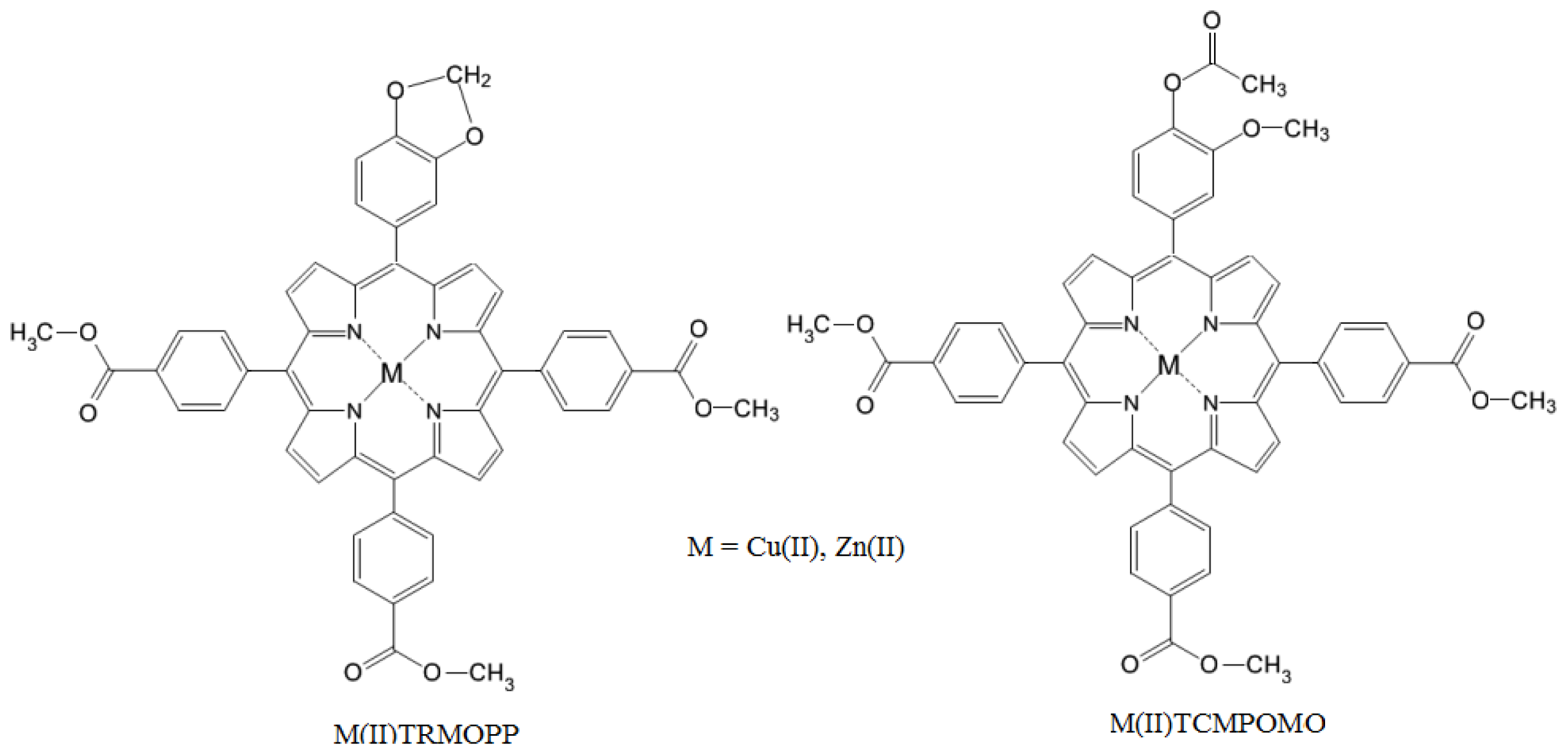
Synthesis and Spectral Evaluation of Some Unsymmetrical Mesoporphyrinic Complexes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Infrared Spectra
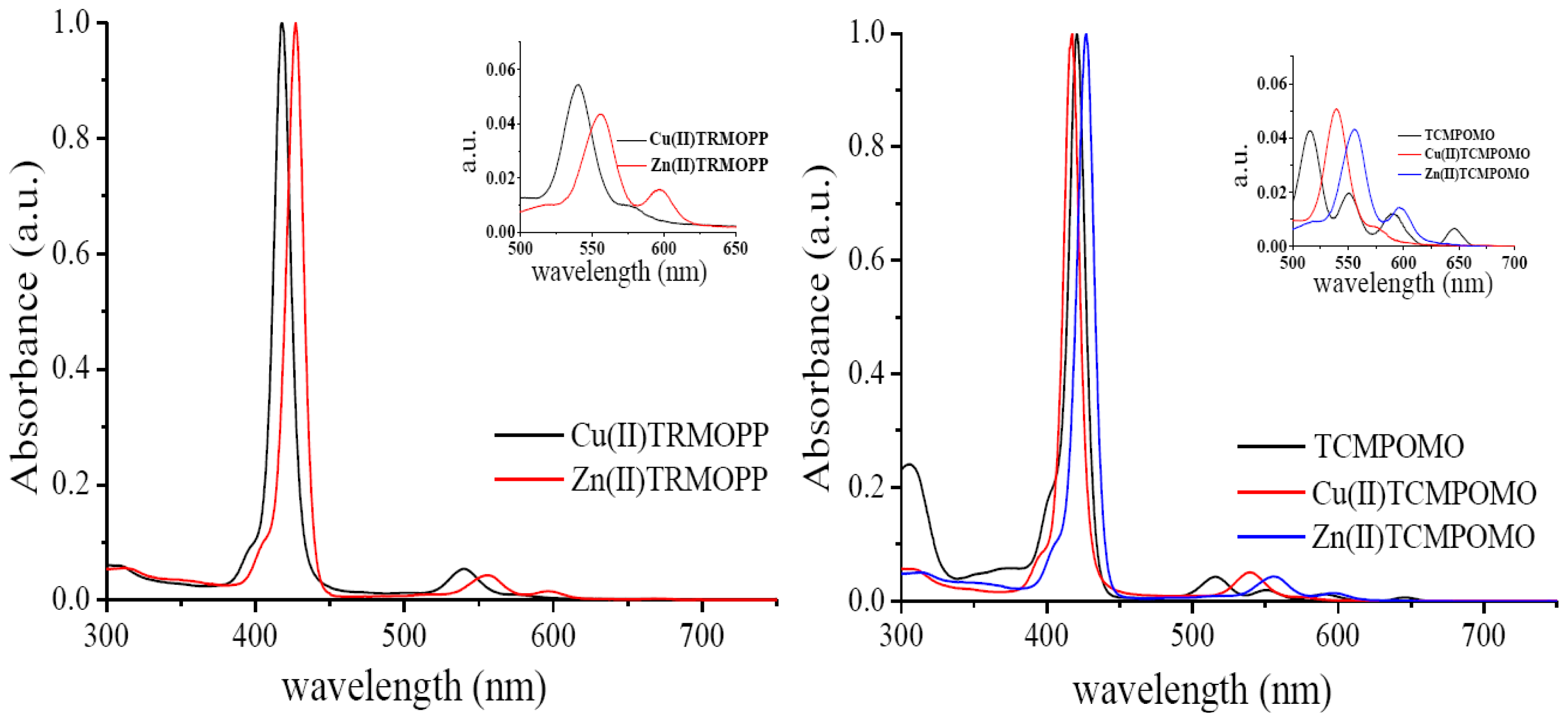
2.2. Absorption Spectra
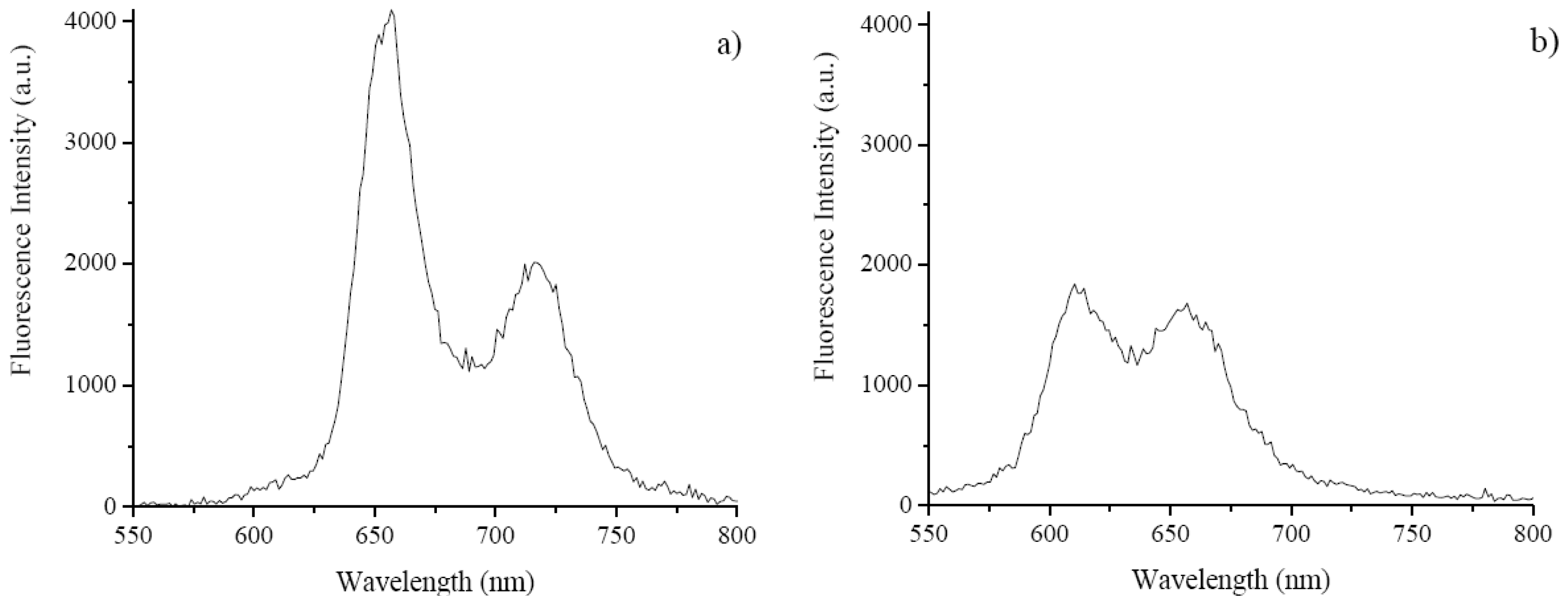
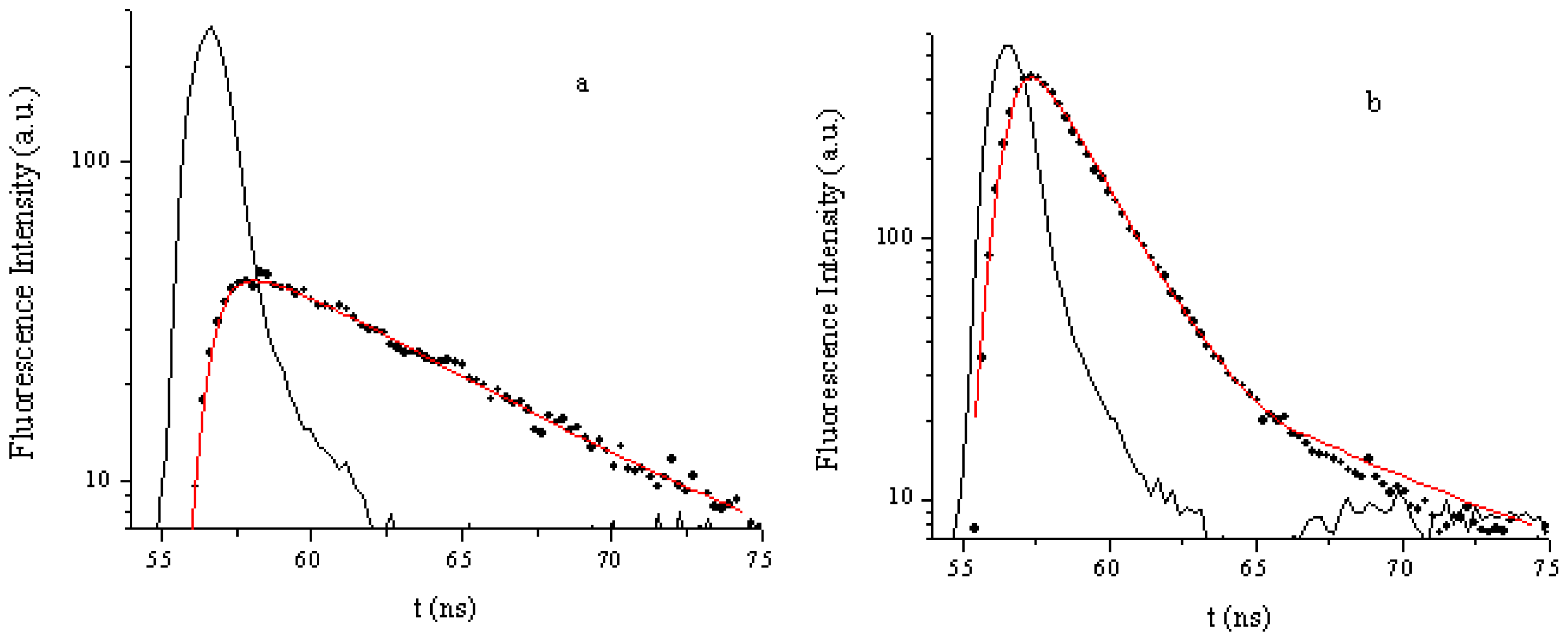
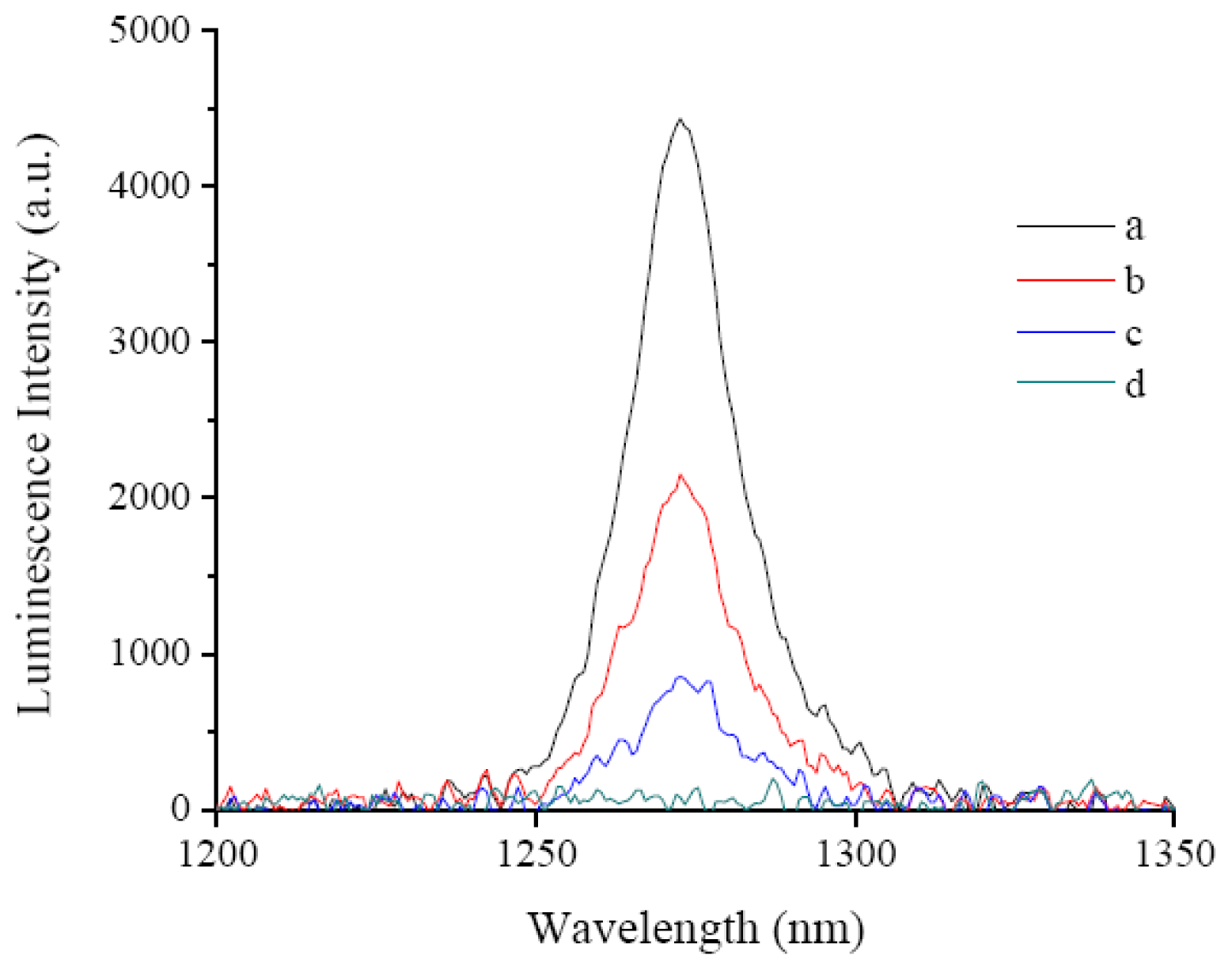
2.3. Fluorescence Emissions, Lifetimes and Singlet Oxygen Formation
3. Experimental Section
3.1. Materials and Methods
3.2. Synthesis of Zn(II)-5-(4-Acetoxy-3-methoxyphenyl)-10,15,20-tris-(4-carboxymethylphenyl)porphyrin (Zn(II)TCMPOMO) and Zn(II)-5-[(3,4-Methylenedioxy)phenyl]-10,15,20-tris-(4-carboxymethylphenyl) porphyrin (Zn(II)TRMOPP)
3.3. Synthesis of Cu(II)-5-(4-Acetoxy-3-methoxyphenyl)-10,15,20-tris-(4-carboxymethylphenyl)porphyrin (Cu(II)TCMPOMO) and Cu(II)-5-[(3,4-Methylenedioxy)phenyl]-10,15,20-tris-(4-carboxymethylphenyl) porphyrin (Cu(II)TRMOPP)
4. Conclusions
Acknowledgements
References
- Milgrom, L.R. What porphyrins are and what they do. In The Colours of Life: An Introduction to the Chemistry of Porphyrins and Related Compounds; Oxford University Press: Oxford, UK, 1977; Volume 1, pp. 1–22. [Google Scholar]
- Milgrom, L.R.; O’Neill, F. Porphyrins. In The Chemistry of Natural Products; Thomson,, R.H, Ed.; Blackie Academic & Professional: London, UK, 1993; Volume 8, pp. 329–376. [Google Scholar]
- Detty, M.R.; Gibson, S.L.; Wagner, S.J. Current clinical and preclinical photosensitizers for use in photodynamic therapy. J. Med. Chem 2004, 47, 3897–3195. [Google Scholar]
- Kamuhabwa, A.; Agostinis, P.; Ahmed, B.; Landuyt, W.; van Cleynenbreugel, B.; van Poppel, H.; de Witte, P. Hypericin as a potential phototherapeutic agent in superficial transitional cell carcinoma of the bladder. Photochem. Photobiol. Sci 2004, 3, 772–780. [Google Scholar]
- Hilderbrand, S.; Weissleder, R. Near-infrared fluorescence: Application to in vivo molecular imaging. Curr. Opin. Chem. Biol 2010, 14, 71–79. [Google Scholar]
- Chatterjee, D.K.; Fong, L.S.; Zhang, Y. Nanoparticles in photodynamic therapy: An emerging paradigm. Adv. Drug Deliv. Rev 2008, 60, 1627–1637. [Google Scholar]
- Bonneau, S.; Bizet, C.V.; Mojzisova, H.; Brault, D. Tetrapyrrole-photosensitizers vectorization and plasma LDL: A physic-chemical approach. Int. J. Pharm 2007, 344, 78–87. [Google Scholar]
- Chin, W.W.L.; Lau, O.W.K.; Bhuvaneswari, R.; Heng, P.W.S.; Olivo, M. Chlorin e6-polyvinylpyrrolidone as a fluorescent marker for fluorescence diagnosis of human bladder cancer implanted on the chick chorioallantoic membrane model. Cancer Lett 2007, 245, 127–133. [Google Scholar]
- Stockert, J.C.; Cañete, M.; Juarranz, A.; Villanueva, A.; Horobin, R.W.; Borrell, J.I.; Teixidó, J.; Nonell, S. Porphycenes: Facts and prospects in photodynamic therapy of cancer. Curr. Med. Chem 2007, 14, 997–1026. [Google Scholar]
- Grosseweiner, L.I. The Science of Phototherapy; CRC Press: London, UK, 1994; Volume Chapter 8, pp. 139–155. [Google Scholar]
- Schweiter, C.; Schmidt, R. Physical mechanisms of generation and deactivation of singlet oxygen. Chem. Rev 2003, 103, 1685–1758. [Google Scholar]
- Celli, J.P.; Spring, B.Q.; Rizvi, I.; Evans, C.L.; Samkoe, K.S.; Verma, S.; Pogue, B.W.; Hasan, T. Imaging and photodynamic therapy: Mechanisms, monitoring, and optimization. Chem. Rev 2010, 110, 2795–2838. [Google Scholar]
- Lovell, J.F.; Liu, T.W.; Chen, J.; Zheng, G. Activatable photosensitizers for imaging and therapy. Chem. Rev 2010, 110, 2839–2857. [Google Scholar]
- Berg, K.; Selbo, P.K.; Weyergang, A.; Dietze, A.; Prasmickaite, L.; Bonsted, A. Porphyrin related photosensitizers for cancer imaging and therapeutic applications. J. Microsc 2005, 218, 133–147. [Google Scholar]
- Bonnett, R. Chemical Aspects of Photodynamic Therapy. In Advanced Chemistry Texts; Gordon and Breach Science Publishers: Amsterdam, The Netherlands, 2000; Volume 1, pp. 57–112. [Google Scholar]
- Postino, F.; Mora, M.; DeMadariaga, M.A.; Nonell, S.; Sagrista, M.L. Incorporation of hydrophobic porphyrins into liposomes: Characterization and structural requirements. Int. J. Pharm 2004, 278, 239–254. [Google Scholar]
- Scalise, I.; Durantini, E.N. Photodynamic effect of metallo 5-(4-carboxyphenyl)-10,15,20-tris(4- methylphenyl) porphyrins in biomimetic AOT reverse micelles containing urease. J. Photochem. Photobiol. A 2004, 162, 105–113. [Google Scholar]
- Boyle, R.B.; Dolphin, D. Structure and biodistribution relationships of photodynamic sensitizers. Photochem. Photobiol 1996, 64, 469–485. [Google Scholar]
- Mac Donald, I.J.; Dougherty, T.J. Basic principles of photodynamic therapy. J. Porphyr. Phthalocyanines 2001, 5, 105–129. [Google Scholar]
- Nyman, E.S.; Hynninen, P.H. Research advances in the use of tetrapyrrolic photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B 2004, 73, 1–28. [Google Scholar]
- Rosenkranz, A.A.; Jans, D.A.; Sobolev, A.S. Targeted intracellular delivery of photosensitizers to enhance photodynamic efficiency. Immunol. Cell Biol 2002, 78, 452–464. [Google Scholar]
- Osterloh, J.; Vicente, M.G.H. Mechanisms of porphyrinoid localization in tumors. J. Porphyr. Phthalocyanines 2002, 5, 305–325. [Google Scholar]
- Milgrom, L.; MacRobert, S. Light years ahead. Chem. Br 1998, 34, 45–50. [Google Scholar]
- Boscencu, R.; Socoteanu, R.; Oliveira, A.S.; Vieira Ferreira, L.F.; Nacea, V.; Patrinoiu, G. Synthesis and characterization of some unsymmetrically-substituted mesoporphyrinic mono-hydroxyphenyl complexes of Copper(II). Pol. J. Chem 2008, 82, 509–522. [Google Scholar]
- Boscencu, R.; Socoteanu, R.; Oliveira, A.S.; Ferreira, L.F.V. Studies on Zn(II) monohydroxyphenyl mesoporphyrinic complexes. Synthesis and characterization. J. Serb. Chem. Soc 2008, 73, 713–726. [Google Scholar]
- Chandrasekharam, M.; Rao, C.S.; Singh, P.; Kantam, M.L.; Reddy, M.R.; Reddy, P.Y.; Toru, T. Microwave-assisted synthesis of metalloporphyrazines. Tetrahedron Lett 2007, 48, 2627–2630. [Google Scholar]
- Collman, J.P.; Decreau, R.A. Microwave-assisted synthesis of corroles. Tetrahedron Lett 2003, 44, 1207–1210. [Google Scholar]
- Liu, M.O.; Hu, A.T. Microwave-assisted synthesis of phthalocyanine-porphyrin complex and its photoelectric conversion properties. J. Organomet. Chem 2004, 689, 2450–2455. [Google Scholar]
- Samaroo, D.; Soll, C.E.; Todaro, L.J.; Drain, C.M. Efficient microwave-assisted synthesis of amine substituted pentafluorophenylporphyrin. Org Lett 2006, 8, 4985–4988. [Google Scholar]
- Kappe, C.O.; Dallinger, D. The impact of microwave synthesis on drug discovery. Nat. Rev. Drug Discov 2006, 5, 51–63. [Google Scholar]
- Liu, M.O.; Tai, C.H.; Hu, A.T. Synthesis of metalloporphyrins by microwave irradiation and their fluorescent properties. Mater. Chem. Phys 2005, 92, 322–326. [Google Scholar]
- Lindsey, J.S. Synthetic routes to meso-patterned porphyrins. Acc. Chem. Res 2010, 43, 300–311. [Google Scholar]
- Senge, M.O. Nucleophilic substitution as a tool for the synthesis of unsymmetrical porphyrins. Acc. Chem. Res 2005, 38, 733–743. [Google Scholar]
- Senge, M.O.; Shaker, Y.M.; Pintea, M.; Ryppa, C.; Hatscher, S.S.; Ryan, A.; Sergeeva, Y. Synthesis of meso-substituted ABCD-type porphyrins by functionalization reactions. Eur. J. Org. Chem 2010, 2, 237–258. [Google Scholar]
- Boscencu, R.; Ilie, M.; Socoteanu, R.; Oliveira, A.S.; Constantin, C.; Neagu, M.; Manda, G.; Vieira Ferreira, L.F. Microwave synthesis, basic spectral and biological evaluation of some copper(II) mesoporphyrinic complexes. Molecules 2010, 15, 3731–3743. [Google Scholar]
- Boscencu, R.; Socoteanu, R.; Ilie, M.; Oliveira, A.S.; Constantin, C.; Vieira Ferreira, L.F. Synthesis, spectral and biological evaluation of some mesoporphyrinic Zn(II) complexes. Rev. Chim 2009, 10, 1006–1011. [Google Scholar]
- Mack, J.; Stilman, M.J. Electronic Structure of Metal Phtalocyanine and Porphyrin Complexes from Analysys of UV-Visible Absorption and Magnetic Circular Dichroism Spectra and Molecular Orbital Calculations. In The Porphyrin Handbook; Kadish, K.M., Smith, K.M., Guilard, R., Eds.; Academic Press: San Diego, CA, USA, 2003; Volume 16, pp. 43–52. [Google Scholar]
- Gouterman, M. Optical Spectra and Electronic Structure of Porphyrins and Related Rings. In The Porphyrins; Dolphin, D., Ed.; Academic Press: New York, NY, USA, 1978; Volume III, pp. 1–165. [Google Scholar]
- Boscencu, R.; Ilie, M.; Socoteanu, R. Spectroscopic behavior of some A3B type tetrapyrrolic complexes in several organic solvents and micellar media. Int. J. Mol. Sci 2011, 12, 5552–5564. [Google Scholar]
- Lu, F.T.; Gao, L.N.; Li, H.H.; Ding, L.P.; Fang, Y. Molecular engineered silica surfaces with an assembled anthracene monolayer as a fluorescent sensor for organic copper(II) salts. Appl. Surf. Sci 2007, 253, 4123–4131. [Google Scholar]
- Branco, T.J.F.; do Rego, A.M.B.; Machado, I.F.; Ferreira, L.F.V. A luminescence lifetime distributions analysis in heterogeneous systems by the use of Excel’s Solver. J. Phys. Chem. B 2005, 109, 15958–15967. [Google Scholar]
- Do Rego, A.M.B.; Ferreira, L.F.V. Photonic and Electronic Spectroscopies for the Characterization of Organic Surfaces and Organic Molecules Adsorbed on Surfaces. In Handbook of Surfaces and Interfaces of Materials; Nalwa, H.S., Ed.; Academic Press: San Diego, CA, USA, 2001; Volume Chapter 7, pp. 275–313. [Google Scholar]
- Lin, W.C. Electron Spin Resonance and Electronic Structure of Metalloporphyrins. In The Porphyrins; Dolphin, D., Ed.; Academic Press: New York, NY, USA, 1978; Volume 4, pp. 358–364. [Google Scholar]
- Manoharan, P.T.; Roger, M.T. ESR Study of Copper(II) and Silver(II) Tetraphenylporphyrin. In Electron Spin Resonance of Metal Complexes; Yen, T.F., Ed.; Plenum Press: New York, NY, USA, 1969; pp. 143–173. [Google Scholar]
- Kivelson, D.; Neiman, R.R. ESR studies on the bonding in copper complexes. J. Chem. Phys 1961, 35, 149–155. [Google Scholar]
| Assignments | Zn(II)TCMPOMO | Cu(II)TCMPOMO | Zn(II)TRMOPP | Cu(II)TRMOPP |
|---|---|---|---|---|
| νC–H | 2920m | 2921m | 2922w | 2924w |
| νC–H from –O–CH2–O– | - | - | 2915m | 2916m |
| νC–H from –O–CH3 | 2850m | 2851m | 2850m | 2850m |
| νC = O | 1723m | 1724m | 1722m | 1725m |
| νC–N | 1606m | 1602m | 1605m | 1606m |
| νC = N | 1510m | 1500m | 1492m | 1496m |
| νC–H pyrrole | 1456w | 1458w | 1433w | 1433w |
| νC–O | 1190s | 1188m | 1198s | 1195s |
| δC–H | 997m | 1000m | 996m | 999m |
| γC–C | 867w | 865w | 867w | 867w |
| γC–N pyrrole | 795m | 799m | 780m | 792m |
| Solvent | λmax (nm) [lg ɛ (L·mol−1·cm−1)] | ||
|---|---|---|---|
| Soret band B(0,0) | Q bands Qy(0,0) | Qx(1,0) | |
| Zn(II)-5-(4-acetoxy-3-methoxyphenyl)-10,15,20-tris-(4-carboxymethylphenyl)porphyrin | |||
| EtOH | 426.2 [5.646] | 558.6 [4.203] | 599.3 [3.783] |
| iso-PrOH | 427.0 [5.641] | 558.6 [4.271] | 599.1 [3.932] |
| CHCl3 | 422.6 [5.584] | 549.8 [4.261] | 589.5 [3.690] |
| DMF | 428.9 [5.602] | 560.3 [4.201] | 601.4 [3.914] |
| DMSO | 431.5 [5.588] | 562.0 [4.227] | 602.7 [3.979] |
| Cu(II)-5-(4-acetoxy-3-methoxyphenyl)-10,15,20-tris-(4-carboxymethylphenyl)porphyrin | |||
| EtOH | 413.0 [5.629] | 537.0 [4.095] | - |
| iso-PrOH | 414.1 [5.507] | 537.1 [4.174] | - |
| CHCl3 | 416.3 [5.539] | 539.9 [4.325] | - |
| DMF | 417.0 [5.565] | 540.1 [4.142] | - |
| DMSO | 420.8 [5.479] | 544.0 [4.261] | - |
| Zn(II)-5-[(3,4-methylenedioxy)phenyl]-10,15,20-tris-(4-carboxymethylphenyl)porphyrin | |||
| EtOH | 426.0 [5.687] | 558.2 [4.292] | 598.7 [3.924] |
| iso-PrOH | 426.3 [5.671] | 558.4 [4.274] | 598.7 [3.903] |
| CHCl3 | 422.0 [5.572] | 549.3 [4.271] | 587.2 [3.680] |
| DMF | 429.0 [5.635] | 559.8 [4.255] | 601.0 [3.944] |
| DMSO 1 | 431.0 [5.632] | 561.9 [4.246] | 603.0 [3.964] |
| Cu(II)-5-[(3,4-methylenedioxy)phenyl]-10,15,20-tris-(4-carboxymethylphenyl)porphyrin | |||
| EtOH | 414.1 [5.778] | 538.0 [4.447] | - |
| iso-PrOH | 414.1 [5.772] | 538.0 [4.435] | - |
| CHCl3 | 416.2 [5.698] | 539.4 [4.387] | - |
| DMF | 418.2 [5.668] | 540.8 [4.394] | - |
| DMSO 1 | 422.3 [5.652] | 544.4 [4.408] | - |
| Porphyrinic compounds | ΦΔ | ΦF | τF (ns) |
|---|---|---|---|
| TCMPOMO | 0.42 | 0.09 | 7.89 |
| Cu(II)TCMPOMO | 0.08 | <0.01 | 1.90 |
| Zn(II)TCMPOMO | 0.16 | 0.06 | 1.75 |
| Cu(II)TRMOPP | 0.04 | <lod * | <lod * |
| Zn(II)TRMOPP | 0.24 | 0.06 | 1.71 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Boscencu, R.; Oliveira, A.S.; Ferreira, D.P.; Ferreira, L.F.V. Synthesis and Spectral Evaluation of Some Unsymmetrical Mesoporphyrinic Complexes. Int. J. Mol. Sci. 2012, 13, 8112-8125. https://doi.org/10.3390/ijms13078112
Boscencu R, Oliveira AS, Ferreira DP, Ferreira LFV. Synthesis and Spectral Evaluation of Some Unsymmetrical Mesoporphyrinic Complexes. International Journal of Molecular Sciences. 2012; 13(7):8112-8125. https://doi.org/10.3390/ijms13078112
Chicago/Turabian StyleBoscencu, Rica, Anabela Sousa Oliveira, Diana P. Ferreira, and Luís Filipe Vieira Ferreira. 2012. "Synthesis and Spectral Evaluation of Some Unsymmetrical Mesoporphyrinic Complexes" International Journal of Molecular Sciences 13, no. 7: 8112-8125. https://doi.org/10.3390/ijms13078112
APA StyleBoscencu, R., Oliveira, A. S., Ferreira, D. P., & Ferreira, L. F. V. (2012). Synthesis and Spectral Evaluation of Some Unsymmetrical Mesoporphyrinic Complexes. International Journal of Molecular Sciences, 13(7), 8112-8125. https://doi.org/10.3390/ijms13078112




