Environmental Enrichment Decreases Asphyxia-Induced Neurobehavioral Developmental Delay in Neonatal Rats
Abstract
:1. Introduction
2. Results and Discussion
2.1. Somatic Development
2.2. Appearance of Physical Signs
2.3. Appearance of Reflexes
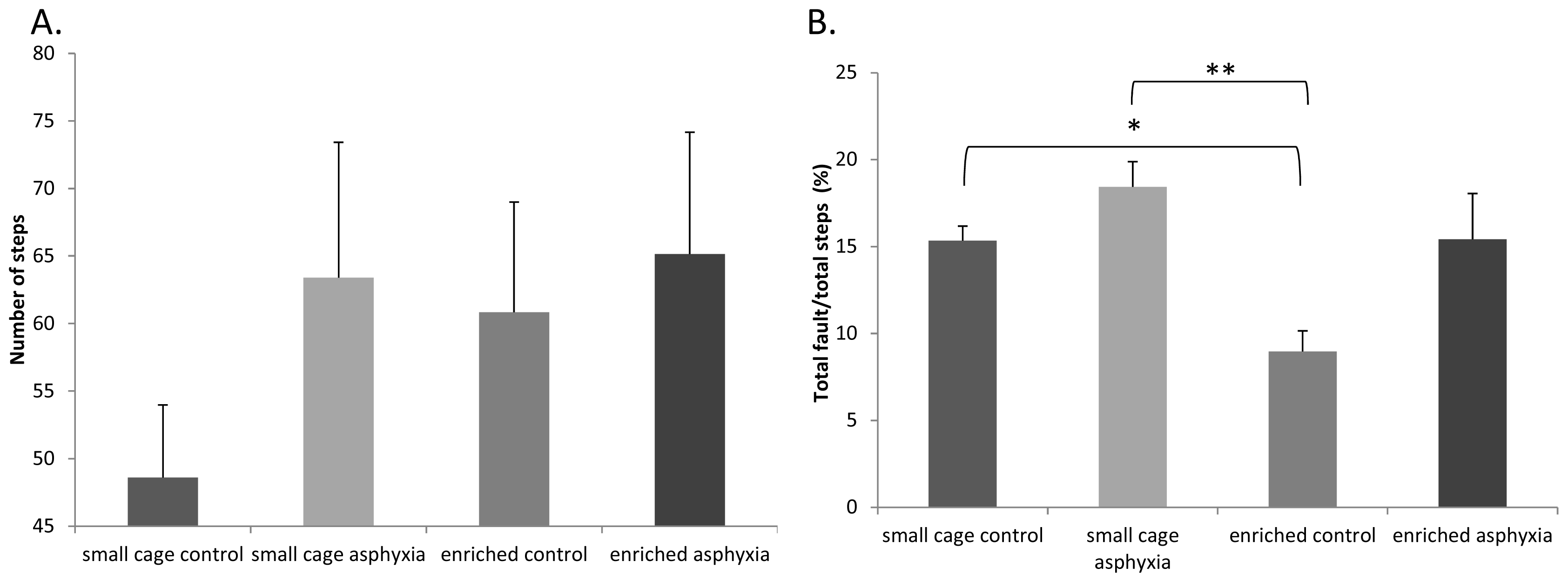
2.4. Motor Coordination
2.5. Discussion
3. Experimental Section
3.1. Experimental Animals
3.2. Asphyxia
3.3. Environmental Enrichment
3.4. Examination of Neurobehavioral Development
3.5. Motor Coordination Tests
3.6. Statistical Analysis
4. Conclusions




Acknowledgments
Conflicts of Interest
References
- De Haan, M.; Wyatt, J.S.; Roth, S.; Vargha-Khadem, F.; Gadian, D.; Mishkin, M. Brain and cognitive-behavioural development after asphyxia at term birth. Dev. Sci 2006, 9, 350–358. [Google Scholar]
- Morales, P.; Fiedler, J.L.; Andres, S.; Berrios, C.; Huaiquin, P.; Bustamante, D.; Cardenas, S.; Parra, E.; Herrera-Marschitz, M. Plasticity of hippocampus following perinatal asphyxia: Effects on postnatal apoptosis and neurogenesis. J. Neurosci. Res 2008, 86, 2650–2662. [Google Scholar]
- Vannucci, R.C. Hypoxic-ischmic encephalopathy. Am. J. Perinatol 2000, 17, 113–120. [Google Scholar]
- Herrera-Marschitz, M.; Morales, P.; Leyton, L.; Bustamante, D.; Klawitter, V.; Espina-Marchant, P. Perinatal asphyxia: Current status and approaches towards neuroprotective strategies, with focus on sentinel proteins. Neurotox. Res 2011, 19, 603–627. [Google Scholar]
- Sarnat, H.B.; Sarnat, M.S. Neonatal encephalopathy following fetal distress. A clinical and electroencephalographic study. Arch. Neurol 1976, 33, 696–705. [Google Scholar]
- Leuthner, S.R.; Das, U.G. Low Apgar scores and the definition of birth asphyxia. Pediatr. Clin. North. Am 2004, 51, 737–745. [Google Scholar]
- Northington, F.J.; Zelaya, M.E.; O’Riordan, D.P.; Blomgren, K.; Flock, D.L.; Hagberg, H.; Ferriero, D.M.; Martin, L.J. Failure to complete apoptosis following neonatal hypoxia-ischemia manifests as “continuum” phenotype of cell death and occurs with multiple manifestations of mitochondrial dysfunction in rodent forebrain. Neuroscience 2007, 149, 822–833. [Google Scholar]
- Hagberg, H.; Mallard, C.; Rousset, C.I.; Xiaoyang, W. Apoptotic mechanisms in the immature brain: Involvement of mitochondria. J. Child Neurol 2009, 24, 1141–1146. [Google Scholar]
- Ginet, V.; Puyal, J.; Clarke, P.G.; Truttmann, A.C. Enhancement of autophagic flux after neonatal cerebral hypoxia-ischemia and its region-specific relationship to apoptotic mechanisms. Am. J. Pathol 2009, 175, 1962–1974. [Google Scholar]
- Eisenberg-Lerner, A.; Bialik, S.; Simon, H.U.; Kimchi, A. Life and death partners: Apoptosis, autophagy and the cross-talk between them. Cell. Death Differ 2009, 16, 966–975. [Google Scholar]
- Holopainen, I.E.; Laurén, H.B. Glutamate signaling in the pathophysiology and therapy of prenatal insults. Pharmacol. Biochem. Behav 2012, 100, 825–834. [Google Scholar]
- Chen, Y.; Herrera-Marschitz, M.; Bjelke, B.; Blum, M.; Gross, J.; Andersson, K. Perinatal asphyxia-induced changes in rat brain tyrosine hydroxylase-immunoreactive cell body number: Effects of nicotine treatment. Neurosci. Lett 1997, 221, 77–80. [Google Scholar]
- Riikonen, R.S.; Kero, P.O.; Simell, O.G. Excitatory amino acids in cerebrospinal fluid in neonatal asphyxia. Pediatr. Neurol 1992, 8, 37–40. [Google Scholar]
- Hagberg, H.; Thornberg, E.; Blennow, M.; Kjellmer, I.; Lagercrantz, H.; Thiringer, K.; Hamberger, A.; Sandberg, M. Excitatory amino acids in the cerebrospinal fluid of asphyxiated infants: Relationship to hypoxic-ischemic encephalopathy. Acta Paediatr 1993, 82, 925–929. [Google Scholar]
- Ziebell, J.M.; Morganti-Kossmann, M.C. Review Involvement of pro- and anti-inflammatory cytokines and chemokines in the pathophysiology of traumatic brain injury. Neurotherapeutics 2010, 7, 22–30. [Google Scholar]
- Lehnardt, S.; Lehmann, S.; Kaul, D.; Tschimmel, K.; Hoffmann, O.; Cho, S. Toll-like receptor 2 mediates CNS injury in focal cerebral ischemia. J. Neuroimmunol 2007, 190, 28–33. [Google Scholar]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms underlying inflammation in neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar]
- Tracey, K.J. Physiology and immunology of the cholinergic antiinflammatory pathway. J. Clin. Invest 2007, 117, 289–296. [Google Scholar]
- Vannucci, R.C.; Christensen, M.A.; Yager, J.Y. Nature, time-course, and extent of cerebral edema in perinatal hypoxic-ischemic brain damage. Pediatr. Neurol 1993, 9, 29–34. [Google Scholar]
- Allende-Castro, C.; Espina-Marchant, P.; Bustamante, D.; Rojas-Mancilla, E.; Neira, T.; Gutierrez-Hernandez, M.A.; Esmar, D.; Valdes, J.L.; Morales, P.; Gebicke-Haerter, P.J.; et al. Further studies on the hypothesis of PARP-1 inhibition as a strategy for lessening the long-term effects produced by perinatal asphyxia: Effects of nicotinamide and theophylline on PARP-1 activity in brain and peripheral tissue: Nicotinamide and theophylline on PARP-1 activity. Neurotox. Res 2012, 22, 79–90. [Google Scholar]
- Ujhazy, E.; Dubovicky, M.; Navarova, J.; Sedlackova, N.; Danihel, L.; Brucknerova, I.; Mach, M. Subchronic perinatal asphyxia in rats: Embryo-foetal assessment of a new model of oxidative stress during critical period of development. Food Chem. Toxicol 2013, in press. [Google Scholar]
- Morales, P.; Huaiquin, P.; Bustamante, D.; Fiedler, J.; Herrera-Marschitz, M. Perinatal asphyxia induces neurogenesis in hippocampus: An organotypic study. Neurotox. Res 2007, 12, 81–84. [Google Scholar]
- Yang, T.; Zhuang, L.; Terrando, N.; Wu, X.; Jonhson, M.R.; Maze, M.; Ma, D. A clinically relevant model of perinatal global ischemic brain damage in rats. Brain Res 2011, 1383, 317–323. [Google Scholar]
- Ten, V.S.; Bradley-Moore, M.; Gingrich, J.A.; Stark, R.I.; Pinsky, D.J. Brain injury and neurofunctional deficit in neonatal mice with hypoxic-ischemic encephalopathy. Behav. Brain Res 2003, 145, 209–219. [Google Scholar]
- Lubics, A.; Reglodi, D.; Tamas, A.; Kiss, P.; Szalai, M.; Szalontay, L.; Lengvari, I. Neurological reflexes and early motor behavior in rats subjected to neonatal hypoxic/ischemic injury. Behav. Brain Res 2005, 157, 157–165. [Google Scholar]
- Kiss, P.; Tamas, A.; Lubics, A.; Szalai, M.; Szalontay, L.; Lengvari, I.; Reglodi, D. Development of neurological reflexes and motor coordination in rats neonatally treated with monosodium glutamate. Neurotox. Res 2005, 8, 235–244. [Google Scholar]
- Kiss, P.; Hauser, D.; Tamas, A.; Lubics, A.; Racz, B.; Horvath, Z.; Farkas, J.; Zimmermann, F.; Stepien, A.; Lengvari, I.; et al. Changes in open-field activity and novelty-seeking behavior in periadolescent rats neonatally treated with monosodium glutamate. Neurotox. Res 2007, 12, 85–93. [Google Scholar]
- Kiss, P.; Szogyi, D.; Reglodi, D.; Horvath, G.; Farkas, J.; Lubics, A.; Tamas, A.; Atlasz, T.; Szabadfi, K.; Babai, N.; et al. Effects of perinatal asphyxia on the neurobehavioral and retinal development of newborn rats. Brain Res 2009, 1255, 42–50. [Google Scholar]
- Cebral, E.; Loidl, C.F. Changes in neostriatal and hippocampal synaptic densities in perinatal asphyctic male and female young rats: Role of hypothermia. Brain Res. Bull 2011, 84, 31–38. [Google Scholar]
- Morales, P.; Simola, N.; Bustamante, D.; Lisboa, F.; Fiedler, J.; Gebicke-Haerter, P.J.; Morelli, M.; Tasker, R.A.; Herrera-Marschitz, M. Nicotinamide prevents the long-term effects of perinatal asphyxia on apoptosis, non-spatial working memory and anxiety in rats. Exp. Brain Res 2010, 202, 1–14. [Google Scholar]
- Vlassaks, E.; Strackx, E.; Vles, J.S.; Nikiforou, M.; Martinez-Martinez, P.; Kramer, B.W.; Gavilanes, A.W. Fetal asphyctic preconditioning modulates the acute cytokine response thereby protecting against perinatal asphyxia in neonatal rats. J. Neuroinflammation 2013, 10, 14. [Google Scholar]
- Kumral, A.; Genc, S.; Ozer, E.; Yilmaz, O.; Gokmen, N.; Koroglu, T.F.; Duman, N.; Genc, K.; Ozkan, H. Erythropoietin downregulates bax and DP5 proapoptotic gene expression in neonatal hypoxic-ischemic brain injury. Biol. Neonate 2006, 89, 205–210. [Google Scholar]
- Alonso-Alconada, D.; Alvarez, A.; Lacalle, J.; Hilario, E. Histological study of the protective effect of melatonin on neural cells after neonatal hypoxia-ischemia. Histol. Histopathol 2012, 27, 771–783. [Google Scholar]
- Kajta, M.; Makarewicz, D.; Ziemińska, E.; Jantas, D.; Domin, H.; Lasoń, W.; Kutner, A.; £azarewicz, J.W. Neuroprotection by co-treatment and post-treating with calcitriol following the ischemic and excitotoxic insult in vivo and in vitro. Neurochem. Int. 2009, 55, 265–274. [Google Scholar]
- Hebb, D.O. The effects of early experience on problem solving at maturity. Am. Psychol 1947, 2, 306–307. [Google Scholar]
- Nithianantharajah, J.; Hannan, A.J. Enriched environments, experience-dependent plasticity and disorders of the nervous system. Nat. Rev. Neurosci 2006, 7, 697–709. [Google Scholar]
- Szabadfi, K.; Atlasz, T.; Horvath, G.; Kiss, P.; Hamza, L.; Farkas, J.; Tamas, A.; Lubics, A.; Gabriel, R.; Reglodi, D. Early postnatal enriched environment decreases retinal degeneration induced by monosodium glutamate treatment. Brain Res 2009, 1259, 107–112. [Google Scholar]
- Kiss, P.; Szabadfi, K.; Horvath, G.; Tamas, A.; Farkas, J.; Gabriel, R.; Reglodi, D. Gender-dependent effects of enriched environment and social isolation in ischemic retinal lesion in adult rats. Int. J. Mol. Sci 2013, 14, 16111–16123. [Google Scholar]
- Horvath, G.; Reglodi, D.; Vadasz, G.; Farkas, J.; Kiss, P. Exposure to enriched environment decreases neurobehavioral deficits induced by neonatal glutamate toxicity. Int. J. Mol. Sci 2013, 14, 19054–19066. [Google Scholar]
- Farkas, J.; Reglodi, D.; Gaszner, B.; Szogyi, D.; Horvath, G.; Lubics, A.; Tamas, A.; Frank, F.; Besirevic, D.; Kiss, P. Effects of maternal separation on the neurobehavioral development of newborn Wistar rats. Brain Res. Bull 2009, 79, 208–214. [Google Scholar]
- Frizzo, J.K.; Cardoso, M.P.; de Assis, A.M.; Perry, M.L.; Volonté, C.; Frizzo, M.E. Effects of acute perinatal asphyxia in the rat hippocampus. Cell. Mol. Neurobiol 2010, 30, 683–692. [Google Scholar]
- Souza, S.K.; Martins, T.L.; Ferreira, G.D.; Vinagre, A.S.; Silva, R.S.; Frizzo, M.E. Metabolic effects of perinatal asphyxia in the rat cerebral cortex. Metab. Brain Dis 2013, 28, 25–32. [Google Scholar]
- Bustamante, D.; Morales, P.; Pereyra, J.T.; Goiny, M.; Herrera-Marschitz, M. Nicotinamide prevents the effect of perinatal asphyxia on dopamine release evaluated with in vivo microdialysis 3 months after birth. Exp. Brain Res 2007, 177, 358–369. [Google Scholar]
- Dell’Anna, E.; Chen, Y.; Engidawork, E.; Andersson, K.; Lubec, G.; Luthman, J.; Herrera-Marschitz, M. Delayed neuronal death following perinatal asphyxia in rat. Exp. Brain Res 1997, 115, 105–115. [Google Scholar]
- Keilhoff, G.; John, R.; Langnaese, K.; Schweizer, H.; Ebmeyer, U. Triggered by asphyxia neurogenesis seems not to be an endogenous repair mechanism, gliogenesis more like it. Neuroscience 2010, 171, 869–884. [Google Scholar]
- Capani, F.; Saraceno, G.E.; Botti, V.; Aon-Bertolino, L.; de Oliveira, D.M.; Barreto, G.; Galeano, P.; Giraldez-Alvarez, L.D.; Coirini, H. Protein ubiquitination in postsynaptic densities after hypoxia in rat neostriatum is blocked by hypothermia. Exp. Neurol 2009, 219, 404–413. [Google Scholar]
- Bonestroo, H.J.; Nijboer, C.H.; van Velthoven, C.T.; Kavelaars, A.; Hack, C.E.; van Bel, F.; Heijnen, C.J. Cerebral and hepatic inflammatory response after neonatal hypoxia-ischemia in newborn rats. Dev. Neurosci 2013, 35, 197–211. [Google Scholar]
- Maślińska, D.; Laure-Kamionowska, M.; Kalisze, K.A.; Makarewicz, D. Proinflammatory cytokines in injured rat brain following perinatal asphyxia. Folia Neuropathol 2002, 40, 177–182. [Google Scholar]
- Klawitter, V.; Morales, P.; Bustamante, D.; Gomez-Urquijo, S.; Hokfelt, T.; Herrera-Marschitz, M. Plasticity of basal ganglia neurocircuitries following perinatal asphyxia: Effect of nicotinamide. Exp. Brain Res 2007, 180, 139–152. [Google Scholar]
- Van de Berg, W.D.; Schmitz, C.; Steinbusch, H.W.; Blanco, C.E. Perinatal asphyxia induced neuronal loss by apoptosis in the neonatal rat striatum: A combined TUNEL and stereological study. Exp. Neurol 2002, 174, 29–36. [Google Scholar]
- Strata, F.; Stoianov, I.P.; de Villers-Sidani, E.; Bonham, B.; Martone, T.; Kenet, T.; Chang, E.F.; Vincenti, V.; Merzenich, M.M. Perinatal asphyxia affects rat auditory processing: Implications for auditory perceptual impairments in neurodevelopmental disorders. PLoS One 2010, 5, e15326. [Google Scholar]
- Landi, S.; Ciucci, F.; Maffei, L.; Berardi, N.; Cenni, M.C. Setting the pace for retinal development: Environmental enrichment acts through insulin-like growth factor 1 and brain-derived neurotrophic factor. J. Neurosci 2009, 29, 10809–10819. [Google Scholar]
- Ortuzar, N.; Argandoña, E.G.; Bengoetxea, H.; Lafuente, J.V. Combination of intracortically administered VEGF and environmental enrichment enhances brain protection in developing rats. J. Neural Transm 2011, 118, 135–144. [Google Scholar]
- Reynolds, S.; Urruela, M.; Devine, D.P. Effects of environmental enrichment on repetitive behaviors in the BTBR T + tf/J mouse model of autism. Autism Res 2013. [Google Scholar] [CrossRef]
- Fernández-Teruel, A.; Escorihuela, R.M.; Castellano, B.; González, B.; Tobeña, A. Neonatal handling and environmental enrichment effects on emotionality, novelty/reward seeking, and age-related cognitive and hippocampal impairments: Focus on the Roman rat lines. Behav. Genet 1997, 27, 513–526. [Google Scholar]
- Richter, S.H.; Zeuch, B.; Riva, M.A.; Gass, P.; Vollmayr, B. Environmental enrichment ameliorates depressive-like symptoms in young rats bred for learned helplessness. Behav. Brain Res 2013, 252, 287–292. [Google Scholar]
- Ravenelle, R.; Byrnes, E.M.; Byrnes, J.J.; McInnis, C.; Park, J.H.; Donaldson, S.T. Environmental enrichment effects on the neurobehavioral profile of selective outbred trait anxiety rats. Behav. Brain Res 2013, 252, 49–57. [Google Scholar]
- Pattison, K.F.; Laude, J.R.; Zentall, T.R. Environmental enrichment affects suboptimal, risky, gambling-like choice by pigeons. Anim. Cogn 2013, 16, 429–434. [Google Scholar]
- Schneider, T.; Lee, M.H.; Anderson, D.W.; Zuck, L.; Lidsky, T.I. Enriched environment during development is protective against lead-induced neurotoxicity. Brain Res 2001, 896, 48–55. [Google Scholar]
- Johnson, E.M.; Traver, K.L.; Hoffman, S.W.; Harrison, C.R.; Herman, J.P. Environmental enrichment protects against functional deficits caused by traumatic brain injury. Front. Behav. Neurosci 2013. [Google Scholar] [CrossRef]
- Nobrega, J.N.; Saari, M.J.; Armstrong, J.N.; Reed, T. Neonatal 6-OHDA lesions and rearing in complex environments: Regional effects on adult brain 14C-2-deoxyglucose uptake revealed by exposure to novel stimulation. Dev. Psychobiol 1992, 25, 183–198. [Google Scholar]
- Monaco, C.M.; Mattiola, V.V.; Folweiler, K.A.; Tay, J.K.; Yelleswarapu, N.K.; Curatolo, L.M.; Matter, A.M.; Cheng, J.P.; Kline, A.E. Environmental enrichment promotes robust functional and histological benefits in female rats after controlled cortical impact injury. Exp. Neurol 2013, 247, 410–418. [Google Scholar]
- Rojas, J.J.; Deniz, B.F.; Miguel, P.M.; Diaz, R.; Hermel Edo, E.; Achaval, M.; Nettó, C.A.; Pereira, L.O. Effects of daily environmental enrichment on behavior and dendritic spine density in hippocampus following neonatal hypoxia-ischemia in the rat. Exp. Neurol 2013, 241, 25–33. [Google Scholar]
- Imanaka, A.; Morinobu, S.; Toki, S.; Yamamoto, S.; Matsuki, A.; Kozuru, T.; Yamawaki, S. Neonatal tactile stimulation reverses the effect of neonatal isolation on open-field and anxiety-like behavior, and pain sensitivity in male and female adult Sprague-Dawley rats. Behav. Brain Res 2008, 186, 91–97. [Google Scholar]
- Baroncelli, L.; Braschi, C.; Maffei, L. Visual depth perception in normal and deprived rats: Effects of environmental enrichment. Neuroscience 2013, 236, 313–319. [Google Scholar]
- Kiss, P.; Atlasz, T.; Szabadfi, K.; Horvath, G.; Griecs, M.; Farkas, J.; Matkovits, A.; Toth, G.; Lubics, A.; Tamas, A.; et al. Comparison between PACAP- and enriched environment-induced retinal protection in MSG-treated newborn rats. Neurosci. Lett 2011, 487, 400–405. [Google Scholar]
- Comeau, W.; Gibb, R.; Hastings, E.; Cioe, J.; Kolb, B. Therapeutic effects of complex rearing or bFGF after perinatal frontal lesions. Dev. Psychobiol 2008, 50, 134–146. [Google Scholar]
- Hannigan, J.H.; O’leary-Moore, S.K.; Berman, R.F. Postnatal environmental or experiential amelioration of neurobehavioral effects of perinatal alcohol exposure in rats. Neurosci. Biobehav. Rev 2007, 31, 202–11. [Google Scholar]
- Imanaka, A.; Morinobu, S.; Toki, S.; Yamawaki, S. Importance of early environment in the development of post-traumatic stress disorder-like behaviors. Behav. Brain Res 2006, 173, 129–137. [Google Scholar]
- Pereira, L.O.; Arteni, N.S.; Petersen, R.C.; da Rocha, A.P.; Achaval, M.; Netto, C.A. Effects of daily environmental enrichment on memory deficits and brain injury following neonatal hypoxia-ischemia in the rat. Neurobiol. Learn. Mem 2007, 87, 101–108. [Google Scholar]
- Adriani, W.; Giannakopoulou, D.; Bokulic, Z.; Jernej, B.; Alleva, E.; Laviola, G. Response to novelty, social and self-control behaviors, in rats exposed to neonatal anoxia: Modulatory effects of an enriched environment. Psychopharmacology (Berl.) 2006, 184, 155–165. [Google Scholar]
- Robertson, C.L.; Scafidi, S.; McKenna, M.C.; Fiskum, G. Mitochondrial mechanisms of cell death and neuroprotection in pediatric ischemic and traumatic brain injury. Exp. Neurol 2009, 218, 371–380. [Google Scholar]
- Sun, X.; Zhou, H.; Luo, X.; Li, S.; Yu, D.; Hua, J.; Mu, D.; Mao, M. Neuroprotection of brain-derived neurotrophic factor against hypoxic injury in vitro requires activation of extracellular signal-regulated kinase and phosphatidylinositol 3-kinase. Int. J. Dev. Neurosci 2008, 26, 363–370. [Google Scholar]
- Yu, K.; Wu, Y.; Jia, J.; Hu, Y.; Zhang, Q.; Xie, H.; Liu, G.; Chen, Y.; Guo, Z. Neuroprotective effects of prior exposure to enriched environment on cerebral ischemia/reperfusion injury in rats: The possible molecular mechanism. Brain Res 2013, in press. [Google Scholar]
- Baldini, S.; Restani, L.; Baroncelli, L.; Coltelli, M.; Franco, R.; Cenni, M.C.; Maffei, L.; Berardi, N. Enriched early life experiences reduce adult anxiety-like behavior in rats: A role for insulin-like growth factor 1. J. Neurosci 2013, 33, 11715–11723. [Google Scholar]
- Vazquez-Sanroman, D.; Sanchis-Segura, C.; Toledo, R.; Hernandez, M.E.; Manzo, J.; Miquel, M. The effects of enriched environment on BDNF expression in the mouse cerebellum depending on the length of exposure. Behav. Brain Res 2013, 243, 118–128. [Google Scholar]
- Olsson, T.; Mohammed, A.H.; Donaldson, L.F.; Henriksson, B.G.; Seckl, J.R. Glucocorticoid receptor and NGFI-A gene expression are induced in the hippocampus after environmental enrichment in adult rats. Brain Res. Mol. Brain Res 1994, 23, 349–353. [Google Scholar]
- Kondo, M.; Takei, Y.; Hirokawa, N. Motor protein KIF1A is essential for hippocampal synaptogenesis and learning enhancement in an enriched environment. Neuron 2012, 73, 743–757. [Google Scholar]
- Kusaka, K.; Morinobu, S.; Kawano, K.; Yamawaki, S. Effect of neonatal isolation on the noradrenergic transduction system in the rat hippocampal slice. Synapse 2004, 54, 223–232. [Google Scholar]
- Clayton, K.; Fleming, J.M.; Copley, J. Behavioral responses to tactile stimuli in children with cerebral palsy. Phys. Occup. Ther. Pediatr 2003, 23, 43–62. [Google Scholar]
- Simola, N.; Bustamante, D.; Pinna, A.; Pontis, S.; Morales, P.; Morelli, M.; Herrera-Marschitz, M. Acute perinatal asphyxia impairs non-spatial memory and alters motor coordination in adult male rats. Exp. Brain Res 2008, 185, 595–601. [Google Scholar]
- Altman, J.; Sudarshan, K. Postnatal development of locomotion in the laboratory rat. Anim. Behav 1975, 23, 896–920. [Google Scholar]
- Dam, K.; Seidler, F.J.; Slotkin, T.A. Chlorpyrifos exposure during a critical neonatal period elicits gender-selective deficits in the development of coordination skills and locomotor activity. Dev. Brain Res 2000, 121, 179–187. [Google Scholar]
- Hill, J.M.; Gozes, I.; Hill, J.L.; Fridkin, M.; Brenneman, D.E. Vasoactive intestinal peptide antagonist retards the development of neonatal behaviors in the rat. Peptides 1991, 12, 187–192. [Google Scholar]
- Reglodi, D.; Kiss, P.; Tamas, A.; Lengvari, I. The effects of PACAP and PACAP antagonist on the neurobehavioral development of newborn rats. Behav. Brain Res 2003, 140, 131–139. [Google Scholar]
- Smart, J.L.; Dobbing, J. Vulnerability of developing brain. II. Effects of early nutritional deprivation on reflex ontogeny and development on behavior in the rat. Brain Res 1971, 28, 85–95. [Google Scholar]
- Smart, J.L.; Dobbing, J. Vulnerability of developing brain. VI. Relative effects of foetal and early postnatal undernutrition on reflex ontogeny and development of behavior in the rat. Brain Res 1971, 33, 303–314. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kiss, P.; Vadasz, G.; Kiss-Illes, B.; Horvath, G.; Tamas, A.; Reglodi, D.; Koppan, M. Environmental Enrichment Decreases Asphyxia-Induced Neurobehavioral Developmental Delay in Neonatal Rats. Int. J. Mol. Sci. 2013, 14, 22258-22273. https://doi.org/10.3390/ijms141122258
Kiss P, Vadasz G, Kiss-Illes B, Horvath G, Tamas A, Reglodi D, Koppan M. Environmental Enrichment Decreases Asphyxia-Induced Neurobehavioral Developmental Delay in Neonatal Rats. International Journal of Molecular Sciences. 2013; 14(11):22258-22273. https://doi.org/10.3390/ijms141122258
Chicago/Turabian StyleKiss, Peter, Gyongyver Vadasz, Blanka Kiss-Illes, Gabor Horvath, Andrea Tamas, Dora Reglodi, and Miklos Koppan. 2013. "Environmental Enrichment Decreases Asphyxia-Induced Neurobehavioral Developmental Delay in Neonatal Rats" International Journal of Molecular Sciences 14, no. 11: 22258-22273. https://doi.org/10.3390/ijms141122258




