Antioxidative Properties of Crude Polysaccharides from Inonotus obliquus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Major Chemical Contents
2.2. Antioxidative Effects on Chemicals-Induced ROS
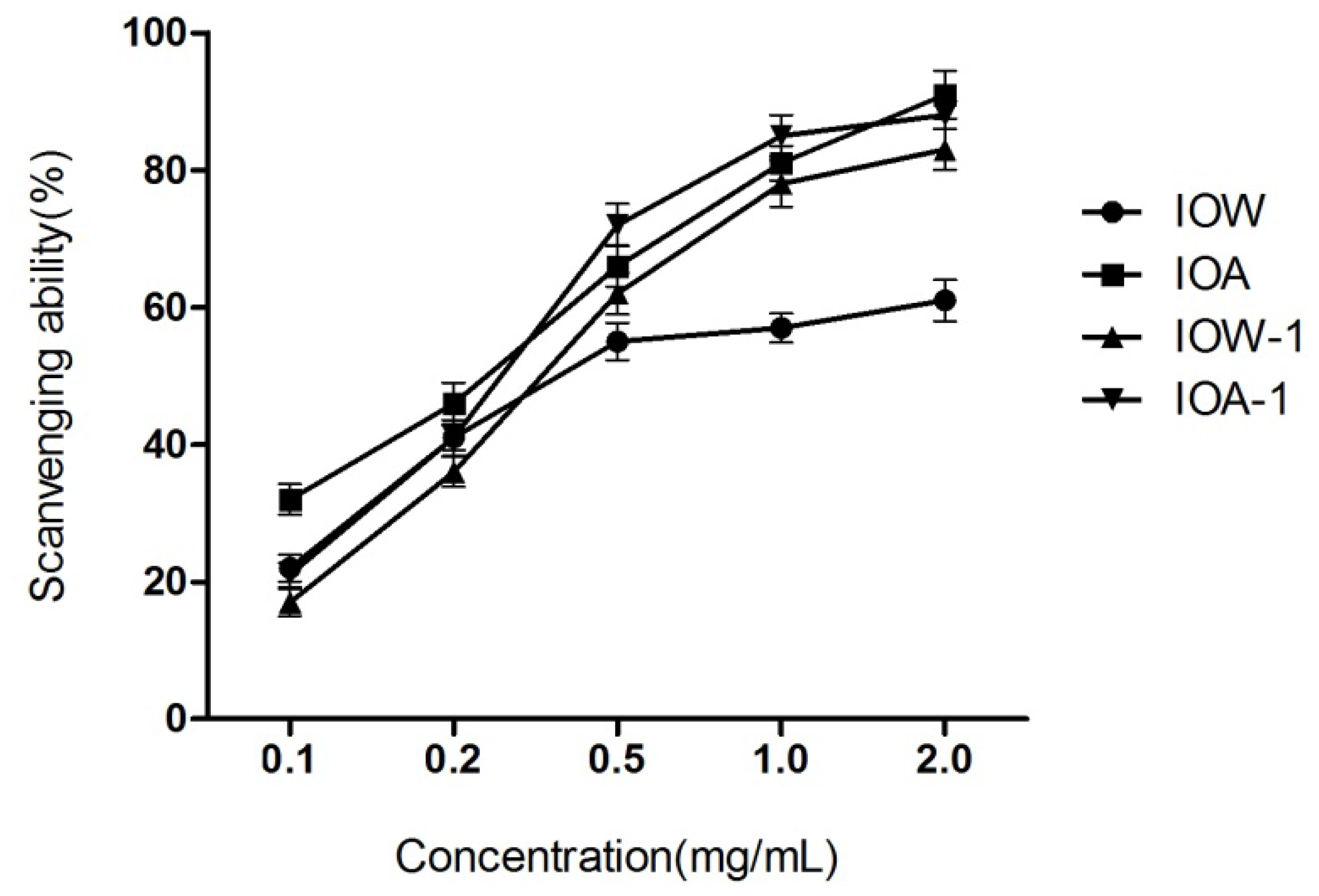
2.2.1. DPPH Radical Scavenging Capability
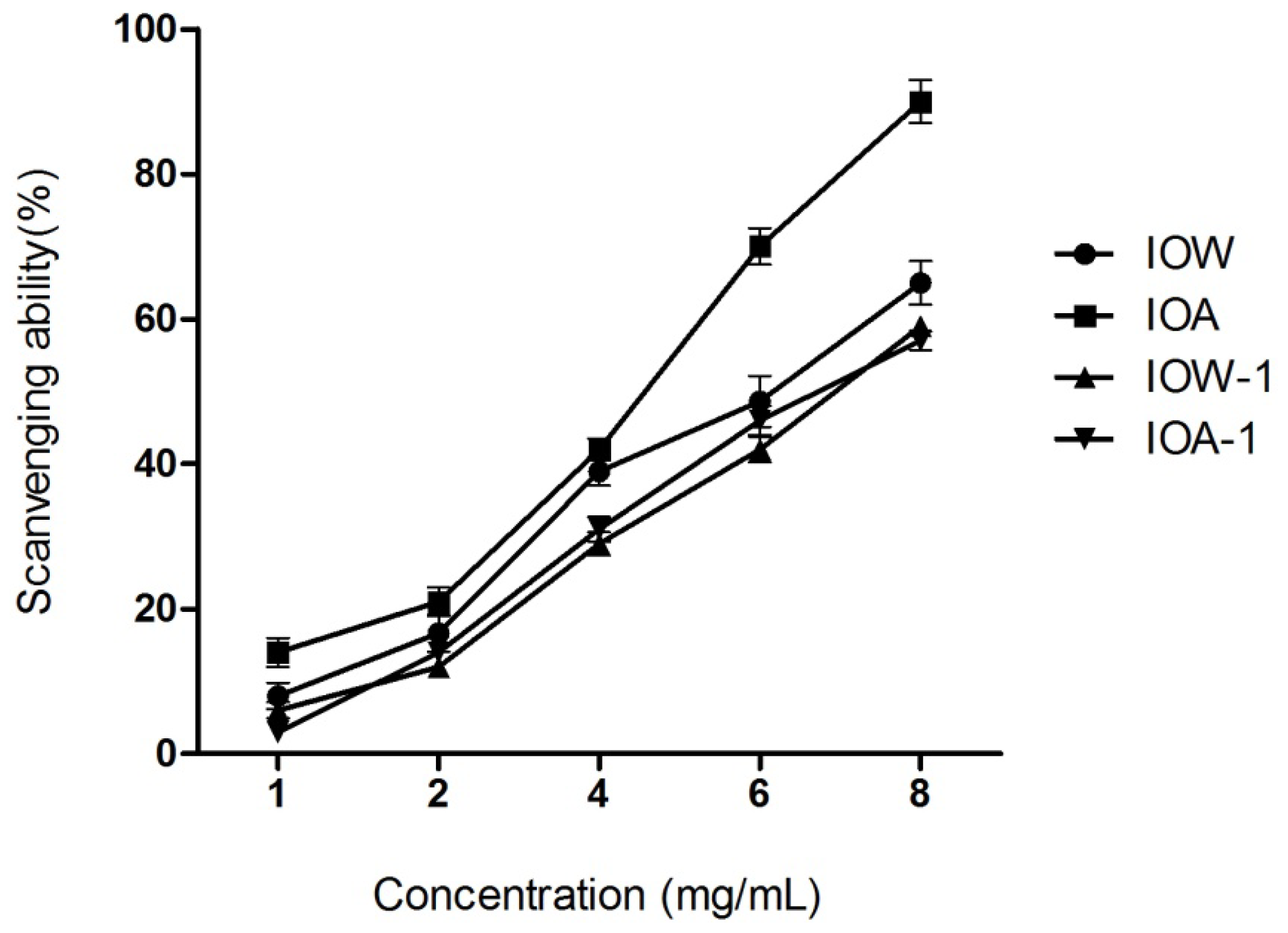
2.2.2. Hydroxyl Radical Scavenging Activity
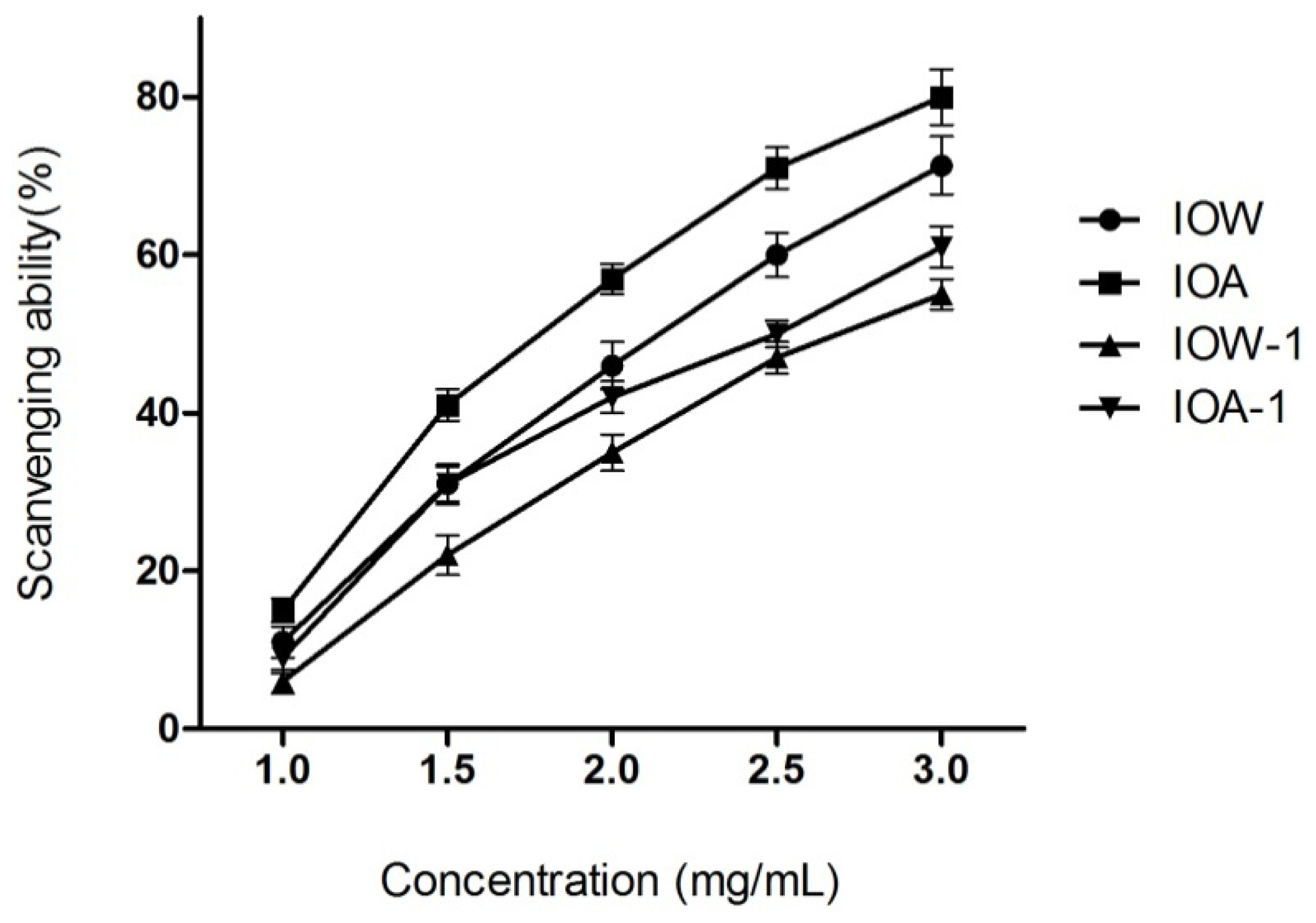
2.2.3. Superoxide Anion Scavenging Capacity
2.2.4. EC50 Values
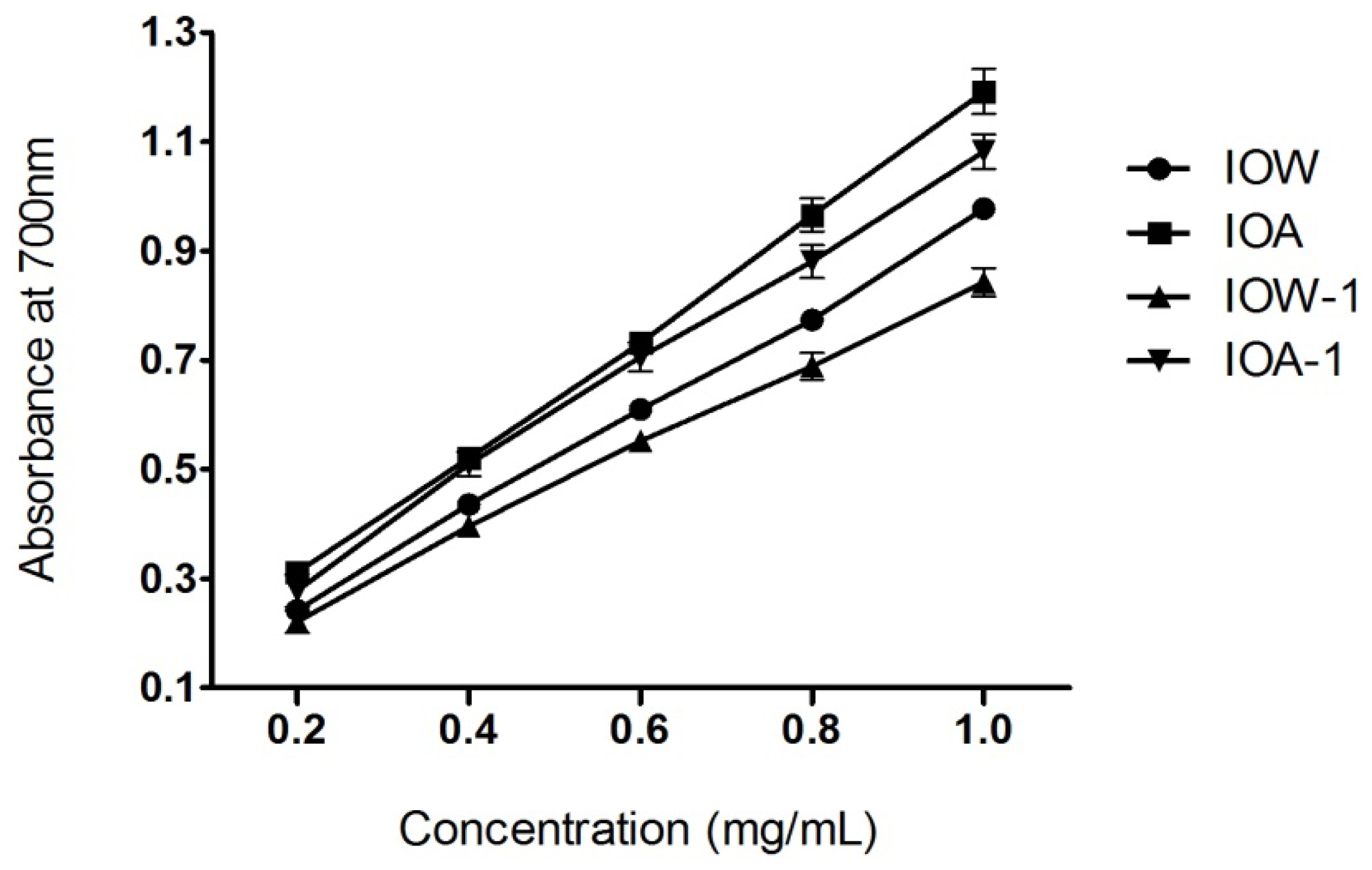
2.3. Ferric Reducing Power Assay
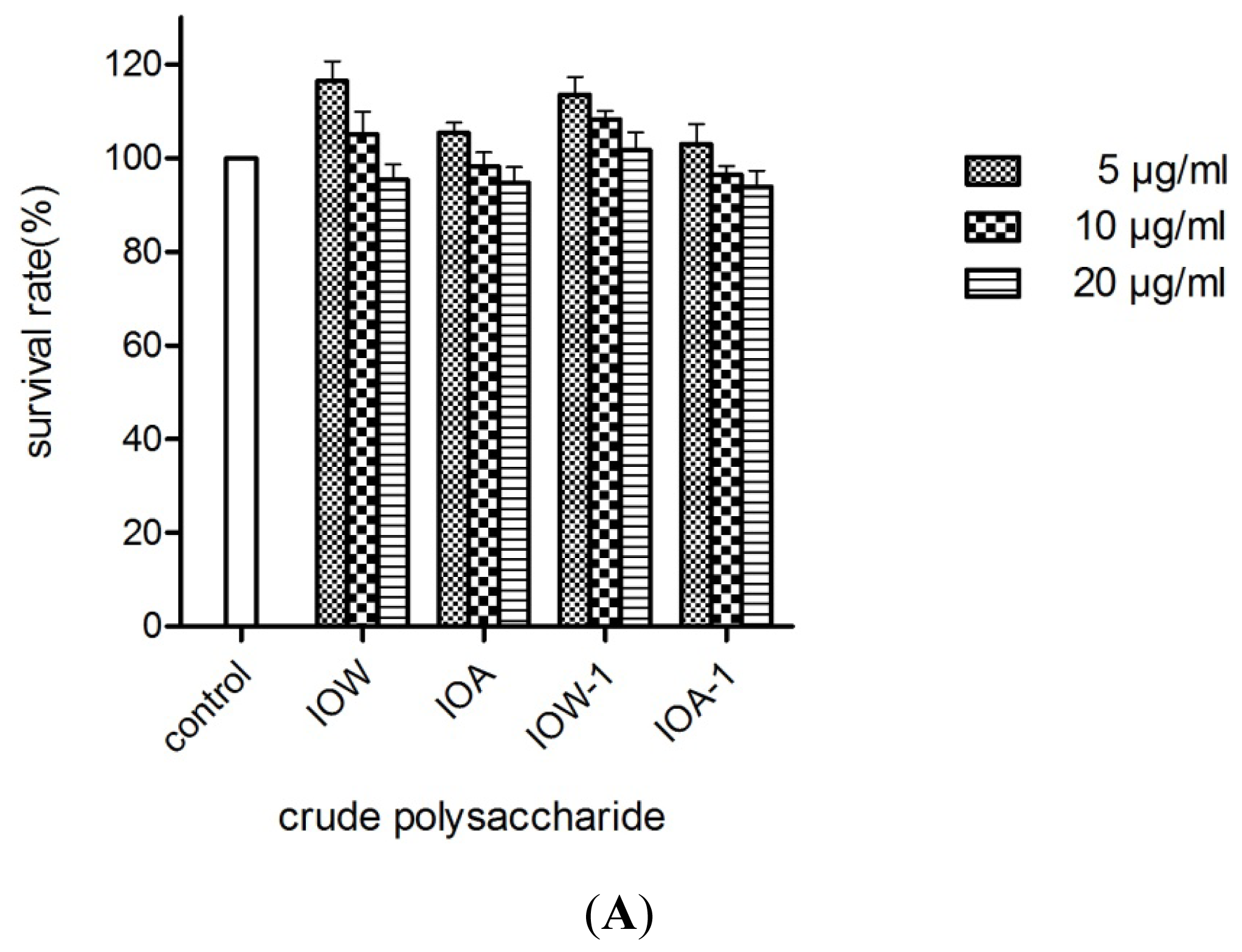
2.4. Protective Effects on H2O2-Induced PC12 Cell Death
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Preparation of Crude Polysaccharides from I. obliquus
3.3. Properties of Crude Polysaccharides
3.3.1. Determination of Total Neutral Carbohydrate Contents
3.3.2. Determination of Uronic Acid Contents
3.3.3. Determination of Protein Contents
3.4. Antioxidant Activity Assay
3.4.1. DPPH Radical Scavenging Assay
3.4.2. Hydroxyl Radical Scavenging Assay
3.4.3. Superoxide Anion Scavenging Capacity
3.4.4. Ferric Reducing Power Assay
3.5. Cell Cytotoxicity and Protective Effects on H2O2-Induced PC12 Cell Death
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Kim, Y.O.; Park, H.W.; Kim, J.H.; Lee, J.Y.; Moon, S.H.; Shin, C.S. Anti-cancer effect and structural characterization of endo-polysaccharide from cultivated mycelia of Inonotus obliquus. Life Sci 2006, 79, 72–80. [Google Scholar]
- Sun, J.E.; Ao, Z.H.; Lu, Z.M.; Xu, H.Y.; Zhang, X.M.; Dou, W.F.; Xu, Z.H. Antihyperglycemic and antilipidperoxidative effects of dry matter of culture broth of Inonotus obliquus in submerged culture on normal and alloxan-diabetes mice. J. Ethnopharmacol 2008, 118, 7–13. [Google Scholar]
- Lindequist, U.; Niedermeyer, T.H.J.; Jülich, W.-D. The pharmacological potential of mushrooms. Evid. Based Complement. Altern. Med 2005, 2, 285–299. [Google Scholar]
- Chen, Y.; Gu, X.; Huang, S.-Q.; Li, J.; Wang, X.; Tang, J. Optimization of ultrasonic/microwave assisted extraction (UMAE) of polysaccharides from Inonotus obliquus and evaluation of its anti-tumor activities. Int. J. Biol. Macromol 2010, 46, 429–435. [Google Scholar]
- Rzymowska, J. The effect of aqueous extracts from Inonotus obliquus on the mitotic index and enzyme activities. Boll. Chim. Farm 1998, 137, 13–15. [Google Scholar]
- Song, Y.; Hui, J.; Kou, W.; Xin, R.; Jia, F.; Wang, N.; Hu, F.; Zhang, H.; Liu, H. Identification of inonotus obliquus and analysis of antioxidation and antitumor activities of polysaccharides. Curr. Microbiol 2008, 57, 454–462. [Google Scholar]
- Kukulyanskaya, T.A.; Kurchenko, N.V.; Kurchenko, V.P.; Babitskaya, V.G. Physicochemical properties of melanins produced by the sterile form of inonotus obliquus(“chaga”) in natural and cultivated fungus. Appl. Biochem. Microbiol 2002, 38, 58–61. [Google Scholar]
- Wasser, S.P.; Weis, A.L. Therapeutic effects of substances occurring in higher Basidiomycetes mushrooms: A modern perspective. Crit. Rev. Immunol 1999, 19, 65–96. [Google Scholar]
- Park, E.; Jeon, K.-.I.; Byun, B.-H. Ethanol extract of Chaga shows antigenotoxic effect on hydrogen peroxide induced DNA damage in human lymphocytes. Cancer Prev. Res 2005, 10, 54–59. [Google Scholar]
- Fu, L.; Chen, H.; Dong, P.; Zhang, X.; Zhang, M. Effects of ultrasonic treatment on the physicochemical properties and DPPH radical scavenging activity of polysaccharides from mushroom Inonotus obliquus. J. Food Sci 2010, 75, C322–C327. [Google Scholar]
- Kim, Y.O.; Han, S.B.; Lee, H.W.; Ahn, H.J.; Yoon, Y.D.; Jung, J.K.; Kim, H.M.; Shin, C.S. Immuno-stimulating effect of the endo-polysaccharide produced by submerged culture of Inonotus obliquus. Life Sci 2005, 77, 2438–2456. [Google Scholar]
- Duan, J.; Kasper, D.L. Oxidative depolymerization of polysaccharides by reactive oxygen/nitrogen species. Glycobiology 2010, 21, 401–409. [Google Scholar]
- Liu, F.; Ng, T.B. Antioxidative and free radical scavenging activities of selected medicinal herbs. Life Sci 2000, 66, 725–735. [Google Scholar]
- Valentao, P.; Fernandes, E.; Carvalho, F.; Andrade, P.B.; Seabra, R.M.; Bastos, M.L. Antioxidative properties of cardoon (Cynara cardunculus L.) infusion against superoxide radical, hydroxyl radical, and hypochlorous acid. J. Agric. Food Chem 2002, 50, 4989–4993. [Google Scholar]
- Luo, D. Identification of structure and antioxidant activity of a fraction of polysaccharide purified from Dioscorea nipponica Makino. Carbohydr. Polym 2008, 71, 544–549. [Google Scholar]
- Wang, J.; Zhang, Q.; Zhang, Z.; Li, Z. Antioxidant activity of sulfated polysaccharide fractions extracted from Laminaria japonica. Int. J. Biol. Macromol 2008, 42, 127–132. [Google Scholar]
- Pasanphan, W.; Buettner, G.R.; Chirachanchai, S. Chitosan gallate as a novel potential polysaccharide antioxidant: an EPR study. Carbohydr. Res 2010, 345, 132–140. [Google Scholar]
- Hongmei, S. Evaluation of antioxidant activity of polysaccharides isolated from Camellia sinensis (tea) in exhausting training mice. J. Med. Plants Res 2011, 5, 791–795. [Google Scholar]
- Li, X.M.; Ma, Y.L.; Liu, X.J. Effect of the Lycium barbarum polysaccharides on age-related oxidative stress in aged mice. J. Ethnopharmacol 2007, 111, 504–511. [Google Scholar]
- Diplock, A.T. Will the “good fairies” please prove to us that vitamin E lessens human degenerative disease? Free Radic. Res 1997, 27, 511–532. [Google Scholar]
- Xu, X.; Wu, Y.; Chen, H. Comparative antioxidative characteristics of polysaccharide-enriched extracts from natural sclerotia and cultured mycelia in submerged fermentation of Inonotus obliquus. Food Chem 2011, 127, 74–79. [Google Scholar]
- Matthäus, B. Antioxidant activity of extracts obtained from residues of different oilseeds. J. Agric. Food Chem 2002, 50, 3444–3452. [Google Scholar]
- Reiter, R.J.; Melchiorri, D.; Sewerynek, E.; Poeggeler, B.; Barlow-Walden, L.; Chuang, J.; Ortiz, G.G.; AcuñaCastroviejo, D. A review of the evidence supporting melatonin’s role as an antioxidant. J. Pineal Res 1995, 18, 1–11. [Google Scholar]
- Duan, J.; Kasper, D.L. Regulation of T cells by gut commensal microbiota. Curr. Opin. Rheumatol 2011, 23, 372–376. [Google Scholar]
- Luo, A.; Fan, Y.; Luo, A. In vitro free radicals scavenging activities of polysaccharide from Polygonum Multiflorum Thunb. J. Med. Plants Res 2011, 5, 966–972. [Google Scholar]
- Muller, F.L.; Lustgarten, M.S.; Jang, Y.; Richardson, A.; van Remmen, H. Trends in oxidative aging theories. Free Radic. Biol. Med 2007, 43, 477–503. [Google Scholar]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem 1996, 239, 70–76. [Google Scholar]
- Nordberg, J.; Arnér, E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med 2001, 31, 1287–1312. [Google Scholar]
- Tang, X.-Q.; Feng, J.-Q.; Chen, J.; Chen, P.-X.; Zhi, J.-L.; Cui, Y.; Guo, R.-X.; Yu, H.-M. Protection of oxidative preconditioning against apoptosis induced by H2O2 in PC12 cells: Mechanisms via MMP, ROS, and Bcl-2. Brain Res 2005, 1057, 57–64. [Google Scholar]
- Shui, G.; Bao, Y.-M.; Bo, J.; An, L.-J. Protective effect of protocatechuic acid from Alpinia oxyphylla on hydrogen peroxide-induced oxidative PC12 cell death. Eur. J. Pharmacol 2006, 538, 73–79. [Google Scholar]
- Xue, H.-Y.; Gao, G.-Z.; Lin, Q.-Y.; Jin, L.-J.; Xu, Y.-P. Protective effects of aucubin on H2O2-induced apoptosis in PC12 cells. Phytother. Res 2012, 26, 369–374. [Google Scholar]
- Crispo, J.A.G.; Piché, M.; Ansell, D.R.; Eibl, J.K.; Tai, I.T.; Kumar, A.; Ross, G.M.; Tai, T.C. Protective effects of methyl gallate on H2O2-induced apoptosis in PC12 cells. Biochem. Biophys. Res. Commun 2010, 393, 773–778. [Google Scholar]
- Masuko, T.; Minami, A.; Iwasaki, N.; Majima, T.; Nishimura, S.; Lee, Y.C. Carbohydrate analysis by a phenol-sulfuric acid method in microplate format. Anal. Biochem 2005, 339, 69–72. [Google Scholar]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem 1973, 54, 484–489. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Xu, W.; Zhang, F.; Luo, Y.; Ma, L.; Kou, X.; Huang, K. Antioxidant activity of a water-soluble polysaccharide purified from Pteridium aquilinum. Carbohydr. Res 2009, 344, 217–222. [Google Scholar]
- Xiong, S.L.; Li, A.; Huang, N.; Lu, F.; Hou, D. Antioxidant and immunoregulatory activity of different polysaccharide fractions from tuber of Ophiopogon japonicus. Carbohydr. Polym 2011, 86, 1273–1280. [Google Scholar]
- Li, B.; Lu, F.; Suo, X.; Nan, H.; Li, B. Antioxidant properties of cap and stipe from coprinus comatus. Molecules 2010, 15, 1473–1486. [Google Scholar]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem 1974, 47, 469–474. [Google Scholar]
- Yen, W.-J.; Chang, L.-W.; Duh, P.-D. Antioxidant activity of peanut seed testa and its antioxidative component, ethyl protocatechuate. LWT Food Sci. Technol 2005, 38, 193–200. [Google Scholar]
- Lee, S.-M.; Yoon, M.-Y.; Park, H.-R. Protective effects of paeonia lactiflora pall on hydrogen peroxide-induced apoptosis in PC12 cells. Biosci. Biotechnol. Biochem 2008, 72, 1272–1277. [Google Scholar]


| IOA | IOA-1 | IOW | IOW-1 | |
|---|---|---|---|---|
| Carbohydrate content (wt.%) | 22.28 ± 0.31 | 50.13 ± 0.47 | 21.23 ± 0.42 | 61.21 ± 0.29 |
| Protein content (wt.%) | 10.60 ± 0.92 | 6.28 ± 0.84 | 14.07 ± 1.05 | 7.69 ± 0.81 |
| Uronic acid content (wt.%) | 3.79 ± 0.43 | 4.12 ± 0.51 | 4.60 ± 0.36 | 4.51 ± 0.20 |
| EC50 (mg/mL) | ||||
|---|---|---|---|---|
| IOW | IOA | IOW-1 | IOA-1 | |
| DPPH radicals | 0.56 ± 0.06 | 0.23 ± 0.03 | 0.37 ± 0.05 | 0.27 ± 0.04 |
| Hydroxyl radicals | 5.66 ± 0.34 | 3.44 ± 0.31 | 7.00 ± 0.22 | 6.45 ± 0.17 |
| Superoxide anion | 2.12 ± 0.12 | 1.80 ± 0.08 | 2.62 ± 0.07 | 2.39 ± 0.09 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mu, H.; Zhang, A.; Zhang, W.; Cui, G.; Wang, S.; Duan, J. Antioxidative Properties of Crude Polysaccharides from Inonotus obliquus. Int. J. Mol. Sci. 2012, 13, 9194-9206. https://doi.org/10.3390/ijms13079194
Mu H, Zhang A, Zhang W, Cui G, Wang S, Duan J. Antioxidative Properties of Crude Polysaccharides from Inonotus obliquus. International Journal of Molecular Sciences. 2012; 13(7):9194-9206. https://doi.org/10.3390/ijms13079194
Chicago/Turabian StyleMu, Haibo, Amin Zhang, Wuxia Zhang, Guoting Cui, Shunchun Wang, and Jinyou Duan. 2012. "Antioxidative Properties of Crude Polysaccharides from Inonotus obliquus" International Journal of Molecular Sciences 13, no. 7: 9194-9206. https://doi.org/10.3390/ijms13079194




