Molecular Mechanisms of Aging and Immune System Regulation in Drosophila
Abstract
:1. Introduction
2. Molecular Mechanisms of Aging in Drosophila
3. Immune Signaling Pathways in Drosophila
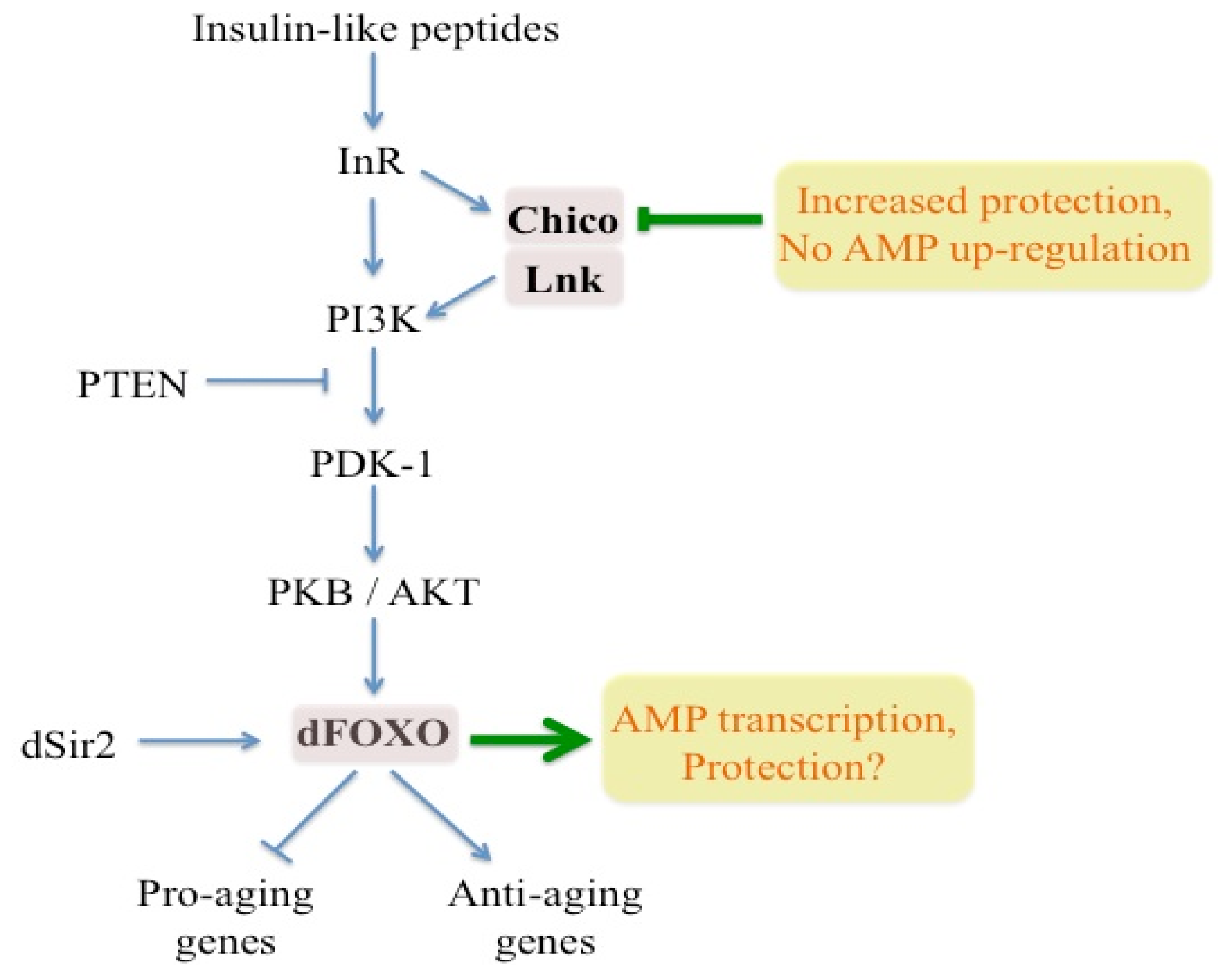
4. Relationship between Aging and Immunity in Drosophila
5. Major Challenges and Future Perspectives
6. Conclusions
Acknowledgments
References
- Kenyon, C.J. The genetics of ageing. Nature 2010, 464, 504–512. [Google Scholar]
- Paaby, A.; Schmidt, P. Dissecting the genetics of longevity in Drosophila melanogaster. Fly 2009, 3, 29–38. [Google Scholar]
- Ackermann, M.; Chao, L.; Bergstrom, C.T.; Doebeli, M. On the evolutionary origin of aging. Aging Cell 2007, 6, 235–244. [Google Scholar]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar]
- Takeuchi, O.; Akira, S. Signaling pathways activated by microorganisms. Curr. Opin. Cell Biol 2007, 19, 185–191. [Google Scholar]
- Ferrandon, D.; Imler, J.L.; Hetru, C.; Hoffmann, J. The Drosophila systemic immune response: Sensing and signalling during bacterial and fungal infections. Nat. Rev. Immunol 2007, 7, 862–874. [Google Scholar]
- DeVeale, B.; Brummel, T.; Seroude, L. Immunity and aging: The enemy within? Aging Cell 2004, 3, 195–208. [Google Scholar]
- Alper, S. Model systems to the rescue: The relationship between aging and innate immunity. Commun. Integr. Biol 2010, 3, 409–414. [Google Scholar]
- Feldhaar, H.; Gross, R. Immune reactions of insects on bacterial pathogens and mutualists. Microbes Infect 2008, 10, 1082–1088. [Google Scholar]
- Pursall, E.R.; Rolff, J. Immune responses accelerate ageing: Proof-of-principle in an insect model. PLoS One 2011, 6, e19972. [Google Scholar]
- Schneider, D.S. Using Drosophila as a model insect. Nat. Rev. Genet 2000, 1, 218–226. [Google Scholar]
- Grotewiel, M.S.; Martin, I.; Bhandari, P.; Cook-Wiens, E. Functional senescence in Drosophila melanogaster. Ageing Res. Rev 2005, 4, 372–397. [Google Scholar]
- Cho, J.; Hur, J.H.; Walker, D.W. The role of mitochondria in Drosophila aging. Exp. Gerontol 2011, 46, 331–334. [Google Scholar]
- Katewa, D.; Kapahi, P. Role of TOR signaling in aging and related biological processes in Drosophila melanogaster. Exp. Gerontol 2012, 45, 382–390. [Google Scholar]
- Inoki, K.; Guan, K.L. Complexity of the TOR signaling network. Trends Cell Biol 2006, 16, 206–212. [Google Scholar]
- Oldham, S.; Montagne, J.; Radimerski, T.; Thomas, G.; Hafen, E. Genetic and biochemical characterization of dTOR, the Drosophila homolog of the target of rapamycin. Genes Dev 2000, 14, 2689–2694. [Google Scholar]
- Zhang, H.; Stallock, J.P.; Ng, J.C.; Reinhard, C.; Neufeld, T.P. Regulation of cellular growth by the Drosophila target of rapamycin dTOR. Genes Dev 2000, 14, 2712–2724. [Google Scholar]
- Kapahi, P.; Zid, B.M.; Harper, T.; Koslover, D.; Sapin, V.; Benzer, S. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr. Biol 2004, 14, 885–890. [Google Scholar]
- Giannakou, M.E.; Goss, M.; Junger, M.A.; Hafen, E.; Leevers, S.J.; Partridge, L. Long-lived Drosophila with overexpressed dFOXO in adult fat body. Science 2004, 305, 361. [Google Scholar]
- Hwangbo, D.S.; Gershman, B.; Tu, M.P.; Palmer, M.; Tatar, M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature 2004, 429, 562–566. [Google Scholar]
- Lee, J.H.; Budanov, A.V.; Park, E.J.; Birse, R.; Kim, T.E.; Perkins, G.A.; Ocorr, K.; Ellisman, M.H.; Bodmer, R.; Bier, E.; et al. Sestrin as a feedback inhibitor of TOR that prevents age-related pathologies. Science 2010, 327, 1223–1228. [Google Scholar]
- Giannakou, M.E.; Partridge, L. Role of insulin-like signaling in Drosophila lifespan. Trends Biochem. Sci 2007, 32, 180–188. [Google Scholar]
- Partridge, L.; Alic, N.; Bjedov, I.; Piper, M.D.W. Ageing in Drosophila: The role of the insulin/Igf and TOR signaling network. Exp. Gerontol 2011, 46, 376–381. [Google Scholar]
- Gronke, S.; Clarke, D.F.; Broughton, S.; Andrews, T.D.; Partridge, L. Molecular evolution and functional characterization of Drosophila insulin-like peptides. PLoS Genet 2010, 6, e1000857. [Google Scholar]
- Tatar, M.; Kopelman, A.; Epstein, D.; Tu, M.P.; Yin, C.M.; Garofalo, R.S. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 2001, 292, 107–110. [Google Scholar]
- Clancy, D.J.; Gems, D.; Harshman, L.G.; Oldham, S.; Stocker, H.; Hafen, E.; Leevers, S.J.; Partridge, L. Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science 2001, 292, 104–106. [Google Scholar]
- Slack, C.; Werz, C.; Wieser, D.; Alic, N.; Foley, A.; Stocker, H.; Withers, D.J.; Thornton, J.M.; Hafen, E.; Partridge, L. Regulation of lifespan, metabolism, and stress responses by the Drosophila SH2B protein, Lnk. PLoS Genet 2010, 6, e1000881. [Google Scholar]
- Piper, M.D.; Selman, C.; McElwee, J.J.; Partridge, L. Separating cause from effect: How does insulin/IGF signalling control lifespan in worms, flies and mice? J. Intern. Med 2008, 263, 179–191. [Google Scholar]
- McElwee, J.J.; Schuster, E.; Blanc, E.; Piper, M.D.; Thomas, J.H.; Patel, D.S.; Selman, C.; Withers, D.J.; Thornton, J.M.; Partridge, L.; et al. Evolutionary conservation of regulated longevity assurance mechanisms. Genome Biol 2007, 8, R132. [Google Scholar]
- Min, K.J.; Yamamoto, R.; Buch, S.; Pankratz, M.; Tatar, M. Drosophila lifespan control by dietary restriction independent of insulin-like signaling. Aging Cell 2008, 7, 199–206. [Google Scholar]
- Mair, W.; Dillin, A. Aging and survival: The genetics of life span extension by dietary restriction. Annu. Rev. Biochem 2008, 77, 727–754. [Google Scholar]
- Bjedov, I.; Toivonen, J.M.; Kerr, F.; Slack, C.; Jacobson, J.; Foley, A.; Partridge, L. Mechanisms of life span extension by rapamycin in the fruit fly Drosophila melanogaster. Cell Metab 2010, 11, 35–46. [Google Scholar]
- Giannakou, M.E.; Goss, M.; Partridge, L. Role of dFOXO in lifespan extension by dietary restriction in Drosophila melanogaster: Not required, but its activity modulates the response. Aging Cell 2008, 7, 187–198. [Google Scholar]
- Flatt, T. Survival costs of reproduction in Drosophila. Exp. Gerontol 2011, 46, 369–375. [Google Scholar]
- Nishimura, M.; Ocorr, K.; Bodmer, R.; Cartry, J. Drosophila as a model to study cardiac aging. Exp. Gerontol 2011, 46, 326–330. [Google Scholar]
- Jones, M.A.; Grotewiel, M. Drosophila as a model for age-related impairment in locomotor and other behaviors. Exp. Gerontol 2011, 46, 320–325. [Google Scholar]
- Bonner, J.M.; Boulianne, G.L. Drosophila as a model to study age-related neurodegenerative disorders: Alzheimer’s disease. Exp. Gerontol 2011, 46, 335–339. [Google Scholar]
- Lin, Y.-J.; Seroude, L.; Benzer, S. Extended life-span and stress resistance in the Drosophila mutant methuselah. Science 1998, 282, 943–946. [Google Scholar]
- Cvejic, S.; Zhu, Z.; Felice, S.J.; Berman, Y.; Huang, X.-Y. The endogenous ligand Stunted of the GPCR Methuselah extends lifespan in Drosophila. Nat. Cell Biol 2004, 6, 540–546. [Google Scholar]
- Kidd, T.; Abu-Shumays, R.; Katzen, A.; Sisson, J.C.; Jiménez, G.; Pinchin, S.; Sullivan, W.; Ish-Horowicz, D. The epsilon-subunit of mitochondrial ATP synthase is required for normal spindle orientation during the Drosophila embryonic divisions. Genetics 2005, 170, 697–708. [Google Scholar]
- Rogina, B.; Reenan, R.A.; Nilsen, S.P.; Helfand, S.L. Extended life-span conferred by cotransporter gene mutation in Drosophila. Science 2000, 290, 2137–2140. [Google Scholar]
- Toivonen, J.M.; Walker, G.A.; Martinez-Diaz, P.; Bjedov, I.; Driege, Y.; Jacobs, H.T.; Gems, D.; Partridge, L. No influence of Indy on lifespan in Drosophila after correction for genetic and cytoplasmic background effect. PLOS Genet 2007, 3, 95. [Google Scholar]
- North, B.J.; Verdin, E. Sirtuins: Sir2-related NAD-dependent protein deacetylases. Genome Biol 2004, 5, 224. [Google Scholar]
- Longo, V.D.; Kennedy, B.K. Sirtuins in aging and age-related disease. Cell 2006, 126, 257–268. [Google Scholar]
- Parsons, X.H.; Garcia, S.N.; Pillus, L.; Kadonaga, J.T. Histone deacetylation by Sir2 generates a transcriptionally repressed nucleoprotein complex. Proc. Natl. Acad. Sci. USA 2003, 100, 1609–1614. [Google Scholar]
- Frankel, S.; Ziafazeli, T.; Rogina, B. dSir2 and longevity in Drosophila. Exp. Gerontol 2012, 46, 391–396. [Google Scholar]
- Rogina, B.; Helfand, S.L. Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc. Natl. Acad. Sci. USA 2004, 101, 15998–16003. [Google Scholar]
- Rogina, B.; Helfand, S.L.; Frankel, S. Longevity regulation by Drosophila Rpd3 deacetylase and caloric restriction. Science 2002, 298, 1745. [Google Scholar]
- Bunett, G.; Valentini, S.; Cabreiro, F.; Goss, M.; Somogyvari, M.; Piper, M.D.; Hoddinott, M.; Sutphin, G.L.; Leko, V.; McElwee, J.J.; et al. Absence of effects of Sir2 overxpression on lifespan in C. elegans and Drosophila. Nature 2011, 477, 482–485. [Google Scholar]
- Reinhardt, H.C.; Schumacher, B. The p53 network: Cellular and systemic DNA damage responses in aging and cancer. Trends Genet 2012, 28, 128–136. [Google Scholar]
- Rutkowski, R.; Hofmann, K.; Gartner, A. Phylogeny and function of the invertebrate p53 superfamily. Cold Spring Harb. Perspect. Biol 2010, 2, a001131. [Google Scholar]
- Levine, A.J.; Feng, Z.; Mak, T.W.; You, H.; Jin, S. Coordination and communication of between the p53 and IGF-1-AKT-TOR signal transduction pathways. Genes Dev 2006, 20, 267–275. [Google Scholar]
- Donehower, L.A.; Levine, A.J. p53, Cancer, and Longevity. In Molecular Biology of Aging; Guarente, L.P., Partridge, L., Wallace, D.C., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2008; pp. 127–152. [Google Scholar]
- Bauer, J.H.; Poon, P.C.; Glatt-Deeley, H.; Abrams, J.M.; Helfand, S.L. Neuronal expression of p53 dominant-negative proteins in adult Drosophila melanogaster extends life span. Curr. Biol 2005, 15, 2063–2068. [Google Scholar]
- Bauer, J.H.; Chang, C.; Morris, S.N.; Hozier, S.; Andersen, S.; Waitzman, J.S.; Helfand, S.L. Expression of dominant-negative Dmp53 in the adult fly brain inhibits insulin signaling. Proc. Natl. Acad. Sci. USA 2007, 104, 13355–13360. [Google Scholar]
- Weston, C.R.; Davis, R.J. The JNK signal transduction pathway. Curr. Opin. Genet. Dev 2002, 12, 14–21. [Google Scholar]
- Park, J.M.; Brady, H.; Ruocco, M.G.; Sun, H.; Williams, D.; Lee, S.J.; Kato, T., Jr; Richards, N.; Chan, K.; Mercurio, F.; et al. Targeting of TAK1 by the NF-κB protein Relish regulates the JNK-mediated immune response in Drosophila. Genes Dev. 2004, 18, 584–594. [Google Scholar]
- Davis, R.J. Signal transduction by the JNK group of MAP kinases. Cell 2000, 103, 239–252. [Google Scholar]
- Biteau, B.; Karpac, J.; Hwangbo, D.S.; Jasper, H. Regulation of Drosophila lifespan by JNK signaling. Exp. Gerontol 2012, 46, 349–354. [Google Scholar]
- Wang, M.C.; Bohmann, D.; Jasper, H. JNK signaling confers tolerance to oxidative stress and extends lifespan in Drosophila. Dev. Cell 2003, 5, 811–816. [Google Scholar]
- Wang, M.C.; Bohmann, D.; Jasper, H. JNK extends life span and limits growth by antagonizing cellular and organism-wide responses to insulin signaling. Cell 2005, 121, 115–125. [Google Scholar]
- Karpac, J.; Jasper, H. Insulin and JNK: Optimizing metabolic homeostasis and lifespan. Trends Endocrinol. Metab 2009, 20, 100–106. [Google Scholar]
- Garrido, C.; Paul, C.; Seigneuric, R.; Kampinga, H.H. The small heat shock proteins family: The long forgotten chaperones. Int. J. Biochem. Cell Biol 2012. [Google Scholar] [CrossRef]
- Tower, J. Heat shock proteins and Drosophila aging. Exp. Gerontol 2011, 46, 355–362. [Google Scholar]
- Tatar, M.; Khazaeli, A.A.; Curtsinger, J.W. Chaperoning extended life. Nature 1997, 390, 30. [Google Scholar]
- Minois, N.; Khazaeli, A.A.; Curtsinger, J.W. Locomotor activity as a function of age and life span in Drosophila melanogaster overexpressing hsp70. Exp. Gerontol 2001, 36, 1137–1153. [Google Scholar]
- Wang, H.D.; Kazemi-Esfarjani, P.; Benzer, S. Multiple-stress analysis for isolation of Drosophila longevity genes. Proc. Natl. Acad. Sci. USA 2004, 101, 12610–12615. [Google Scholar]
- Morrow, G.; Samson, M.; Michaud, S.; Tanguay, R.M. Overexpression of the small mitochondrial Hsp22 extends Drosophila life span and increases resistance to oxidative stress. FASEB J 2004, 18, 598–599. [Google Scholar]
- Cuervo, A.M. Autophagy and aging: Keeping that old broom working. Trends Genet 2008, 24, 604–612. [Google Scholar]
- Madeo, F.; Tavernarakis, N.; Kroemer, G. Can autophagy promote longevity? Nat. Cell Biol 2010, 12, 842–846. [Google Scholar]
- Juhasz, G.; Erdi, B.; Neufeld, T.P. Atg7-dependent autophagy promotes neuronal health, stress tolerance, and longevity but is dispensable for metamorphosis in Drosophila. Genes Dev 2007, 21, 3061–3066. [Google Scholar]
- Simonsen, A.; Cumming, R.C.; Brech, A.; Isakson, P.; Schubert, D.R.; Finley, K.D. Promoting basal levels of autophagy in the nervous system enhances longevity and oxidant resistance in adult Drosophila. Autophagy 2008, 4, 176–184. [Google Scholar]
- Simonsen, A.; Cumming, R.C.; Finley, K.D. Linking lysosomal trafficking defects with changes in aging and stress response in Drosophila. Autophagy 2007, 3, 499–501. [Google Scholar]
- Madeo, F.; Eisenberg, T.; Buttner, S.; Ruckenstuhl, C.; Kroemer, G. Spermidine: A novel autophagy inducer and longevity elixir. Autophagy 2010, 6, 160–162. [Google Scholar]
- Ferrandon, D.; Imler, J.L.; Hetru, C.; Hoffmann, J. The Drosophila systemic immune response: Sensing and signalling during bacterial and fungal infections. Nat. Rev. Immunol 2007, 7, 862–874. [Google Scholar]
- Dionne, M.S.; Schneider, D.S. Models of infectious diseases in the fruit fly Drosophila melanogaster. Dis. Models Mech 2008, 1, 43–49. [Google Scholar]
- Kurata, S. Fly immunity: Recognition of pathogens and induction of immune responses. Adv. Exp. Med. Biol 2010, 708, 205–217. [Google Scholar]
- Lemaitre, B.; Hoffmann, J. The host defense of Drosophila melanogaster. Annu. Rev. Immunol 2007, 25, 697–743. [Google Scholar]
- Hetru, C.; Hoffmann, J.A. NF-κB in the immune response of Drosophila. Cold Spring Harb. Perspect. Biol 2009, 1, a000232. [Google Scholar]
- Hultmark, D. Drosophila immunity: Paths and patterns. Curr. Opin. Immunol 2003, 15, 12–19. [Google Scholar]
- Marek, L.R.; Kagan, J.C. Phosphoinositide binding by the Toll adaptor dMyD88 controls antibacterial responses in Drosophila. Immunity 2012, 36, 612–622. [Google Scholar]
- Valanne, S.; Wang, J.H.; Rämet, M. The Drosophila Toll signaling pathway. J. Immunol 2011, 186, 649–656. [Google Scholar]
- Leulier, F.; Lemaitre, B. Toll-like receptors—Taking an evolutionary approach. Nat. Rev. Genet 2008, 9, 165–178. [Google Scholar]
- Rämet, M. The fruit fly Drosophila melanogaster unfolds the secrets of innate immunity. Acta Paediatr 2012, 101, 900–905. [Google Scholar]
- Hoffmann, J.A.; Reichhart, J.-M. Drosophila innate immunity: An evolutionary perspective. Nat. Immunol 2002, 3, 121–126. [Google Scholar]
- Georgel, P.; Naitza, S.; Kappler, C.; Ferrandon, D.; Zachary, D.; Swimmer, C.; Kopczynski, C.; Duyk, G.; Reichhart, J.M.; Hoffmann, J.A. Drosophila immune deficiency (IMD) is a death domain protein that activates antibacterial defense and can promote apoptosis. Dev. Cell 2001, 1, 503–514. [Google Scholar]
- Ganesan, S.; Aggarwal, K.; Paquette, N.; Silverman, N. NF-κB/Rel proteins and the humoral immune responses of Drosophila melanogaster. Curr. Top. Microbiol. Immunol 2011, 349, 25–60. [Google Scholar]
- Broderick, N.A.; Welchman, D.P.; Lemaitre, B. Recognition and response to microbial infection in Drosophila. In Insect Infection and Immunity: Evolution, Ecology, and Mechanisms; Rolff, R., Ed.; Oxford University Press: Oxford, UK, 2009; pp. 16–33. [Google Scholar]
- Uvell, H.; Engstrom, Y.A. Multilayered defense against infection: Combinatorial control of insect immune genes. Trends Genet 2007, 23, 342–349. [Google Scholar]
- Aggarwal, K.; Silverman, N. Positive and negative regulation of the Drosophila immune response. BMB Rep 2008, 41, 267–277. [Google Scholar]
- Zerofsky, M.; Harel, E.; Silverman, N.; Tatar, M. Aging of the innate immune response in Drosophila melanogaster. Aging Cell 2005, 4, 103–108. [Google Scholar]
- Landis, G.N.; Abdueva, D.; Skvortsov, D.; Yang, J.; Rabin, B.E.; Carrick, J.; Tavare, S.; Tower, J. Similar gene expression patterns characterize aging and oxidative stress in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2004, 101, 7663–7668. [Google Scholar]
- Moskalev, A.; Shaposhnikov, M. Pharmacological inhibition of NF-κB prolongs lifespan of Drosophila melanogaster. Aging 2011, 3, 391–394. [Google Scholar]
- Pletcher, S.D.; Macdonald, S.J.; Marguerie, R.; Certa, U.; Stearns, S.C.; Goldstein, D.B.; Partridge, L. Genome-wide transcript profiles in aging and calorically restricted Drosophila melanogaster. Curr. Biol 2002, 12, 712–723. [Google Scholar]
- Seroude, L.; Brummel, T.; Kapahi, P.; Benzer, S. Spatio-temporal analysis of gene expression during aging in Drosophila melanogaster. Aging Cell 2002, 1, 47–56. [Google Scholar]
- Kim, H.; Kim, J.; Lee, Y.; Yang, J.; Han, K. Transcriptional regulation of the Methuselah gene by Dorsal protein in Drosophila melanogaster. Mol. Cell 2006, 21, 261–268. [Google Scholar]
- Schneider, D.S.; Ayres, J.S. Two ways to survive infection: What resistance and tolerance can teach us about treating infectious diseases. Nat. Rev. Immunol 2008, 8, 889–895. [Google Scholar]
- Kim, Y.S.; Nam, H.J.; Chung, H.Y.; Kim, N.D.; Ryu, J.H.; Lee, W.J.; Arking, R.; Yoo, M.A. Role of xanthine dehydrogenase and aging on the innate immune response of Drosophila. Age (Omaha) 2001, 24, 187–193. [Google Scholar]
- Libert, S.; Chao, Y.; Chu, X.; Pletcher, S.D. Trade-offs between longevity and pathogen resistance in Drosophila melanogaster are mediated by NF-κB signaling. Aging Cell 2006, 5, 533–543. [Google Scholar]
- Min, K.T.; Benzer, S. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc. Natl. Acad. Sci. USA 1997, 94, 10792–10796. [Google Scholar]
- Fry, A.J.; Rand, D.M. Wolbachia interactions that determine Drosophila melanogaster survival. Evolution 2002, 56, 1976–1981. [Google Scholar]
- Siozios, S.; Sapountzis, P.; Ioannidis, P.; Bourtzis, K. Wolbachia symbiosis and insect immune response. Insect Sci 2008, 15, 89–100. [Google Scholar]
- Brummel, T.; Ching, A.; Seroude, L.; Simon, A.F.; Benzer, S. Drosophila lifespan enhancement by exogenous bacteria. Proc. Natl. Acad. Sci. USA 2004, 101, 12974–12979. [Google Scholar]
- Ren, C.; Webster, P.; Finkel, S.E.; Tower, J. Increased internal and external bacterial load during Drosophila aging without life-span trade-off. Cell Metab 2007, 6, 144–152. [Google Scholar]
- Ramsden, S.; Cheung, Y.Y.; Seroude, L. Functional analysis of the Drosophila immune response during aging. Aging Cell 2008, 7, 225–236. [Google Scholar]
- Le Bourg, E. Combined effects of suppressing live yeast and of a cold pretreatment on longevity, aging and resistance to several stresses in Drosophila melanogaster. Biogerontology 2010, 11, 245–254. [Google Scholar]
- Le Bourg, E.; Massou, I.; Gobert, V. Cold stress increases resistance to fungal infection throughout life in Drosophila melanogaster. Biogerontology 2009, 10, 613–625. [Google Scholar]
- Fauvarque, M.O.; Williams, M.J. Drosophila cellular immunity: A story of migration and adhesion. J. Cell Sci. 2011, 124, 1373–1382. [Google Scholar]
- Mackenzie, D.K.; Bussiere, L.F.; Tinsley, M.C. Senescence of the cellular immune response in Drosophila melanogaster. Exp. Gerontol 2011, 46, 853–859. [Google Scholar]
- Libert, S.; Chao, Y.; Zwiener, J.; Pletcher, S.D. Realized immune response is enhanced in long-lived puc and chico mutants but is unaffected by dietary restriction. Aging Cell 2008, 45, 810–817. [Google Scholar]
- Becker, T.; Loch, G.; Beyer, M.; Zinke, I.; Aschenbrenner, A.C.; Carrera, P.; Inhester, T.; Schultze, J.L.; Hoch, M. FOXO-dependent regulation of innate immune homeostasis. Nature 2010, 463, 369–373. [Google Scholar]
- DiAngelo, J.R.; Bland, M.L.; Bambina, S.; Cherry, S.; Birnbaum, M.J. The immune response attenuates growth and nutrient storage in Drosophila by reducing insulin signaling. Proc. Natl. Acad. Sci. USA 2009, 106, 20853–20858. [Google Scholar]
- Lesser, K.J.; Paiusi, I.C.; Leips, J. Naturally occurring genetic variation in the age-specific immune response of Drosophila melanogaster. Aging Cell 2006, 5, 293–295. [Google Scholar]
- Cho, I.; Horn, L.; Felix, T.M.; Foster, L.; Gwendlyn, G.; Starz-Gaiano, M.; Chambers, M.M.; de Luca, M.; Leips, J. Age-and diet-specific effects on variation at S6 kinase on life history, metabolic, and immune response traits in Drosophila melanogaster. DNA Cell Biol 2010, 29, 473–485. [Google Scholar]
- Felix, T.M.; Hughes, K.A.; Stone, E.A.; Drnevich, J.M.; Leips, J. Age-specific variation in immune response in Drosophila melanogaster has a genetic basis. Genetics 2012. [Google Scholar] [CrossRef]
- Ren, C.; Finkel, S.E.; Tower, J. Conditional inhibition of autophagy genes in adult Drosophila impairs immunity without compromising longevity. Exp. Gerontol 2009, 44, 228–235. [Google Scholar]
- Croker, B.; Crozat, K.; Berger, M.; Xia, Y.; Sovath, S.; Schaffer, L.; Eleftherianos, I.; Imler, J.L.; Beutler, B. ATP-sensitive potassium channels mediate survival during infection in mammals and insects. Nat. Genet 2007, 39, 1453–1460. [Google Scholar]
- Eleftherianos, I.; Won, S.; Chtarbanova, S.; Squiban, B.; Ocorr, K.; Bodmer, R.; Beutler, B.; Hoffmann, J.A.; Imler, J.A. ATP-sensitive potassium channel (KATP)-dependent regulation of cardiotropic viral infections. Proc. Natl. Acad. Sci. USA 2011, 108, 12024–12029. [Google Scholar]
- Akasaka, T.; Klinedinst, S.; Ocorr, K.; Bustamante, E.L.; Kim, S.K.; Bodmer, R. The ATP-sensitive potassium (KATP) channel-encoded dSUR gene is required for Drosophila heart function and is regulated by Tinman. Proc. Natl. Acad. Sci. USA 2006, 103, 11999–12004. [Google Scholar]
- Burger, J.M.; Promislow, D.E. Sex-specific effects of interventions that extend fly life span. Sci. Aging Knowl. Environ 2004, 2004, pe30. [Google Scholar]
- Taylor, K.; Kimbrell, D.A. Host immune response and differential survival of the sexes in Drosophila. Fly 2007, 1, 197–204. [Google Scholar]
- Shirazu-Hiza, M.M.; Schneider, D.S. Confronting physiology: How do infected flies die? Cell. Microbiol 2007, 9, 2775–2783. [Google Scholar]
- Alper, S. Model systems to the rescue: The relationship between aging and innate immunity. Commun. Integr. Biol 2010, 3, 409–414. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Eleftherianos, I.; Castillo, J.C. Molecular Mechanisms of Aging and Immune System Regulation in Drosophila. Int. J. Mol. Sci. 2012, 13, 9826-9844. https://doi.org/10.3390/ijms13089826
Eleftherianos I, Castillo JC. Molecular Mechanisms of Aging and Immune System Regulation in Drosophila. International Journal of Molecular Sciences. 2012; 13(8):9826-9844. https://doi.org/10.3390/ijms13089826
Chicago/Turabian StyleEleftherianos, Ioannis, and Julio Cesar Castillo. 2012. "Molecular Mechanisms of Aging and Immune System Regulation in Drosophila" International Journal of Molecular Sciences 13, no. 8: 9826-9844. https://doi.org/10.3390/ijms13089826
APA StyleEleftherianos, I., & Castillo, J. C. (2012). Molecular Mechanisms of Aging and Immune System Regulation in Drosophila. International Journal of Molecular Sciences, 13(8), 9826-9844. https://doi.org/10.3390/ijms13089826




