Ultrasound-Assisted Extraction of Carnosic Acid and Rosmarinic Acid Using Ionic Liquid Solution from Rosmarinus officinalis
Abstract
:1. Introduction
2. Results and Discussion
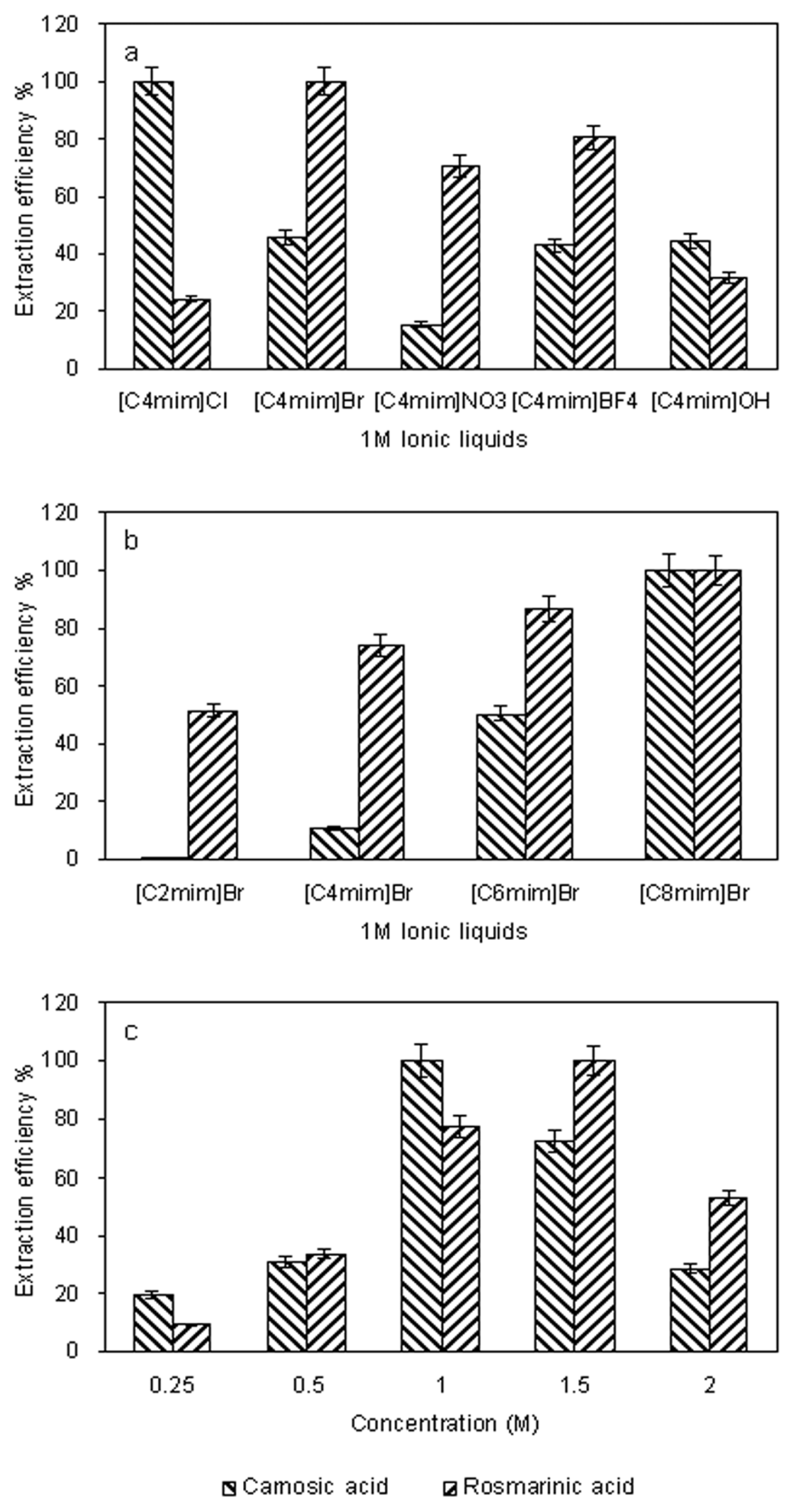
2.1. Screening of the Ionic Liquid-Based Extracting Solvent
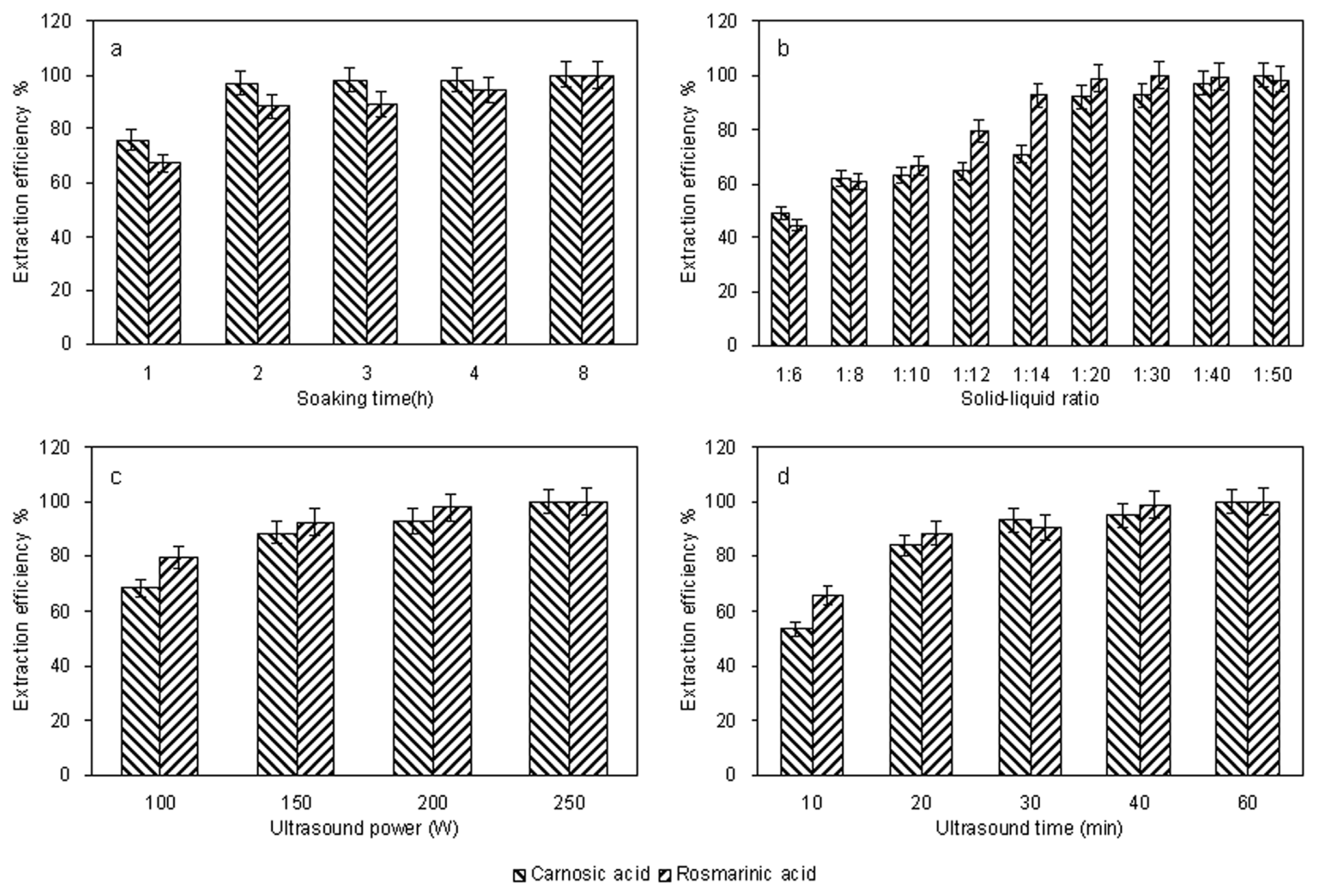
2.2. Optimization of Extraction Conditions
2.2.1. Single Factor Experiments
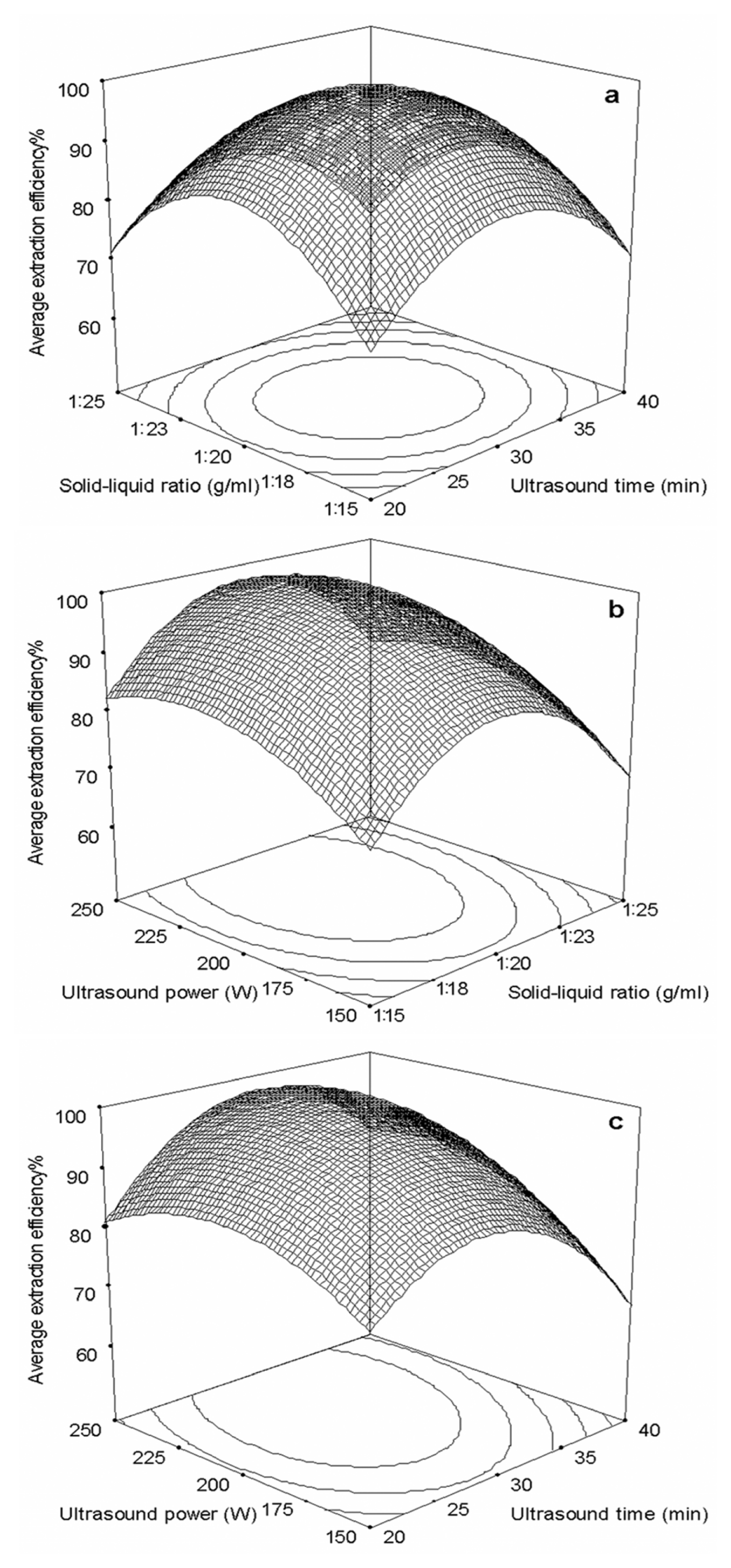
2.2.2. Optimization Parameters by Response Surface Methodology
2.3. Verification Tests
2.4. Comparison of ILUAE Approach with the Reference and Conventional Methods
2.5. Method Validation
3. Experimental Section
3.1. Materials and Reagents
3.2. Apparatus
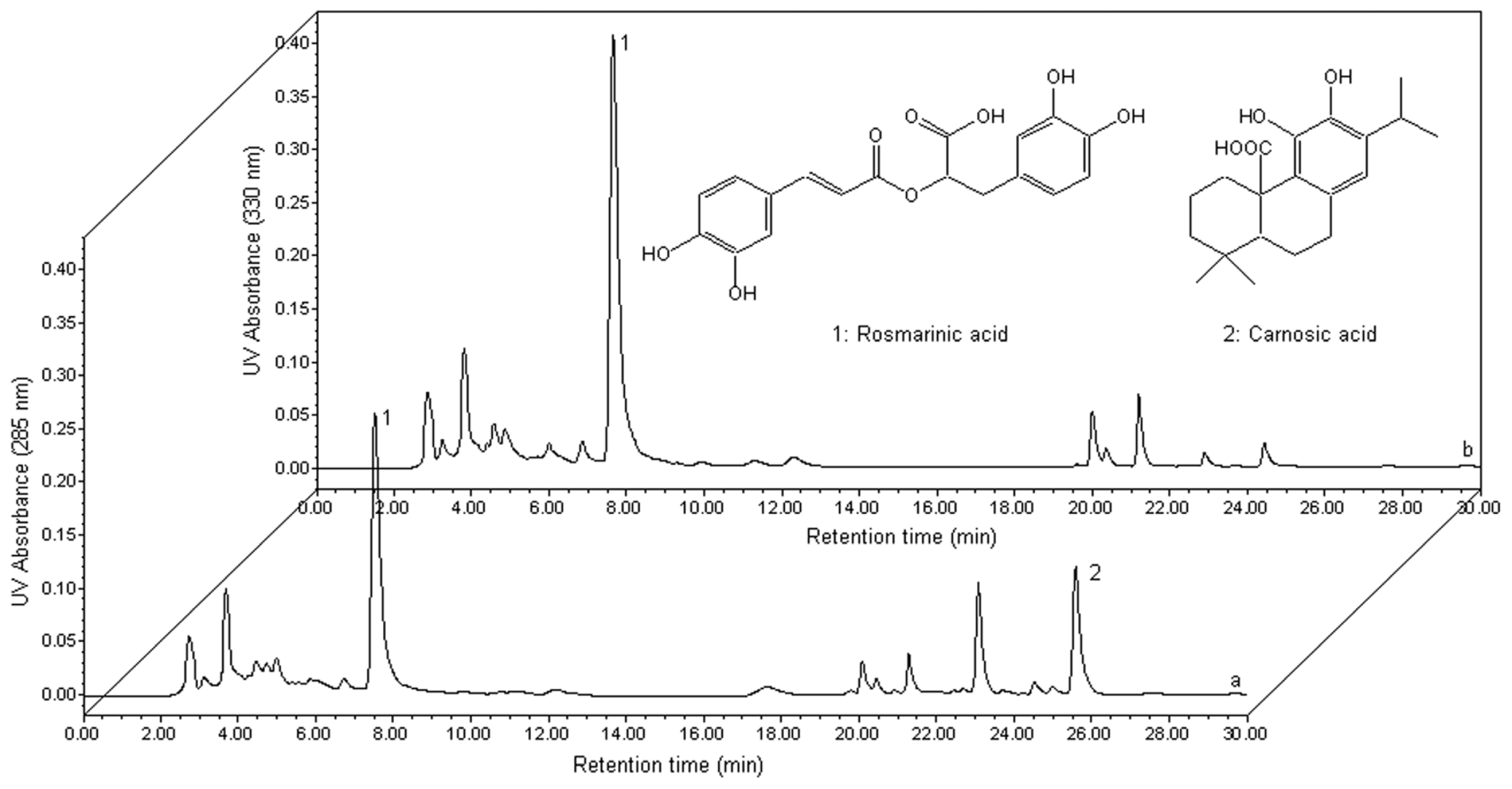
3.3. HPLC Analysis and Quantification
3.4. Ionic Liquid-Based Ultrasound-Assisted Extraction (ILUAE)
3.5. Optimization of ILUAE by Response Surface Methodology
3.6. Method Validation
3.7. Reference and Conventional Extraction Methods
3.8. Statistical Analysis
4. Conclusions
References
- Zunin, P.; Leardi, R.; Bisio, A.; Boggia, R.; Romussi, G. Oxidative stability of virgin olive oil enriched with carnosic acid. Food Res. Int 2010, 43, 1511–1516. [Google Scholar]
- Wang, H.; Zu, G.; Yang, L.; Zu, Y.; Wang, H.; Zhang, Z.; Zhang, Y.; Zhang, L.; Wang, H. Effects of heat and ultraviolet radiation on the stability of pine nut oil supplemented with carnosic acid. J. Agric. Food Chem 2011, 59, 13018–13025. [Google Scholar]
- Wang, H.; Wang, H.; Yang, L.; Zu, Y.; Liu, F.; Liu, T. Comparative effect of carnosic acid, BHT and α-tocopherol on the stability of squalene under heating and UV irradiation. Food Res. Int 2011, 44, 2370–2374. [Google Scholar]
- Wang, H.; Liu, F.; Yang, L.; Zu, Y.; Wang, H.; Qu, S.; Zhang, Y. Oxidative stability of fish oil supplemented with carnosic acid compared with synthetic antioxidants during long-term storage. Food Chem 2011, 128, 93–99. [Google Scholar]
- Bernardes, W.A.; Lucarini, R.; Tozatti, M.G.; Souza, M.G.M.; Silva, M.L.A.; Filho, A.A.S.; Martins, C.H.G.; Crotti, A.E.M.; Pauletti, P.M.; Groppo, M.; et al. Antimicrobial activity of Rosmarinus officinalis against oral pathogens: Relevance of carnosic acid and carnosol. Chem. Biodivers 2010, 7, 1835–1840. [Google Scholar]
- Poeckel, D.; Greiner, C.; Verhoff, M.; Rau, O.; Tausch, L.; Hörnig, C.; Steinhilber, D.; Schubert-Zsilavecz, M.; Werz, O. Carnosic acid and carnosol potently inhibit human 5-lipoxygenase and suppress pro-inflammatory responses of stimulated human polymorphonuclear leukocytes. Biochem. Pharm 2008, 76, 91–97. [Google Scholar]
- Yesil-Celiktas, O.; Sevimli, C.; Bedir, E.; Vardar-Sukan, F. Inhibitory effects of rosemary extracts, carnosic acid and rosmarinic acid on the growth of various human cancer cell lines. Plant Food Hum. Nutr 2010, 65, 158–163. [Google Scholar]
- Manoharan, S.; Selvan, M.; Selvan, V.; Silvan, S.; Baskaran, N.; Singh, A.K.; Kumar, V.V. Carnosic acid: A potent chemopreventive agent against oral carcinogenesis. Chem. Biol. Interact 2010, 188, 616–622. [Google Scholar]
- Satoh, T.; Kosaka, K.; Itoh, K.; Kobayashi, A.; Yamamoto, M.; Shimojo, Y.; Kitajima, C.; Cui, J.; Kamins, J.; Okamoto, S.; et al. Carnosic acid, a catechol-type electrophilic compound, protects neurons both in vitro and in vivo through activation of the Keap1/Nrf2 pathway via S-alkylation of targeted cysteines on Keap1. J. Neurochem 2008, 104, 1116–1131. [Google Scholar]
- Ninomiya, K.; Matsuda, H.; Shimoda, H.; Nishida, N.; Kasajima, N.; Yoshino, T.; Morikawa, T.; Yoshikawa, M. Carnosic acid, a new class of lipid absorption inhibitor from sage. Bioorg. Med. Chem. Lett 2004, 14, 1943–1946. [Google Scholar]
- Shanlou, Q.; Weihua, L.; Ryoko, T.; Miyako, H.; Keiko, M.; Fumio, T.; Yukio, N.; Masataka, Y. Rosmarinic acid inhibits the formation of reactive oxygen and nitrogen species in RAW264.7 macrophages. Free Radic. Res 2005, 39, 995–1003. [Google Scholar]
- Sui, X.; Liu, T.; Ma, C.; Yang, L.; Zu, Y.; Zhang, L.; Wang, H. Microwave irradiation to pretreat rosemary (Rosmarinus officinalis L.) for maintaining antioxidant content during storage and to extract essential oil simultaneously. Food Chem 2012, 131, 1399–1405. [Google Scholar]
- Osakabe, N.; Takano, H.; Sanbongi, C.; Yasuda, A.; Yanagisawa, R.; Inoue, K.; Yoshikawa, T. Anti-inflammatory and anti-allergic effect of rosmarinic acid (RA); inhibition of seasonal allergic rhinoconjunctivitis (SAR) and its mechanism. Biol. Factors 2004, 21, 127–131. [Google Scholar]
- Furtado, M.A.; de Almeida, L.C.F.; Furtado, R.A.; Cunha, W.R.; Tavares, D.C. Antimutagenicity of rosmarinic acid in Swiss mice evaluated by the micronucleus assay. Mutat. Res. Gen. Toxicol. Environ 2008, 657, 150–154. [Google Scholar]
- Huang, S.S.; Zheng, R.L. Rosmarinic acid inhibits angiogenesis and its mechanism of action in vitro. Cancer Lett 2006, 239, 271–280. [Google Scholar]
- Kim, D.S.; Kim, H.R.; Woo, E.R.; Hong, S.T.; Chae, H.J.; Chae, S.W. Inhibitory effects of rosmarinic acid on adriamycin-induced apoptosis in H9c2 cardiac muscle cells by inhibiting reactive oxygen species and the activations of c-Jun N-terminal kinase and extracellular signal-regulated kinase. Biochem. Pharm 2005, 70, 1066–1078. [Google Scholar]
- Li, G.S.; Jiang, W.L.; Tian, J.W.; Qu, G.W.; Zhu, H.B.; Fu, F.H. In vitro and in vivo antifibrotic effects of rosmarinic acid on experimental liver fibrosis. Phytomedicine 2010, 17, 282–288. [Google Scholar]
- Debersac, P.; Vernevaut, M.F.; Amiot, M.J.; Suschetet, M.; Siess, M.H. Effects of a water-soluble extract of rosemary and its purified component rosmarinic acid on xenobiotic-metabolizing enzymes in rat liver. Food Chem. Toxicol 2001, 39, 109–117. [Google Scholar]
- Fallarini, S.; Miglio, G.; Paoletti, T.; Minassi, A.; Amoruso, A.; Bardelli, C.; Brunelleschi, S.; Lombardi, G. Clovamide and rosmarinic acid induce neuroprotective effects in vitro models of neuronal death. Br. J. Pharm 2009, 157, 1072–1084. [Google Scholar]
- Lee, J.; Jung, E.; Koh, J.; Kim, Y.S.; Park, D. Effect of rosmarinic acid on atopic dermatitis. J. Dermatol 2008, 35, 768–771. [Google Scholar]
- Psotova, J.; Svobodova, A.; Kolarova, H.; Walterova, D. Photoprotective properties of Prunella vulgaris and rosmarinic acid on humankeratinocytes. J. Photochem. Photobiol. B Biol 2006, 84, 167–174. [Google Scholar]
- Hamaguchi, T.; Ono, K.; Murase, A.; Yamada, M. Phenolic compounds prevent Alzheimer’s pathology through different effects on the amyloid-β aggregation pathway. Am. J. Pathol 2009, 175, 2557–2565. [Google Scholar]
- Ma, C.; Wang, S.; Yang, L.; Zu, Y. Ionic liquid-based ultrasonic-assisted extraction of camptothecin and 10-hydroxycamptothecin from samara of Camptotheca acuminate. Chem. Eng. Process. Process Intensif 2012, 57–58, 59–64. [Google Scholar]
- Wang, S.; Yang, L.; Zu, Y.; Zhao, C.; Sun, X.; Zhang, L.; Zhang, Z. Design and performance evaluation of ionic liquids-microwave based environmental-friendly extraction technique for camptothecin and 10-hydroxycamptothecin from samara of Camptotheca acuminate. Ind. Eng. Chem. Res 2011, 50, 13620–13627. [Google Scholar]
- Liu, T.; Sui, X.; Zhang, R.; Yang, L.; Zu, Y.; Zhang, L.; Zhang, Y.; Zhang, Z. Application of ionic liquids based microwave-assisted simultaneous extraction of carnosic acid, rosmarinic acid and essential oil from Rosmarinus officinalis. J. Chromatogr. A 2011, 1218, 8480–8489. [Google Scholar]
- Jiao, Y.; Zuo, Y. Ultrasonic extraction and HPLC determination of anthraquinones, aloe-emodine, emodine, rheine, chrysophanol, and physcione, in Radix Polygoni multiflori. Phytochem. Anal 2009, 20, 272–278. [Google Scholar]
- Zuo, Y.; Zhang, L.; Wu, J.; Fritz, J.W.; Medeiros, S.; Rego, C. Ultrasonic extraction and capillary gas chromatography determination of nicotine in pharmaceutical formulations. Anal. Chim. Acta 2004, 526, 35–39. [Google Scholar]
- Chemat, F.; Zill-e-Huma; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem 2011, 18, 813–835. [Google Scholar]
- Rodrigues, S.; Pinto, G.A.S. Ultrasound extraction of phenolic compounds from coconut (Cocos nucifera) shell powder. J. Food Eng 2007, 80, 869–872. [Google Scholar]
- Pereiro, A.B.; Rodríguez, A. An ionic liquid proposed as solvent in aromatic hydrocarbon separation by liquid extraction. AIChE J 2010, 56, 381–386. [Google Scholar]
- Hernández-Fernández, F.J.; de los Ríos, A.P.; Gómez, D.; Rubio, M.; Víllora, G. Selective extraction of organic compounds from transesterification reaction mixtures by using ionic liquids. AIChE J 2010, 56, 1213–1217. [Google Scholar]
- Yang, L.; Wang, H.; Zu, Y.; Zhao, C.; Zhang, L.; Chen, X.; Zhang, Z. Ultrasound-assisted extraction of the three terpenoid indole alkaloids vindoline, catharanthine and vinblastine using ionic liquid solution from Catharanthus roseus. Chem. Eng. J 2011, 172, 705–712. [Google Scholar]
- Cao, X.; Ye, X.; Lu, Y.; Yu, Y.; Mo, W. Ionic liquid-based ultrasonic-assisted extraction of piperine from white pepper. Anal. Chim. Acta 2009, 640, 47–51. [Google Scholar]
- Du, F.Y.; Xiao, X.H.; Li, G.K. Application of ionic liquids in the microwave-assisted extraction of trans-resveratrol from Rhizma Polygoni Cuspidati. J. Chromatogr. A 2007, 1140, 56–62. [Google Scholar]
- Yang, L.; Sun, X.; Yang, F.; Zhao, C.; Zhang, L.; Zu, Y. Application of ionic liquids in the microwave-assisted extraction of proanthocyanidins from Larix gmelini bark. Int. J. Mol. Sci 2012, 13, 5163–5178. [Google Scholar]
- Wu, K.K.; Zhang, Q.L.; Liu, Q.; Tang, F.; Long, Y.M.; Yao, S.Z. Ionic liquid surfactant-mediated ultrasonic-assisted extraction coupled to HPLC: Application to analysis of tanshinones in Salvia miltiorrhiza Bunge. J. Sep. Sci 2009, 32, 4220–4226. [Google Scholar]
- Ma, C.; Liu, T.; Yang, L.; Zu, Y.; Wang, S.; Zhang, R. Study on ionic liquid-based ultrasonic-assisted extraction of biphenyl cyclooctene lignans from the fruit of Schisandra chinensis Baill. Anal. Chim. Acta 2011, 689, 110–116. [Google Scholar]
- Ma, C.; Liu, T.; Yang, L.; Zu, Y.; Chen, X.; Zhang, L.; Zhang, Y.; Zhao, C. Ionic liquid based microwave simultaneous extraction of essential oil and biphenyl cyclooctene lignans from Schisandra chinensis Baill fruits. J. Chromatogr. A 2011, 1218, 8573–8580. [Google Scholar]
- Yang, L.; Liu, Y.; Zu, Y.; Zhao, C.; Zhang, L.; Chen, X.; Zhang, Z. Optimize the process of ionic liquid-based ultrasonic-assisted extraction of aesculin and aesculetin from Cortex Fraxini by response surface methodology. Chem. Eng. J 2011, 175, 539–547. [Google Scholar]
- Manic, M.S.; Najdanovic-Visak, V.; da Ponte, M.N.; Visak, Z.P. Extraction of free fatty acids from soybean oil using ionic liquids or poly(ethyleneglycol)s. AIChE J 2011, 57, 1344–1355. [Google Scholar]
- Balachandran, S.; Kentish, S.E.; Mawson, R.; Ashokkumar, M. Ultrasonic enhancement of the supercritical extraction from ginger. Ultrason. Sonochem 2006, 13, 471–479. [Google Scholar]
- Li, H.; Pordesimo, L.O.; Weiss, J. High-intensity ultrasound assisted extraction of oil from soybeans. Food Res. Int 2004, 37, 731–738. [Google Scholar]
- Chemat, F.; Grondin, I.; Costes, P.; Moutoussamy, L.; Sing, A.S.C.; Smadja, J. High power ultrasound effects on lipid oxidation of refined sunflower oil. Ultrason. Sonochem 2004, 11, 281–285. [Google Scholar]
- Chemat, F.; Grondin, I.; Sing, A.S.C.; Smadja, J. Deterioration of edible oils during food processing by ultrasound. Ultrason. Sonochem 2004, 11, 13–15. [Google Scholar]
| Run | Independent variables | Response | ||
|---|---|---|---|---|
| X1 (min) | X2 (g/mL) | X3 (W) | Average extraction efficiency (%) a | |
| 1 | 30 (0) | 1:20 (0) | 200 (0) | 98.75 |
| 2 | 40 (1) | 1:20 (0) | 250 (1) | 86.00 |
| 3 | 40 (1) | 1:25 (1) | 200 (0) | 65.00 |
| 4 | 20 (−1) | 1:25 (1) | 200 (0) | 72.10 |
| 5 | 30 (0) | 1:20 (0) | 200 (0) | 99.30 |
| 6 | 30 (0) | 1:25 (1) | 150 (−1) | 68.04 |
| 7 | 40 (1) | 1:20 (0) | 150 (−1) | 67.77 |
| 8 | 20 (−1) | 1:20 (0) | 250 (1) | 80.40 |
| 9 | 30 (0) | 1:20 (0) | 200 (0) | 97.94 |
| 10 | 30 (0) | 1:20 (0) | 200 (0) | 99.40 |
| 11 | 30 (0) | 1:20 (0) | 200 (0) | 100.00 |
| 12 | 30 (0) | 1:15 (−1) | 250 (1) | 82.99 |
| 13 | 40 (1) | 1:15 (−1) | 200 (0) | 70.31 |
| 14 | 20 (−1) | 1:20 (0) | 150 (−1) | 77.68 |
| 15 | 30 (0) | 1:25 (1) | 250 (1) | 81.04 |
| 16 | 20 (−1) | 1:15 (−1) | 200 (0) | 70.37 |
| 17 | 30 (0) | 1:15 (−1) | 150 (−1) | 71.83 |
| Source | Sum of squares | Degree of freedom | Mean square | F-value | p-value |
|---|---|---|---|---|---|
| Model b | 2652.90 | 9 | 294.77 | 389.49 | <0.0001 |
| X1 | 16.41 | 1 | 16.41 | 21.69 | 0.0023 |
| X2 | 10.86 | 1 | 10.86 | 14.36 | 0.0068 |
| X3 | 254.26 | 1 | 254.26 | 335.97 | <0.0001 |
| X1X2 | 12.42 | 1 | 12.42 | 16.41 | 0.0049 |
| X1X3 | 60.24 | 1 | 60.24 | 79.60 | <0.0001 |
| X2X3 | 0.85 | 1 | 0.85 | 1.12 | 0.3249 |
| X12 | 804.65 | 1 | 804.65 | 1063.23 | <0.0001 |
| X22 | 1052.30 | 1 | 1052.30 | 1390.46 | <0.0001 |
| X32 | 224.08 | 1 | 224.08 | 296.09 | <0.0001 |
| Residual | 5.30 | 7 | 0.76 | ||
| Lack of Fit | 2.90 | 3 | 0.97 | 1.61 | 0.3207 |
| Pure Error | 2.40 | 4 | 0.60 | ||
| Cor Total | 2658.19 | 16 | |||
| Pred R2 | 0.981 | ||||
| Adj R2 | 0.995 | ||||
| Adeq Precision | 50.947 | ||||
| No. | Solvent | Methods | Extraction time (h) | Solvent consumption (mL/g) | Extraction efficiency ± SD (%) a | ||
|---|---|---|---|---|---|---|---|
| Carnosic acid | Rosmarinic acid | Average | |||||
| 1 | Pure water | UAE | 0.5 | 20 | 0 | 61.03 ± 3.45 | 61.03 ± 3.45 |
| 2 | 1 M NaBr | UAE | 0.5 | 20 | 0 | 53.54 ± 3.38 | 53.54 ± 3.38 |
| 3 | 80% Ethanol | UAE | 0.5 | 20 | 100.00 ± 5.44 | 64.69 ± 4.23 | 82.35 ± 4.84 |
| 4 | 1 M [C8mim]Br | UAE | 0.5 | 20 | 66.23 ± 3.85 | 100.00 ± 4.76 | 83.12 ± 4.31 |
| 5 | 80% Ethanol | HRE | 4 | 20 | 99.05 ± 4.87 | 67.61 ± 3.42 | 74.87 ± 4.15 |
| 6 | 80% Ethanol | ME | 48 | 20 | 95.04 ± 4.33 | 40.08 ± 2.66 | 48.69 ± 2.50 |
| 7 | 80% Ethanol | SE | 24 | 20 | 96.80 ± 3.96 | 67.03 ± 3.78 | 65.96 ± 3.87 |
| 8 | 80% Ethanol | SLE | 24 | 20 | 95.47 ± 3.34 | 62.22 ± 4.05 | 70.35 ± 3.70 |
| Compounds | Initial concentration (mg/mL) | Recovered concentration after UAE (mg/mL) | RSD% (n = 3) | Average recovery (%) | Recovered concentration after 3 day (mg/mL) | RSD% (n = 3) | Average recovery (%) | Recovered concentration after 15 day (mg/mL) | RSD% (n = 3) | Average recovery (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Carnosic acid | 14.16 | 14.14 | 3.21 | 99.86 | 13.66 | 1.06 | 96.48 | 12.48 | 2.11 | 88.17 |
| Rosmarinic acid | 1.28 | 1.27 | 0.78 | 99.22 | 1.25 | 2.49 | 98.01 | 1.20 | 1.60 | 93.93 |
| Sample | Phenolcarboxylic acids content of the sample determined (mg) | The amount of added phenolcarboxylic acids standards (mg) | The amount of the sample determined with added phenolcarboxylic acids standards (mg) | Recovery (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Carnosic acid | Rosmarinic acid | Carnosic acid | Rosmarinic acid | Carnosic acid | Rosmarinic acid | Carnosic acid | Rosmarinic acid | |
| 1 | 7.8 | 12.0 | 5.0 | 10.0 | 13.3 | 21.5 | 103.9 | 97.7 |
| 2 | 7.8 | 12.0 | 10.0 | 15.0 | 17.2 | 26.6 | 96.6 | 98.5 |
| 3 | 7.8 | 12.0 | 15.0 | 20.0 | 23.6 | 33.5 | 103.5 | 101.6 |
| Average | - | - | - | - | - | - | 101.3 | 99.3 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zu, G.; Zhang, R.; Yang, L.; Ma, C.; Zu, Y.; Wang, W.; Zhao, C. Ultrasound-Assisted Extraction of Carnosic Acid and Rosmarinic Acid Using Ionic Liquid Solution from Rosmarinus officinalis. Int. J. Mol. Sci. 2012, 13, 11027-11043. https://doi.org/10.3390/ijms130911027
Zu G, Zhang R, Yang L, Ma C, Zu Y, Wang W, Zhao C. Ultrasound-Assisted Extraction of Carnosic Acid and Rosmarinic Acid Using Ionic Liquid Solution from Rosmarinus officinalis. International Journal of Molecular Sciences. 2012; 13(9):11027-11043. https://doi.org/10.3390/ijms130911027
Chicago/Turabian StyleZu, Ge, Rongrui Zhang, Lei Yang, Chunhui Ma, Yuangang Zu, Wenjie Wang, and Chunjian Zhao. 2012. "Ultrasound-Assisted Extraction of Carnosic Acid and Rosmarinic Acid Using Ionic Liquid Solution from Rosmarinus officinalis" International Journal of Molecular Sciences 13, no. 9: 11027-11043. https://doi.org/10.3390/ijms130911027





