Chick Chorioallantoic Membrane (CAM) Assay as an In Vivo Model to Study the Effect of Newly Identified Molecules on Ovarian Cancer Invasion and Metastasis
Abstract
:1. Introduction
2. Results
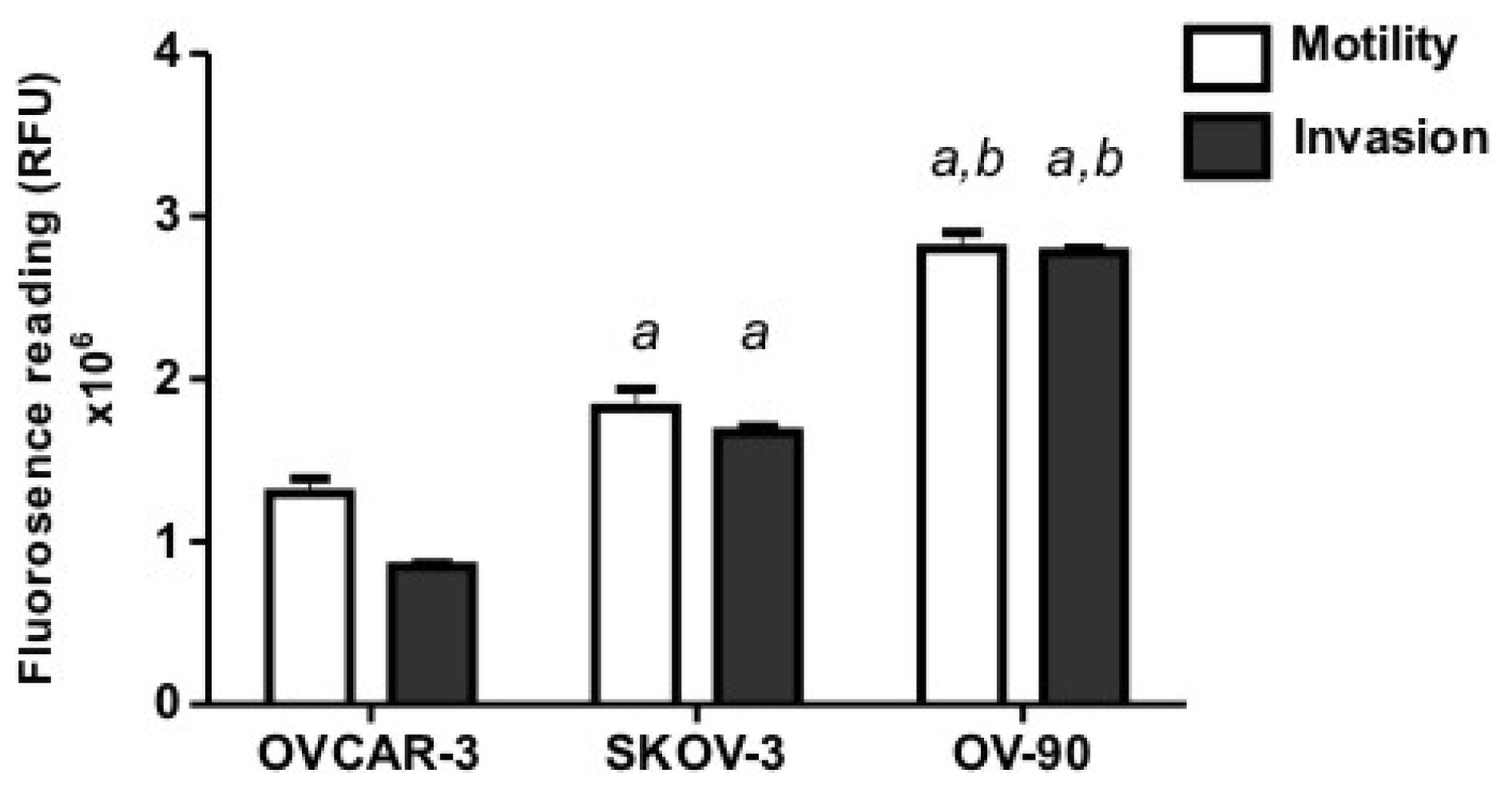
2.1. Human Ovarian Cancer Cell Motility and Invasion In Vitro
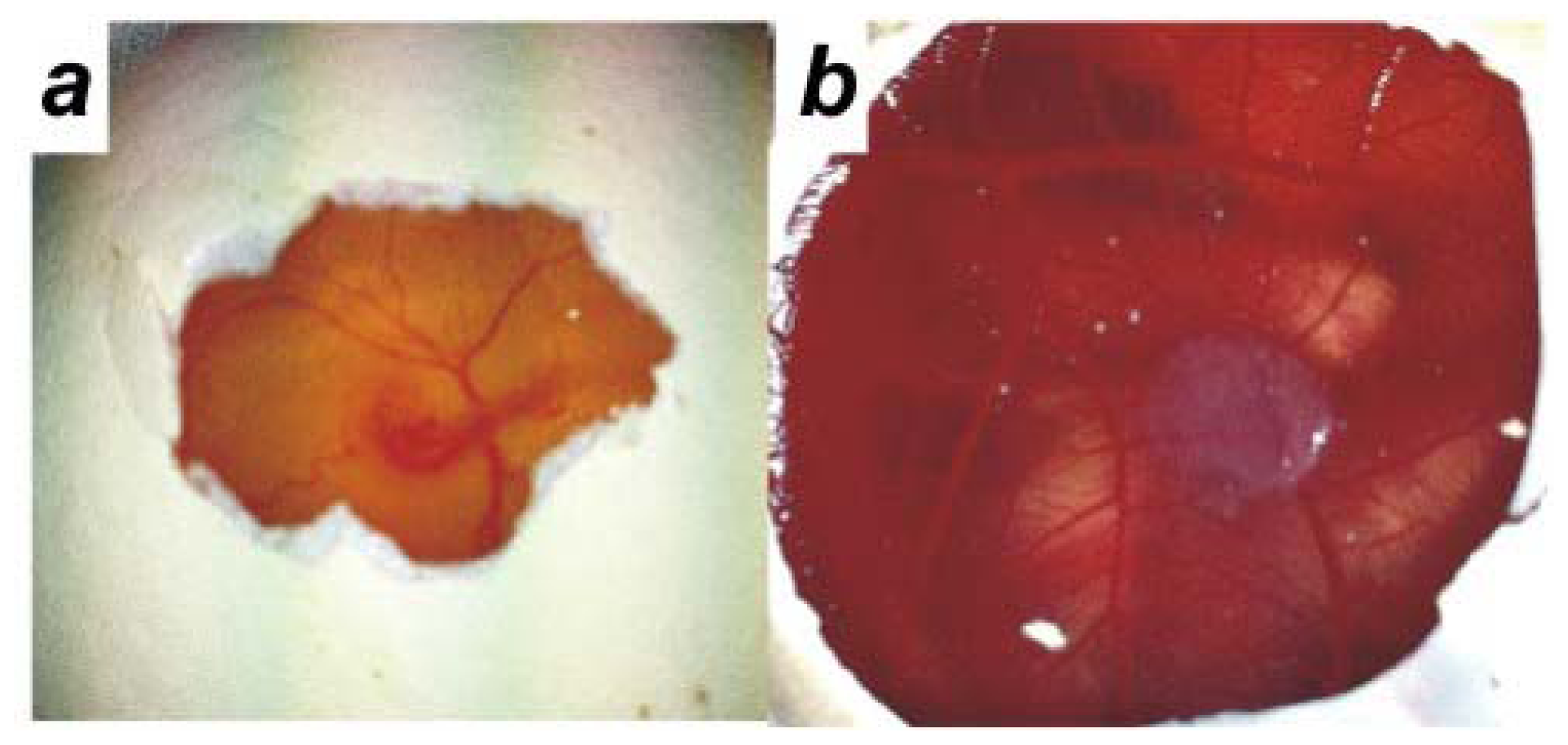
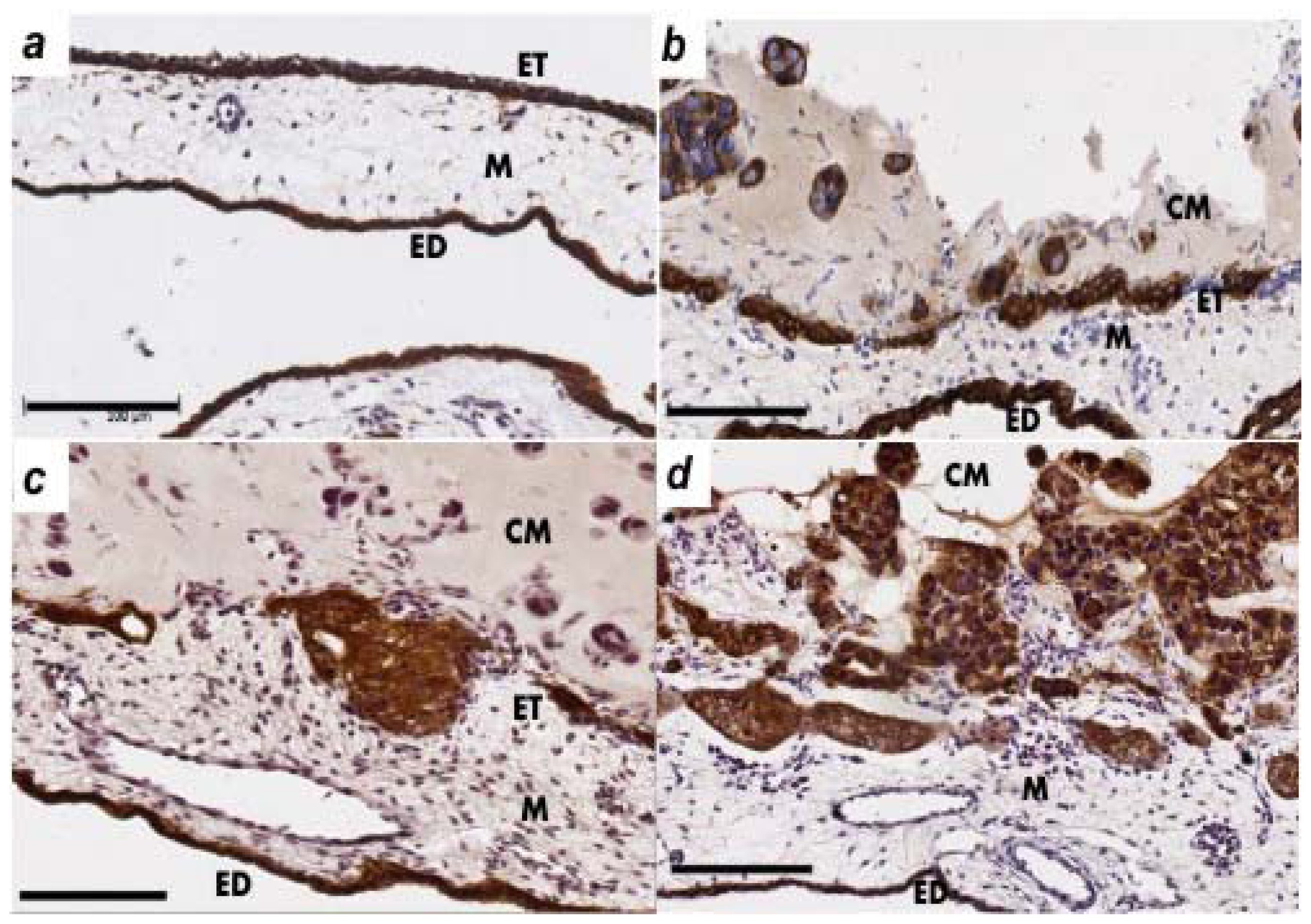
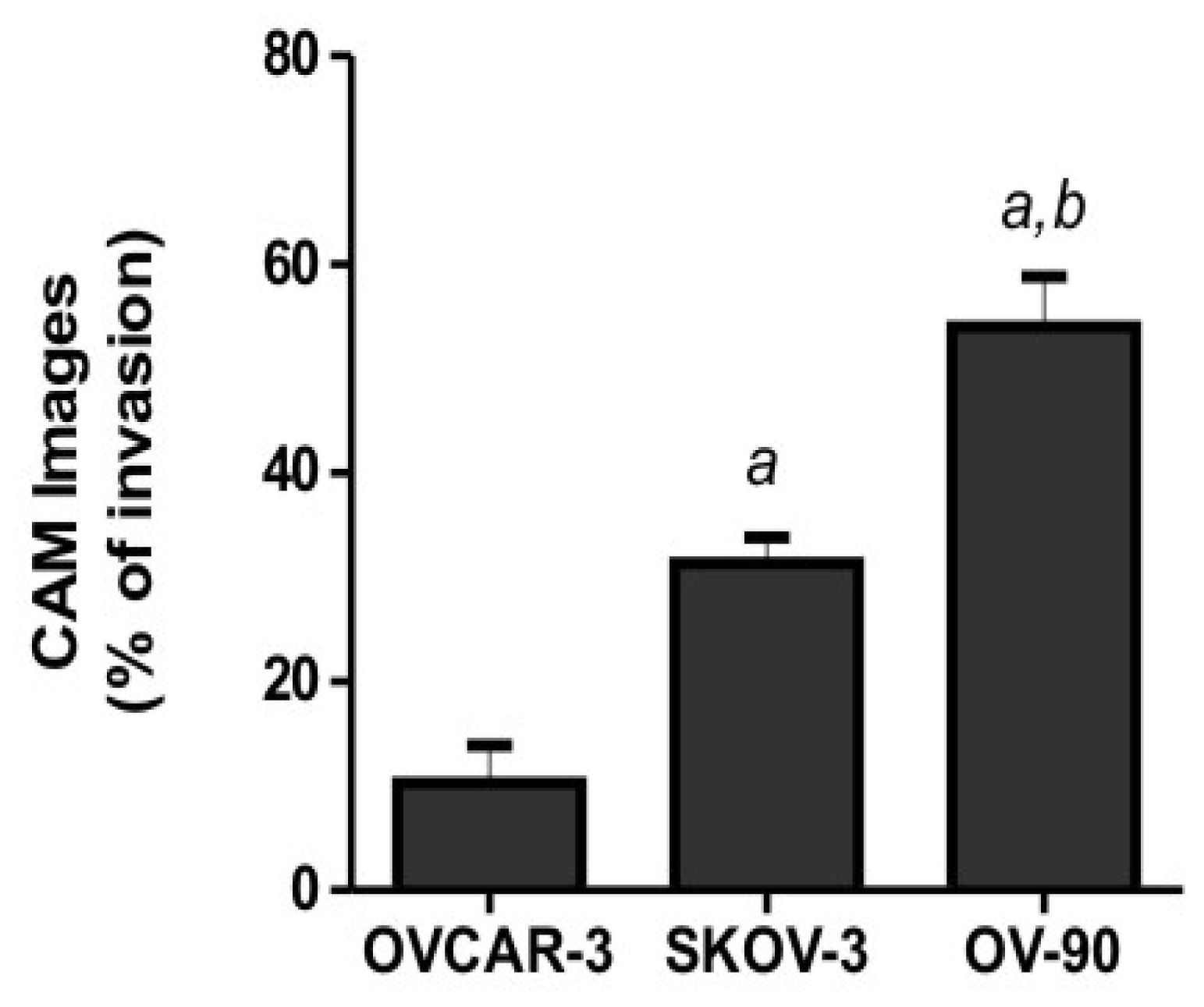
2.2. Human Ovarian Cancer Cell Invasion into the Chick Chorioallantoic Membrane (CAM)
2.3. The Effects of Protein a Neutralizing Antibody on OV-90 Cancer Cell Invasion into the CAM
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Motility and Invasion Assays
4.3. Chick Chorioallantoic Membrane Assay (CAM Assay)
4.4. Immunohistochemistry
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
References
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol 2010, 177, 1053–1064. [Google Scholar]
- Richardson, M.; Singh, G. Observations on the use of the avian chorioallantoic membrane (CAM) model in investigations into angiogenesis. Curr. Drug Targets-Cardiovasc. Hematol. Disord 2003, 3, 155–185. [Google Scholar]
- Deryugina, E.I.; Quigley, J.P. Chick embryo chorioallantoic membrane model systems to study and visualize human tumor cell metastasis. Histochem. Cell Biol 2008, 130, 1119–1130. [Google Scholar]
- Tufan, A.C.; Satiroglu-Tufan, N.L. The chick embryo chorioallantoic membrane as a model system for the study of tumor angiogenesis, invasion and development of anti-angiogenic agents. Curr. Cancer Drug Targets 2005, 5, 249–266. [Google Scholar]
- Ossowski, L. In vivo invasion of modified chorioallantoic membrane by tumor cells: The role of cell surface-bound urokinase. J. Cell Biol 1988, 107, 2437–2445. [Google Scholar]
- Zhai, Y.; Kuick, R.; Nan, B.; Ota, I.; Weiss, S.J.; Trimble, C.L.; Fearon, E.R.; Cho, K.R. Gene expression analysis of preinvasive and invasive cervical squamous cell carcinomas identifies HOXC10 as a key mediator of invasion. Cancer Res 2007, 67, 10163–10172. [Google Scholar]
- Cimpean, A.M.; Ribatti, D.; Raica, M. The chick embryo chorioallantoic membrane as a model to study tumor metastasis. Angiogenesis 2008, 11, 311–319. [Google Scholar]
- Valdes, T.I.; Kreutzer, D.; Moussy, F. The chick chorioallantoic membrane as a novel in vivo model for the testing of biomaterials. J. Biomed. Mater. Res 2002, 62, 273–282. [Google Scholar]
- Giannopoulou, E.; Katsoris, P.; Hatziapostolou, M.; Kardamakis, D.; Kotsaki, E.; Polytarchou, C.; Parthymou, A.; Papaioannou, S.; Papadimitriou, E. X-Rays modulate extracellular matrix in vivo. Int. J. Cancer 2001, 94, 690–698. [Google Scholar]
- Subauste, M.C.; Kupriyanova, T.A.; Conn, E.M.; Ardi, V.C.; Quigley, J.P.; Deryugina, E.I. Evaluation of metastatic and angiogenic potentials of human colon carcinoma cells in chick embryo model systems. Clin. Exp. Metastasis 2009, 26, 1033–1047. [Google Scholar]
- Demir, R.; Dimmler, A.; Naschberger, E.; Demir, I.; Papadopoulos, T.; Melling, N.; Sturzl, M.; Hohenberger, W. Malignant progression of invasive tumour cells seen in hypoxia present an accumulation of beta-catenin in the nucleus at the tumour front. Exp. Mol. Pathol 2009, 87, 109–116. [Google Scholar]
- Strojnik, T.; Kavalar, R.; Barone, T.A.; Plunkett, R.J. Experimental model and immunohistochemical comparison of U87 human glioblastoma cell xenografts on the chicken chorioallantoic membrane and in rat brains. Anticancer Res 2010, 30, 4851–4860. [Google Scholar]
- Balciuniene, N.; Tamasauskas, A.; Valanciute, A.; Deltuva, V.; Vaitiekaitis, G.; Gudinaviciene, I.; Weis, J.; von Keyserlingk, D.G. Histology of human glioblastoma transplanted on chicken chorioallantoic membrane. Medicina (Kaunas) 2009, 45, 123–131. [Google Scholar]
- Hagedorn, M.; Javerzat, S.; Gilges, D.; Meyre, A.; de Lafarge, B.; Eichmann, A.; Bikfalvi, A. Accessing key steps of human tumor progression in vivo by using an avian embryo model. Proc. Natl. Acad. Sci. USA 2005, 102, 1643–1648. [Google Scholar]
- Kobayashi, T.; Koshida, K.; Endo, Y.; Imao, T.; Uchibayashi, T.; Sasaki, T.; Namiki, M. A chick embryo model for metastatic human prostate cancer. Eur. Urol 1998, 34, 154–160. [Google Scholar]
- Wittig-Blaich, S.M.; Kacprzyk, L.A.; Eismann, T.; Bewerunge-Hudler, M.; Kruse, P.; Winkler, E.; Strauss, W.S.; Hibst, R.; Steiner, R.; Schrader, M.; et al. Matrix-dependent regulation of AKT in Hepsin-overexpressing PC3 prostate cancer cells. Neoplasia 2011, 13, 579–589. [Google Scholar]
- Conn, E.M.; Botkjaer, K.A.; Kupriyanova, T.A.; Andreasen, P.A.; Deryugina, E.I.; Quigley, J.P. Comparative analysis of metastasis variants derived from human prostate carcinoma cells: Roles in intravasation of VEGF-mediated angiogenesis and uPA-mediated invasion. Am. J. Pathol 2009, 175, 1638–1652. [Google Scholar]
- Taizi, M.; Deutsch, V.R.; Leitner, A.; Ohana, A.; Goldstein, R.S. A novel and rapid in vivo system for testing therapeutics on human leukemias. Exp. Hematol 2006, 34, 1698–1708. [Google Scholar]
- Balke, M.; Neumann, A.; Kersting, C.; Agelopoulos, K.; Gebert, C.; Gosheger, G.; Buerger, H.; Hagedorn, M. Morphologic characterization of osteosarcoma growth on the chick chorioallantoic membrane. BMC Res. Notes 2010, 3. [Google Scholar] [CrossRef]
- Chang, H.L.; Pieretti-Vanmarcke, R.; Nicolaou, F.; Li, X.; Wei, X.; MacLaughlin, D.T.; Donahoe, P.K. Mullerian inhibiting substance inhibits invasion and migration of epithelial cancer cell lines. Gynecol. Oncol 2011, 120, 128–134. [Google Scholar]
- Ween, M.P.; Lokman, N.A.; Hoffmann, P.; Rodgers, R.J.; Ricciardelli, C.; Oehler, M.K. Transforming growth factor-beta-induced protein secreted by peritoneal cells increases the metastatic potential of ovarian cancer cells. Int. J. Cancer 2011, 128, 1570–1584. [Google Scholar]
- Lokman, N.A.; Ween, M.P.; Oehler, M.K.; Ricciardelli, C. The role of annexin A2 in tumorigenesis and cancer progression. Cancer Microenviron 2011, 4, 199–208. [Google Scholar]
- Deryugina, E.I.; Quigley, J.P. Chapter 2. Chick embryo chorioallantoic membrane models to quantify angiogenesis induced by inflammatory and tumor cells or purified effector molecules. Methods Enzymol 2008, 444, 21–41. [Google Scholar]
- Manenti, L.; Riccardi, E.; Marchini, S.; Naumova, E.; Floriani, I.; Garofalo, A.; Dossi, R.; Marrazzo, E.; Ribatti, D.; Scanziani, E.; et al. Circulating plasma vascular endothelial growth factor in mice bearing human ovarian carcinoma xenograft correlates with tumor progression and response to therapy. Mol. Cancer Ther 2005, 4, 715–725. [Google Scholar]
- Fang, J.; Zhou, Q.; Liu, L.Z.; Xia, C.; Hu, X.; Shi, X.; Jiang, B.H. Apigenin inhibits tumor angiogenesis through decreasing HIF-1α and VEGF expression. Carcinogenesis 2006, 28, 858–864. [Google Scholar]
- Luo, H.; Rankin, G.O.; Liu, L.; Daddysman, M.K.; Jiang, B.H.; Chen, Y.C. Kaempferol inhibits angiogenesis and VEGF expression through both HIF dependent and independent pathways in human ovarian cancer cells. Nutr. Cancer 2009, 61, 554–563. [Google Scholar]
- Liu, L.Z.; Jing, Y.; Jiang, L.L.; Jiang, X.E.; Jiang, Y.; Rojanasakul, Y.; Jiang, B.H. Acacetin inhibits VEGF expression, tumor angiogenesis and growth through AKT/HIF-1alpha pathway. Biochem. Biophys. Res. Commun 2011, 413, 299–305. [Google Scholar]
- Kisker, O.; Onizuka, S.; Banyard, J.; Komiyama, T.; Becker, C.M.; Achilles, E.G.; Barnes, C.M.; O’Reilly, M.S.; Folkman, J.; Pirie-Shepherd, S.R. Generation of multiple angiogenesis inhibitors by human pancreatic cancer. Cancer Res 2001, 61, 7298–7304. [Google Scholar]
- Petitclerc, E.; Brooks, P.C. The chick embryo metastasis model. Methods Mol. Med 2001, 58, 173–187. [Google Scholar]
- Zijlstra, A.; Mellor, R.; Panzarella, G.; Aimes, R.T.; Hooper, J.D.; Marchenko, N.D.; Quigley, J.P. A quantitative analysis of rate-limiting steps in the metastatic cascade using human-specific real-time polymerase chain reaction. Cancer Res 2002, 62, 7083–7092. [Google Scholar]
- Aguirre-Ghiso, J.A.; Ossowski, L.; Rosenbaum, S.K. Green fluorescent protein tagging of extracellular signal-regulated kinase and p38 pathways reveals novel dynamics of pathway activation during primary and metastatic growth. Cancer Res 2004, 64, 7336–7345. [Google Scholar]
- Lyu, M.A.; Choi, Y.K.; Park, B.N.; Kim, B.J.; Park, I.K.; Hyun, B.H.; Kook, Y.H. Over-expression of urokinase receptor in human epidermoid-carcinoma cell line (HEp3) increases tumorigenicity on chorio-allantoic membrane and in severe-combined-immunodeficient mice. Int. J. Cancer 1998, 77, 257–263. [Google Scholar]
- Ray, P.; Lewin, S.A.; Mihalko, L.A.; Schmidt, B.T.; Luker, K.E.; Luker, G.D. Noninvasive imaging reveals inhibition of ovarian cancer by targeting CXCL12-CXCR4. Neoplasia (New York, NY, USA) 2011, 13, 1152–1161. [Google Scholar]
- Sale, S.; Orsulic, S. Models of ovarian cancer metastasis: Murine models. Drug Discov. Today Dis. Models 2006, 3, 149–154. [Google Scholar]
- Bekes, E.M.; Deryugina, E.I.; Kupriyanova, T.A.; Zajac, E.; Botkjaer, K.A.; Andreasen, P.A.; Quigley, J.P. Activation of pro-uPA is critical for initial escape from the primary tumor and hematogenous dissemination of human carcinoma cells. Neoplasia 2011, 13, 806–821. [Google Scholar]
- Mullany, L.K.; Richards, J.A.S. Minireview: Animal models and mechanisms of ovarian cancer development. Endocrinology 2012, 153, 1585–1592. [Google Scholar]
- Ween, M.P.; Hummitzsch, K.; Rodgers, R.J.; Oehler, M.K.; Ricciardelli, C. Versican induces a pro-metastatic ovarian cancer cell behavior which can be inhibited by small hyaluronan oligosaccharides. Clin. Exp. Metastasis 2011, 28, 113–125. [Google Scholar]

| In vivo model | Advantages | Reference | Limitations | Reference |
|---|---|---|---|---|
| CAM | Short experimental assay (days) | Short observation period (days) | ||
| Inexpensive | Cannot examine cancer-immune cell interactions | |||
| Easily reproducible and high throughput | Rapid morphological changes | [4] | ||
| Closed system—allows assessment of small quantities of therapeutic agents | Limited antibodies to chicken tissues for characterization | [7] | ||
| Naturally immunodeficient | [7] | |||
| Multiple tests per individual CAM | [4] | |||
| Allows large scale screening | ||||
| Biology and physiology well known | ||||
| Availability of in vivo imaging | [3] | |||
| Allows direct visualization | [8] | |||
| Animals do not have to be restrained | ||||
| Can be used with primary human cell lines | ||||
| Mouse | Longer observation period (weeks to months) | Long experimental length (months to years) | [4] | |
| Biology and physiology well known, but also complex | [4] | Costly | ||
| Availability of in vivo imaging | [33] | Mature immune system | ||
| Defined genetic background | [34] | Reproducibility is expensive | ||
| Large number of animals required | ||||
| Animals have to be restrained | [8] | |||
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lokman, N.A.; Elder, A.S.F.; Ricciardelli, C.; Oehler, M.K. Chick Chorioallantoic Membrane (CAM) Assay as an In Vivo Model to Study the Effect of Newly Identified Molecules on Ovarian Cancer Invasion and Metastasis. Int. J. Mol. Sci. 2012, 13, 9959-9970. https://doi.org/10.3390/ijms13089959
Lokman NA, Elder ASF, Ricciardelli C, Oehler MK. Chick Chorioallantoic Membrane (CAM) Assay as an In Vivo Model to Study the Effect of Newly Identified Molecules on Ovarian Cancer Invasion and Metastasis. International Journal of Molecular Sciences. 2012; 13(8):9959-9970. https://doi.org/10.3390/ijms13089959
Chicago/Turabian StyleLokman, Noor A., Alison S. F. Elder, Carmela Ricciardelli, and Martin K. Oehler. 2012. "Chick Chorioallantoic Membrane (CAM) Assay as an In Vivo Model to Study the Effect of Newly Identified Molecules on Ovarian Cancer Invasion and Metastasis" International Journal of Molecular Sciences 13, no. 8: 9959-9970. https://doi.org/10.3390/ijms13089959
APA StyleLokman, N. A., Elder, A. S. F., Ricciardelli, C., & Oehler, M. K. (2012). Chick Chorioallantoic Membrane (CAM) Assay as an In Vivo Model to Study the Effect of Newly Identified Molecules on Ovarian Cancer Invasion and Metastasis. International Journal of Molecular Sciences, 13(8), 9959-9970. https://doi.org/10.3390/ijms13089959






