Evaluation of Antioxidant Properties and Mineral Composition of Purslane (Portulaca oleracea L.) at Different Growth Stages
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction Using Different Solvents
2.2. Growth of Purslane at Different Stages
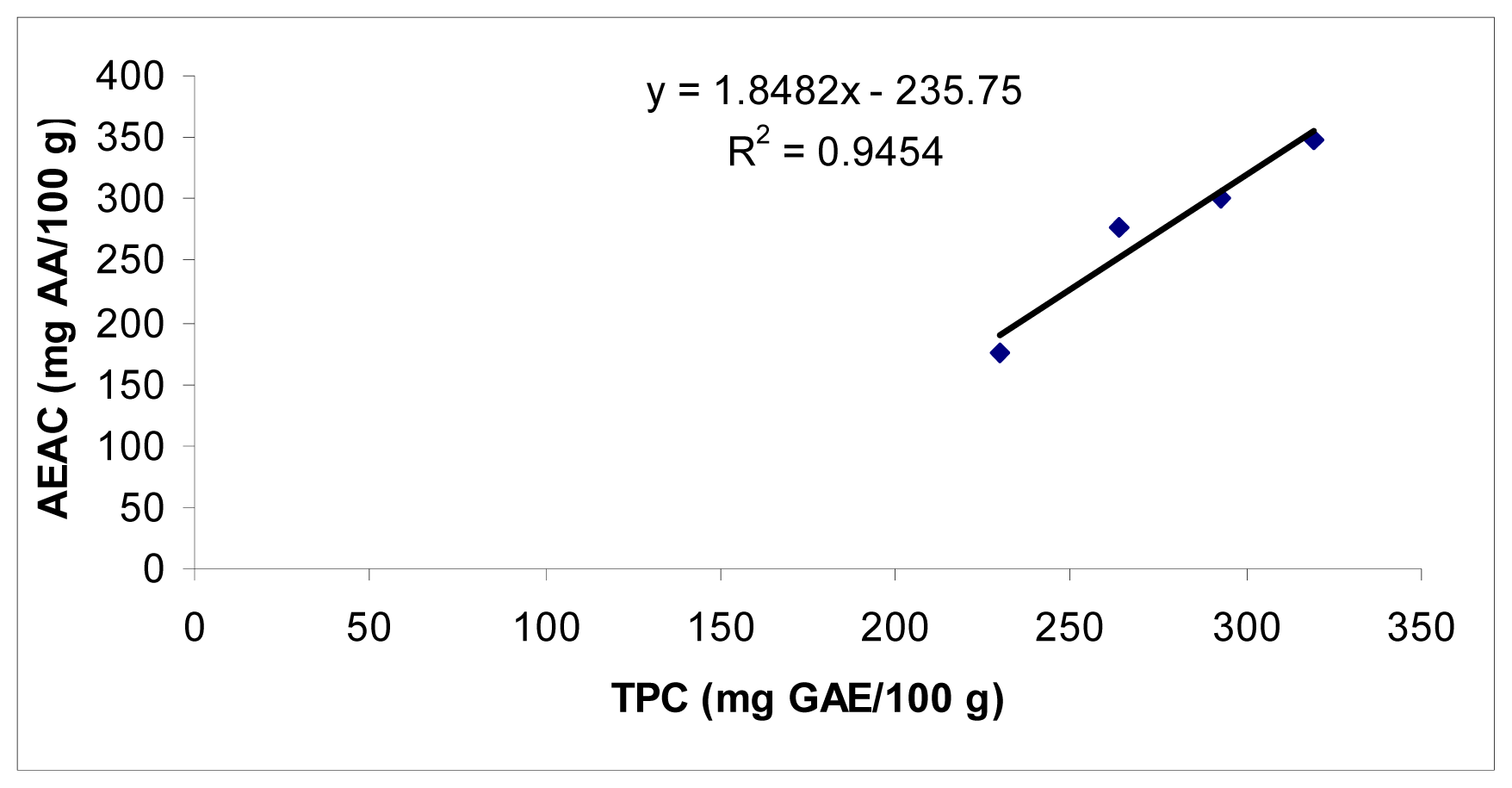
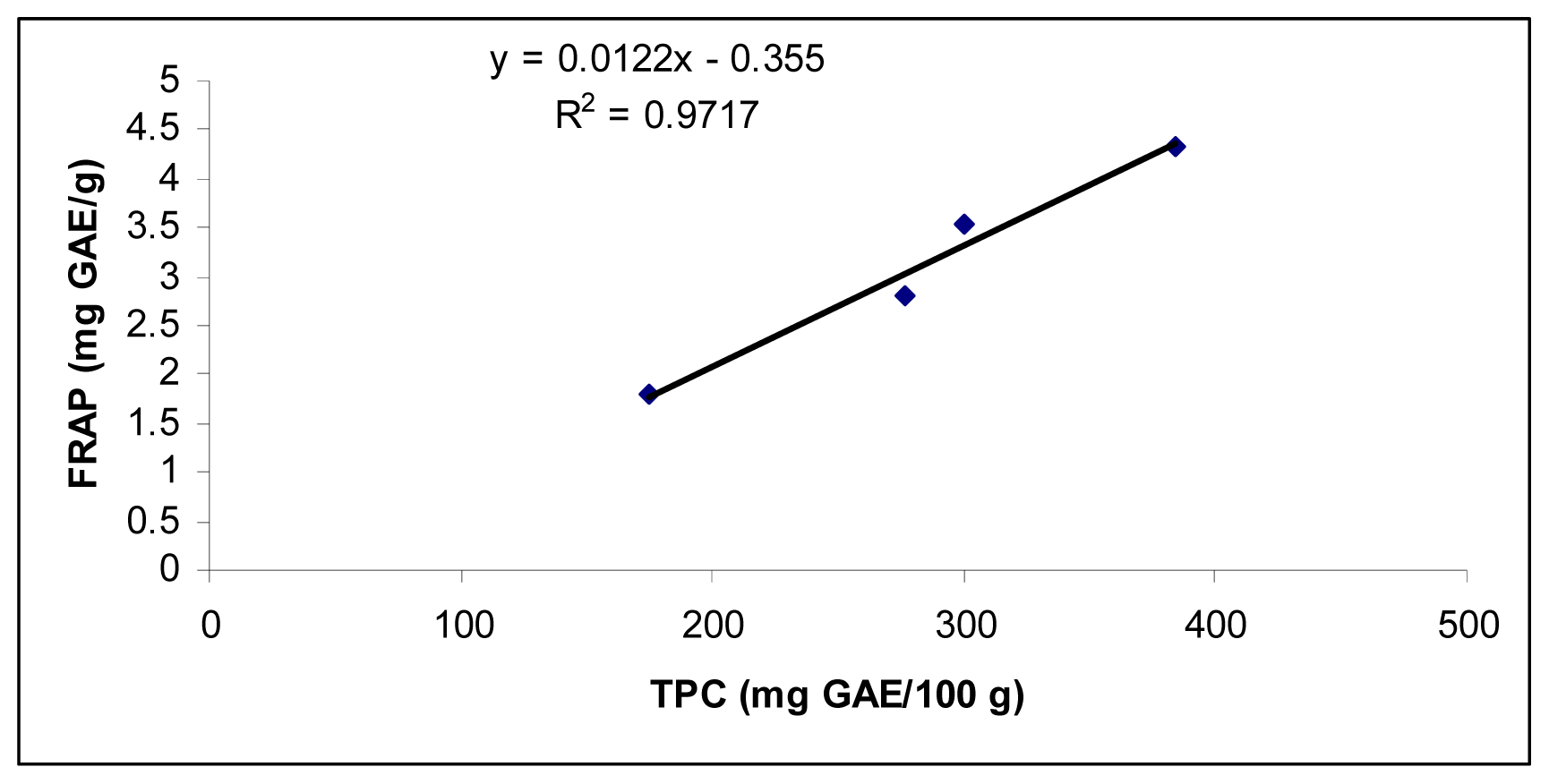
2.3. Comparative Study of Antioxidant at Different Plant Growth Stages
2.4. Mineral Composition
3. Experimental Section
3.1. Plant Materials
3.2. Preparation of Purslane Extracts
3.3. Total Phenolic Content
3.4. Total Flavonoid Content
3.5. Antioxidant Activity (DPPH Free Radical Scavenging Activity)
3.6. Ferric Reducing Antioxidant Power (FRAP)
3.7. Chemical Analysis of Leaf Samples
3.8. Statistical Analysis
4. Conclusions
Acknowledgment
References
- Samy, J.; Sugumaran, M.; Lee, K.L.W. Herbs of Malaysia: An Introduction to the Medicinal, Culinary, Aromatic and Cosmetic Use of Herbs; Wong, K.M., Ed.; Times Editions: Shah Alam, Malaysia, 2005. [Google Scholar]
- Uddin, M.K.; Juraimi, A.S.; Begum, M.; Ismail, M.R.; Rahim, A.A.; Otheman, R. Floristic composition of weed community in turfgrass area of West Peninsular Malaysia. Int. J. Agric. Biol 2009, 11, 13–20. [Google Scholar]
- Uddin, M.K.; Juraimi, A.S.; Ismail, M.R.; Brosnan, J.B. Characterizing weed populations in different turfgrass sites throughout the Klang Valley of Western Peninsular Malaysia. Weed Technol 2010, 24, 173–181. [Google Scholar]
- Chu, Y.F.; Sun, J.; Wu, X.; Liu, R.H. Antioxidant and antiproliferative activities of common vegetables. J. Agric. Food Chem 2002, 50, 6910–6916. [Google Scholar]
- Palaniswamy, U.R.; McAvoy, R.J.; Bible, B.B. Stage of harvest and polyunsaturated essential fatty acid concentrations in purslane (Portulaca oleracea) leaves. J. Agric. Food Chem. 2001, 49, 3490–3493. [Google Scholar]
- Palaniswamy, U.R.; Bible, B.B.; McAvoy, R.J. Effect of Nitrate: Ammonium Nitrogen Ratio on Oxalate Levels of Purslane. In Trends in New Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, VA, USA, 2002; pp. 453–455. [Google Scholar]
- Wenzel, G.E.; Fontana, J.D.; Correa, J.B.C. The viscous mucilage from the weed Portulaca oleracea L. Appl. Biochem. Biotech 1990, 24–25, 341–353. [Google Scholar]
- Simopoulos, A.P. Omega-3 fatty acids and antioxidants in edible wild plants. Biol. Res 2004, 37, 263–277. [Google Scholar]
- Yen, G.C.; Chen, H.Y.; Peng, H.H. Evaluation of the cytotoxicity, mutagenicity and antimutagenicity of emerging edible plants. Food Chem. Toxicol 2001, 39, 1045–1053. [Google Scholar]
- Kaur, C.; Kapoor, H.C. Antioxidants in fruits and vegetables-the millennium’s health. Int. J. Food Sci. Tech 2001, 36, 703–725. [Google Scholar]
- Vinson, J.A.; Su, X.; Zubik, L.; Bose, P. Phenol antioxidant quantity and quality in foods: Fruits. J. Agric. Food Chem 2001, 49, 5315–5321. [Google Scholar]
- Li, B.B.; Smith, B.; Hossain, M.M. Extraction of phenolics from citrus peles: I. solvent extraction method. Sep. Purif. Technol 2006, 48, 182–188. [Google Scholar]
- Yao, L.; Jiang, Y.; Datta, N.; Singanusong, R.; Liu, X.; Duan, J.; Raymont, K.; Lisle, A.; Xu, Y. HPLC analyses of flavonols and phenolic acid in the fresh young shoots of tea (Camellia sisnensis) grown in Australia. Food Chem 2004, 84, 253–263. [Google Scholar]
- Abas, F.; Lajis, N.H.; Israf, D.A.; Khozirah, S.; Kalsom, Y.U. Antioxidant and nitric oxide inhibition activities of selected activities of selected Malay traditional vegetables. Food Chem 2006, 95, 566–573. [Google Scholar]
- Sakai, N.; Inada, K.; Okamoto, M.; Shizuri, Y.; Fukuyama, Y. Portuloside A, a monoterpene glucoside from Portulaca oleracea. Phytochemistry 1996, 42, 1625–1628. [Google Scholar]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with cancer. Life Sci 2004, 74, 2157–2184. [Google Scholar]
- Oboh, G. Effect of blanching on the antioxidant properties of some tropical green leafy vegetables. LWT-Food Sci. Technol 2005, 38, 513–517. [Google Scholar]
- Witzell, J.; Gref, R.; Näsholm, T. Plant-part specific and temporal variation in phenolic compounds of boreal bilberry (Vaccinium myrtillus) plants. Biochem. Syst. Ecol 2003, 31, 115–127. [Google Scholar]
- López-Vélez, M.; Martínez-Martínez, F.; Del Velli-Ribes, C. The study of phenolic compounds as natural antioxidant in wine. Crit. Rev. Food Sci. Nutr 2003, 43, 233–244. [Google Scholar]
- Wang, S.Y.; Lin, H.-S. Antioxidant activity in fruits and leaves of blackberry, raspberry and strawberry varies with cultivar and developmental stages. J. Agric. Food Chem 2000, 48, 140–146. [Google Scholar]
- Del Rio, J.A.; Fuster, M.D.; Gómez, P.; Porras, I.; García-Lídon, A.; Ortuño, A. Citrus Limon: a source of flavinoids of pharmaceutical interest. Food Chem 2004, 84, 457–461. [Google Scholar]
- Del Bano, M.J.; Lorente, J.; Castillo, J.; Benavete-Garcia, O.; Del Rio, J.A.; Ortuno, A. Phenolic deterpenes, flavones and rosmarinic acid distribution during the development of leaves, flowers, stems, and roots of Rosmarinus officinalis: Antioxidant activity. J. Agric. Food Chem 2003, 51, 4247–4253. [Google Scholar]
- Siriamornpun, S.; Suttajit, M. Microchemical components and antioxidant activity of different morphological parts of Thai wild purslane (Portulaca oleracea). Weed Sci 2010, 58, 182–188. [Google Scholar]
- Crozier, A.; Lean, M.E.J.; McDonald, M.S.; Black, C. Quantitative analysis of the flavonoid content of commercial tomatoes, onions, lettuce and celery. J. Agric. Food Chem 1997, 45, 590–595. [Google Scholar]
- Gülcin, Í.; Şat, G.; Beydemir, Ş.; Elmastaş, M.; Küfrevioğlu, Ö.İ. Comparison of antioxidant activity of clove (Eugenia caryophylata Thunb) buds and lavender (Lavandula stoechs L.). Food Chem. 2004, 87, 393–400. [Google Scholar]
- Halicia, M.; Odabasoglua, F.; Suleymanb, H.; Cakirc, A.; Asland, A.; Bayir, Y. Effects of water extract of Usnea longissima on antioxidant enzyme activity and mucosal damage caused by indomethacin in rats. Phytomedicine 2005, 12, 656–662. [Google Scholar]
- Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Jia, Z.; Tang, M.; Wu, J. The determination of flavinoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 1999, 64, 555–559. [Google Scholar]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food Chem 1994, 43, 27–32. [Google Scholar]
- Ma, T.; Zuazaga, G. Micro-Kjeldahl determination of nitrogen. A new indicator and an improved rapid method. Ind. Eng. Chem. Anal. Ed 1942, 14, 280–282. [Google Scholar]


| Solvent | Total phenolic content 1 | Total flavonoid content 2 |
|---|---|---|
| Ethanol | 276.8 ± 5.5 | 41.30 ± 4.0 |
| Methanol | 360.3 ± 8.9 | 49.2 ± 3.4 |
| Water | 142.8 ± 8.7 | 28.7 ± 2.1 |
| Day intervals | FW (g) | DW (g) | Shoot length (cm) | RWC (%) |
|---|---|---|---|---|
| 15 | 11.97 ± 1.10 | 0.93 ± 0.05 | 4.12 ± 0.42 | 90 ± 2.69 |
| 30 | 20.52 ± 0.83 | 1.23 ± 0.037 | 8.50 ± 0.64 | 88 ± 2.53 |
| 45 | 26.30 ± 1.06 | 1.33 ± 0.023 | 14.75 ± 0.85 | 79 ± 2.41 |
| 60 | 29.15 ± 0.73 | 1.97 ± 0.053 | 20.25 ± 0.86 | 74 ± 2.71 |
| Day intervals | TPC (mg GAE/100 g) | AAC (mg/100 g) | IC50 (mg/mL) | AEAC (mg AA/100 g) | FRAP (mg GAE/g) |
|---|---|---|---|---|---|
| 15 | 174.5 ± 8.5 | 86.5 ± 3.9 | 1.71 ± 0.04 | 229.5 ± 7.9 | 1.8 ± 0.1 |
| 30 | 276.8 ± 5.5 | 84.0 ± 4.2 | 1.46 ± 0.03 | 263.8 ± 8.3 | 2.8 ± 0.1 |
| 45 | 300.5 ± 6.2 | 72.3 ± 2.7 | 1.38 ± 0.01 | 293.0 ± 8.2 | 3.6 ± 0.1 |
| 60 | 348.5 ± 7.9 | 60.5 ± 2.1 | 1.30 ± 0.04 | 319.3 ± 8.7 | 4.3 ± 0.1 |
| Minerals | 15 Day | 30 Day | 45 Day | 60 Day |
|---|---|---|---|---|
| Ca | 1612 ± 27 | 1742 ± 22 | 1892 ± 27 | 1945 ± 30 |
| Mg | 2127 ± 23 | 2196 ± 16 | 2250 ± 21 | 2443 ± 27 |
| Na | 356 ± 4 | 332 ± 7 | 306 ± 3 | 278 ± 8 |
| K | 1257 ± 10 | 1289 ± 7 | 1323 ± 11 | 1526 ± 31 |
| Fe | 218 ± 8 | 248 ± 5 | 252 ± 4 | 262 ± 3 |
| Zn | 128 ± 2 | 136 ± 1 | 142 ± 2 | 160 ± 1 |
| Cl | 82 ± 2 | 71 ± 1 | 62 ± 2 | 53 ± 2 |
| Factor | Ca | Mg | Na | K | Cl | Fe | Zn |
|---|---|---|---|---|---|---|---|
| Ca | 1 | ||||||
| Mg | 0.799 ** | 1 | |||||
| Na | −0.872 ** | −0.896 ** | 1 | ||||
| K | 0.735 ** | 0.892 ** | −0.837 ** | 1 | |||
| Cl | −0.901 ** | −0.829 ** | 0.892 ** | −0.755 ** | 1 | ||
| Fe | 0.836 ** | 0.619 ** | −0.759 ** | 0.683 ** | −0.720 ** | 1 | |
| Zn | 0.813 ** | 0.924 ** | −0.826 ** | 0.888 ** | −0.865 ** | 0.690 ** | 1 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Uddin, M.K.; Juraimi, A.S.; Ali, M.E.; Ismail, M.R. Evaluation of Antioxidant Properties and Mineral Composition of Purslane (Portulaca oleracea L.) at Different Growth Stages. Int. J. Mol. Sci. 2012, 13, 10257-10267. https://doi.org/10.3390/ijms130810257
Uddin MK, Juraimi AS, Ali ME, Ismail MR. Evaluation of Antioxidant Properties and Mineral Composition of Purslane (Portulaca oleracea L.) at Different Growth Stages. International Journal of Molecular Sciences. 2012; 13(8):10257-10267. https://doi.org/10.3390/ijms130810257
Chicago/Turabian StyleUddin, Md. Kamal, Abdul Shukor Juraimi, Md. Eaqub Ali, and Mohd Razi Ismail. 2012. "Evaluation of Antioxidant Properties and Mineral Composition of Purslane (Portulaca oleracea L.) at Different Growth Stages" International Journal of Molecular Sciences 13, no. 8: 10257-10267. https://doi.org/10.3390/ijms130810257




