Metal-Sulfate Induced Generation of ROS in Human Brain Cells: Detection Using an Isomeric Mixture of 5- and 6-Carboxy-2′,7′-Dichlorofluorescein Diacetate (Carboxy-DCFDA) as a Cell Permeant Tracer
Abstract
:1. Introduction
2. Results
3. Discussion
4. Experimental Section
4.1. Reagents and Antibodies
4.2. Ultrapure Water and Minimization of Extraneous Contamination
4.3. Human Neuronal-Glial (HNG) Cells in Primary Culture
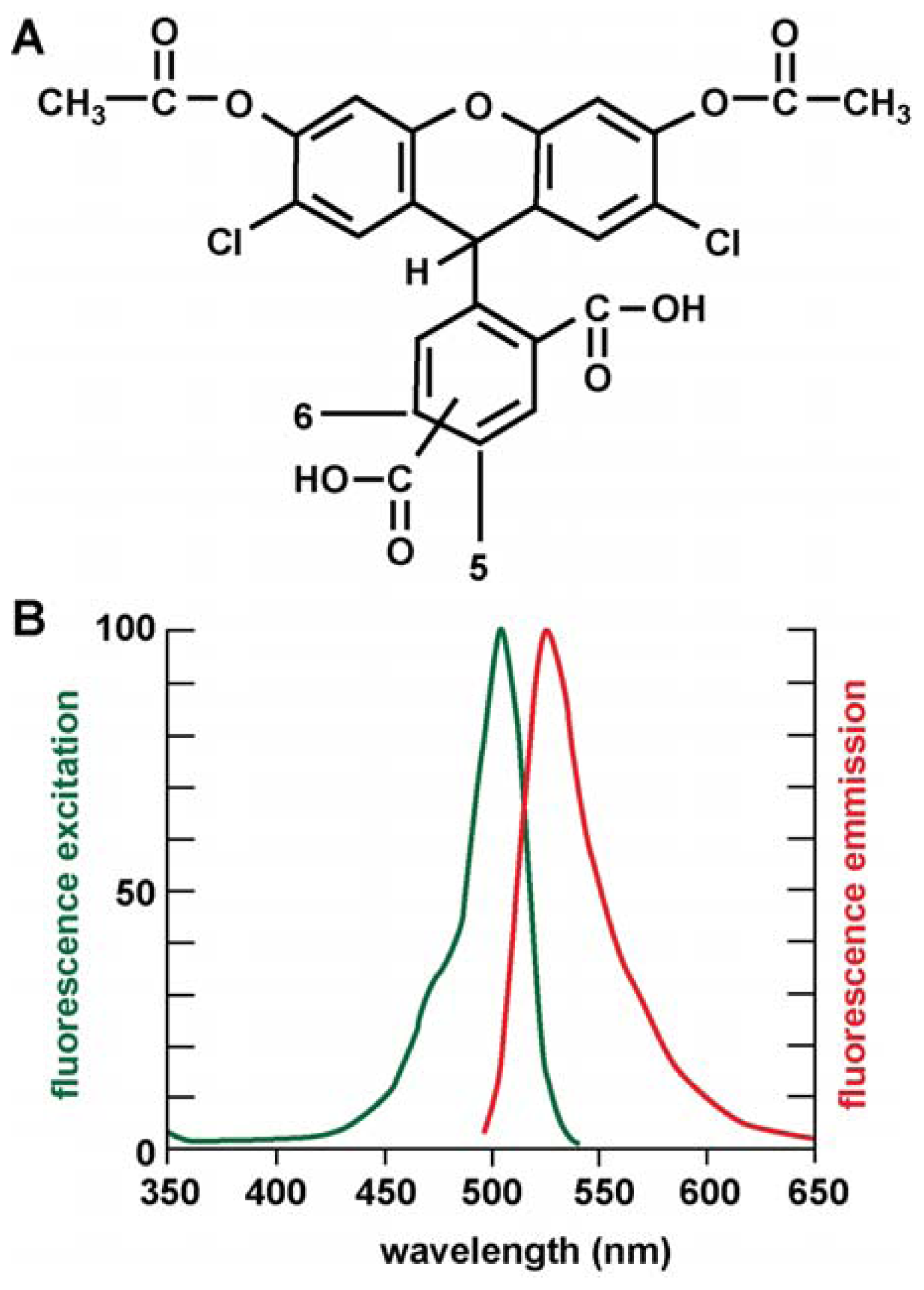
4.4. ROS Assay Using the Novel CDCFDA [5-(and-6)-darboxy-2′,7′-Dichlorofluorescein Diacetate] “Mixed Isomers”
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
References
- Harman, D. The aging process. Proc. Natl. Acad. Sci. USA 1981, 78, 7124–7128. [Google Scholar]
- Oliveira, B.F.; Nogueira-Machado, J.A.; Chaves, M.M. The role of oxidative stress in the aging process. Sci. World J 2010, 10, 1121–1128. [Google Scholar]
- Butterfield, A.; Dalle-Donne, I. Redox proteomics. Antioxid. Redox Signal 2012. [Google Scholar] [CrossRef]
- Mariani, E.; Polidori, M.C.; Cherubini, A.; Mecocci, P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: An overview. J. Chromatogr. B 2005, 827, 65–75. [Google Scholar]
- Sasaki, T. Analysis of aging-related oxidative stress status in normal aging animals and development of anti-aging interventions. Yakugaku Zasshi 2010, 130, 29–42. [Google Scholar]
- Benzi, G.; Moretti, A. Are reactive oxygen species involved in Alzheimer’s disease? Neurobiol. Aging 1995, 16, 661–674. [Google Scholar]
- Massaad, C.A. Neuronal and vascular oxidative stress in Alzheimer’s disease. Curr. Neuropharmacol 2011, 9, 662–673. [Google Scholar]
- Alexandrov, P.N.; Zhao, Y.; Pogue, A.I.; Tarr, M.A.; Kruck, T.P.; Percy, M.E.; Cui, J.G.; Lukiw, W.J. Synergistic effects of iron and aluminum on stress-related gene expression in primary human neural cells. J. Alzheimers Dis 2005, 8, 117–127. [Google Scholar]
- Rana, A.; Gnaneswari, D.; Bansal, S.; Kundu, B. Prion metal interaction: Is prion pathogenesis a cause or a consequence of metal imbalance? Chem. Biol. Interact 2009, 181, 282–291. [Google Scholar]
- Haigh, C.L.; McGlade, A.R.; Lewis, V.; Masters, C.L.; Lawson, V.A.; Collins, S.J. Acute exposure to prion infection induces transient oxidative stress progressing to be cumulatively deleterious with chronic propagation in vitro. Free Radic. Biol. Med 2011, 51, 594–608. [Google Scholar]
- Kell, D.B. Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson’s, Huntington’s, Alzheimer’s, prions, bactericides, chemical toxicology and others as examples. Arch. Toxicol 2010, 84, 825–889. [Google Scholar]
- Allsop, D.; Mayes, J.; Moore, S.; Masad, A.; Tabner, B.J. Metal-dependent generation of reactive oxygen species from amyloid proteins implicated in neurodegenerative disease. Biochem. Soc. Trans 2008, 36, 1293–1298. [Google Scholar]
- Nunomura, A.; Hofer, T.; Moreira, P.I.; Castellani, R.J.; Smith, M.A.; Perry, G. RNA oxidation in Alzheimer disease and related neurodegenerative disorders. Acta Neuropathol 2009, 118, 151–166. [Google Scholar]
- Lukiw, W.J.; Bhattacharjee, S.; Zhao, Z.; Pogue, A.I.; Percy, M.E. Generation of reactive oxygen species (ROS) and pro-inflammatory signaling in human brain cells in primary culture. J. Alzheimers Dis 2012. [Google Scholar] [CrossRef]
- Lukiw, W.J.; LeBlanc, H.J.; Carver, L.A.; McLachlan, D.R.; Bazan, N.G. Run-on gene transcription in human neocortical nuclei. Inhibition by nanomolar aluminum and implications for neurodegenerative disease. J. Mol. Neurosci 1998, 11, 67–78. [Google Scholar]
- Cui, J.G.; Zhao, Y.; Lukiw, W.J. Isolation of high spectral quality RNA using run-on gene transcription; application to gene expression profiling of human brain. Cell. Mol. Neurobiol 2005, 25, 789–794. [Google Scholar]
- Shibata, N.; Kobayashi, M. The role for oxidative stress in neurodegenerative diseases. Brain Nerve 2008, 60, 157–170. [Google Scholar]
- Lukiw, W.J.; Percy, M.E.; Kruck, T.P. Nanomolar aluminum induces pro-inflammatory and pro-apoptotic gene expression in human brain cells in primary culture. J. Inorg. Biochem 2005, 99, 1895–1898. [Google Scholar]
- Pogue, A.I.; Li, Y.Y.; Cui, J.G.; Zhao, Y.; Kruck, T.P.; Percy, M.E.; Tarr, M.A.; Lukiw, W.J. Characterization of an NF-κB-regulated, miRNA-146a-mediated down-regulation of complement factor H (CFH) in metal-sulfate-stressed human brain cells. J. Inorg. Biochem 2009, 103, 1591–1595. [Google Scholar]
- Kruck, T.P.; Cui, J.G.; Percy, M.E.; Lukiw, W.J. Molecular shuttle chelation: The use of ascorbate, desferrioxamine and Feralex-G in combination to remove nuclear bound aluminum. Cell. Mol. Neurobiol 2004, 24, 443–459. [Google Scholar]
- Lukiw, W.J.; Pappolla, M.; Pelaez, R.P.; Bazan, N.G. Alzheimer’s disease—A dysfunction in cholesterol and lipid metabolism. Cell. Mol. Neurobiol 2005, 25, 475–483. [Google Scholar]
- Zhao, Y.; Calon, F.; Julien, C.; Winkler, J.W.; Petasis, N.A.; Lukiw, W.J.; Bazan, N.G. Docosahexaenoic acid-derived neuroprotectin D1 induces neuronal survival via secretase- and PPARγ-mediated mechanisms in Alzheimer’s disease models. PLoS One 2011, 6, e15816. [Google Scholar]
- Riazanskaia, N.; Lukiw, W.J.; Grigorenko, A.; Korovaitseva, G.; Dvoryanchikov, G.; Moliaka, Y.; Nicolaou, M.; Farrer, L.; Bazan, N.G.; Rogaev, E. Regulatory region variability in the human presenilin-2 (PSEN2) gene: Potential contribution to the gene activity and risk for AD. Mol. Psychiatry 2002, 7, 891–898. [Google Scholar]
- Higaki, S.; Gebhardt, B.M.; Lukiw, W.J.; Thompson, H.W.; Hill, J.M. Effect of immunosuppression on gene expression in the HSV-1 latently infected mouse trigeminal ganglion. Invest. Ophthalmol. Vis. Sci 2002, 43, 1862–1869. [Google Scholar]
- Zhao, Y.; Cui, J.G.; Lukiw, W.J. Natural secretory products of human neural and microvessel endothelial cells: Implications in pathogenic “spreading” and Alzheimer’s disease. Mol. Neurobiol 2006, 34, 181–192. [Google Scholar]
- Hirtz, D.; Thurman, D.J.; Gwinn-Hardy, K.; Mohamed, M.; Chaudhuri, A.R.; Zalutsky, R. How common are the “common” neurologic disorders? Neurology 2007, 68, 326–337. [Google Scholar]
- Lukiw, W.J.; Krishnan, B.; Wong, L.; Kruck, T.P.A.; Bergeron, C.; McLachlan, D.R.C. Nuclear compartmentalization of aluminum in Alzheimer’s disease. Neurobiol. Aging 1992, 13, 115–121. [Google Scholar]
- Kruck, T.P.; Percy, M.E.; Lukiw, W.J. Metal sulfate-mediated induction of pathogenic genes and repression by phenyl butyl nitrone and Feralex-G. Neuroreport 2008, 19, 245–249. [Google Scholar]
- Lukiw, W.J.; Zhao, Y.; Cui, J.G. An NF-κB-sensitive miRNA-146a-mediated inflammatory circuit in Alzheimer disease and in stressed human brain cells. J. Biol. Chem 2008, 283, 31315–31322. [Google Scholar]
- Percy, M.E.; Kruck, T.P.A.; Pogue, A.I.; Lukiw, W.J. Towards the prevention of potential aluminum toxic effects and an effective treatment for Alzheimer’s disease. J. Inorg. Biochem 2011, 105, 1505–1512. [Google Scholar]
- Lukiw, W.J.; Bazan, N.G. Neuroinflammatory signaling upregulation in Alzheimer’s disease. Neurochem. Res 2000, 25, 1173–1184. [Google Scholar]
- Lukiw, W.J.; Bazan, N.G. Strong nuclear factor-κB-DNA binding parallels cyclooxygenase-2 gene transcription in aging and in sporadic Alzheimer’s disease superior temporal lobe neocortex. J. Neurosci. Res 1998, 53, 583–592. [Google Scholar]
- Flood, P.M.; Qian, L.; Peterson, L.J.; Zhang, F.; Shi, J.S.; Gao, H.M.; Hong, J.S. Transcriptional factor NF-κB as a target for therapy in Parkinson’s disease. Parkinsons Dis 2011. [Google Scholar] [CrossRef]
- Campbell, A.; Yang, E.Y.; Tsai-Turton, M.; Bondy, S.C. Pro-inflammatory effects of aluminum in human glioblastoma cells. Brain Res 2002, 933, 60–65. [Google Scholar]
- Bondy, S.C. The neurotoxicity of environmental aluminum is still an issue. Neurotoxicology 2010, 31, 575–581. [Google Scholar]
- Lukiw, W.J. Micro-RNA speciation in fetal, adult and Alzheimer’s disease hippocampus. Neuroreport 2007, 18, 297–300. [Google Scholar]
- Lukiw, W.J.; Pogue, A.I. Induction of specific micro RNA (miRNA) species by ROS-generating metal sulfates in primary human brain cells. J. Inorg. Biochem 2007, 101, 1265–1269. [Google Scholar]
- Pogue, A.I.; Cui, J.G.; Li, Y.Y.; Zhao, Y.; Culicchia, F.; Lukiw, W.J. Micro RNA-125b (miRNA-125b) function in astrogliosis and glial cell proliferation. Neurosci. Lett 2010, 476, 18–22. [Google Scholar]
- Pogue, A.I.; Percy, M.E.; Cui, J.G.; Li, Y.Y.; Bhattacharjee, S.; Hill, J.M.; Kruck, T.P.; Zhao, Y.; Lukiw, W.J. Up-regulation of NF-κB-sensitive miRNA-125b and miRNA-146a in metal sulfate-stressed human astroglial (HAG) primary cell cultures. J. Inorg. Biochem 2011, 105, 1434–1437. [Google Scholar]
- McLachlan, D.R.; Lukiw, W.J.; Kruck, T.P. New evidence for an active role of aluminum in Alzheimer’s disease. Can. J. Neurol. Sci 1989, 16, 490–497. [Google Scholar]
- Lukiw, W.J.; Bazan, N.G. Survival signalling in Alzheimer’s disease. Biochem. Soc. Trans 2006, 34, 1277–1282. [Google Scholar]
- Exley, C.; Korchazhkina, O.; Job, D.; Strekopytov, S.; Polwart, A.; Crome, P. Non-invasive therapy to reduce the body burden of aluminium in Alzheimer’s disease. J. Alzheimers Dis 2006, 10, 17–24. [Google Scholar]
- Sittig’s Handbook of Pesticides and Agricultural Chemicals; Green, S.A.; Pohanish, R.P. (Eds.) William Andrew Publishing: East Norwich, NY, USA, 2005.
- Bargar, T.A.; Fisher, S.W. Molluscicide acute and sub-chronic toxicity to the three non-target organisms Hexagenia limbata, Ceriodaphnia dubia, and Pimephelas promelas, and neutralization of that toxicity by bentonite clay. Bull. Environ. Contam. Toxicol 1997, 58, 275–282. [Google Scholar]
- Aluminum Sulfate; MSDS Number 76324; Sciencelab.com, Inc.: Houston, TX, USA, 2005. Available online: http://www.sciencelab.com/msds.php?msdsId=9922864 accessed on 27 July 2012.
- Lukiw, W.J. Evidence supporting a biological role for aluminum in brain chromatin compaction and epigenetics. J. Inorg. Biochem 2010, 104, 1010–1012. [Google Scholar]
- Lukiw, W.J.; Kruck, T.P.; McLachlan, D.R. Linker histone-DNA complexes: enhanced stability in the presence of aluminum lactate and implications for Alzheimer’s disease. FEBS Lett 1989, 253, 59–62. [Google Scholar]
- Ershov, A.V.; Lukiw, W.J.; Bazan, N.G. Selective transcription factor induction in retinal pigment epithelial cells during photoreceptor phagocytosis. J. Biol. Chem 1996, 271, 28458–28462. [Google Scholar]
- Flaten, T.P. Aluminium as a risk factor in Alzheimer’s disease, with emphasis on drinking water. Brain Res. Bull 2001, 55, 187–196. [Google Scholar]
- Frisardi, V.; Solfrizzi, V.; Capurso, C.; Kehoe, P.G.; Imbimbo, B.P.; Santamato, A.; Dellegrazie, F.; Seripa, D.; Pilotto, A.; Capurso, A.; et al. Aluminum in the diet and Alzheimer’s disease: From current epidemiology to possible disease-modifying treatment. J. Alzheimers Dis 2010, 20, 17–30. [Google Scholar]
- Guilarte, T.R. Manganese and Parkinson’s disease: A critical review and new findings. Cien Saude Colet 2011, 16, 4549–4566. [Google Scholar]
- Ramalingam, M.; Kim, S.J. Reactive oxygen/nitrogen species and their functional correlations in neurodegenerative diseases. J. Neural. Transm 2012, 119, 891–910. [Google Scholar]

| Metal sulfate | ROS intensity (raw signal at Em 530 nm) | Relative induction of ROS |
|---|---|---|
| Na | 1 | 0 |
| Mg | 1.2 | 0 |
| Hg | 18 | 1.5 |
| Sn | 26 | 2 |
| Cd | 36 | 3 |
| Cu | 36 | 3 |
| Ga | 36 | 3 |
| Pb | 42 | 3.5 |
| Ni | 42 | 3.5 |
| Zn | 48 | 4 |
| Mn | 53 | 4.5 |
| Fe | 73 | 6 |
| Al | 121 | 10 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pogue, A.I.; Jones, B.M.; Bhattacharjee, S.; Percy, M.E.; Zhao, Y.; Lukiw, W.J. Metal-Sulfate Induced Generation of ROS in Human Brain Cells: Detection Using an Isomeric Mixture of 5- and 6-Carboxy-2′,7′-Dichlorofluorescein Diacetate (Carboxy-DCFDA) as a Cell Permeant Tracer. Int. J. Mol. Sci. 2012, 13, 9615-9626. https://doi.org/10.3390/ijms13089615
Pogue AI, Jones BM, Bhattacharjee S, Percy ME, Zhao Y, Lukiw WJ. Metal-Sulfate Induced Generation of ROS in Human Brain Cells: Detection Using an Isomeric Mixture of 5- and 6-Carboxy-2′,7′-Dichlorofluorescein Diacetate (Carboxy-DCFDA) as a Cell Permeant Tracer. International Journal of Molecular Sciences. 2012; 13(8):9615-9626. https://doi.org/10.3390/ijms13089615
Chicago/Turabian StylePogue, Aileen I., Brandon M. Jones, Surjyadipta Bhattacharjee, Maire E. Percy, Yuhai Zhao, and Walter J. Lukiw. 2012. "Metal-Sulfate Induced Generation of ROS in Human Brain Cells: Detection Using an Isomeric Mixture of 5- and 6-Carboxy-2′,7′-Dichlorofluorescein Diacetate (Carboxy-DCFDA) as a Cell Permeant Tracer" International Journal of Molecular Sciences 13, no. 8: 9615-9626. https://doi.org/10.3390/ijms13089615
APA StylePogue, A. I., Jones, B. M., Bhattacharjee, S., Percy, M. E., Zhao, Y., & Lukiw, W. J. (2012). Metal-Sulfate Induced Generation of ROS in Human Brain Cells: Detection Using an Isomeric Mixture of 5- and 6-Carboxy-2′,7′-Dichlorofluorescein Diacetate (Carboxy-DCFDA) as a Cell Permeant Tracer. International Journal of Molecular Sciences, 13(8), 9615-9626. https://doi.org/10.3390/ijms13089615




