New 14-Membered Cyclopeptide Alkaloids from Zizyphus oxyphylla Edgew
Abstract
:1. Introduction
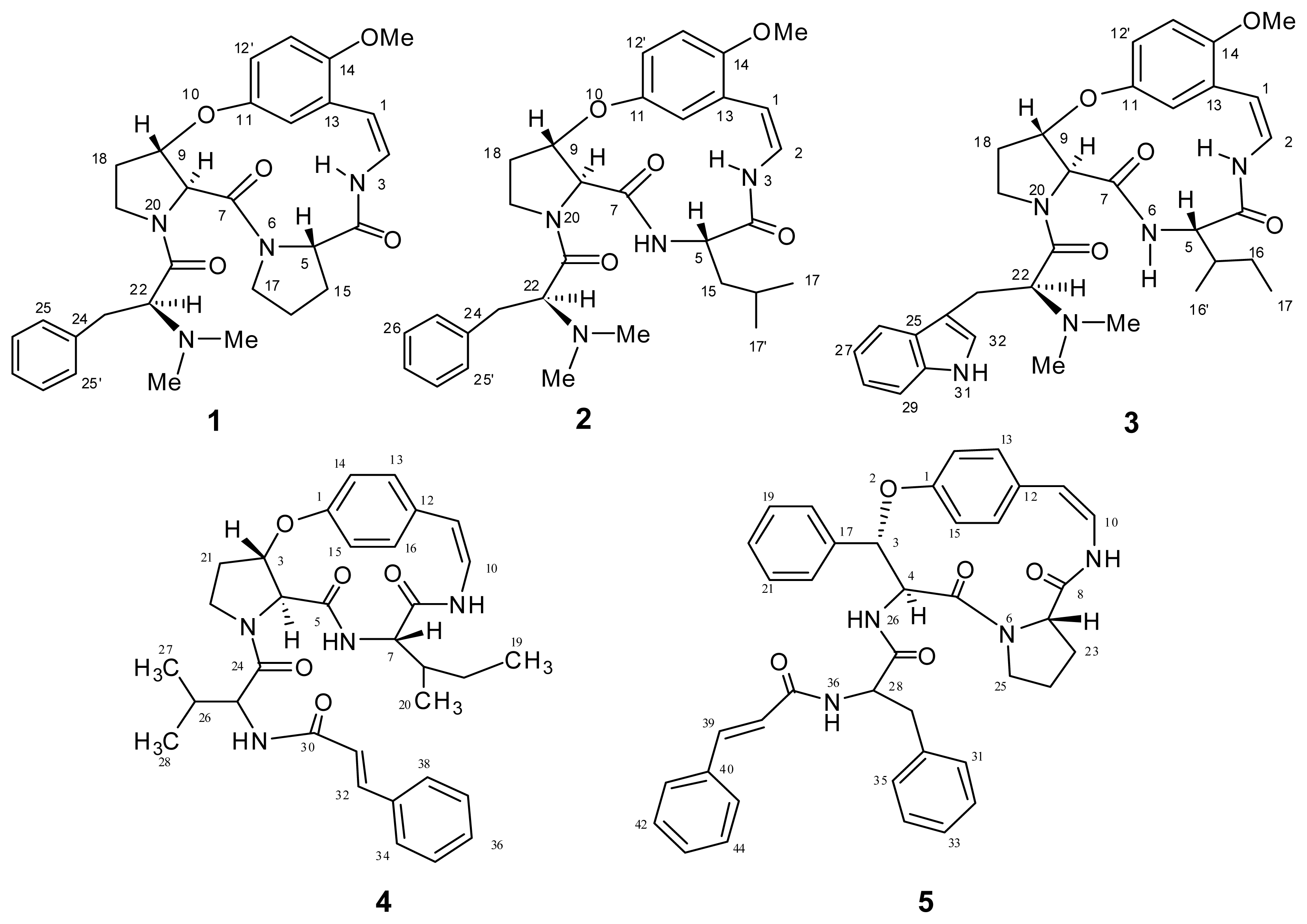
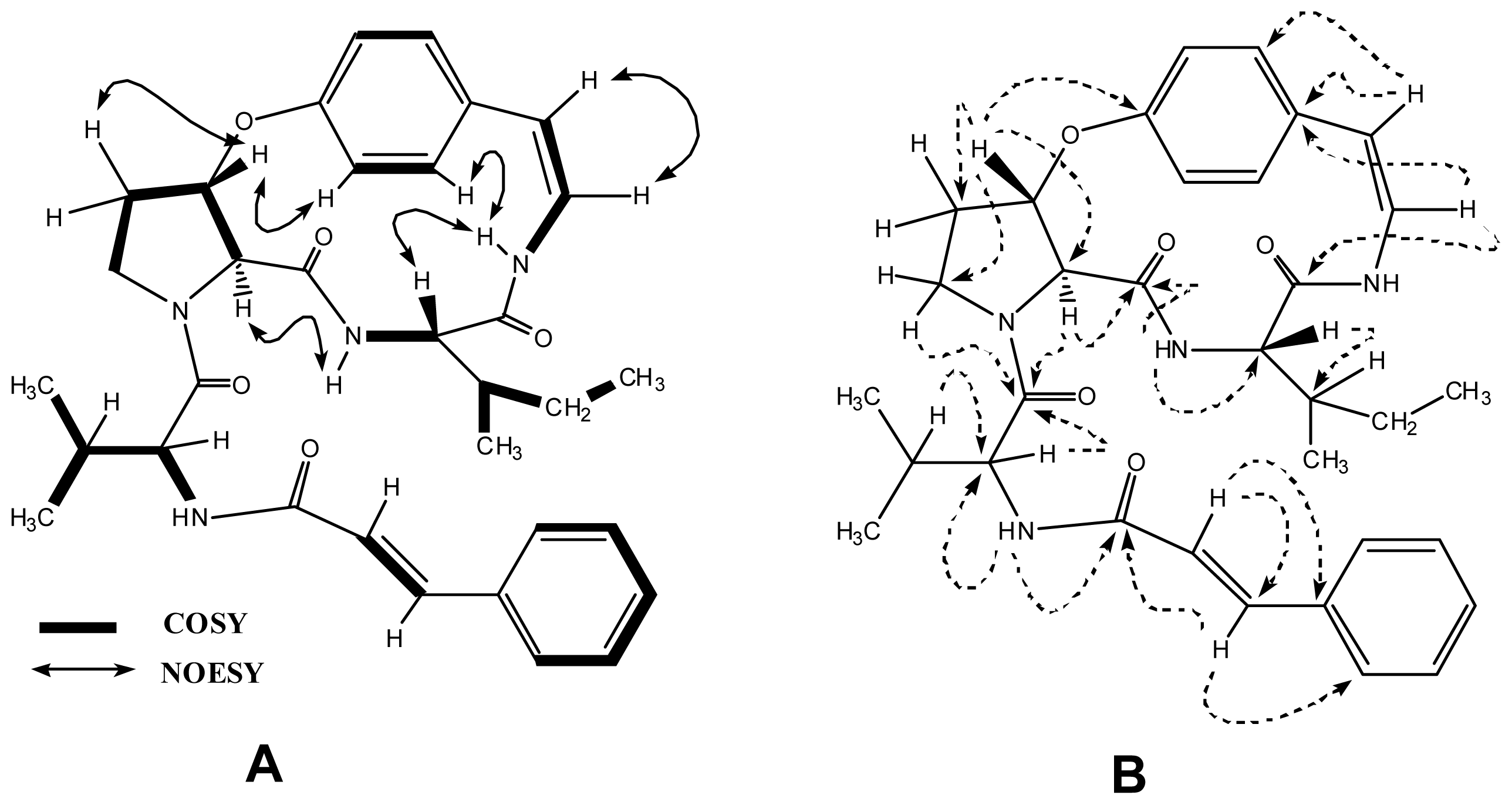
2. Results and Discussion
3. Experimental Section
3.1. General Procedures
3.2. Plant Material Extraction and Isolation of Compounds
3.3. Antibacterial Activity
4. Conclusions
References
- Shah, A.H.; Qureshi, S.; Tariq, M.; Ageel, A.M. Toxicity studies on six plants used in Traditional Arab System of Medicine. Phytother. Res 1989, 3, 25–29. [Google Scholar]
- Nisar, M.; Adzu, B.; Inamullah, K.; Bashir, A.; Ihsan, A.; Gilani, A.H. Antinociceptive and antipyretic activities of the Zizyphus oxyphylla Edgew leaves. Phytother. Res 2007, 21, 693–695. [Google Scholar]
- Schmidt, U.; Lieberknecht, A.; Haslinger, E. The Alkuloids; Academic Press: New York, 1985. [Google Scholar]
- Zia-Ul-Haq, M.; Ahmad, M.; Mehjabeen; Jehan, N.; Ahmad, S.; Qayum, M.; Marwat, I.K. Antimicrobial screening of selected flora of Pakistan. Arch. Biol. Sci 2011, 63, 691–695. [Google Scholar]
- Nisar, M.; Kaleem, W.A.; Qayum, M.; Hussain, A.; Zia-Ul-Haq, M.; Ali, I.; Choudhary, M.I. Biological screening of Zizyphus oxyphylla Edgew leaves. Pak. J. Bot 2010, 42, 4063–4069. [Google Scholar]
- Nisar, M.; Kaleem, W.A.; Adhikari, A.; Ali, Z.; Hussain, N.; Khan, I.; Qayum, M.; Choudhary, M.I. Stereochemistry and NMR data assignment of cyclopeptide alkaloids from Zizyphus oxyphylla. Nat. Product Commun 2010, 5, 1205–1208. [Google Scholar]
- Nisar, M.; Kaleem, W.A.; Qayum, M.; Marwat, I.K.; Zia-UL-Haq, M.; Ali, I.; Choudhary, M.I. Biological screening of Zizyphus oxyphylla Edgew stem. Pak. J. Bot 2011, 43, 311–317. [Google Scholar]
- Inayat-ur-Rahman; Khan, M.A.; Arfan, M.; Akhtar, G.; Khan, L.; Ahmad, V.U. A new 14-membered cyclopeptide alkaloid from Zizyphus oxyphylla. Nat. Product. Res 2007, 21, 243–253. [Google Scholar]
- Tschesche, R.; Miana, G.A.; Eckhardt, G. Nummularin-A, -B und -C, drei neue 13 gliedrige peptidalkaloide aus Ziziphus nummularia. Chem. Ber 1974, 107, 3180–3185. [Google Scholar]
- Devi, S.; Pandey, V.B.; Singh, J.P.; Shah, A.H. Peptide alkaloids from Zizyphus species. Phytochemistry 1987, 26, 3374–3375. [Google Scholar]
- Triphati, Y.C.; Maurya, S.K.; Singh, V.P.; Pandey, V.B. Cyclopeptide alkaloids from Zizyphus rugosa bark. Phytochemistry 1988, 28, 1563–1565. [Google Scholar]
- Morel, A.F.; Araujo, C.A.; da Silva, U.F.; Hoelzel, S.C.S.M.; Zachia, R.; Bastos, N.R. Antibacterial cyclopeptide alkaloids from the bark of Condalia buxifolia. Phytochemistry 2002, 61, 561–566. [Google Scholar]
- Khaliq Jan, A.; Shah, M.R.; Anis, I.; Marwat, I.K. In vitro antifungal and antibacterial activities of extracts of Galium tricornutum subsplongipedunculatum. J. Enzym. Inhib. Med. Chem 2009, 24, 192–196. [Google Scholar]
- Nisar, M.; Khan, I.; Ahmad, B.; Ali, I.; Ahmad, W.; Choudhary, M.I. Antifungal and antibacterial activities of Taxus wallichiana Zucc. J. Enzym. Inhib. Med. Chem 2008, 23, 256–260. [Google Scholar]
- Nisar, M.; Tariq, S.A.; Marwat, I.K.; Shah, M.R.; Khan, I.A. Antibacterial, antifungal, insecticidal, cytotoxicity and phytotoxicity studies on Indigofera gerardiana. J. Enzym. Inhib. Med. Chem 2009, 24, 224–229. [Google Scholar]


| Position | Oxyphylline B (4) | Oxyphylline C (5) | ||
|---|---|---|---|---|
| δH (J in Hz) | δC, Multiplicity | δH (J in Hz) | δC, Multiplicity | |
| 1 | - | 158.7, C | - | 156.8, C |
| 3 | 5.45 ddd (7.0, 5.5, 3.0) | 85.3, CH | 5.80 d (J3,4 = 7.2 Hz) | 83.8, CH |
| 4 | 4.26 d (5.5) | 65.4, CH | 4.80 dd (J4,26 = 10.2, J4,3 = 7.2 Hz) | 57.0, CH |
| 5 | - | 172.1, C | 172.0, CO | |
| 6 | 6.90 d (4.0), NH | - | ||
| 7 | 4.10 dd (9.5, 4.0) | 59.2,CH | 3.95 br d (J7,23 = 7.8 Hz) | 59.8, CH |
| 8 | - | 168.1, C | _ | 167.7, CO |
| 9 | 7.54 d (9.5) | -, NH | 7.51 d (J9,10 = 10.0 Hz) | _, NH |
| 10 | 6.10 dd (9.5, 7.5) | 126.6, CH | 6.65 dd (J10,9 = 10.0 Hz, J10,11 = 7.2 Hz) | 126.7, CH |
| 11 | 6.38 d (7.5) | 117.3, CH | 6.42 d (J11,10 = 7.2 Hz) | 116.7, CH |
| 12 | - | 133.4, C | - | 133.0, C |
| 13 | 7.18 dd (8.5, 2.5) | 123.6, CH | 7.35 dd (J13,14 = 8.4 Hz, J13,16 = 3.0 Hz) | 123.2, CH |
| 14 | 7.06 dd (8.5, 2.5) | 131.5, CH | 7.1 dd (J14,13 = 8.4 Hz, J14,15 = 3.0 Hz) | 131.4, CH |
| 15 | 7.37 (8.5,2.5) | 130.3, CH | 7.0 dd (J15,16 = 8.4 Hz, J15,14 = 3.0 Hz) | 132.5, CH |
| 16 | 7.30 dd (8.5, 2.5) | 122.7, CH | 7.27 dd (J16,15 = 8.4 Hz, J16,13 = 3.0 Hz) | 123.9, CH |
| 17 | 1.99 m | 37.1, CH | _ | 139.0, C |
| 18 | 1.11 m, 1.59 m | 24.8, CH2 | 7.71 br. d (J18,19 = 7.2 Hz) | 129.0, CH |
| 19 | 0.78 t (7.5) | 16.2, CH3 | 7.15 br t (7.2) | 128.6, CH |
| 20 | 0.77 d (7.5) | 12.3, CH3 | 7.29 br t (7.2) | 128.7, CH |
| 21 | 1.28 m, 2.15 m | 32.8, CH2 | 7.15 br t (7.2) | 128.6, CH |
| 22 | 3.50 m, 4.48 m | 47.2, CH2 | 7.71 br. d (J18,19 = 7.2 Hz) | 129.0, CH |
| 23 | - | 1.51 m, 2.11 m | 25.9, CH2 | |
| 24 | - | 172.0, C | 1.35 m, 1.74 m | 25.1, CH2 |
| 25 | 4.50 t (8.5) | 56.6, CH | 3.07 t (8.4), 3.28 m | 47.4, CH2 |
| 26 | 1.97 m | 31.8, CH | 7.78 d (J26,4 = 10.2 Hz) | NH |
| 27 | 0.84 d (6.5) | 19.4, CH3 | _ | 169.7, CO |
| 28 | 0.94 d (6.5) | 19.1, CH3 | 4.39 m | 56.1,CH |
| 29 | 7.6 d (8.5) | -, NH | 2.73 dd (11.4,10.8), 3.29 dd ( 11.4,3.6) | 37.0, CH2 |
| 30 | - | 166.2, C | - | 138.5, C |
| 31 | 6.85 d (15.5) | 122.3, CH | 7.12 br d (7.2) | 129.8, CH |
| 32 | 7.58 d (15.5) | 141.0, CH | 7.20 br t (7.2) | 129.3, CH |
| 33 | - | 136.3, C | 7.15 br t (7.2) | 130.7, CH |
| 34 | 7.39 br d (7.5) | 129.7, CH | 7.20 br t (7.2) | 129.3,CH |
| 35 | 7.57 br t (7.5) | 128.5, CH | 7.12 br d (7.2) | 129.8,CH |
| 36 | 7.07 br t (7.5) | 132.6, CH | 7.11 d (7.5) | _, NH |
| 37 | 7.39 br d (7.5) | 129.7, CH | - | 166.4, CO |
| 38 | 7.57 br t (7.5) | 128.5, CH | 6.65 d (15.0) | 120.0, CH |
| 39 | 7.53 d (15.0) | 142.7, CH | ||
| 40 | 136.2, C | |||
| 41,45 | 7.10 br d (7.2) | 129.0, CH | ||
| 42,44 | 7.20 br t (7.2) | 128 .6, CH | ||
| 43 | 7.07 br t (7.2) | 129.8, CH | ||
| MIC (μg/mL) | ||||||
|---|---|---|---|---|---|---|
| Compound | Escherichia coli | Bacillus subtilis | Shigella flexeneri | Staphylococcus aureus | Pseudomonas aeruginosa | Salmonella typhi |
| 1 | 10 | 25 | 50 | 25 | 50 | 25 |
| 2 | 10 | 25 | 100 | 25 | 100 | 10 |
| 3 | 25 | 10 | 100 | 50 | 100 | 50 |
| 4 | 5 | 10 | 25 | 25 | 50 | 10 |
| 5 | 10 | 50 | 50 | 10 | 50 | 50 |
| Standard | 0.0002 | 0.0005 | 0.0003 | 0.0009 | 0.0021 | 0.0014 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kaleem, W.A.; Nisar, M.; Qayum, M.; Zia-Ul-Haq, M.; Adhikari, A.; De Feo, V. New 14-Membered Cyclopeptide Alkaloids from Zizyphus oxyphylla Edgew. Int. J. Mol. Sci. 2012, 13, 11520-11529. https://doi.org/10.3390/ijms130911520
Kaleem WA, Nisar M, Qayum M, Zia-Ul-Haq M, Adhikari A, De Feo V. New 14-Membered Cyclopeptide Alkaloids from Zizyphus oxyphylla Edgew. International Journal of Molecular Sciences. 2012; 13(9):11520-11529. https://doi.org/10.3390/ijms130911520
Chicago/Turabian StyleKaleem, Waqar Ahmad, Muhammad Nisar, Mughal Qayum, Muhammad Zia-Ul-Haq, Achyut Adhikari, and Vincenzo De Feo. 2012. "New 14-Membered Cyclopeptide Alkaloids from Zizyphus oxyphylla Edgew" International Journal of Molecular Sciences 13, no. 9: 11520-11529. https://doi.org/10.3390/ijms130911520







