Effects of Purified Saccharomyces cerevisiae (1→3)-β-Glucan on Venous Ulcer Healing
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structural Characterization
2.2. Study Subjects
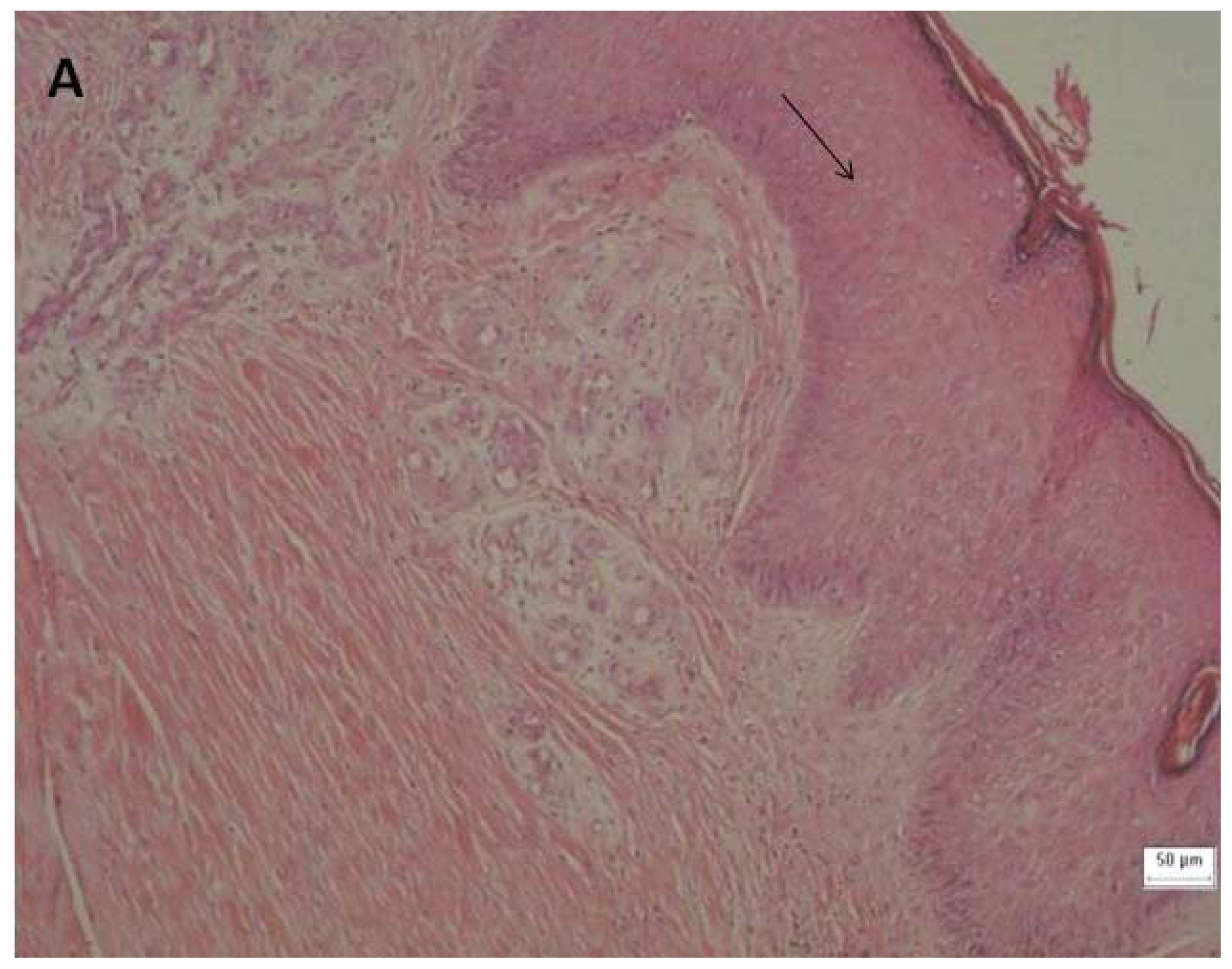
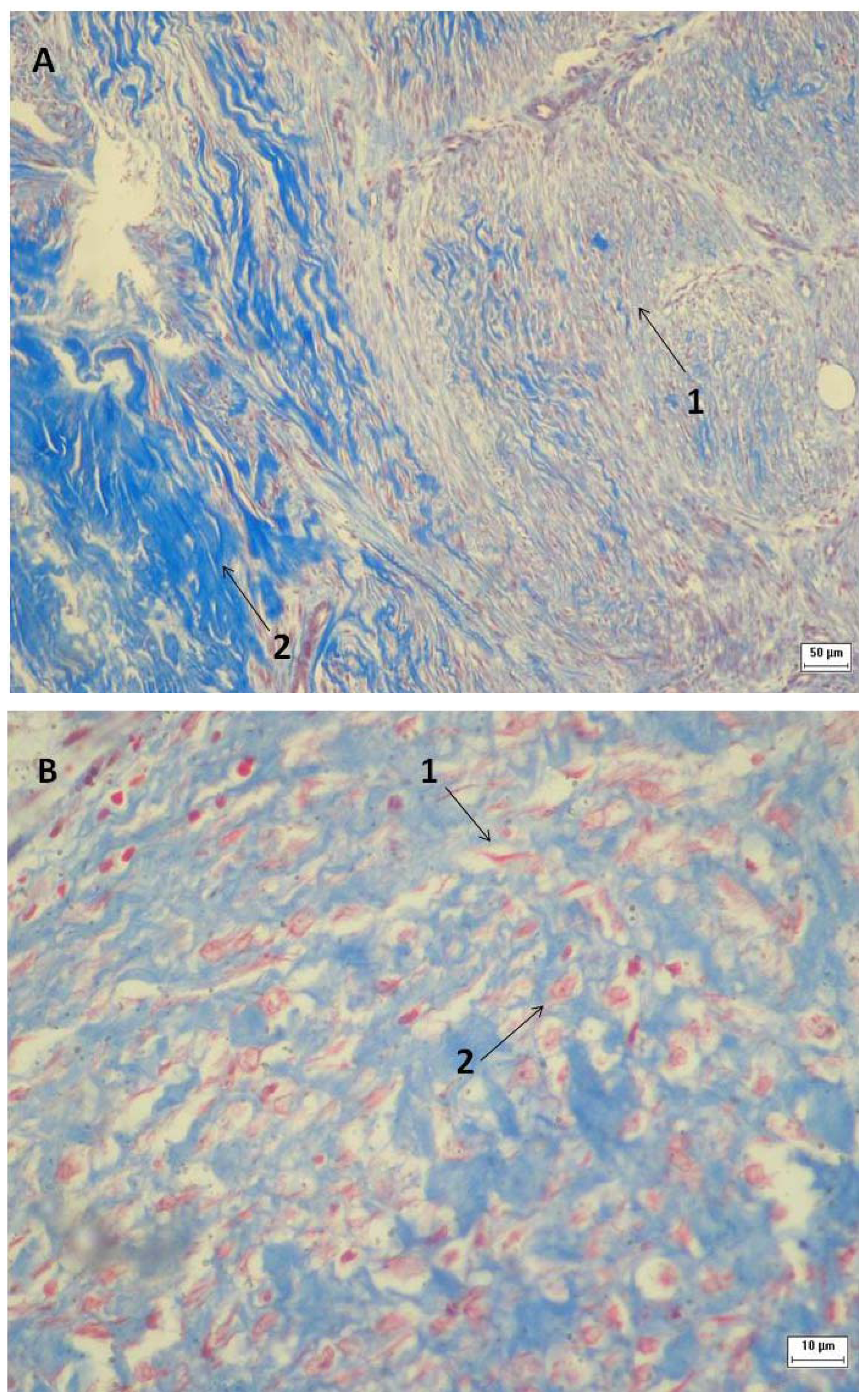
2.3. Tissue Sample Collection and Histopathological Analysis
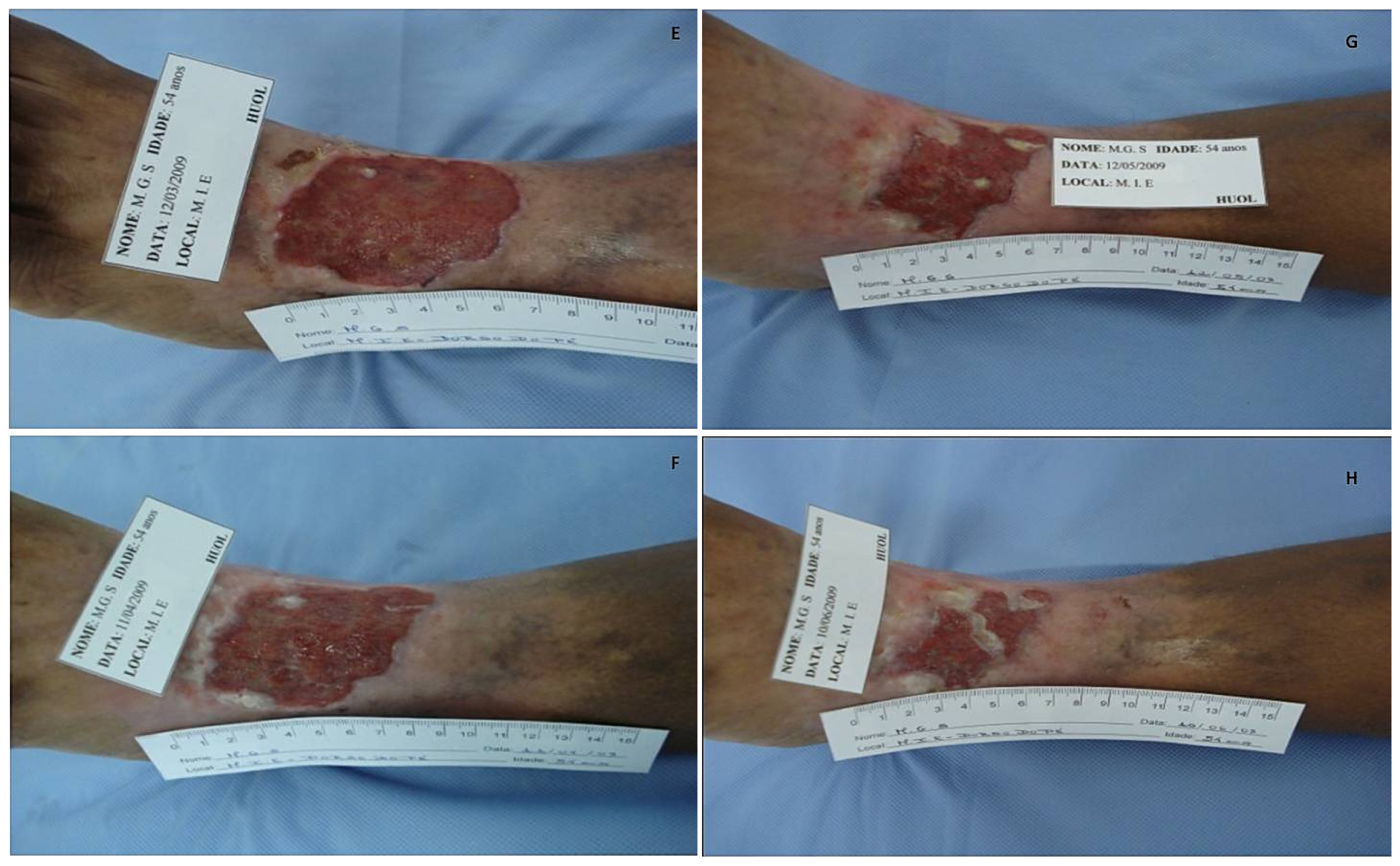
2.4. Ulcer Area Imaging and Measurement
3. Experimental Section
3.1. Materials
3.2. Isolation of Water-Insoluble (1→3)-β-Glucan
3.3. Structural Characterization
3.4. Study Subjects
3.5. (1→3)-β-Glucan Treatment
3.6. Tissue Sample Collection and Histopathological Analysis
3.7. Ulcer Imaging and Quantification of the Ulcer Area
4. Conclusions
Acknowledgments
References
- Rodero, M.P.; Khosrotehrani, K. Skin wound healing modulation by macrophages. Int. J. Clin. Exp. Pathol 2010, 3, 643–653. [Google Scholar]
- Gillitzer, R.; Goebeler, M. Chemokines in cutaneous wound healing. J. Leukocyte Biol 2001, 69, 513–521. [Google Scholar]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar]
- Park, J.E.; Barbul, A. Understanding the role of immune regulation in wound healing. Am. J. Surg 2004, 187, 11–16. [Google Scholar]
- Singer, A.J.; Clark, R.A.F. Mechanisms of disease: Cutaneous wound healing. N. Engl. J. Med 1999, 341, 738–746. [Google Scholar]
- Werner, S.; Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev 2003, 83, 835–870. [Google Scholar]
- Boyce, D.E.; Jones, W.D.; Ruge, F.; Harding, K.G.; Moore, K. The role of lymphocytes in human dermal wound healing. Br. J. Dermatol 2000, 143, 59–65. [Google Scholar]
- Iwata, Y.; Yoshizaki, A.; Komura, K.; Shimizu, K.; Ogawa, F.; Hara, T.; Muroi, E.; Bae, S.; Takenaka, M.; Yukami, T.; et al. CD19, A response regulator of b lymphocytes, regulates wound healing through hyaluronan-induced TLR4 signaling. Am. J. Pathol 2009, 175, 649–660. [Google Scholar]
- Chen, J.; Seviour, R. Medicinal importance of fungal β-(1→3),(1→6) glucans. Mycol. Res 2007, 111, 635–652. [Google Scholar]
- Manners, D.J.; Masson, A.J.; Patterson, J.C. The heterogeneity of glucan preparations from the walls of various yeasts. J. Gen. Microbiol 1974, 80, 411–417. [Google Scholar]
- Kim, Y.T.; Kim, E.H.; Cheong, C.; Williams, D.L.; Kim, C.W.; Lim, S.T. Structural characterization of β-D-(1→3,1→6)-linked Glucans using NMR spectroscopy. Carbohydr. Res 2000, 328, 331–341. [Google Scholar]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. β-Glucan Recognition by the Innate Immune System. Immunol. Rev 2009, 230, 38–50. [Google Scholar]
- Wei, D.; Williams, D.; Browder, W. Activation of AP-1 and SP1 correlates with wound growth factor gene expression in glucan-treated human fibroblasts. Int. Immunopharmacol 2002, 2, 1163–1172. [Google Scholar]
- Berdal, M.; Appelbom, H.I.; Eikrem, J.H.; Lund, A.; Zykova, S.; Busund, L.T.; Seljelid, R.; Jenssen, T. Aminated β-1,3-D-glucan improves wound healing in diabetic db/db mice. Wound. Repair. Regen 2007, 15, 825–832. [Google Scholar]
- Lee, S.B.; Jeon, H.W.; Lee, Y.W.; Lee, Y.M.; Song, K.W.; Park, M.H.; Nam, Y.S.; Ahn, H.C. Bio-artificial skin composed of gelatin and (1→3),(1→6)-β-glucan. Biomaterials 2003, 24, 2503–2511. [Google Scholar]
- Toklu, H.Z.; Sener, G.; Jahovic, N.; Uslu, B.; Arbak, S.; Yegen, B.C. β-glucan protects against burn-induced oxidative organ damage in rats. Int. Immunopharmacol 2006, 6, 156–169. [Google Scholar]
- Delatte, S.J.; Evans, J.; Hebra, A.; Adamson, W.; Othersen, H.B.; Tagge, E.P. Effectiveness of beta-glucan collagen for treatment of partial-thickness burns in children. J. Pediatr. Surg 2001, 36, 113–118. [Google Scholar]
- Simon, D.A.; Dix, F.P.; McCollum, C.N. Management of venous leg ulcers. Br. Med. J 2004, 328, 1358–1362. [Google Scholar]
- Bi, H.; Ni, Z.; Liu, X.; Iteku, J.; Tai, G.; Zhou, Y.; Zhao, J. A novel water-soluble β-(1→6)-D-glucan isolated from the fruit bodies of Bulgaria inquinans (Fries). Carbohydr. Res 2009, 344, 1254–1258. [Google Scholar]
- Ensley, H.E.; Tobias, B.; Pretus, H.A.; McNamee, R.B.; Jones, E.L.; Browder, I.W.; Williams, D.L. NMR spectral analysis of a water-insoluble (1→3)-β-D-glucan isolated from Saccharomyces cerevisiae. Carbohydr. Res 1994, 258, 307–311. [Google Scholar]
- Freimund, S.; Sauter, M.; Käppeli, O.; Dutler, H. A new non-degrading isolation process for 1,3-β-D-glucan of high purity from baker’s yeast Saccharomyces cerevisiae. Carbohydr. Polym 2003, 54, 159–171. [Google Scholar]
- Roubroeks, J.P.; ersson, R.; Aman, P. Structural features of (1→3),(1→4) β-D-glucan and arabinoxylan fractions isolated from rye bran. Carbohydr. Polym 2000, 42, 3–11. [Google Scholar]
- Tada, R.; Harada, T.; Nagi-Miura, N.; Adachi, Y.; Nakajima, M.; Yadomae, T.; Ohno, N. NMR characterization of the structure of a β-(1→3)-D-glucan isolate from cultured fruit bodies of Sparassis crispa. Carbohydr. Res 2007, 342, 2611–2618. [Google Scholar]
- Williams, D.L.; McNamee, R.B.; Jones, E.L.; Pretus, H.A.; Ensley, H.E.; Browder, I.W.; di Luzio, N.R. A method for the solubilization of a (1→3)-β-D-glucan isolated from Saccharomyces cerevisiae. Carbohydr. Res 1991, 219, 203–213. [Google Scholar]
- Shivakumar, S.; Vijayendra, S.V.N. Production of exopolysaccharides by Agrobacterium sp. CFR-24 using coconut water—a byproduct of food industry. Lett. Appl. Microbiol 2006, 42, 477–482. [Google Scholar]
- Kenyon, W.J.; Buller, C.S. Structural analysis of the curdlan-like exopolysaccharide produced by Cellulomonas flavigena KU. J. Ind. Microbiol. Biotechnol 2002, 29, 200–203. [Google Scholar]
- Wang, Y.; Zhang, M.; Ruan, D.; Shashkov, A.S.; Kilcoyne, M.; Savage, A.V.; Zhang, L. Chemical components and molecular mass of six polysaccharides isolated from the sclerotium of Poria cocos. Carbohydr. Res 2004, 339, 327–334. [Google Scholar]
- Coleridge-Smith, P.D. Leg ulcer treatment. J. Vasc. Surg 2009, 49, 804–808. [Google Scholar]
- O’Meara, S.; Cullum, N.A.; Nelson, E.A. Compression for venous leg ulcers. Cochrane Database Syst. Rev 2009, 1. [Google Scholar] [CrossRef]
- Leibovich, S.J.; Danon, D. Promotion of wound repair in mice by application of glucan. J. Reticuloendothel. Soc 1980, 27, 1–11. [Google Scholar]
- Wolk, M.; Danon, D. Promotion of wound healing by yeast glucan evaluated on single animals. Med. Biol 1985, 63, 73–80. [Google Scholar]
- Browder, W.; Williams, D.; Lucore, P.; Pretus, H.; Jones, E.; McNamee, R. Effect of enhanced macrophage function on early wound healing. Surgery 1988, 104, 224–230. [Google Scholar]
- Portera, C.A.; Love, E.J.; Memore, L.; Zhang, L.; Muller, A.; Browder, W.; Williams, D.L. Effect of macrophage stimulation on collagen biosynthesis in the healing wound. Am. Surg 1997, 63, 125–130. [Google Scholar]
- Hassid, W.Z.; Joslyn, M.A.; McCready, R.M. The molecular constitution of an insoluble polysaccharide from yeast, Saccharomyces cerevisiae. J. Am. Chem. Soc 1941, 63, 295–298. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric methods for determination of sugars and related substances. Anal. Chem 1956, 28, 350–356. [Google Scholar]
- Spector, J. Refinement of the comassie blue method of protein qualification. A simple and linear spectrofotometric assay of 0.5 to 50 μg of protein. Anal. Chem 1978, 86, 142–146. [Google Scholar]
- Melo-Silveira, R.F.; Fidelis, G.P.; Costa, M.S.S.P.; Telles, C.B.S.; Dantas-Santos, N.; Elias, S.O.; Ribeiro, V.B.; Barth, A.L.; Macedo, A.J.; Leite, E.L.; Rocha, H.A.O. In vitro antioxidant, anticoagulant and antimicrobial activity and in inhibition of cancer cell proliferation by xylan extracted from corn cobs. Int. J. Mol. Sci 2012, 13, 409–426. [Google Scholar]
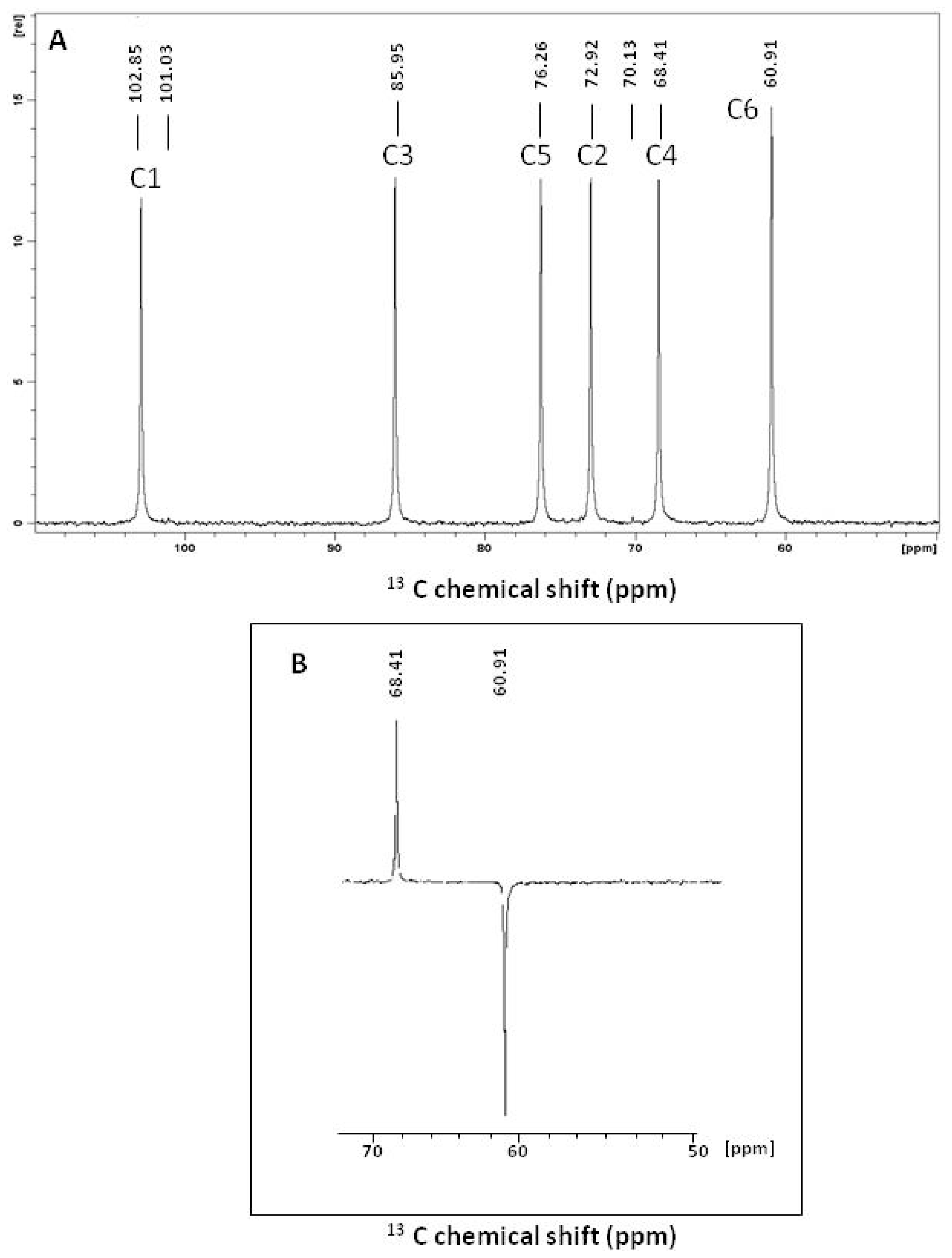




| Sugar Residue | 13C/1H (ppm) | Ref. | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |||
| 6a | 6b | |||||||
| →3)-β-Glc-(1→ | 102.85 | 72.94 | 85.95 | 68.41 | 76.26 | 60.91 | 3.49 | a |
| 4.53 | 3.32 | 3.50 | 3.27 | 3.27 | 3.71 | |||
| →3)-β-Glc-(1→ | 102.49 | 72.46 | 85.76 | 68.18 | 76.02 | 60.67 | 3.51 | b |
| 4.55 | 3.34 | 3.51 | 3.31 | 3.31 | 3.74 | |||
| →6)-β-Glc-(1→ | 104.66 | 74.71 | 76.57 | 71.14 | 77.25 | 70.48 | 3.75 | c |
| 4.42 | 3.22 | 3.40 | 3.35 | 3.53 | 4.12 | |||
| →3,6)-β-Glc-(1→ | 102.96 | 73.04 | 85.76 | 68.53 | 74.96 | 68.68 | 3.58 | d |
| 4.54 | 3.34 | 3.54 | 3.26 | 3.52 | 4.08 | |||
| →3,4)-β-Glc-(1→ | 103.02 | 73.69 | 85.34 | 68.99 | 76.48 | 61.97 | e | |
| - | - | - | - | - | - | |||
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Medeiros, S.D.V.; Cordeiro, S.L.; Cavalcanti, J.E.C.; Melchuna, K.M.; Lima, A.M.d.S.; Filho, I.A.; Medeiros, A.C.; Rocha, K.B.F.; Oliveira, E.M.; Faria, E.D.B.; et al. Effects of Purified Saccharomyces cerevisiae (1→3)-β-Glucan on Venous Ulcer Healing. Int. J. Mol. Sci. 2012, 13, 8142-8158. https://doi.org/10.3390/ijms13078142
Medeiros SDV, Cordeiro SL, Cavalcanti JEC, Melchuna KM, Lima AMdS, Filho IA, Medeiros AC, Rocha KBF, Oliveira EM, Faria EDB, et al. Effects of Purified Saccharomyces cerevisiae (1→3)-β-Glucan on Venous Ulcer Healing. International Journal of Molecular Sciences. 2012; 13(7):8142-8158. https://doi.org/10.3390/ijms13078142
Chicago/Turabian StyleMedeiros, Sarah Dantas Viana, Sara Lima Cordeiro, Jéssica Escorel Chaves Cavalcanti, Karina Mendes Melchuna, Aleida Maria da Silva Lima, Irami Araújo Filho, Aldo Cunha Medeiros, Keyla Borges Ferreira Rocha, Elizabeth Maia Oliveira, Eduardo Dantas Baptista Faria, and et al. 2012. "Effects of Purified Saccharomyces cerevisiae (1→3)-β-Glucan on Venous Ulcer Healing" International Journal of Molecular Sciences 13, no. 7: 8142-8158. https://doi.org/10.3390/ijms13078142





