Colorectal Laterally Spreading Tumors by Computed Tomographic Colonography
Abstract
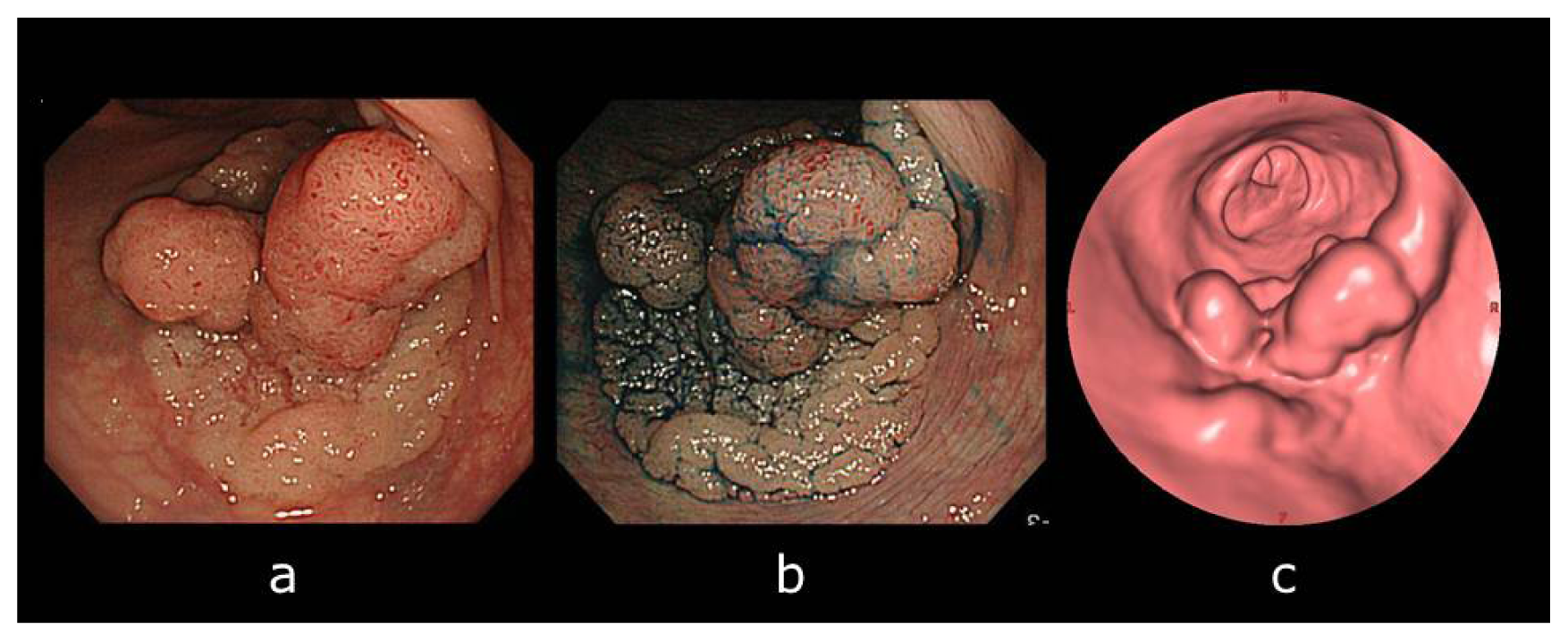
:1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Overall Visibility Rate with CTC
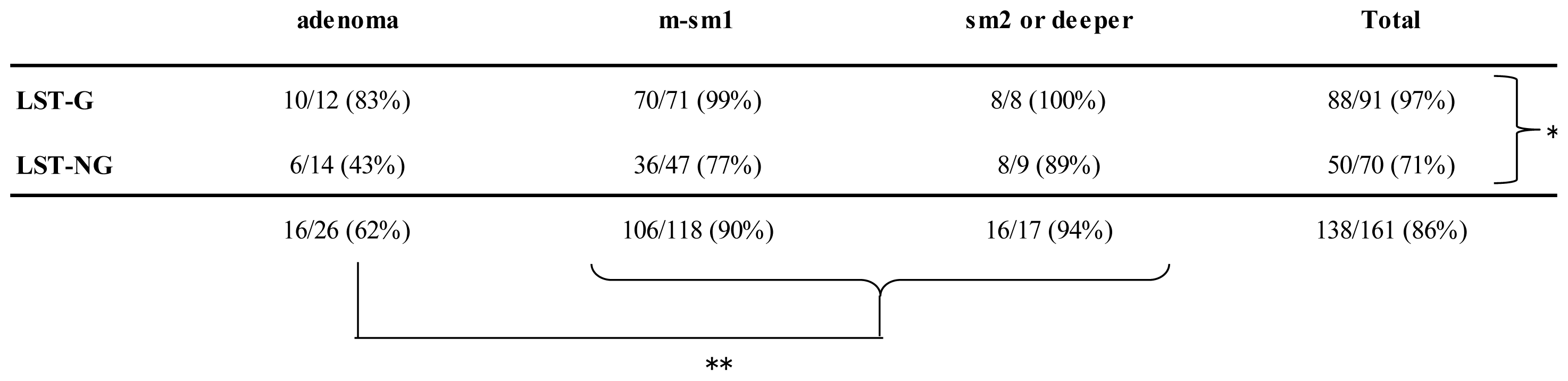
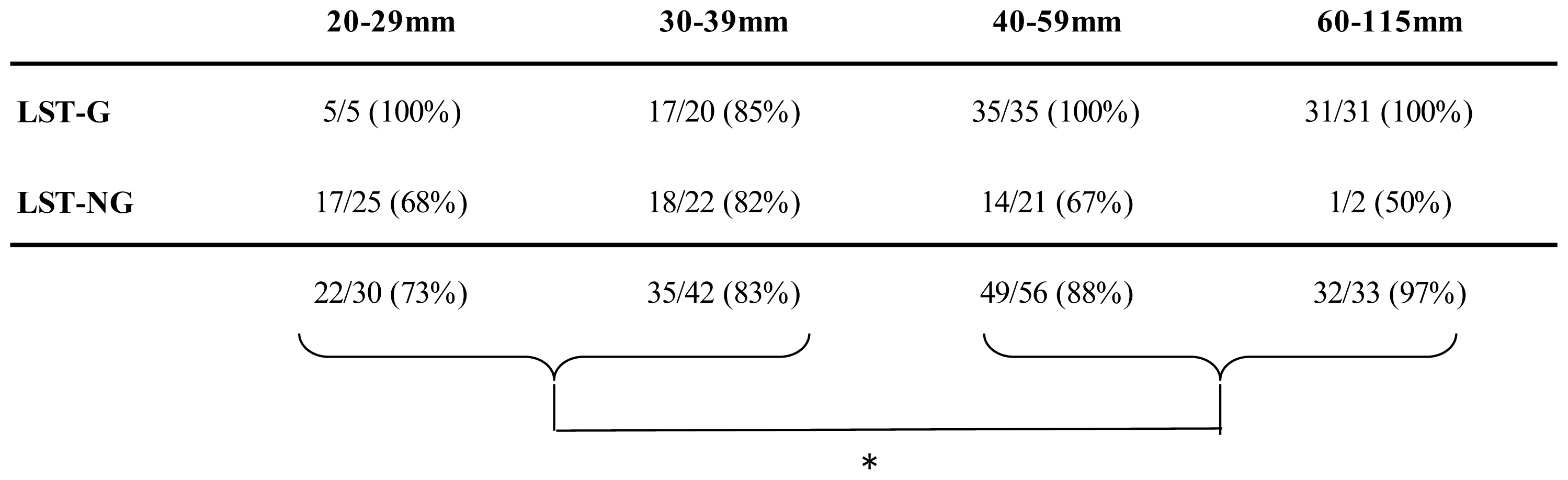
2.3. Visibility Rates According to Factors
3. Discussion
4. Materials and Methods
4.1. Study Patients
4.2. Endoscopic Procedure
4.3. CTC Procedure
4.4. Pathological Examination
4.5. Statistical Analysis
5. Conclusions
Conflicts of Interest
References
- Winawer, S.J.; Zauber, A.G.; O’Brien, M.J.; Ho, M.N.; Gottlieb, L.; Sternberg, S.S.; Waye, J.D.; Bond, J.; Schapiro, M.; Stewart, E.T.; et al. Randomized comparison of surveillance intervals after colonoscopic removal of newly diagnosed adenomatous polyps. The National Polyp Study Workgroup. N. Engl. J. Med 1993, 328, 901–906. [Google Scholar]
- Winawer, S.J.; Zauber, A.G.; Ho, M.N.; O’Brien, M.J.; Gottlieb, L.S.; Sternberg, S.S.; Waye, J.D.; Schapiro, M.; Bond, J.H.; Panish, J.F.; et al. Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup. N. Engl. J. Med 1993, 329, 1977–1981. [Google Scholar]
- Banerjee, S.; van Dam, J. CT colonography for colon cancer screening. Gastrointest. Endosc 2006, 63, 121–133. [Google Scholar]
- Johnson, C.D.; Chen, M.H.; Toledano, A.Y.; Heiken, J.P.; Dachman, A.; Kuo, M.D.; Menias, C.O.; Siewert, B.; Cheema, J.I.; Obregon, R.G.; et al. Accuracy of CT colonography for detection of large adenomas and cancers. N. Engl. J. Med 2008, 359, 1207–1217. [Google Scholar]
- Pickhardt, P.J.; Taylor, A.J.; Kim, D.H.; Reichelderfer, M.; Gopal, D.V.; Pfau, P.R. Screening for colorectal neoplasia with CT colonography: Initial experience from the 1st year of coverage by third-party payers. Radiology 2006, 241, 417–425. [Google Scholar]
- Fidler, J.L.; Johnson, C.D.; MacCarty, R.L.; Welch, T.J.; Hara, A.K.; Harmsen, W.S. Detection of flat lesions in the colon with CT colonography. Abdom. Imaging 2002, 27, 292–300. [Google Scholar]
- Galdino, G.M.; Yee, J. Carpet lesion on CT colonography: A potential pitfall. AJR Am. J. Roentgenol 2003, 180, 1332–1334. [Google Scholar]
- Pickhardt, P.J.; Nugent, P.A.; Choi, J.R.; Schindler, W.R. Flat colorectal lesions in asymptomatic adults: Implications for screening with CT virtual colonoscopy. AJR Am. J. Roentgenol 2004, 183, 1343–1347. [Google Scholar]
- Park, S.H.; Lee, S.S.; Choi, E.K.; Kim, S.Y.; Yang, S.K.; Kim, J.H.; Ha, H.K. Flat colorectal neoplasms: Definition, importance, and visualization on CT colonography. AJR Am. J. Roentgenol 2007, 188, 953–959. [Google Scholar]
- European Society of Gastrointestinal and Abdominal Radiology CT Colonography Group Investigators. Effect of directed training on reader performance for CT colonography: Multicenter study. Radiology 2007, 242, 152–161.
- Pickhardt, P.J.; Wise, S.M.; Kim, D.H. Positive predictive value for polyps detected at screening CT colonography. Eur. Radiol 2010, 20, 1651–1656. [Google Scholar]
- Fidler, J.; Johnson, C. Flat polyps of the colon: Accuracy of detection by CT colonography and histologic significance. Abdom. Imaging 2009, 34, 157–171. [Google Scholar]
- Summers, R.M.; Liu, J.; Yao, J.; Brown, L.; Choi, J.R.; Pickhardt, P.J. Automated measurement of colorectal polyp height at CT colonography: Hyperplastic polyps are flatter than adenomatous polyps. AJR Am. J. Roentgenol 2009, 193, 1305–1310. [Google Scholar]
- Pickhardt, P.J.; Kim, D.H.; Robbins, J.B. Flat (nonpolypoid) colorectal lesions identified at CT colonography in a U.S. screening population. Acad. Radiol 2010, 17, 784–790. [Google Scholar]
- Pickhardt, P.J.; Kim, D.H. Performance of CT colonography for detecting small, diminutive, and flat polyps. Gastrointest. Endosc. Clin. N. Am 2010, 20, 209–226. [Google Scholar]
- Lostumbo, A.; Suzuki, K.; Dachman, A.H. Flat lesions in CT colonography. Abdom. Imaging 2010, 35, 578–583. [Google Scholar]
- Xu, Y.; Cai, W.; Nappi, J.; Yoshida, H. Fecal-tagging CT colonography with structure-analysis electronic cleansing for detection of colorectal flat lesions. Eur. J. Radiol 2012, 81, 1712–1716. [Google Scholar]
- Pickhardt, P.J.; Lam, V.P.; Weiss, J.M.; Kennedy, G.D.; Kim, D.H. Carpet lesions detected at CT colonography: Clinical, imaging, and pathologic features. Radiology 2013, 13. in press. [Google Scholar]
- Kudo, S.; Kashida, H.; Nakajima, T.; Tamura, S.; Nakajo, K. Endoscopic diagnosis and treatment of early colorectal cancer. World J. Surg 1997, 21, 694–701. [Google Scholar]
- Kudo, S.; Kashida, H.; Tamura, T.; Kogure, E.; Imai, Y.; Yamano, H.; Hart, A.R. Colonoscopic diagnosis and management of nonpolypoid early colorectal cancer. World J. Surg 2000, 24, 1081–1090. [Google Scholar]
- Tamura, S.; Nakajo, K.; Yokoyama, Y.; Ohkawauchi, K.; Yamada, T.; Higashidani, Y.; Miyamoto, T.; Ueta, H.; Onishi, S. Evaluation of endoscopic mucosal resection for laterally spreading rectal tumors. Endoscopy 2004, 36, 306–312. [Google Scholar]
- Uraoka, T.; Saito, Y.; Matsuda, T.; Ikehara, H.; Gotoda, T.; Saito, D.; Fujii, T. Endoscopic indications for endoscopic mucosal resection of laterally spreading tumours in the colorectum. Gut 2006, 55, 1592–1597. [Google Scholar]
- Matsuda, T.; Saito, Y.; Fu, K.I.; Uraoka, T.; Kobayashi, N.; Nakajima, T.; Ikehara, H.; Mashimo, Y.; Shimoda, T.; Murakami, Y.; et al. Does autofluorescence imaging videoendoscopy system improve the colonoscopic polyp detection rate?—A pilot study. Am. J. Gastroenterol 2008, 103, 1926–1932. [Google Scholar]
- The Paris endoscopic classification of superficial neoplastic lesions: Esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest. Endosc 2003, 58, S3–S43.
- Iinuma, G.; Miyake, M.; Uchida, K.; Arai, Y.; Muramatsu, Y.; Moriyama, N. The challenge: Detection of early-stage superficial colorectal lesions. In Virtual colonoscopy: A practical guide, 2nd ed.; Lefere, P., Gryspeerdt, S., Eds.; Springer-Verlag: Berlin, Germany, 2010; pp. 153–163. [Google Scholar]
- Matsuda, T.; Fujii, T.; Saito, Y.; Nakajima, T.; Uraoka, T.; Kobayashi, N.; Ikehara, H.; Ikematsu, H.; Fu, K.I.; Emura, F.; et al. Efficacy of the invasive/non-invasive pattern by magnifying chromoendoscopy to estimate the depth of invasion of early colorectal neoplasms. Am. J. Gastroenterol 2008, 103, 2700–2706. [Google Scholar]
- Saito, Y.; Uraoka, T.; Yamaguchi, Y.; Hotta, K.; Sakamoto, N.; Ikematsu, H.; Fukuzawa, M.; Kobayashi, N.; Nasu, J.; Michida, T.; et al. A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest. Endosc 2010, 72, 1217–1225. [Google Scholar]
- Schlemper, R.J.; Riddell, R.H.; Kato, Y.; Borchard, F.; Cooper, H.S.; Dawsey, S.M.; Dixon, M.F.; Fenoglio-Preiser, C.M.; Fléjou, J.F.; Geboes, K.; et al. The Vienna classification of gastrointestinal epithelial neoplasia. Gut 2000, 47, 251–255. [Google Scholar]

| Age | Median (Range) | 64 (32–83) years old |
| Gender | Male/Female | 86/71 |
| Macroscopic Type | LST-G/LST-NG | 91/70 lesions |
| Location | Cecum/Right Colon/Left Colon/Rectum | 17/58/39/47 lesions |
| Size | Median (Range) | 40 (20–115) mm |
| Histopathological Diagnosis | Adenoma | 26 lesions |
| Cancer | 135 lesions | |
| m | 99 lesions | |
| sm1 | 19 lesions | |
| sm2 or deeper | 17 lesions |
| Cecum | Right Colon | Left Colon | Rectum | |
|---|---|---|---|---|
| LST-G | 15/15 (100%) | 20/22 (91%) | 12/13 (92%) | 41/41 (100%) |
| LST-NG | 2/2 (100%) | 23/36 (64%) | 19/26 (73%) | 6/6 (100%) |
| 17/17 (100%) | 43/58 (74%) | 31/39 (79%) | 47/47 (100%) | |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kakugawa, Y.; Saito, Y.; Matsuda, T.; Nakajima, T.; Miyake, M.; Iinuma, G. Colorectal Laterally Spreading Tumors by Computed Tomographic Colonography. Int. J. Mol. Sci. 2013, 14, 23629-23638. https://doi.org/10.3390/ijms141223629
Kakugawa Y, Saito Y, Matsuda T, Nakajima T, Miyake M, Iinuma G. Colorectal Laterally Spreading Tumors by Computed Tomographic Colonography. International Journal of Molecular Sciences. 2013; 14(12):23629-23638. https://doi.org/10.3390/ijms141223629
Chicago/Turabian StyleKakugawa, Yasuo, Yutaka Saito, Takahisa Matsuda, Takeshi Nakajima, Mototaka Miyake, and Gen Iinuma. 2013. "Colorectal Laterally Spreading Tumors by Computed Tomographic Colonography" International Journal of Molecular Sciences 14, no. 12: 23629-23638. https://doi.org/10.3390/ijms141223629




