Photophysical Property and Photocatalytic Activity of New Gd2InSbO7 and Gd2FeSbO7 Compounds under Visible Light Irradiation
Abstract
:1. Introduction
2. Results and Discussion
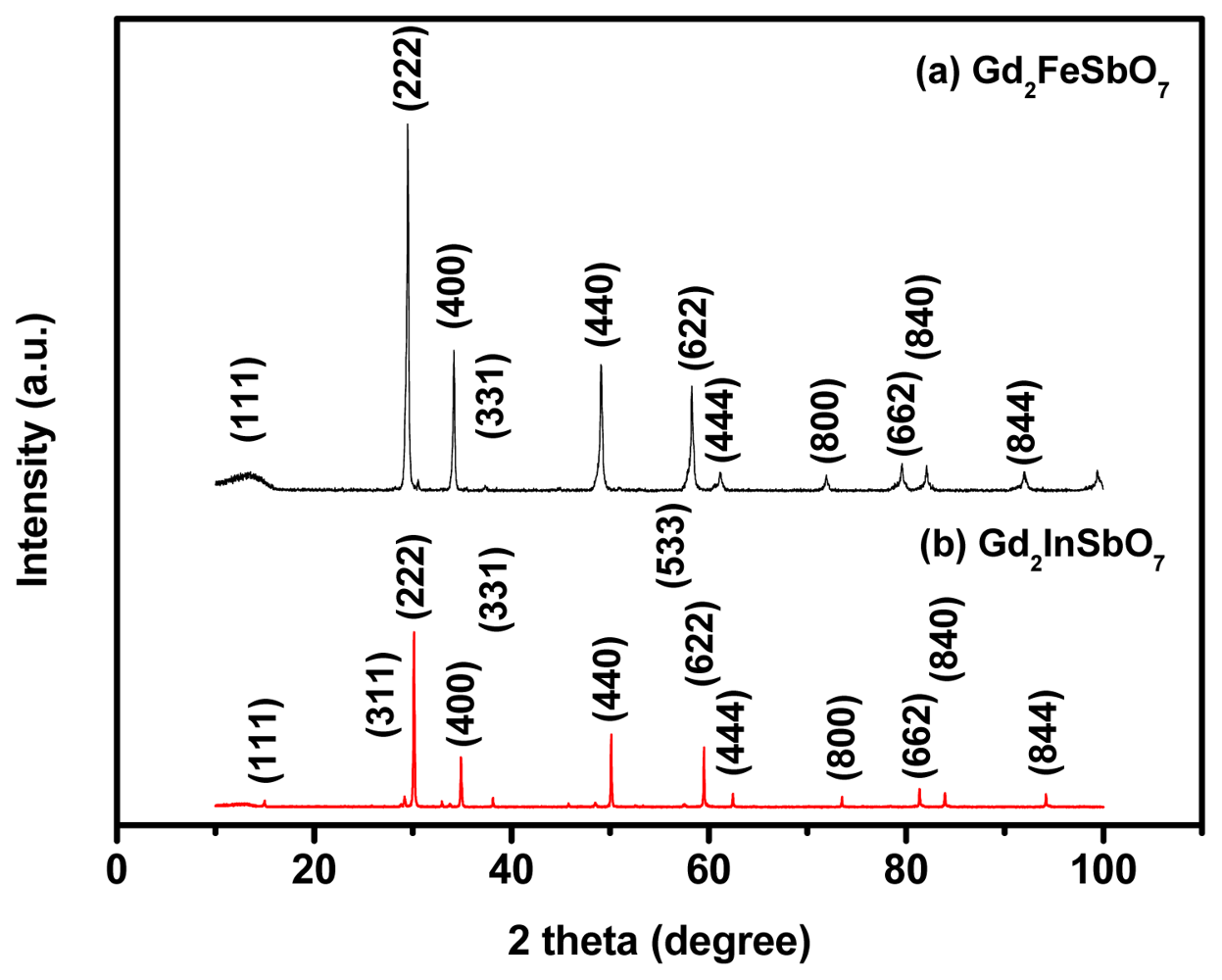
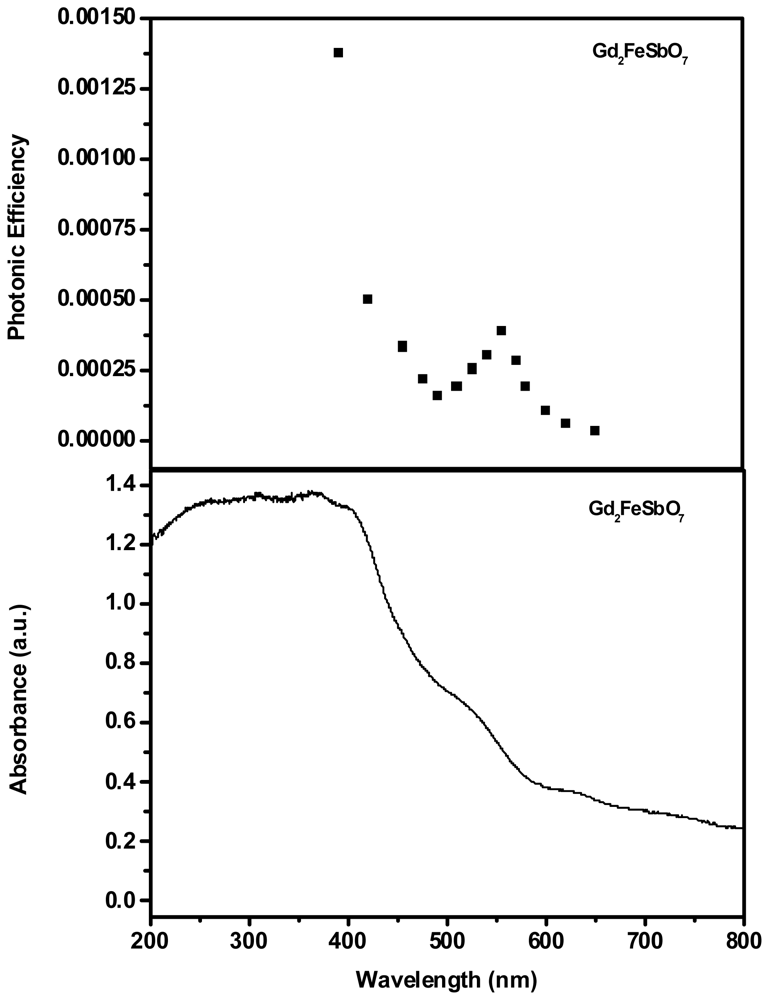
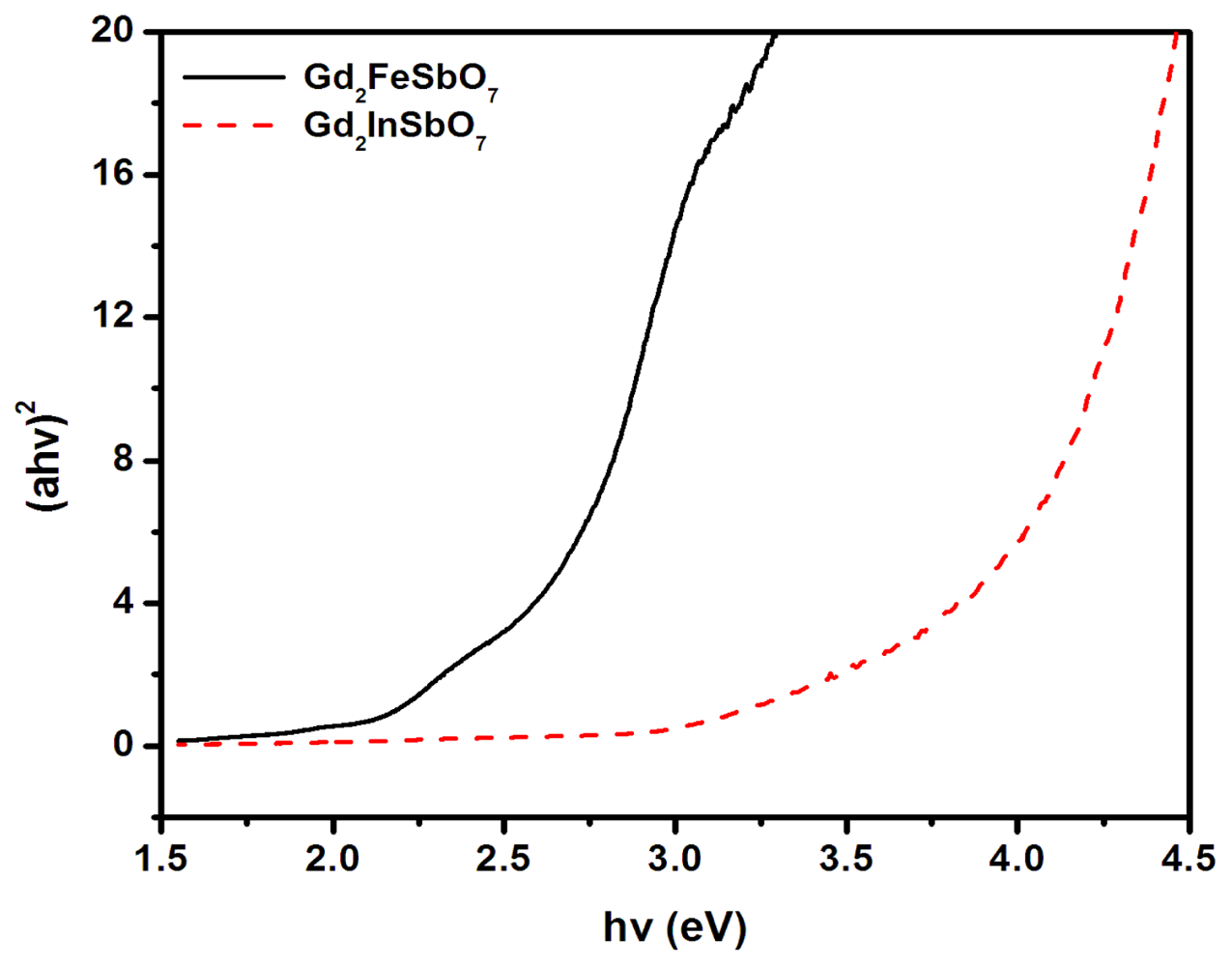
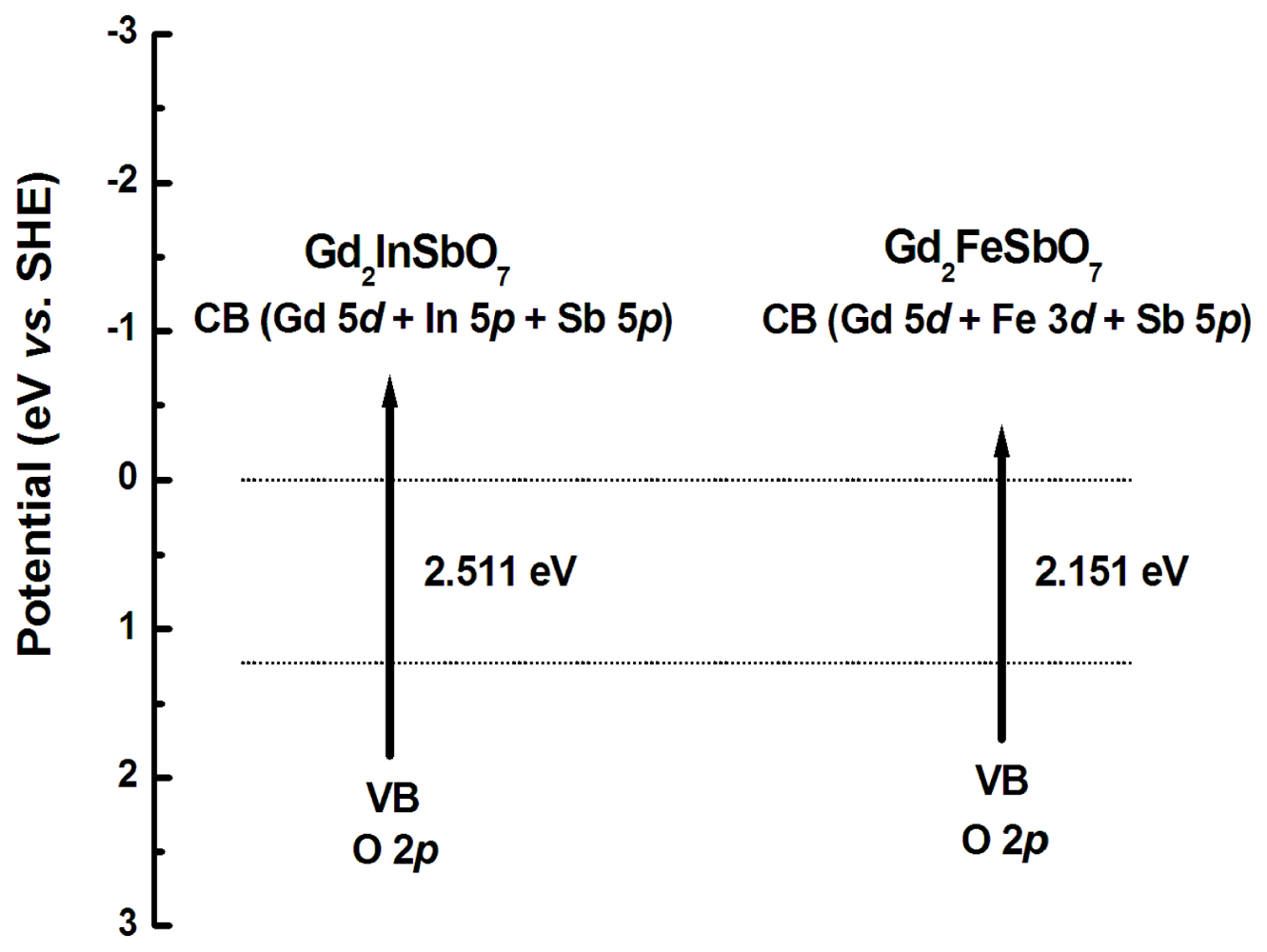
2.1. Characterization
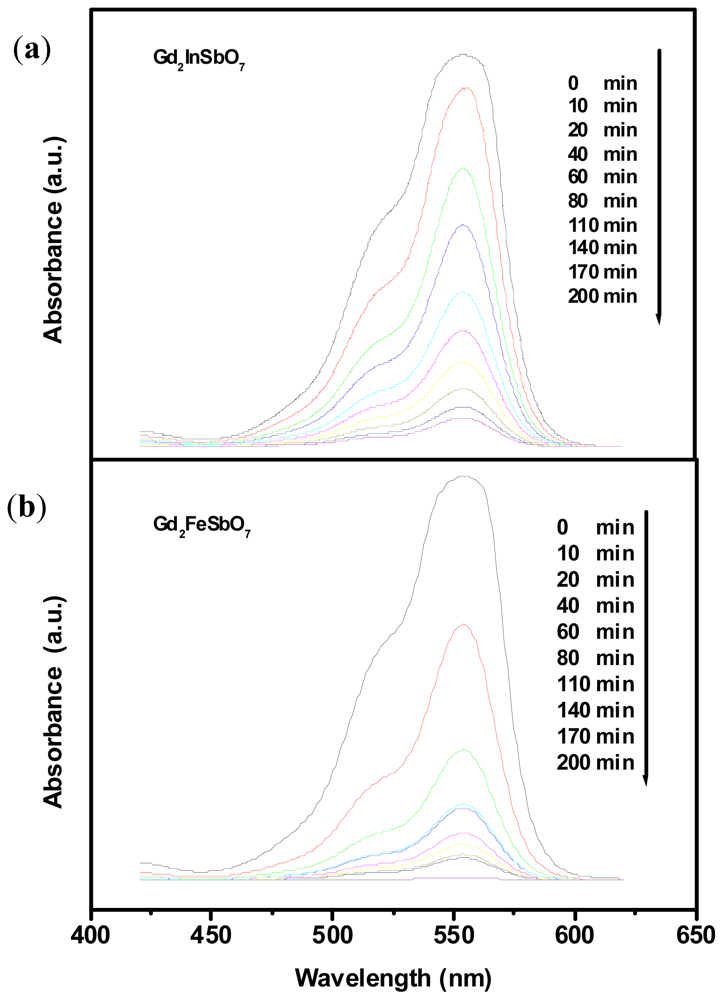
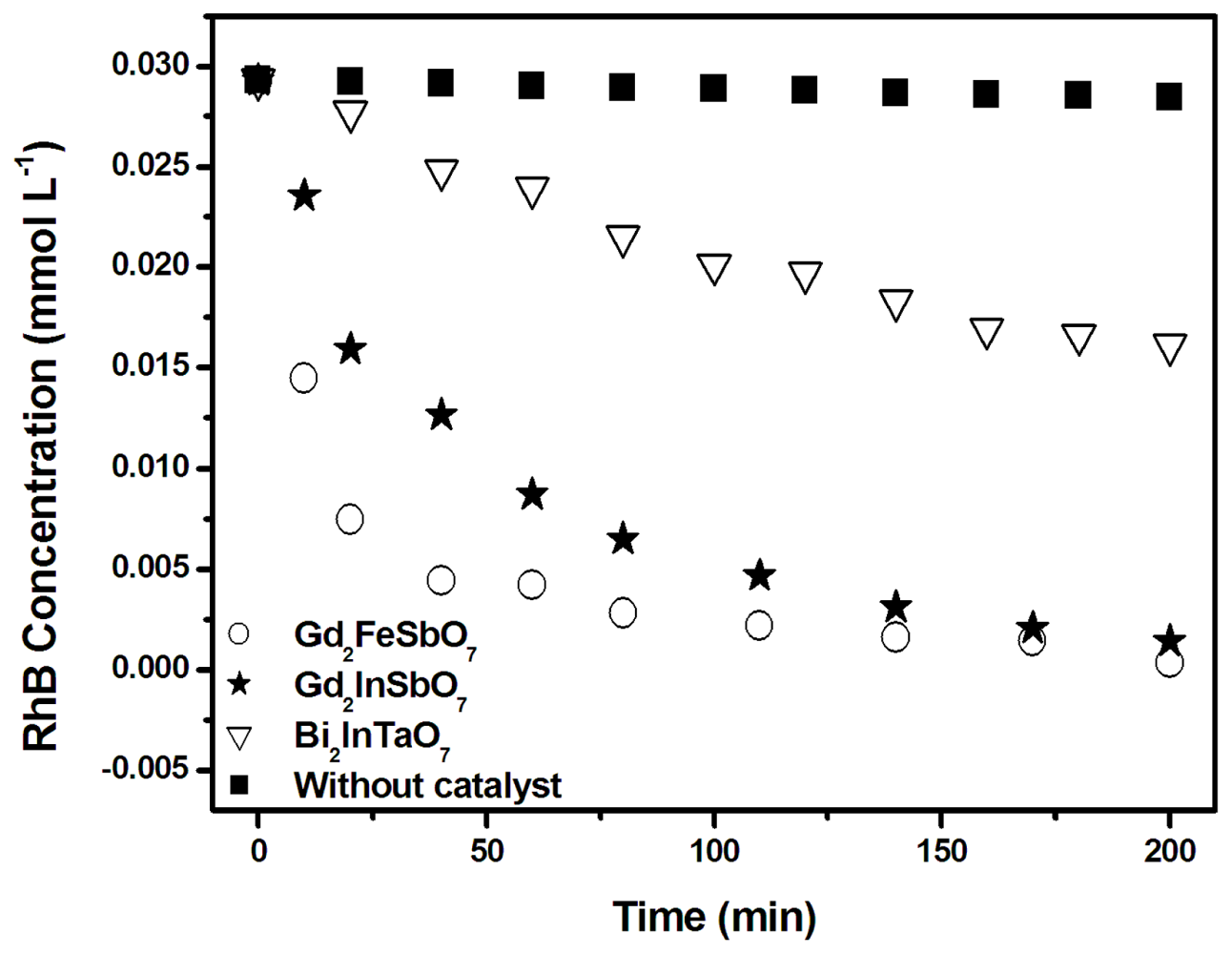
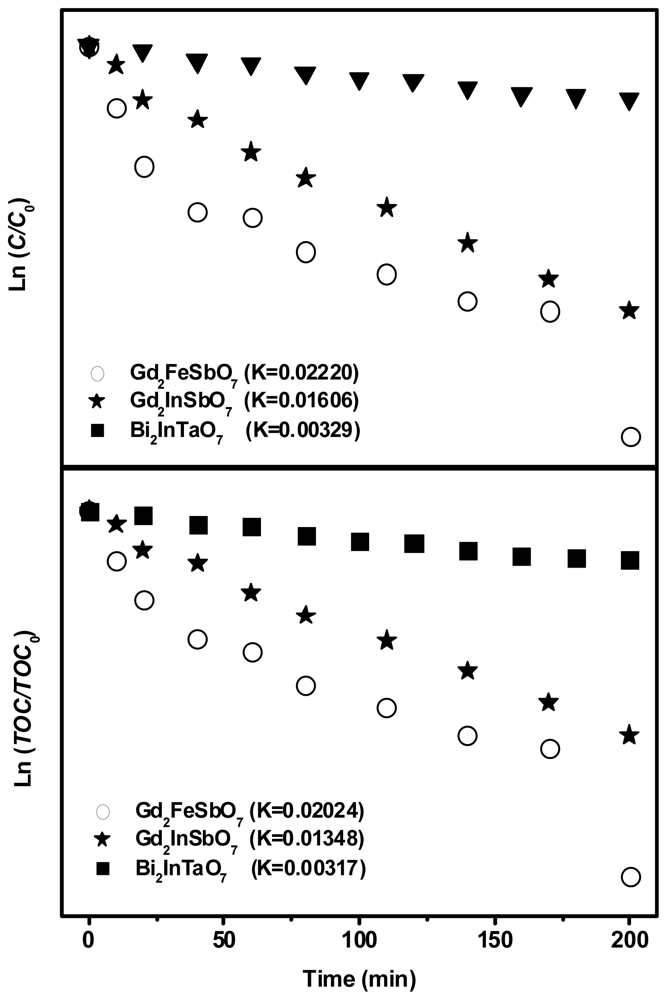
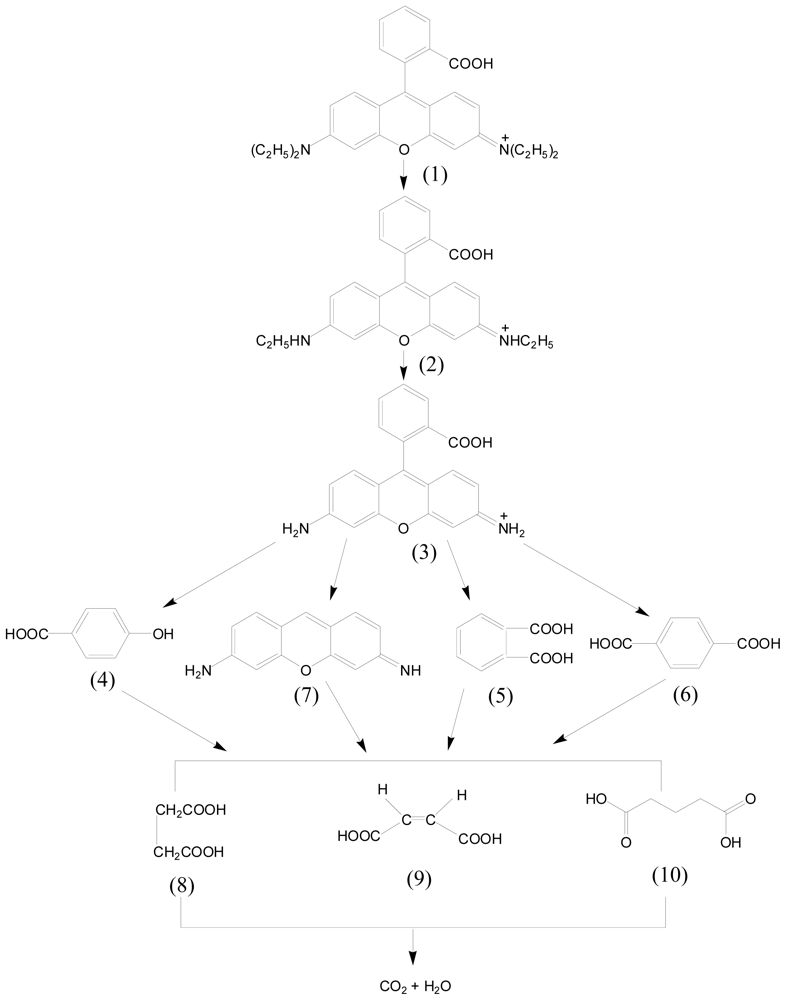
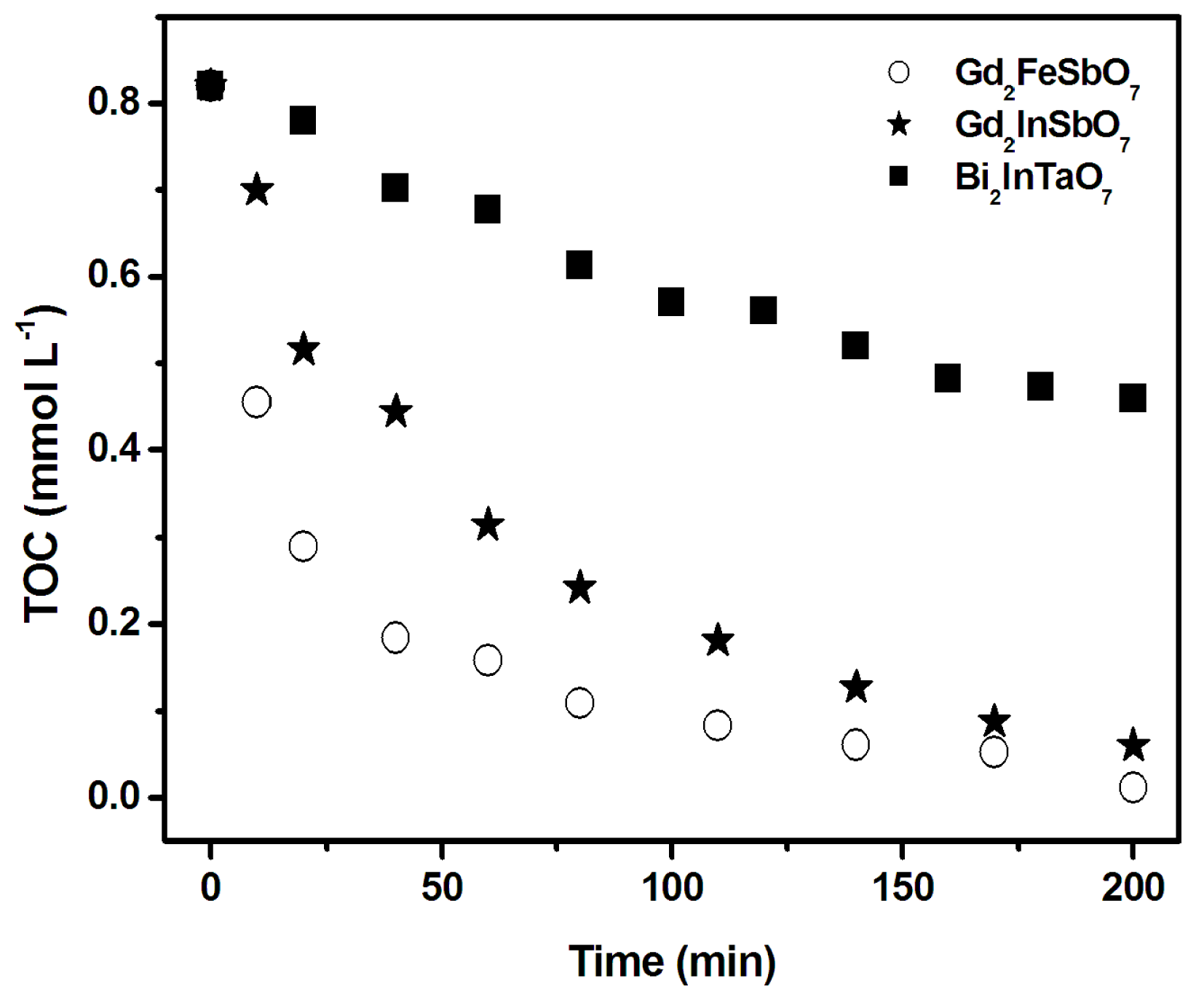
2.2. Photocatalytic Activity
3. Experimental Section
4. Conclusions
Acknowledgments
References
- Bhatnagar, A.; Jain, A.K. A comparative adsorption study with different industrial wastes as adsorbents for the removal of cationic dyes from water. J. Colloid Interface Sci 2005, 281, 49–55. [Google Scholar]
- Fu, H.B.; Pan, C.S.; Yao, W.Q.; Zhu, Y.F. Visible-light-induced degradation of rhodamine B by nanosized Bi2WO6. J. Phys. Chem. B 2005, 109, 22432–22439. [Google Scholar]
- Guo, Y.P.; Zhao, J.Z.; Zhang, H.; Yang, S.F.; Qi, J.R.; Wang, Z.C.; Xu, H.D. Use of rice husk-based porous carbon for adsorption of Rhodamine B from aqueous solutions. Dyes Pigm 2005, 66, 123–128. [Google Scholar]
- Gupta, V.K.; Ali, I.; Saini, V.K. Removal of rhodamine B, fast green and methylene blue from wastewater using red mud, an aluminum industry waste. Ind. Eng. Chem. Res 2004, 43, 1740–1747. [Google Scholar]
- Fujii, T.; Nishikiori, H.; Tamura, T. Absorption-Spectra of Rhodamine-B dimers in dip-coated thin-films prepared by the sol-gel method. Chem. Phy. Lett 1995, 233, 424–429. [Google Scholar]
- Wang, S.B.; Li, H.T. Dye adsorption on unburned carbon: Kinetics and equilibrium. J. Hazard. Mater 2005, 126, 71–77. [Google Scholar]
- Wang, S.B.; Boyjoo, Y.; Choueib, A. A comparative study of dye removal using fly ash treated by different methods. Chemosphere 2005, 60, 1401–1407. [Google Scholar]
- Gupta, V.K.; Jain, R.; Malathi, S.; Nayak, A. Adsorption-desorption studies of indigocarmine from industrial effluents by using deoiled mustard and its comparison with charcoal. J. Colloid Interface Sci 2010, 348, 628–633. [Google Scholar]
- Ohsaki, H.; Kanai, N.; Fukunaga, Y.; Suzuki, M.; Watanabe, T.; Hashimoto, K. Photocatalytic properties of SnO2/TiO2 multilayers. Thin Solid Films 2006, 502, 138–142. [Google Scholar]
- Davit, P.; Martra, G.; Coluccia, S.; Augugliaro, V.; Lopez, E.G.; Loddo, V.; Marci, G.; Palmisano, L.; Schiavello, M. Adsorption and photocatalytic degradation of acetonitrile: FT-IR investigation. J. Mol. Catal. A 2003, 204, 693–701. [Google Scholar]
- Kim, S.; Choi, W. Dual photocatalytic pathways of trichloroacetate degradation on TiO2: Effects of nanosized platinum deposits on kinetics and mechanism. J. Phys. Chem. B 2002, 106, 13311–13317. [Google Scholar]
- Luan, J.F.; Ma, K.; Wang, S.; Hu, Z.T.; Li, Y.M.; Pan, B.C. Research on Photocatalytic Degradation Pathway and Degradation Mechanisms of Organics. Curr. Org. Chem 2010, 14, 645–682. [Google Scholar]
- Chatterjee, D.; Mahata, A. Visible light induced photodegradation of organic pollutants on dye adsorbed TiO2 surface. J. Photochem. Photobiol. A 2002, 153, 199–204. [Google Scholar]
- Fabbri, D.; Prevot, A.B.; Zelano, V.; Ginepro, M.; Pramauro, E. Removal and degradation of aromatic compounds from a highly polluted site by coupling soil washing with photocatalysis. Chemosphere 2008, 71, 59–65. [Google Scholar]
- Sohrabi, M.R.; Ghavami, M. Comparison of Direct Yellow 12 dye degradation efficiency using UV/semiconductor and UV/H(2)O(2)/semiconductor systems. Desalination 2010, 252, 157–162. [Google Scholar]
- Franch, M.I.; Peral, J.; Domenech, X.; Howe, R.F.; Ayllon, J.A. Enhancement of photocatalytic activity of TiO2 by adsorbed aluminium(III). Appl. Catal. B 2005, 55, 105–113. [Google Scholar]
- Yamashita, H.; Nose, H.; Kuwahara, Y.; Nishida, Y.; Yuan, S.; Mori, K. TiO2 photocatalyst loaded on hydrophobic Si3N4 support for efficient degradation of organics diluted in water. Appl. Catal. A 2008, 350, 164–168. [Google Scholar]
- Neppolian, B.; Yamashita, H.; Okada, Y.; Nishijima, H.; Anpo, M. Preparation of unique TiO2 nano-particle photocatalysts by a multi-gelation method for control of the physicochemical parameters and reactivity. Catal. Lett 2005, 105, 111–117. [Google Scholar]
- An, H.Q.; Zhou, J.; Li, J.X.; Zhu, B.L.; Wang, S.R.; Zhang, S.M.; Wu, S.H.; Huang, W.P. Deposition of Pt on the stable nanotubular TiO2 and its photocatalytic performance. Catal. Commun 2009, 11, 175–179. [Google Scholar]
- Davit, P.; Martra, G.; Coluccia, S. Photocatalytic degradation of organic compounds on TiO2 powders: FT-IR investigation of surface reactivity and mechanistic aspects. J. Jpn. Pet. Inst 2004, 47, 359–376. [Google Scholar]
- Cernigoj, U.; Stangar, U.L.; Trebse, P. Degradation of neonicotinoid insecticides by different advanced oxidation processes and studying the effect of ozone on TiO2 photocatalysis. Appl. Catal. B 2007, 75, 229–238. [Google Scholar]
- Sohrabi, M.R.; Ghavami, M. Photocatalytic degradation of Direct Red 23 dye using UV/TiO2: Effect of operational parameters. J. Hazard. Mater 2008, 153, 1235–1239. [Google Scholar]
- Franch, M.I.; Peral, J.; Domenech, X.; Ayllon, J.A. Aluminium(III) adsorption: A soft and simple method to prevent TiO2 deactivation during salicylic acid photodegradation. Chem. Commun 2005, 14, 1851–1853. [Google Scholar]
- Hammami, S.; Bellakhal, N.; Oturan, N.; Oturan, M.A.; Dachraoui, M. Degradation of Acid Orange 7 by electrochemically generated (OH)-O-center dot radicals in acidic aqueous medium using a boron-doped diamond or platinum anode: A mechanistic study. Chemosphere 2008, 73, 678–684. [Google Scholar]
- Luan, J.F.; Pan, B.C.; Paz, Y.; Li, Y.M.; Wu, X.S.; Zou, Z.G. Structural, photophysical and photocatalytic properties of new Bi2SbVO7 under visible light irradiation. Phys. Chem. Chem. Phys 2009, 11, 6289–6298. [Google Scholar]
- Arana, J.; Rendon, E.T.; Rodriguez, J.M.D.; Melian, J.A.H.; Diaz, O.G.; Pena, J.P. FTIR study of the photocatalytic degradation of NH4+ determination wastes. J. Photochem. Photobiol. A 2002, 148, 215–222. [Google Scholar]
- Arana, J.; Nieto, J.L.M.; Melian, J.A.H.; Rodriguez, J.M.D.; Diaz, O.G.; Pena, J.P.; Bergasa, O.; Alvarez, C.; Mendez, J. Photocatalytic degradation of formaldehyde containing wastewater from veterinarian laboratories. Chemosphere 2004, 55, 893–904. [Google Scholar]
- Vijayabalan, A.; Selvam, K.; Velmurugan, R.; Swaminathan, M. Photocatalytic activity of surface fluorinated TiO2-P25 in the degradation of Reactive Orange 4. J. Hazard. Mater 2009, 172, 914–921. [Google Scholar]
- Arana, J.; Rodriguez, J.M.D.; Diaz, O.G.; Melian, J.A.H.; Pena, J.P. Comparative study on the photocatalytic mineralization of homologous aliphatic acids and alcohols. Appl. Surface Sci 2006, 252, 8193–8202. [Google Scholar]
- Eslami, A.; Nasseri, S.; Yadollahi, B.; Mesdaghinia, A.; Vaezi, F.; Nabizadeh, R.; Nazmara, S. Photocatalytic degradation of methyl tert-butyl ether (MTBE) in contaminated water by ZnO nanoparticles. J. Chem. Technol. Biotechnol 2008, 83, 1447–1453. [Google Scholar]
- Arana, J.; Cabo, C.G.I.; Rodriguez, C.F.; Melian, J.A.H.; Mendez, J.A.O.; Rodriguez, J.M.D.; Pena, J.P. Combining TiO2-photocatalysis and wetland reactors for the efficient treatment of pesticides. Chemosphere 2008, 71, 788–794. [Google Scholar]
- Li, J.P.; Zhang, X.; Ai, Z.H.; Jia, F.L.; Zhang, L.Z.; Lin, J. Efficient visible light degradation of rhodamine B by a photo-electrochemical process based on a Bi2WO6 nanoplate film electrode. J. Phys. Chem. C 2007, 111, 6832–6836. [Google Scholar]
- Li, X.; Ye, J.H. Photocatalytic degradation of rhodamine B over Pb3Nb4O13/fumed SiO2 composite under visible light irradiation. J. Phys. Chem. C 2007, 111, 13109–13116. [Google Scholar]
- Wu, J.M. Photodegradation of rhodamine B in water assisted by titania nanorod thin films subjected to various thermal treatments. Environ. Sci. Technol 2007, 41, 1723–1728. [Google Scholar]
- Wu, J.M.; Zhang, T.W. Photodegradation of rhodamine B in water assisted by titania films prepared through a novel procedure. J. Photochem. Photobiol. A 2004, 162, 171–177. [Google Scholar]
- Li, J.Q.; Li, L.P.; Zheng, L.; Xian, Y.Z.; Jin, L.T. Photoelectrocatalytic degradation of rhodamine B using Ti/TiO2 electrode prepared by laser calcination method. Electrochim. Acta 2006, 51, 4942–4949. [Google Scholar]
- Qu, P.; Zhao, J.C.; Shen, T.; Hidaka, H. TiO2-assisted photodegradation of dyes: A study of two competitive primary processes in the degradation of RB in an aqueous TiO2 colloidal solution. J. Mol. Catal. A 1998, 129, 257–268. [Google Scholar]
- Zhao, J.C.; Wu, T.X.; Wu, K.Q.; Oikawa, K.; Hidaka, H.; Serpone, N. Photoassisted degradation of dye pollutants. 3. Degradation of the cationic dye rhodamine B in aqueous anionic surfactant/TiO2 dispersions under visible light irradiation: Evidence for the need of substrate adsorption on TiO2 particles. Environ. Sci. Technol 1998, 32, 2394–2400. [Google Scholar]
- Luan, J.F.; Ma, K.; Zhang, L.Y.; Li, M.; Li, Y.M.; Pan, B.C. Research on Different Preparation Methods of New Photocatalysts. Curr. Org. Chem 2010, 14, 683–698. [Google Scholar]
- Kitano, M.; Takeuchi, M.; Matsuoka, M.; Thomas, J.M.; Anpo, M. Preparation of visible light-responsive TiO2 thin film photocatalysts by an RF magnetron sputtering deposition method and their photocatalytic reactivity. Chem. Lett 2005, 34, 616–617. [Google Scholar]
- Anpo, M.; Yamashita, H.; Ikeue, K.; Fujii, Y.; Zhang, S.G.; Ichihashi, Y.; Park, D.R.; Suzuki, Y.; Koyano, K.; Tatsumi, T. Photocatalytic reduction of CO2 with H2O on Ti-MCM-41 and Ti-MCM-48 mesoporous zeolite catalysts. Catal. Today 1998, 44, 327–332. [Google Scholar]
- Anpo, M.; Takeuchi, M. The design and development of highly reactive titanium oxide photocatalysts operating under visible light irradiation. J. Catal 2003, 216, 505–516. [Google Scholar]
- Hamadanian, M.; Reisi-Vanani, A.; Majedi, A. Preparation and characterization of S-doped TiO2 nanoparticles, effect of calcination temperature and evaluation of photocatalytic activity. Mater. Chem. Phys 2009, 116, 376–382. [Google Scholar]
- Luan, J.F.; Zhao, W.; Feng, J.W.; Cai, H.L.; Zheng, Z.; Pan, B.C.; Wu, X.S.; Zou, Z.G.; Li, Y.M. Structural, photophysical and photocatalytic properties of novel Bi2AlVO7. J. Hazard. Mater 2009, 164, 781–789. [Google Scholar]
- Izumi, F. A software package for the Rietveld analysis of X-ray and neutron diffraction patterns. J. Crystallogr. Assoc. Jpn 1985, 27, 23–26. [Google Scholar]
- Zou, Z.; Ye, J.; Arakawa, H. Preparation, structural and photophysical properties of Bi2InNbO7 compound. J. Mater. Sci. Lett 2000, 19, 1909–1911. [Google Scholar]
- Tauc, J.; Grigorov, R.; Vancu, A. Optical properties and electronic structure of amorphous germanium. Phys. Status Solidi 1966, 15, 627–637. [Google Scholar]
- Butler, M.A. Photoelectrolysis and physical-properties of semiconducting electrode WO3. J. Appl. Phys 1977, 48, 1914–1920. [Google Scholar]
- Horikoshi, S.; Saitou, A.; Hidaka, H.; Serpone, N. Environmental remediation by an integrated microwave/UV illumination method. V. Thermal and nonthermal effects of microwave radiation on the photocatalyst and on the photodegradation of rhodamine-b under UV/Vis radiation. Environ. Sci. Technol 2003, 37, 5813–5822. [Google Scholar]
- Li, G.R.; Qu, D.L.; Arurault, L.; Tong, Y.X. Hierarchically Porous Gd3+-Doped CeO2 Nanostructures for the Remarkable Enhancement of Optical and Magnetic Properties. J. Phys. Chem. C 2009, 113, 1235–1241. [Google Scholar]
- Selvan, R.K.; Gedanken, A.; Anilkumar, P.; Manikandan, G.; Karunakaran, C. Synthesis and Characterization of Rare Earth Orthovanadate (RVO4; R = La, Ce, Nd, Sm, Eu & Gd) Nanorods/Nanocrystals/Nanospindles by a Facile Sonochemical Method and Their Catalytic Properties. J. Cluster Sci 2009, 20, 291–305. [Google Scholar]
- Perles, J.; Fortes-Revilla, C.; Gutierrez-Puebla, E.; Iglesias, M.; Monge, M.A.; Ruiz-Valero, C.; Snejko, N. Synthesis, structure and catalytic properties of rare-earth ternary sulfates. Chem. Mater 2005, 17, 2701–2706. [Google Scholar]
- Wang, W.; Su, C.; Ran, R.; Shao, Z.P. A new Gd-promoted nickel catalyst for methane conversion to syngas and as an anode functional layer in a solid oxide fuel cell. J. Power Sources 2011, 196, 3855–3862. [Google Scholar]
- Nasr, C.; Vinodgopal, K.; Fisher, L.; Hotchandani, S.; Chattopadhyay, A.K.; Kamat, P.V. Environmental photochemistry on semiconductor surfaces. Visible light induced degradation of a textile diazo dye, naphthol blue black, on TiO2 nanoparticles. J. Phys. Chem 1996, 100, 8436–8442. [Google Scholar]
- Wiegel, M.; Middel, W.; Blasse, G. Influence of NS2 ions on the luminescence of niobates and tantalates. J. Mater. Chem 1995, 5, 981–983. [Google Scholar]
- Li, N.; Xiao, H.Y.; Zu, X.T.; Wang, L.M.; Ewing, R.C.; Lian, J.; Gao, F. First-principles study of electronic properties of La2Hf2O7 and Gd2Hf2O7. J. Appl. Phys. 2007, 102, 063704:1–063704:6. [Google Scholar]
- Luan, J.F.; Zhang, L.Y.; Ma, K.; Li, Y.M.; Zou, Z.G. Preparation and property characterization of new Y2FeSbO7 and In2FeSbO7 photocatalysts. Solid State Sci 2011, 13, 185–194. [Google Scholar]
- Charifi, Z.; Reshak, A.H.; Baaziz, H. Phase transition of LaX (X = P, As, Sb and Bi) at high pressure: Theoretical investigation of the structural and electronic properties. Solid State Commun 2008, 148, 139–144. [Google Scholar]
- Vajenine, G.V.; Hoffmann, R.; zurLoye, H.C. The electronic structures and magnetic properties of one-dimensional ABO6 chains in Sr3ABO6 (A = Co,Ni; B = Pt,Ir) and two-dimensional MO3 sheets in InMO3 (M = Fe,Mn). Chem. Phys 1996, 204, 469–478. [Google Scholar]
- Luo, H.Z.; Zhu, Z.Y.; Liu, G.D.; Xu, S.F.; Wu, G.H.; Liu, H.Y.; Qu, J.P.; Li, Y.X. Ab-initio investigation of electronic properties magnetism of half-Heusler alloys XCrAl (X = Fe, Co, Ni) and NiCrZ (Z = Al, Ga, In). Phys. B Condens. Matter 2008, 403, 200–206. [Google Scholar]
- Xiang, H.P.; Wu, Z.J.; Meng, J. Theoretical investigation on the magnetic and electronic properties of Sr2BMoO6 (B = Fe, Co). Phys. Status Solidi B 2005, 242, 1414–1421. [Google Scholar]
- Zhou, A.; Liu, L.S.; Shu, C.C.; Zhai, P.C.; Zhao, W.Y.; Zhang, Q.J. Electronic Structures and Transport Properties of RFe4Sb12 (R = Na, Ca, Nd, Yb, Sn, In). J. Electron. Mater 2011, 40, 974–979. [Google Scholar]
- Oshikiri, M.; Boero, M.; Ye, J.H.; Zou, Z.G.; Kido, G. Electronic structures of promising photocatalysts InMO4 (M = V, Nb, Ta) and BiVO4 for water decomposition in the visible wavelength region. J. Chem. Phys 2002, 117, 7313–7318. [Google Scholar]
- Marugan, J.; Hufschmidt, D.; Sagawe, G.; Selzer, V.; Bahnemann, D. Optical density and photonic efficiency of silica-supported TiO2 photocatalysts. Water Res 2006, 40, 833–839. [Google Scholar]





| Atom | x | y | z | Occupation factor |
|---|---|---|---|---|
| Gd | 0.00000 | 0.00000 | 0.00000 | 1.0 |
| Fe | 0.50000 | 0.50000 | 0.50000 | 0.5 |
| Sb | 0.50000 | 0.50000 | 0.50000 | 0.5 |
| O(1) | −0.20249 | 0.12500 | 0.12500 | 1.0 |
| O(2) | 0.12500 | 0.12500 | 0.12500 | 1.0 |
| Atom | x | y | z | Occupation factor |
|---|---|---|---|---|
| Gd | 0.00000 | 0.00000 | 0.00000 | 1.0 |
| In | 0.50000 | 0.50000 | 0.50000 | 0.5 |
| Sb | 0.50000 | 0.50000 | 0.50000 | 0.5 |
| O(1) | −0.15469 | 0.12500 | 0.12500 | 1.0 |
| O(2) | 0.12500 | 0.12500 | 0.12500 | 1.0 |
| In3d5/2 BE (eV) | Sb3d5/2 BE (eV) | Fe2p3/2 BE (eV) | Gd4d5/2 BE (eV) | O1s BE (eV) |
|---|---|---|---|---|
| 530.85 | 710.81 | 143.93 | 530.35 | |
| 444.65 | 530.82 | 143.85 | 530.12 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luan, J.; Xu, Y. Photophysical Property and Photocatalytic Activity of New Gd2InSbO7 and Gd2FeSbO7 Compounds under Visible Light Irradiation. Int. J. Mol. Sci. 2013, 14, 999-1021. https://doi.org/10.3390/ijms14010999
Luan J, Xu Y. Photophysical Property and Photocatalytic Activity of New Gd2InSbO7 and Gd2FeSbO7 Compounds under Visible Light Irradiation. International Journal of Molecular Sciences. 2013; 14(1):999-1021. https://doi.org/10.3390/ijms14010999
Chicago/Turabian StyleLuan, Jingfei, and Yong Xu. 2013. "Photophysical Property and Photocatalytic Activity of New Gd2InSbO7 and Gd2FeSbO7 Compounds under Visible Light Irradiation" International Journal of Molecular Sciences 14, no. 1: 999-1021. https://doi.org/10.3390/ijms14010999





