Different Effects of Androgen on the Expression of Fut1, Fut2, Fut4 and Fut9 in Male Mouse Reproductive Tract
Abstract
:1. Introduction
2. Results and Discussion
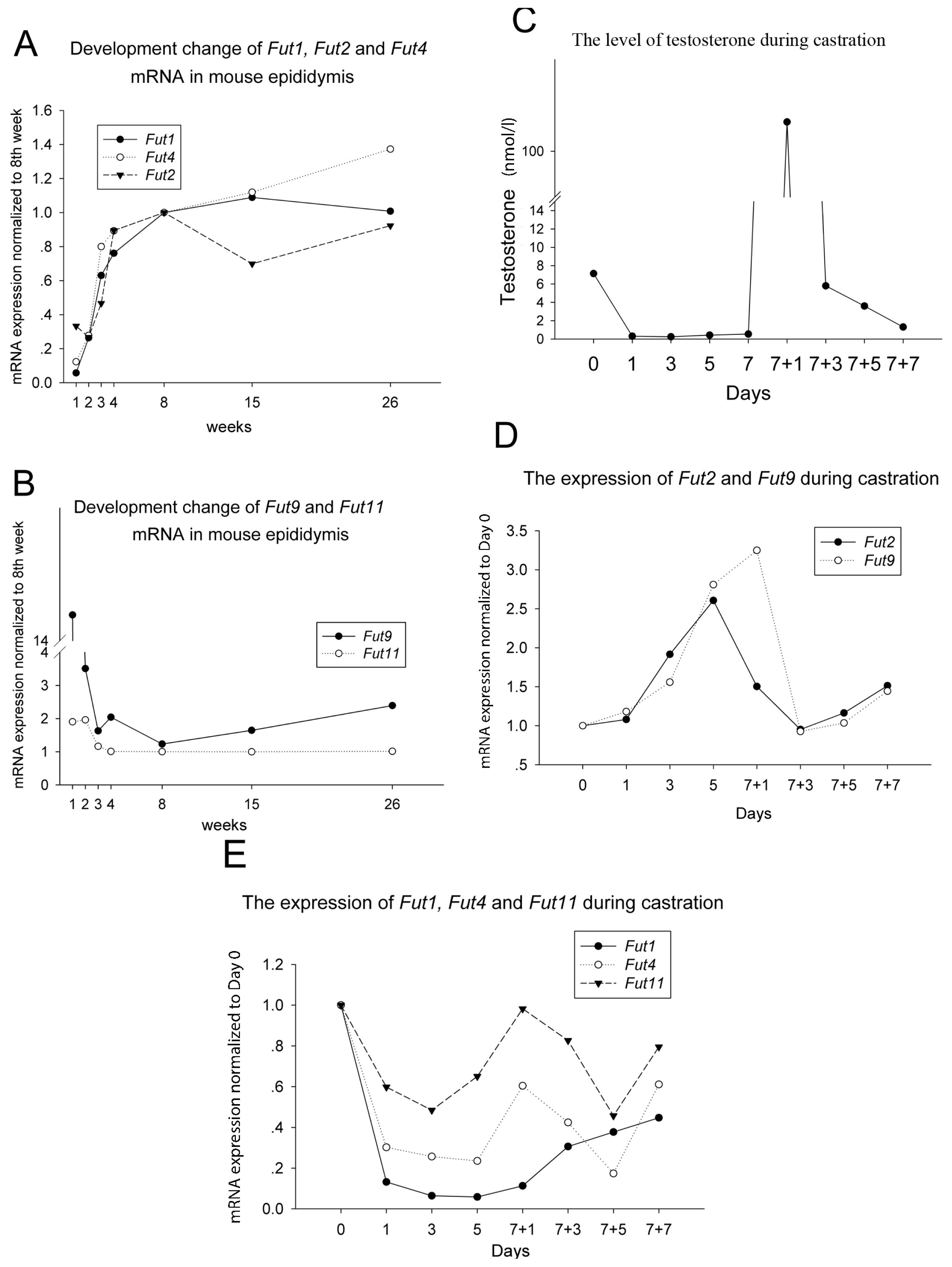
2.1. Fut1 and Fut4 mRNAs Are Upregulated, while Fut2 and Fut9 mRNAs Are Downregulated by Androgen in the Mouse Caput Epididymis
2.2. The Expression of Fut1, Fut2, Fut4 and Fut9 Are Regulated Differently by Androgen in Other Organs of the Male Mouse Reproductive Tract
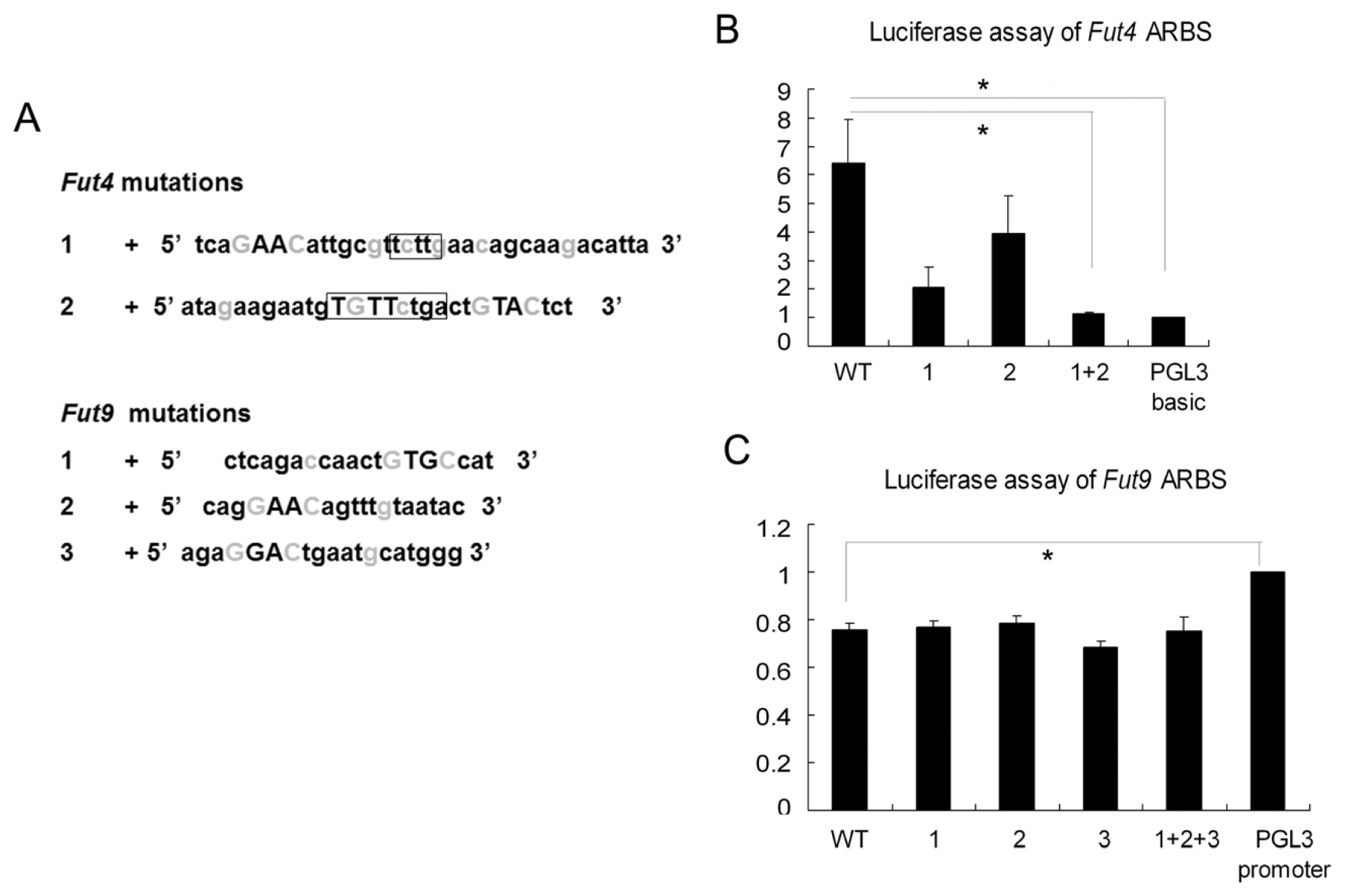
2.3. The AR-ARBS Binding Was Not the Only Reason for the Difference of Androgen Regulation between Epididymis and Prostate
2.4. Luciferase Assay Showed that Fut4 Was Upregulated by Androgen
2.5. Discussion
3. Experimental Section
3.1. Animals
3.2. Mouse Development and Castration Model
3.3. RNA Preparation and Real-Time PCR
3.4. AR-Binding Site Searching and ChIP-PCR
3.5. Luciferase Reporter Assay
4. Conclusions


| Fut1 | sense: 5′ GCATCCGCCCTCATACCT 3′ |
| anti-sense: 5′ GCCAGCGAAGACCACATCA 3′ | |
| Fut2 | sense: 5′ CCCACTTCCTCATCTTTGTCTTT 3′ |
| anti-sense: 5′ TTTGAACCGCCTGTAATTCCTT 3′ | |
| Fut4 | sense: 5′ CAGCCTGCGCTTCAACATC 3′ |
| anti-sense: 5′ CGCCTTATCCGTGCGTTCT 3′ | |
| Fut9 | sense: 5′ ATCCAAGTGCCTTATGGCTTCT 3′ |
| anti-sense: 5′ TGCTCAGGGTTCCAGTTACTCA 3′ | |
| Fut11 | sense: 5′ TAACTTGGAAGACTGCGTTACTG 3′ |
| antisense: 5′ GGCTGAGATACTAGCTCCATACC 3′ | |
| Gapdh | sense: 5′ GGTGAAGCAGGCATCTGAGGG 3′ |
| anti-sense: 5′ GGTGGGTGGTCCAGGGTTT 3′ | |
| Fut1 | 1 sense: 5′ AAAGAAGAAGAAGAAGAAGAAGAAG 3′ |
| anti-sense: 5′ CATTTTGGGCTCTGATAAAGCA 3′ | |
| 2 sense: 5′ AGGATGTTGCATCCTGGTTTGG 3′ | |
| anti-sense: 5′ CTCTGTCCCACAGCCTCACTTTGA 3′ | |
| 3 sense: 5′ ACTCTAGATCTCATCCATTCCATCA 3′ | |
| anti-sense: 5′ ACAGCCATTCACTTTGCCTGAG 3′ | |
| Fut2 | 1 sense: 5′ CCCTCTACCAAAGGAGCATTC 3′ |
| anti-sense: 5′ AACACCAAGTGGAGACGTTCAG 3′ | |
| 2 sense: 5′ AAACCTGCAATTCCAGCCAC 3′ | |
| anti-sense: 5′ CGTGAATCTTGTGTGATGACCG 3′ | |
| 3 sense: 5′ TGTTATCCGGCCCATTGTGT 3′ | |
| anti-sense: 5′ GGGTTACTGGAGCATAGCGC 3′ | |
| Fut4 | 1 sense: 5′ CGTGTGCTGGGATTACAGATACA 3′ |
| anti-sense: 5′ CAAGGACTTAACAAGGCAGGGG 3′ | |
| 2 sense: 5′ CTGCTTTGTGCTTTCTCTTTTGCT 3′ | |
| anti-sense: 5′ ATATTCATTTTCCTGAACACCCAC 3′ | |
| Fut9 | 1 sense: 5′ GACTGCATGGAGCTCTCTGGAAG 3′ |
| anti-sense: 5′ AAGATAGCCACATAACCAAACCCA 3′ | |
| 2 sense: 5′ ATGTTGGCTTTGGTTCATGTCT 3′ | |
| anti-sense: 5′ GCATTCAGTCCTCTGCTATTCAA 3′ | |
| 3 sense: 5′ TGTCTCCCAGGAACAGTTTGTAATA 3′ | |
| anti-sense: 5′ CTTGCCAGCAGTAGTTTCCTATCA 3′ | |
| Fut4 | sense: 5′ CCAGGTACCTCTATTCTCTATTGCTAC 3′ |
| antisense: 5′ CAAGAGCTCAATCTCAGATTCCACT 3′ | |
| Fut9 | sense: 5′ AGGTACCTCTCTGGAAGAAACAAAGA 3′ |
| antisense: 5′ ACCGAGCTCACGCATTTATTTTTAG 3′ |
| Fut4 | 1 sense | 5′TACCTAGGTCCTGTAAGTCAACATAGCAATCAAAATATTGCTTTATTAAATAGCAATACATTACCCATTGAGTCATCTGGCC-3′ |
| antisense | 5′GGCCAGATGACTCAATGGGTAATGTATTGCTATTTAATAAAGCAATATTTTGATTGCTATGTTGACTTACAGGACCTAGGTA-3′ | |
| 2 sense | 5′TTCACTGACCTTAAGTTGGTACAGTCAATATAAGAATGTATTATGACTATATTCTTCCTTGAGTGAAGAGGTAATG-3′ | |
| antisense | 5′CATTACCTCTTCACTCAAGGAAGAATATAGTCATAATACATTCTTATATTGACTGTACCAACTTAAGGTCAGTGAA-3′ | |
| Fut9 | 1 sense | 5′-GAGCTGTGAAAGAAGCTCAGATCAACTATGACATCTGAAAAGCACAGTCTTT-3′ |
| antisense | 5′-AAAGACTGTGCTTTTCAGATGTCATAGTTGATCTGAGCTTCTTTCACAGCTC-3′ | |
| 2 sense | 5′-ATGTTGGCTTTGGTTCATGTCTCCCAGTAAAAGTTTATAATACAATATAATTGGCAATTGAAATG-3′ | |
| antisense | 5′-CATTTCAATTGCCAATTATATTGTATTATAAACTTTTACTGGGAGACATGAACCAAAGCCAACAT-3′ | |
| 3 sense | 5′-AATAGAACTAAAGTTGAATAGCAGATGAATGAATACATGGGTCTTGAAGCTATGTGTCTC-3′ | |
| antisense | 5′-GAGACACATAGCTTCAAGACCCATGTATTCATTCATCTGCTATTCAACTTTAGTTCTATT-3′ | |
Acknowledgements
Conflicts of Interest
Reference
- Becker, D.J.; Lowe, J.B. Fucose: Biosynthesis and biological function in mammals. Glycobiology 2003, 13, 41R–53R. [Google Scholar]
- Ma, B.; Simala-Grant, J.L.; Taylor, D.E. Fucosylation in prokaryotes and eukaryotes. Glycobiology 2006, 16, 158R–184R. [Google Scholar]
- Domino, S.E.; Zhang, L.; Gillespie, P.J.; Saunders, T.L.; Lowe, J.B. Deficiency of reproductive tract α(1,2)fucosylated glycans and normal fertility in mice with targeted deletions of the Fut1 or Fut2 α(1,2)fucosyltransferase locus. Mol. Cell. Biol 2001, 21, 8336–8345. [Google Scholar]
- Homeister, J.W.; Thall, A.D.; Petryniak, B.; Maly, P.; Rogers, C.E.; Smith, P.L.; Kelly, R.J.; Gersten, K.M.; Askari, S.W.; Cheng, G.; et al. The α(1,3)fucosyltransferases Fuct-iv and Fuct-vii exert collaborative control over selectin-dependent leukocyte recruitment and lymphocyte homing. Immunity 2001, 15, 115–126. [Google Scholar]
- Kudo, T.; Kaneko, M.; Iwasaki, H.; Togayachi, A.; Nishihara, S.; Abe, K.; Narimatsu, H. Normal embryonic and germ cell development in mice lacking α 1,3-fucosyltransferase ix (Fut9) which show disappearance of stage-specific embryonic antigen 1. Mol. Cell. Biol 2004, 24, 4221–4228. [Google Scholar]
- Weston, B.W.; Hiller, K.M.; Mayben, J.P.; Manousos, G.; Nelson, C.M.; Klein, M.B.; Goodman, J.L. A cloned cd15s-negative variant of hl60 cells is deficient in expression of Fut7 and does not adhere to cytokine-stimulated endothelial cells. Eur. J. Haematol 1999, 63, 42–49. [Google Scholar]
- Mollicone, R.; Moore, S.E.; Bovin, N.; Garcia-Rosasco, M.; Candelier, J.J.; Martinez-Duncker, I.; Oriol, R. Activity, splice variants, conserved peptide motifs, and phylogeny of two new α1,3-fucosyltransferase families (Fut10 and Fut11). J. Biol. Chem 2009, 284, 4723–4738. [Google Scholar]
- Wang, C.; Huang, C.; Gu, Y.; Zhou, Y.; Zhu, Z.; Zhang, Y. Biosynthesis and distribution of Lewis X- and Lewis Y-containing glycoproteins in the murine male reproductive system. Glycobiology 2011, 21, 225–234. [Google Scholar]
- Jones, R.; Brown, C.R.; von Glos, K.I.; Parker, M.G. Hormonal regulation of protein synthesis in the rat epididymis. Characterization of androgen-dependent and testicular fluid-dependent proteins. Biochem. J 1980, 188, 667–676. [Google Scholar]
- Hinton, B.T.; Lan, Z.J.; Rudolph, D.B.; Labus, J.C.; Lye, R.J. Testicular regulation of epididymal gene expression. J. Reprod. Fertil 1998, 53, 47–57. [Google Scholar]
- Ezer, N.; Robarie, B. Androgenic Regulation of the Structure and Function of the Epididymis. In The Epididymis: From Molecules to Clinical Practice; Robaire, B., Hinton, B.T., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2002; pp. 297–316. [Google Scholar]
- Hu, S.; Yao, G.; Guan, X.; Ni, Z.; Ma, W.; Wilson, E.M.; French, F.S.; Liu, Q.; Zhang, Y. Research resource: Genome-wide mapping of in vivo androgen receptor binding sites in mouse epididymis. Mol. Endocrinol 2010, 24, 2392–2405. [Google Scholar]
- Pearl, C.A.; Berger, T.; Roser, J.F. Estrogen and androgen receptor expression in relation to steroid concentrations in the adult boar epididymis. Domest. Anim. Endocrinol 2007, 33, 451–459. [Google Scholar]
- Chauvin, T.R.; Griswold, M.D. Androgen-regulated genes in the murine epididymis. Biol. Reprod 2004, 71, 560–569. [Google Scholar]
- Barkley, M.S.; Goldman, B.D. A quantitative study of serum testosterone, sex accessory organ growth, and the development of intermale aggression in the mouse. Horm. Behav 1977, 8, 208–218. [Google Scholar]
- Genomatix. Genomatix Region Miner Release 3.2. Available online: http://www.genomatix.de/index.html (accessed on 7 November 2013).
- Sipila, P.; Krutskikh, A.; Pujianto, D.A.; Poutanen, M.; Huhtaniemi, I. Regional expression of androgen receptor coregulators and androgen action in the mouse epididymis. J. Androl 2011, 32, 711–717. [Google Scholar]
- Takagi-Morishita, Y.; Kuhara, A.; Sugihara, A.; Yamada, N.; Yamamoto, R.; Iwasaki, T.; Tsujimura, T.; Tanji, N.; Terada, N. Castration induces apoptosis in the mouse epididymis during postnatal development. Endocr. J 2002, 49, 75–84. [Google Scholar]
- Delmotte, P.; Degroote, S.; Lafitte, J.J.; Lamblin, G.; Perini, J.M.; Roussel, P. Tumor necrosis factor α increases the expression of glycosyltransferases and sulfotransferases responsible for the biosynthesis of sialylated and/or sulfated Lewis X epitopes in the human bronchial mucosa. J. Biol. Chem 2002, 277, 424–431. [Google Scholar]
- Azuma, Y.; Ito, M.; Taniguchi, A.; Matsumoto, K. Expression of cell surface Lewis X and Y antigens and Fut4 mRNA is increased in jurkat cells undergoing apoptosis. Biochim. Biophys. Acta 2004, 1672, 157–163. [Google Scholar]
- Azuma, Y.; Kurusu, Y.; Sato, H.; Higai, K.; Matsumoto, K. Increased expression of Lewis X and Y antigens on the cell surface and Fut4 mrna during granzyme b-induced jurkat cell apoptosis. Biol. Pharm. Bull 2007, 30, 655–660. [Google Scholar]
- Hu, Y.; Zhou, Z.; Xu, C.; Shang, Q.; Zhang, Y.D.; Zhang, Y.L. Androgen downregulated and region-specific expression of germ cell nuclear factor in mouse epididymis. Endocrinology 2003, 144, 1612–1619. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, C.-M.; Hu, S.-G.; Ru, Y.-F.; Yao, G.-X.; Ma, W.-B.; Gu, Y.-H.; Chu, C.; Wang, S.-L.; Zhou, Z.-M.; Liu, Q.; et al. Different Effects of Androgen on the Expression of Fut1, Fut2, Fut4 and Fut9 in Male Mouse Reproductive Tract. Int. J. Mol. Sci. 2013, 14, 23188-23202. https://doi.org/10.3390/ijms141123188
Wang C-M, Hu S-G, Ru Y-F, Yao G-X, Ma W-B, Gu Y-H, Chu C, Wang S-L, Zhou Z-M, Liu Q, et al. Different Effects of Androgen on the Expression of Fut1, Fut2, Fut4 and Fut9 in Male Mouse Reproductive Tract. International Journal of Molecular Sciences. 2013; 14(11):23188-23202. https://doi.org/10.3390/ijms141123188
Chicago/Turabian StyleWang, Chun-Mei, Shuang-Gang Hu, Yan-Fei Ru, Guang-Xin Yao, Wu-Bin Ma, Yi-Hua Gu, Chen Chu, Shou-Lin Wang, Zuo-Min Zhou, Qiang Liu, and et al. 2013. "Different Effects of Androgen on the Expression of Fut1, Fut2, Fut4 and Fut9 in Male Mouse Reproductive Tract" International Journal of Molecular Sciences 14, no. 11: 23188-23202. https://doi.org/10.3390/ijms141123188




