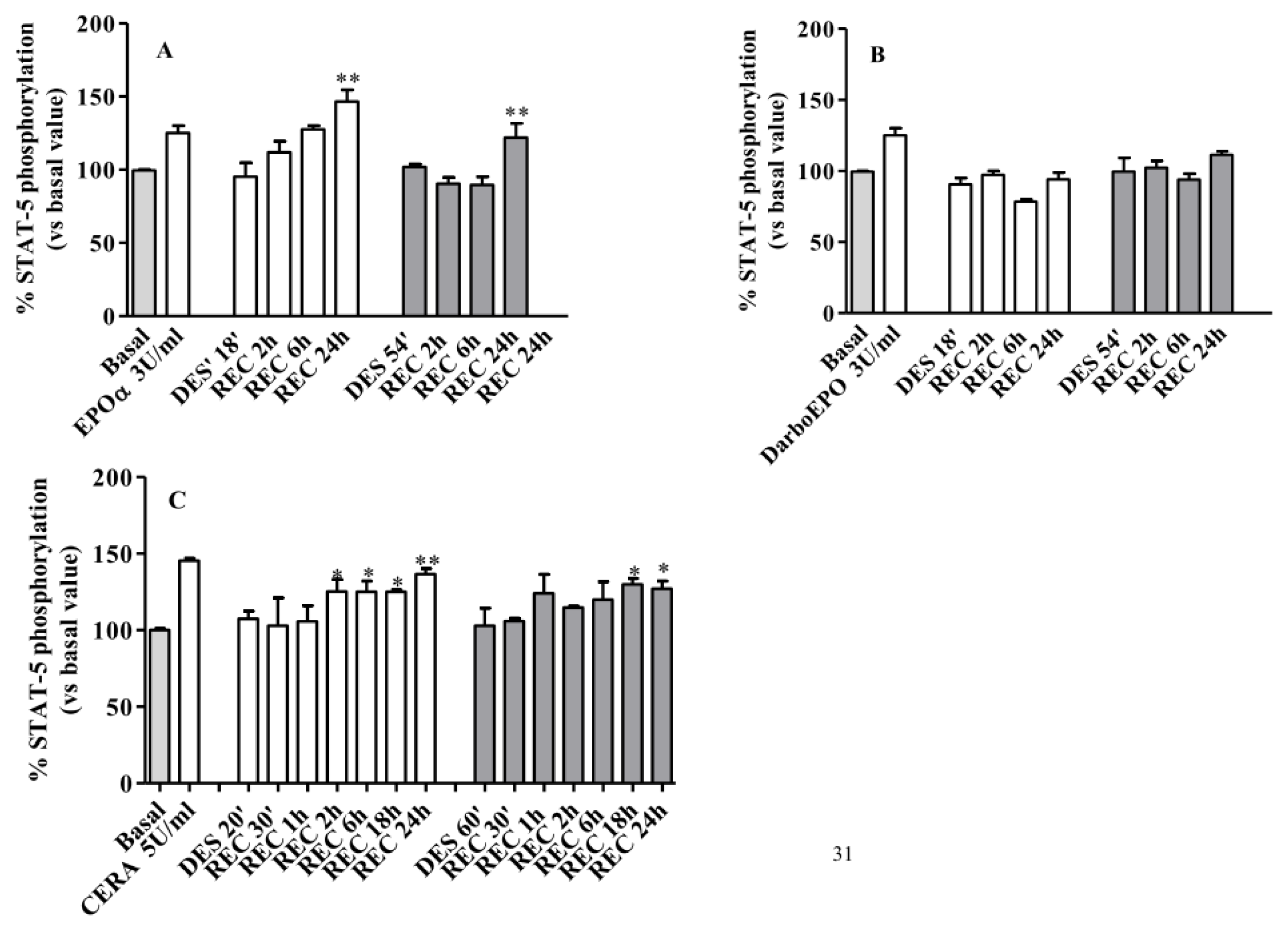
2.5. EPOR Desensitization
2.6. EPOR Resensitization
2.7. Effects of ESAs on EPOR Expression Levels
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Lines and Drug Treatments
4.3. STAT-5 Phosphorylation Assay
4.4. EPO-Mediated STAT-5 Phosphorylation: Concentration and Time Dependence
4.5. Viability Assay
4.6. In Vitro Angiogenesis Model
4.7. EPOR Desensitization
4.8. EPOR Resensitization Experiments
4.9. EPOR mRNA Expression
4.10. Statistics
Acknowledgements
Abbreviations
| EPO | erythropoietin |
| EPOR | erythropoietin receptor |
| DarbEPO | darbepoetin alpha |
| CERA | continuous erythropoietin receptor activator |
| ESAs | erythropoiesis-stimulating agents |
| HUVEC | human umbilical vein endothelial cells |
References
- Erslev, A. Humoral regulation of red cell production. Blood 1953, 8, 349–357. [Google Scholar]
- Lai, P.H.; Everett, R.; Wang, F.F.; Arakawa, T.; Goldwasser, E. Structural characterization of human erythropoietin. J. Biol. Chem 1986, 261, 3116–3121. [Google Scholar]
- Bazan, J.F. Structural design and molecular evolution of a cytokine receptor superfamily. Proc. Natl. Acad. Sci. USA 1990, 87, 6934–6938. [Google Scholar]
- Lin, C.S.; Lim, S.K.; D’Agati, V.; Costantini, F. Differential effects of an erythropoietin receptor gene disruption on primitive and definitive erythropoiesis. Genes Dev 1996, 10, 154–164. [Google Scholar]
- Anagnostou, A.; Liu, Z.; Steiner, M.; Chin, K.; Lee, E.S.; Kessimian, N.; Noguchi, C.T. Erythropoietin receptor mRNA expression in human endothelial cells. Proc. Natl. Acad. Sci. USA 1994, 91, 3974–3978. [Google Scholar]
- Morishita, E.; Masuda, S.; Nagao, M.; Yasuda, Y.; Sasaki, R. Erythropoietin receptor is expressed in rat hippocampal and cerebral cortical neurons and erythropoietin prevents in vitro glutamate-induced neuronal death. Neuroscience 1997, 76, 105–116. [Google Scholar]
- Banerjee, D.; Rodriguez, M.; Nag, M.; Adamson, J.W. Exposure of endothelial cells to recombinant human erythropoietin induces nitric oxide synthase activity. Kidney Int 2000, 57, 1895–1904. [Google Scholar]
- Stasko, J.; Drouet, L.; Soria, C.; Mazoyer, E.; Caen, J.; Kubisz, P. Erythropoietin and granulocyte colony-stimulating factor increase plasminogen activator inhibitor-1 release in HUVEC culture. Thromb. Res 2002, 105, 161–164. [Google Scholar]
- Congote, L.F.; Sadvakassova, G.; Dobocan, M.C.; Difalco, M.R.; Li, Q. Erythropoietin-dependent endothelial proteins: Potential use against erythropoietin resistance. Cytokine 2010, 51, 113–118. [Google Scholar]
- Noguchi, C.T.; Wang, L.; Rogers, H.M.; Teng, R.; Jia, Y. Survival and proliferative roles of erythropoietin beyond the erythroid lineage. Expert Rev. Mol. Med 2008, 10, e36. [Google Scholar]
- Heeschen, C.; Aicher, A.; Lehmann, R.; Fichtlscherer, S.; Vasa, M.; Urbich, C.; Mildner-Rihm, C.; Martin, H.; Zeiher, A.M.; Dimmeler, S. Erythropoietin is a potent physiologic stimulus for endothelial progenitor cell mobilization. Blood 2003, 102, 1340–1346. [Google Scholar]
- Santhanam, A.V.; d’Uscio, L.V.; Peterson, T.E.; Katusic, Z.S. Activation of endothelial nitric oxide synthase is critical for erythropoietin-induced mobilization of progenitor cells. Peptides 2008, 29, 1451–1455. [Google Scholar]
- Westenbrink, B.D.; Lipsic, E.; van der Meer, P.; van der Harst, P.; Oeseburg, H.; Du Marchie Sarvaas, G.J.; Koster, J.; Voors, A.A.; van Veldhuisen, D.J.; van Gilst, W.H.; et al. Erythropoietin improves cardiac function through endothelial progenitor cell and vascular endothelial growth factor mediated neovascularization. Eur. Heart. J. 2007, 28, 2018–2027. [Google Scholar]
- Brines, M.; Cerami, A. Discovering erythropoietin’s extra-hematopoietic functions: Biology and clinical promise. Kidney Int 2006, 70, 246–250. [Google Scholar]
- Haller, H.; Christel, C.; Dannenberg, L.; Thiele, P.; Lindschau, C.; Luft, F.C. Signal transduction of erythropoietin in endothelial cells. Kidney Int 1996, 50, 481–488. [Google Scholar]
- Yang, J.; Xiao, Z.; Li, T.; Gu, X.; Fan, B. Erythropoietin promotes the growth of pituitary adenomas by enhancing angiogenesis. Int. J. Oncol 2012, 40, 1230–1237. [Google Scholar]
- Kawachi, K.; Iso, Y.; Sato, T.; Wakabayashi, K.; Kobayashi, Y.; Takeyama, Y.; Suzuki, H. Effects of erythropoietin on angiogenesis after myocardial infarction in porcine. Heart Vessels 2012, 27, 79–88. [Google Scholar]
- Carlini, R.G.; Reyes, A.A.; Rothstein, M. Recombinant human erythropoietin stimulates angiogenesis in vitro. Kidney Int 1995, 47, 740–745. [Google Scholar]
- Ahn, S.; Min, S.K.; Min, S.I.; Suh, J.H.; Kim, S.J.; Ha, J. Early sustained injections of erythropoietin improve angiogenesis and restoration of perfusion in the ischemic mouse hindlimb. J. Korean Med. Sci 2012, 27, 1073–1078. [Google Scholar]
- Ribatti, D.; Presta, M.; Vacca, A.; Ria, R.; Giuliani, R.; Dell’Era, P.; Nico, B.; Roncali, L.; Dammacco, F. Human erythropoietin induces a pro-angiogenic phenotype in cultured endothelial cells and stimulates neovascularization in vivo. Blood 1999, 93, 2627–2636. [Google Scholar]
- Constantinescu, S.N.; Ghaffari, S.; Lodish, H.F. The erythropoietin receptor: Structure, activation and intracellular signal transduction. Trends Endocrinol. Metab 1999, 10, 18–23. [Google Scholar]
- Fliser, D.; Bahlmann, F.H. Erythropoietin and the endothelium—A promising link? Eur. J. Clin. Invest 2008, 38, 457–461. [Google Scholar]
- Marzo, F.; Lavorgna, A.; Coluzzi, G.; Santucci, E.; Tarantino, F.; Rio, T.; Conti, E.; Autore, C.; Agati, L.; Andreotti, F. Erythropoietin in heart and vessels: Focus on transcription and signaling pathways. J. Thromb. Thrombolysis 2008, 26, 183–187. [Google Scholar]
- Janmaat, M.L.; Heerkens, J.L.; de Bruin, A.M.; Klous, A.; de Waard, V.; de Vries, C.J. Erythropoietin accelerates smooth muscle cell-rich vascular lesion formation in mice through endothelial cell activation involving enhanced PDGF-BB release. Blood 2010, 115, 1453–1460. [Google Scholar]
- Fusté, B.; Serradell, M.; Escolar, G.; Cases, A.; Mazzara, R.; Castillo, R.; Ordinas, A.; Díaz-Ricart, M. Erythropoietin triggers a signaling pathway in endothelial cells and increases the thrombogenicity of their extracellular matrices in vitro. Thromb Haemostasis 2002, 88, 678–685. [Google Scholar]
- Su, K.H.; Shyue, S.K.; Kou, Y.R.; Ching, L.C.; Chiang, A.N.; Yu, Y.B.; Chen, C.Y.; Pan, C.C.; Lee, T.S. β Common receptor integrates the erythropoietin signaling in activation of endothelial nitric oxide synthase. J. Cell Physiol 2011, 226, 3330–3339. [Google Scholar]
- Locatelli, F.; Olivares, J.; Walker, R.; Wilkie, M.; Jenkins, B.; Dewey, C.; Gray, S.J. Novel erythropoiesis stimulating protein for treatment of anemia in chronic renal insufficiency. Kidney Int 2001, 60, 741–747. [Google Scholar]
- Elliott, S.; Pham, E.; Macdougall, I.C. Erythropoietins: A common mechanism of action. Exp. Hematol 2008, 36, 1573–1584. [Google Scholar]
- Klingmuller, U.; Lorenz, U.; Cantley, L.C.; Neel, B.C.; Lodish, H.C. Specific recruitment of SH-PTP1 to the erythropoietin receptor causes inactivation of JAK2 and termination of proliferative signals. Cell 1995, 80, 729–738. [Google Scholar]
- Mufson, R.A.; Gesner, T.G. Binding and internalization of recombinant human erythropoietin in murine erythroid precursor cells. Blood 1987, 69, 1485–1490. [Google Scholar]
- Levin, I.; Cohen, J.; Supino-Rosin, L.; Yoshimura, A.; Watowich, S.S.; Neumann, D. Identification of a cytoplasmic motif in the erythropoietin receptor required for receptor internalization. FEBS Lett 1998, 427, 164–170. [Google Scholar]
- Zuo, G.; Guan, T.; Chen, D.; Li, C.; Jiang, R.; Luo, C.; Hu, X.; Wang, Y.; Wang, J. Total saponins of Panax ginseng induces K562 cell differentiation by promoting internalization of the erythropoietin receptor. Am. J. Chin. Med 2009, 37, 747–757. [Google Scholar]
- Sulahian, R.; Cleaver, O.; Huang, L.J. Ligand-induced EpoR internalization is mediated by JAK2 and p85 and is impaired by mutations responsible for primary familial and congenital polycythemia. Blood 2009, 113, 5287–5297. [Google Scholar]
- Flint-Ashtamker, G.; Eisen-Lev, R.; Cohen, J.; Huang, L.J.; Neumann, D. Amino acid residues 268–276 of the erythropoietin receptor contain an endocytosis motif and are required for erythropoietin-mediated proliferation. FEBS Lett 2002, 518, 189–194. [Google Scholar]
- Walrafen, P.; Verdier, F.; Kadri, Z.; Chrétien, S.; Lacombe, C.; Mayeux, P. Both proteasomes and lysosomes degrade the activated erythropoietin receptor. Blood 2005, 105, 600–608. [Google Scholar]
- Verdier, F.; Walrafen, P.; Hubert, N.; Chretien, S.; Gisselbrecht, S.; Lacombe, C.; Mayeux, P. Proteasomes regulate the duration of erythropoietin receptor activation by controlling downregulation of cell surface receptors. J. Biol. Chem 2000, 275, 18375–18381. [Google Scholar]
- Yen, C.H.; Yang, Y.C.; Ruscetti, S.K.; Kirken, R.A.; Dai, R.M.; Li, C.C. Involvement of the ubiquitin-proteasome pathway in the degradation of nontyrosine kinase-type cytokine receptors of IL-9, IL-2 and erythropoietin. J. Immunol 2000, 165, 6372–6380. [Google Scholar]
- Müller, J.; Sperl, B.; Reindl, W.; Kiessling, A.; Berg, T. Discovery of chromone-based inhibitors of the transcription factor STAT5. ChemBioChem 2008, 9, 723–727. [Google Scholar]
- Małyszko, J.S.; Małyszko, J.; Pawlak, K.; Pawlak, K.; Buczko, W.; Myśliwiec, M. Importance of serotonergic mechanisms in the thrombotic complications in hemodialyzed patients treated with erythropoietin. Nephron 2000, 84, 305–311. [Google Scholar]
- Borawski, J.; Mysliwiec, M. Effects of recombinant erythropoietin therapy on circulating endothelial markers in hemodialysis patients. Clin. Appl. Thromb. Hemostasis 2002, 8, 77–84. [Google Scholar]
- Staško, J.; Kubisz, P. Increased soluble thrombomodulin in hemodialysis patients with long-term erythropoietin treatment: What does it reflect? Clin. Appl. Thromb. Hemostasis 2003, 9, 349–352. [Google Scholar]
- Taylor, J.E.; Belch, J.J.F.; McLaren, M.; Henderson, J.S.; Stewart, W.K. Effect of erythropoietin therapy and withdrawal on blood coagulation and fibrinolysis in hemodialysis patients. Kidney Int 1993, 44, 182–190. [Google Scholar]
- Pawlak, K.; Pawlak, D.; Mysliwiec, M. Long-term erythropoietin therapy does not affect endothelial markers, coagulation activation and oxidative stress in haemodialyzed patients. Thromb. Res 2007, 120, 797–803. [Google Scholar]
- Bahlmann, F.H.; Song, R.; Boehm, S.M.; Mengel, M.; von Wasielewski, R.; Lindschau, C.; Kirsch, T.; de Groot, K.; Laudeley, R.; Niemczyk, E.; et al. Low-dose therapy with the long-acting erythropoietin analogue darbepoetin alpha persistently activates endothelial Akt and attenuates progressive organ failure. Circulation 2004, 110, 1006–1012. [Google Scholar]
- Kanellakis, P.; Pomilio, G.; Agrotis, A.; Gao, X.; Du, X.J.; Curtis, D.; Bobik, A. Darbepoetin-mediated cardioprotection after myocardial infarction involves multiple mechanisms independent of erythropoietin receptor-common beta-chain heteroreceptor. Br. J. Pharmacol 2010, 160, 2085–2096. [Google Scholar]
- Baker, J.E.; Kozik, D.; Hsu, A.K.; Fu, X.; Tweddell, J.S.; Gross, GJ. Darbepoetin alfa protects the rat heart against infarction: Dose-response, phase of action and mechanisms. J. Cardiovasc. Pharmacol 2007, 49, 337–345. [Google Scholar]
- Opie, L.H. Erythropoietin as a cardioprotective agent: Down, but not out. Heart 2011, 97, 1537–1539. [Google Scholar]
- Minamino, T.; Toba, K.; Higo, S.; Nakatani, D.; Hikoso, S.; Umegaki, M.; Yamamoto, K.; Sawa, Y.; Aizawa, Y.; Komuro, I. Design and rationale of low-dose erythropoietin in patients with ST-segment elevation myocardial infarction (EPO-AMI-II study): A randomized controlled clinical trial. Cardiovasc. Drugs Ther 2012, 26, 409–416. [Google Scholar]
- Inrig, J.K.; Bryskin, S.K.; Patel, U.D.; Arcasoy, M.; Szczech, L.A. Association between high-dose erythropoiesis-stimulating agents, inflammatory biomarkers and soluble erythropoietin receptors. BMC Nephrol 2011, 12, 67–77. [Google Scholar]
- Sawyer, S.T.; Penta, K. Association of JAK2 and STAT5 with erythropoietin receptors. Role of receptor phosphorylation in erythropoietin signal transduction. J. Biol. Chem 1996, 271, 32430–32437. [Google Scholar]
- Ji, Y.Q.; Zhang, Y.Q.; Li, M.Q.; Du, M.R.; Wei, W.W.; Li, D.J. EPO improves the proliferation and inhibits apoptosis of trophoblast and decidual stromal cells through activating STAT-5 and inactivating p38 signal in human early pregnancy. Int. J. Clin. Exp. Pathol 2011, 4, 765–774. [Google Scholar]
- Mezentsev, A.; Merks, R.M.; O’Riordan, E.; Chen, J.; Mendelev, N.; Goligorsky, M.S.; Brodsky, S.V. Endothelial microparticles affect angiogenesis in vitro: Role of oxidative stress. Am. J. Physiol. Heart Circ. Physiol 2005, 289, H1106–H1114. [Google Scholar]
- Aranda, E.; Owen, G.I. A semi-quantitative assay to screen for angiogenic compounds and compounds with angiogenic potential using the EA.hy926 endothelial cell line. Biol. Res 2009, 42, 377–389. [Google Scholar]
- El-Komy, M.H.; Schmidt, R.L.; Widness, J.A.; Veng-Pedersen, P. Differential pharmacokinetic analysis of in vivo erythropoietin receptor interaction with erythropoietin and continuous erythropoietin receptor activator in sheep. Biopharm. Drug Dispos 2011, 32, 276–288. [Google Scholar]
- Bulut, G.B.; Sulahian, R.; Ma, Y.; Chi, N.W.; Huang, L.J. Ubiquitination regulates the internalization, endolysosomal sorting and signaling of the erythropoietin receptor. J. Biol. Chem 2011, 286, 6449–6457. [Google Scholar]
- Suzuki, N.; Ohneda, O.; Takahashi, S.; Higuchi, M.; Mukai, H.Y.; Nakahata, T.; Imagawa, S.; Yamamoto, M. Erythroid-specific expression of the erythropoietin receptor rescued its null mutant mice from lethality. Blood 2002, 100, 2279–2288. [Google Scholar]
- Meyer, L.; Deau, B.; Forejtníková, H.; Duménil, D.; Margottin-Goguet, F.; Lacombe, C.; Mayeux, P.; Verdier, F. beta-Trcp mediates ubiquitination and degradation of the erythropoietin receptor and controls cell proliferation. Blood 2007, 109, 5215–5222. [Google Scholar]
- Macdougall, I.C. Novel erythropoiesis-stimulating agents: A new era in anemia management. Clin. J. Am. Soc. Nephrol 2008, 3, 200–207. [Google Scholar]
- Elliott, S.; Egrie, J.; Browne, J.; Lorenzini, T.; Busse, L.; Rogers, N.; Ponting, I. Control of rHuEPO biological activity: The role of carbohydrate. Exp. Hematol 2004, 32, 1146–1155. [Google Scholar]
- Gross, A.W.; Lodish, H.F. Cellular trafficking and degradation of erythropoietin and novel erythropoiesis stimulating protein (NESP). J. Biol. Chem 2006, 281, 2024–2032. [Google Scholar]
- Macdougall, I.C. CERA (Continuous erythropoietin receptor activator): A new erythropoiesis-stimulating agent for the treatment of anemia. Curr. Hematol. Rep 2005, 4, 436–440. [Google Scholar]
- Macdougall, I.C.; Robson, R.; Opatrna, S.; Liogier, X.; Pannier, A.; Jordan, P.; Dougherty, F.C.; Reigner, B. Pharmacokinetics and pharmacodynamics of intravenous and subcutaneous continuous erythropoietin receptor activator (CERA) in patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol 2006, 1, 1211–1215. [Google Scholar]
- Jarsch, M.; Brandt, M.; Lanzendörfer, M.; Haselbeck, A. Comparative erythropoietin receptor binding kinetics of CERA and epoetin-beta determined by surface plasmon resonance and competition binding assay. Pharmacology 2008, 81, 63–69. [Google Scholar]
- Egrie, J.C.; Browne, J.K. Development and characterization of novel erythropoiesis stimulating protein (NESP). Nephrol. Dial. Transplant 2001, 16, 3–13. [Google Scholar]


© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Trincavelli, M.L.; Da Pozzo, E.; Ciampi, O.; Cuboni, S.; Daniele, S.; Abbracchio, M.P.; Martini, C. Regulation of Erythropoietin Receptor Activity in Endothelial Cells by Different Erythropoietin (EPO) Derivatives: An in Vitro Study. Int. J. Mol. Sci. 2013, 14, 2258-2281. https://doi.org/10.3390/ijms14022258
Trincavelli ML, Da Pozzo E, Ciampi O, Cuboni S, Daniele S, Abbracchio MP, Martini C. Regulation of Erythropoietin Receptor Activity in Endothelial Cells by Different Erythropoietin (EPO) Derivatives: An in Vitro Study. International Journal of Molecular Sciences. 2013; 14(2):2258-2281. https://doi.org/10.3390/ijms14022258
Chicago/Turabian StyleTrincavelli, Maria Letizia, Eleonora Da Pozzo, Osele Ciampi, Serena Cuboni, Simona Daniele, Maria Pia Abbracchio, and Claudia Martini. 2013. "Regulation of Erythropoietin Receptor Activity in Endothelial Cells by Different Erythropoietin (EPO) Derivatives: An in Vitro Study" International Journal of Molecular Sciences 14, no. 2: 2258-2281. https://doi.org/10.3390/ijms14022258