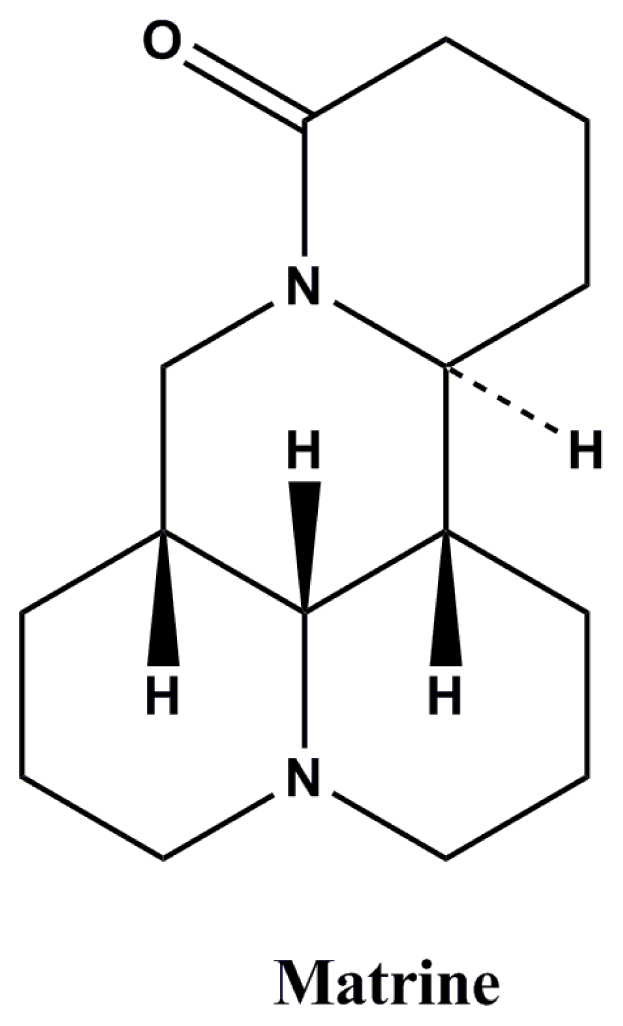
Antiepileptic Potential of Matrine via Regulation the Levels of Gamma-Aminobutyric Acid and Glutamic Acid in the Brain
Abstract
:1. Introduction
2. Results and Discussion
2.1. Result of the Maximal Electroshock Test
2.2. Result of Pentylenetetrazole-Induced Seizure Test
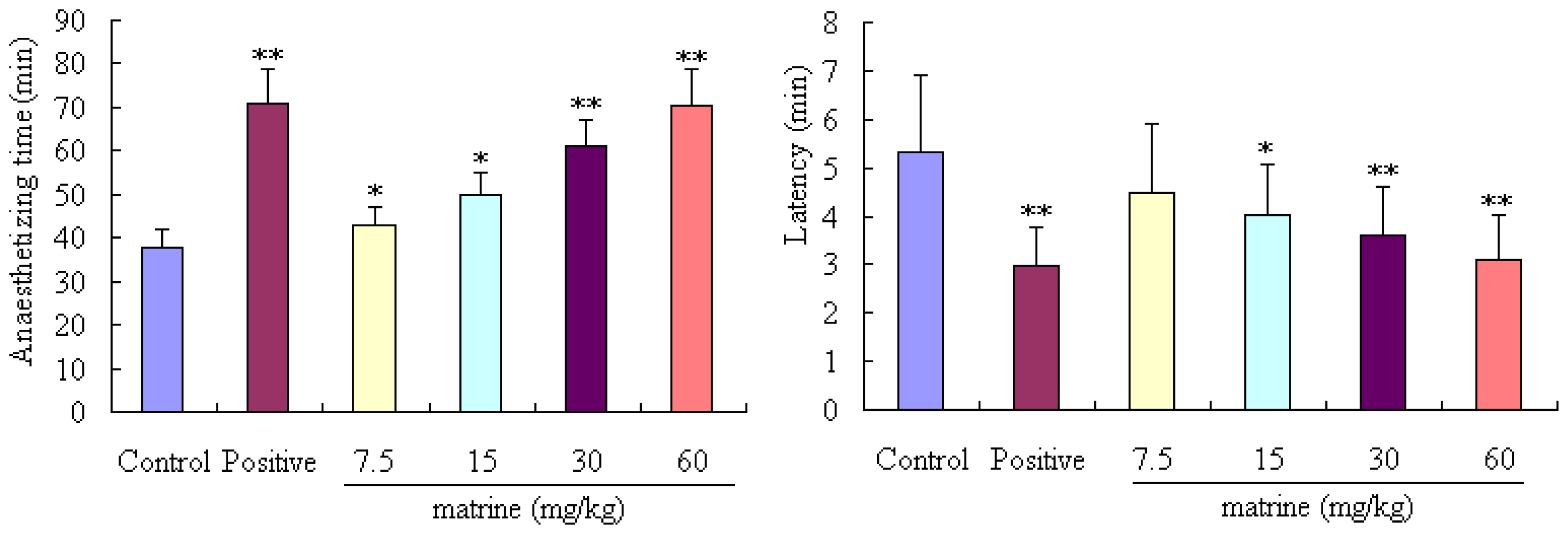
2.3. Result of Pentobarbital Sodium-Induced Anaesthetizing Time in Mice
2.4. Results of Mouse Locomotor Activity Tests
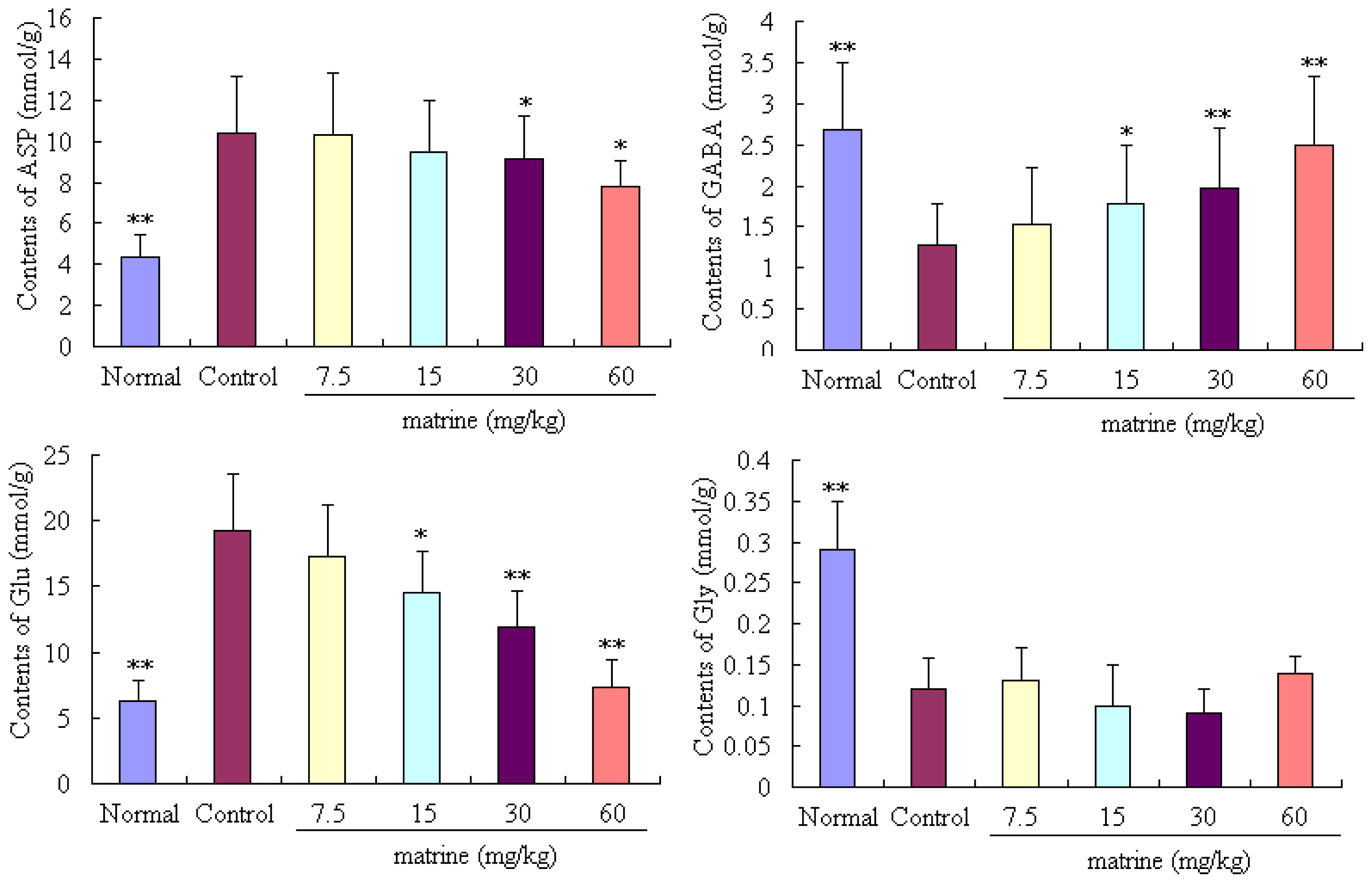
2.5. Results of the Contents of Asp, GABA, Glu, and Gly in Mouse Brain
2.6. Effect of Matrine on Expressions of GAD 65, GABAA, GABAB in the Brain of Epileptic Rats
2.7. Discussion
3. Experimental Section
3.1. Animals
3.2. Drugs and Chemicals
3.3. Protocols
3.4. Maximal Electroshock (MES)-Induced Seizures in Mice
3.5. Pentylenetetrazole (PTZ)-Induced Convulsion in Mice
3.6. Pentobarbital Sodium-Induced Anaesthetizing Time in Mice
3.7. Measurement of Locomotor Activity
3.8. Measurement of the Contents of Asp, GABA, Glu, and Gly in Brain
3.9. Chronic Epileptic Rat Seizure Induced by PTZ and Western Blotting
3.10. Statistical Analysis
4. Conclusions
Conflicts of Interest
References
- Choudhary, N.; Bijjem, K.R.V.; Kalia, A.N. Antiepileptic potential of flavonoids fraction from the leaves of Anisomeles malabarica. J. Ethnopharmacol 2011, 135, 238–242. [Google Scholar]
- Radhakrishnan, K. Challenges in the management of epilepsy in resource-poor conntries. Nat. Rev. Neurol 2009, 5, 323–330. [Google Scholar]
- Privitera, M. Current challenges in the management of epilepsy. Am. J. Manag. Care 2011, 7, S195–S203. [Google Scholar]
- Fukata, Y.; Adesnik, H.; Iwanaga, T.; Bredt, D.S.; Nicoll, R.A.; Fukata, M. Epilepsy-related ligand/receptor complex LGI1 and ADAM22 regulate synaptic transmission. Science 2006, 313, 1792–1795. [Google Scholar]
- Perucca, P.; Mula, M. Antiepileptic drug effects on mood and behavior: Molecular targets. Epilepsy Behav 2013, 26, 440–449. [Google Scholar]
- Qiu, J. Traditional medicine: A culture in the balance. Nature 2007, 448, 126–128. [Google Scholar]
- Peng, W.; Han, T.; Xin, W.B.; Zhang, X.G.; Zhang, Q.Y.; Jia, M. Comparative research of chemical constituents and bioactivities between petroleum ether extracts of the aerial part and the rhizome of Atractylodes macrocephala. Med. Chem. Res 2011, 20, 146–151. [Google Scholar]
- Xu, M.; Yang, L.; Hong, L.Z.; Zhao, X.Y.; Zhang, H.L. Direct protection of neurons and astrocytes by matrine via inhibition of the NF-κB signaling pathway contributes to neuroprotection against focal cerebral ischemia. Brain Res 2012, 1454, 48–64. [Google Scholar]
- Yin, L.L.; Zhu, X.Z. The involvement of central cholinergic system in (+)-matrine-induced antinociception in mice. Pharmacol. Biochem. Behav 2005, 80, 419–425. [Google Scholar]
- Dai, W.H.; Qian, L.W.; Wang, L.L. Toxicity studies in mice of matrine and oxymatrine. Anhui Med. Pharm. J 2012, 16, 904–905. [Google Scholar]
- Sayyah, M.; Nadjafni, L.; Kamalinejad, M. Anticonvulsant activity and chemical composition of Artemisia dracunculus L. essential oil. J. Ethnopharmacol 2004, 94, 283–287. [Google Scholar]
- Garzón-De la Mora, P.; García-López, P.M.; García-Estrada, J.; Navarro-Ruíz, A.; Villanueva-Michel, T.; Villarreal-de Puga, L.M. Casimiroa edulis seed extracts show anticonvulsive properties in rats. J. Ethnopharmacol 1999, 68, 275–282. [Google Scholar]
- Mikael, E.; Pedersen, M.E.; Henrik, T.; Vestergaard, H.T.; Suzanne, L.; Hansen, S.L.; Bah, S.; Diallo, D.; Anna, K.; Jäger, A.K. Pharmacological screening of Malian medicinal plants used against epilepsy and convulsions. J. Ethnopharmacol 2009, 121, 472–475. [Google Scholar]
- Awad, R.; Ahmed, F.; Bourbonnais-Spear, N.; Mullally, M.; Ta, C.A.; Tang, A.; Merali, Z.; Maquin, P.; Caal, F.; Cal, V.; et al. Ethnopharmacology of Q’eqchi’ Maya antiepileptic and anxiolytic plants: Effects on the GABAergic system. J. Ethnopharmacol 2009, 125, 257–264. [Google Scholar]
- Ha, J.H.; Lee, D.U.; Lee, J.T.; Kim, J.S.; Yong, C.S.; Kim, J.A.; Ha, J.S.; Huh, K. 4-Hydroxybenzaldehyde from Gastrodia elata B1. is active in the antioxidation and GABAergic neuromodulation of the rat brain. J. Ethnopharmacol 2000, 73, 329–333. [Google Scholar]
- Yang, D.B.; Wang, L.; Huang, M.; Yu, J.M.; Wang, X.M.; Luo, J.M. Effects of pretreatment with repetitive transcranial magnetic stimulation on development of seizures induced by pilocarpine and expression of GAD65 in rat hippocampus. Chin. J. Clin. Neurosci 2009, 17, 337–340. [Google Scholar]
- Mandegary, A.; Arab-Nozari, M.; Ramiar, H.; Sharififar, F. Anticonvulsant activity of the essential oil and methanolic extract of Bunium persicum (Boiss). B. Fedtsch. J. Ethnopharmacol 2012, 140, 447–451. [Google Scholar]
- Lüttjohann, A.; Fabene, P.F.; van Luijtelaar, G. A revised Racine’s scale for PTZ-induced seizures in rats. Physiol. Behav 2009, 98, 579–586. [Google Scholar]
- Racine, R.J. Modification of seizure activity by electrical stimulation: II. Motor seizure. Electroencephalogr. Clin. Neurophysiol 1972, 32, 281–294. [Google Scholar]
- Gupta, G.; Kazmi, I.; Afzal, M.; Rahman, M.; Saleem, S.; Ashraf, Md.S.; Khusroo, M.J.; Nazeer, K.; Ahmed, S.; Mujeeb, M.; et al. Sedative, antiepileptic and antipsychotic effects of Viscum album L. (Loranthaceae) in mice and rats. J. Ethnopharmacol 2012, 141, 810–816. [Google Scholar]


| Treatment | Dose | Convulsions (n) | Inhibition (%) | Tonic-clonus (%) | Death (n) | Mortality (%) |
|---|---|---|---|---|---|---|
| Control | 20 mL/kg | 15 | 0 | 100 | 3 | 20 |
| Positive | 20 mL/kg | 0 | 100 ** | 0 ** | 0 | 0 |
| Matrine | 7.5 mg/kg | 12 | 20 | 73.3 | 2 | 13.3 |
| 15 mg/kg | 8 | 46.7 * | 40 ** | 0 | 0 | |
| 30 mg/kg | 4 | 73.3 ** | 20 ** | 0 | 0 | |
| 60 mg/kg | 0 | 100 ** | 0 ** | 0 | 0 | |
| Treatment | Dose | Convulsions (n) | Inhibition (%) | Death (n) | Mortality (%) | ||
|---|---|---|---|---|---|---|---|
| IV–V | V | IV–V | V | ||||
| Control | 20 mL/kg | 15 | 15 | 0 | 0 | 15 | 100 |
| Positive | 4 mL/kg | 0 | 0 | 100 ** | 100 ** | 0 | 0 ** |
| Matrine | 7.5 mg/kg | 8 | 7 | 46.7 * | 53.3 * | 9 | 60 * |
| 15 mg/kg | 7 | 5 | 53.3 ** | 66.7 ** | 6 | 40 * | |
| 30 mg/kg | 5 | 3 | 66.7 ** | 80 ** | 4 | 26.6 ** | |
| 60 mg/kg | 4 | 2 | 73.3 ** | 86.6 ** | 2 | 13.3 ** | |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xiang, J.; Jiang, Y. Antiepileptic Potential of Matrine via Regulation the Levels of Gamma-Aminobutyric Acid and Glutamic Acid in the Brain. Int. J. Mol. Sci. 2013, 14, 23751-23761. https://doi.org/10.3390/ijms141223751
Xiang J, Jiang Y. Antiepileptic Potential of Matrine via Regulation the Levels of Gamma-Aminobutyric Acid and Glutamic Acid in the Brain. International Journal of Molecular Sciences. 2013; 14(12):23751-23761. https://doi.org/10.3390/ijms141223751
Chicago/Turabian StyleXiang, Jun, and Yugang Jiang. 2013. "Antiepileptic Potential of Matrine via Regulation the Levels of Gamma-Aminobutyric Acid and Glutamic Acid in the Brain" International Journal of Molecular Sciences 14, no. 12: 23751-23761. https://doi.org/10.3390/ijms141223751




