Green Formation of Spherical and Dendritic Silver Nanostructures under Microwave Irradiation without Reducing Agent
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Characterization Methods
3. Results and Discussion
3.1. MW Irradiation
3.1.1. TEM Images
3.1.2. Photon Correlation Spectroscopy
3.1.3. UV–vis Spectra
3.2. Formation of AgNPs with Conventional Heating
4. Conclusions
Acknowledgement
References
- Sclafani, A.; Herrmann, J. Influence of metallic silver and of platinum silver bimetallic deposits on the photocatalytic activity of titania (anatase and rutile) in organic and aqueous media. J. Photochem. Photobiol. A 1998, 113, 181–188. [Google Scholar]
- Jiang, H.; Moon, K.; Zhang, Z. Variable frequency microwave synthesis of silver nanoparticles. J. Nanopart. Res 2006, 8, 117–124. [Google Scholar]
- Sun, Y.G.; Mayers, B.T.; Herricks, Y. Crystalline silver nanowires by soft solution processing. Nano Lett 2003, 3, 955–960. [Google Scholar]
- Darroudi, M.; Ahmad, M.B. Preparation and characterization of gelatin mediated silver nanoparticles by laser ablation. J. Alloy Compd 2011, 509, 1301–1304. [Google Scholar]
- Carotenuto, G.; Pepe, G. Preparation and characterization of nano-sized Ag/PVP composites for optical applications. Eur. Phys. J. B 2000, 16, 11–17. [Google Scholar]
- Clary, D.R.; Mills, G. Preparation and thermal properties of CuO particles. J. Phys. Chem. C 2011, 115, 14656–14663. [Google Scholar]
- Bilecka, I.; Elser, P.; Niederberger, M. Kinetic and thermodynamic aspects in the microwave-assisted synthesis of ZnO nanoparticles in benzyl alcohol. ACS Nano 2009, 3, 467–477. [Google Scholar]
- Zhu, J.F.; Zhu, Y.J.; Ma, M.G. Simultaneous and rapid microwave synthesis of polyacrylamide-metal sulfide (Ag2S, Cu2S, HgS) nanocomposites. J. Phys. Chem. C 2007, 111, 3920–3926. [Google Scholar]
- Mohamed, M.B.; AbouZeid, K.M.; Abdelsayed, V.A. Growth mechanism of anisotropic gold nanoparticles via microwave synthesis: formation of dioleamide by gold nanocatalysis. ACS Nano 2010, 4, 2766–2772. [Google Scholar]
- Tsuji, M.; Hashimoto, M.; Nishizawa, Y. Microwave rapid heating has received considerable attention as a new promising method for the one-pot synthesis of metallic nanostructures in solutions. Chem. Eur. J 2005, 11, 440–452. [Google Scholar]
- Sun, Y.; Mayers, B.; Herricks, T. Polyol synthesis of uniform silver nanowires: a plausible growth mechanism and the supporting evidence. Nano Lett 2003, 3, 955–960. [Google Scholar]
- Wada, Y.; Kuramoto, H. Microwave-assisted size control of CdS nanocrystallites. J. Mater. Chem 2001, 11, 1936–1940. [Google Scholar]
- Kundu, S.; Peng, L. New route to obtain high-yield multiple-shaped gold nanoparticles in aqueous solution using microwave irradiation. Inorg. Chem 2008, 47, 6344–6352. [Google Scholar]
- Acharya, S.; Singh, K. Microwave-assisted chemical reduction routes for direct synthesis of (fct) L10 phase of Fe-Pt. J. Microw. Power Electromagn. Energy 2011, 45, 63–69. [Google Scholar]
- He, R.; Qian, X.; Yin, J. Formation of silver dendrites under microwave irradiation. Chem. Phys. Lett 2003, 369, 454–458. [Google Scholar]
- Zhou, X.; Huang, X.; Qi, X. In situ synthesis of metal nanoparticles on single-layer graphene oxide and reduced graphene oxide surfaces. J. Phys. Chem. C 2009, 113, 10842–10846. [Google Scholar]
- Hood, E. Nanotechnology: Looking as we leap. Environ. Health Perspect 2004, 112, 740–749. [Google Scholar]
- Acharya, S.A.; Khule, S.M.; Singh, K.; Bhoga, S.S. Direct Synthesis of Fe-Pt Nanoparticles in Ordered Face Centered (fct) LIo Phase by Microwave Assisted Route. Proceedings of the 55th DAE Solid State Physics Symposium, Manipal, India, 26–30 December 2010; pp. 371–372.
- Hsiao, M.; Lin, C.; Chang, Y.; Chen, L. Ultrasonic mixing and closed microwave irradiation-assisted transesterification of soybean oil. Fuel 2010, 89, 3618–3622. [Google Scholar]
- Chen, G.; Zhu, F.; Sun, X.; Sun, S.; Chen, R. Benign synthesis of ceria hollow nanocrystals by a template-free method. CrystEngComm 2011, 13, 2904–2908. [Google Scholar]
- Chung, S.J.; Leonard, J.P. Characterization of ZnO nanoparticle suspension in water: Effectiveness of ultrasonic dispersion. Powder Technol 2009, 194, 75–80. [Google Scholar]
- McConnell, D.R. Energy consumption: A comparison between the microwave oven and the conventional electric rang. J. Microw. Power 1974, 9, 341–347. [Google Scholar]
- Cheng, W.M.; Raghavan, G.S.V.; Ngadi, M.; Wang, N. Microwave power control strategies on the drying process. I. Development and evaluation of new microwave drying system. J. Food Eng 2006, 76, 188–194. [Google Scholar]
- Link, S.; El-Sayed, M.A. Size and temperature dependence of the plasmon absorption of colloidal gold nanoparticles. J. Phys. Chem. B 1999, 103, 4212–4217. [Google Scholar]
- Baruwati, B.; Polshettiwar, V. Glutathione promoted expeditious green synthesis of silver nanoparticles in water using microwaves. Green Chem 2009, 11, 926–930. [Google Scholar]
- Agrawal, V.V.; Kulkarni, G.U. Surfactant-promoted formation of fractal and dendritic nanostructures of gold and silver at the organic–aqueous interface. J. Colloid Interface Sci 2008, 318, 501–506. [Google Scholar]
- Tsuji, T.; Iryo, K. Preparation of silver nanoparticles by laser ablation in solution: Influence of laser wavelength on particle size. Appl. Surf. Sci 2002, 202, 80–85. [Google Scholar]
- Kappe, C.O. Controlled microwave heating in modern organic synthesis. Angew. Chem. Int. Ed 2004, 43, 6250–6284. [Google Scholar]
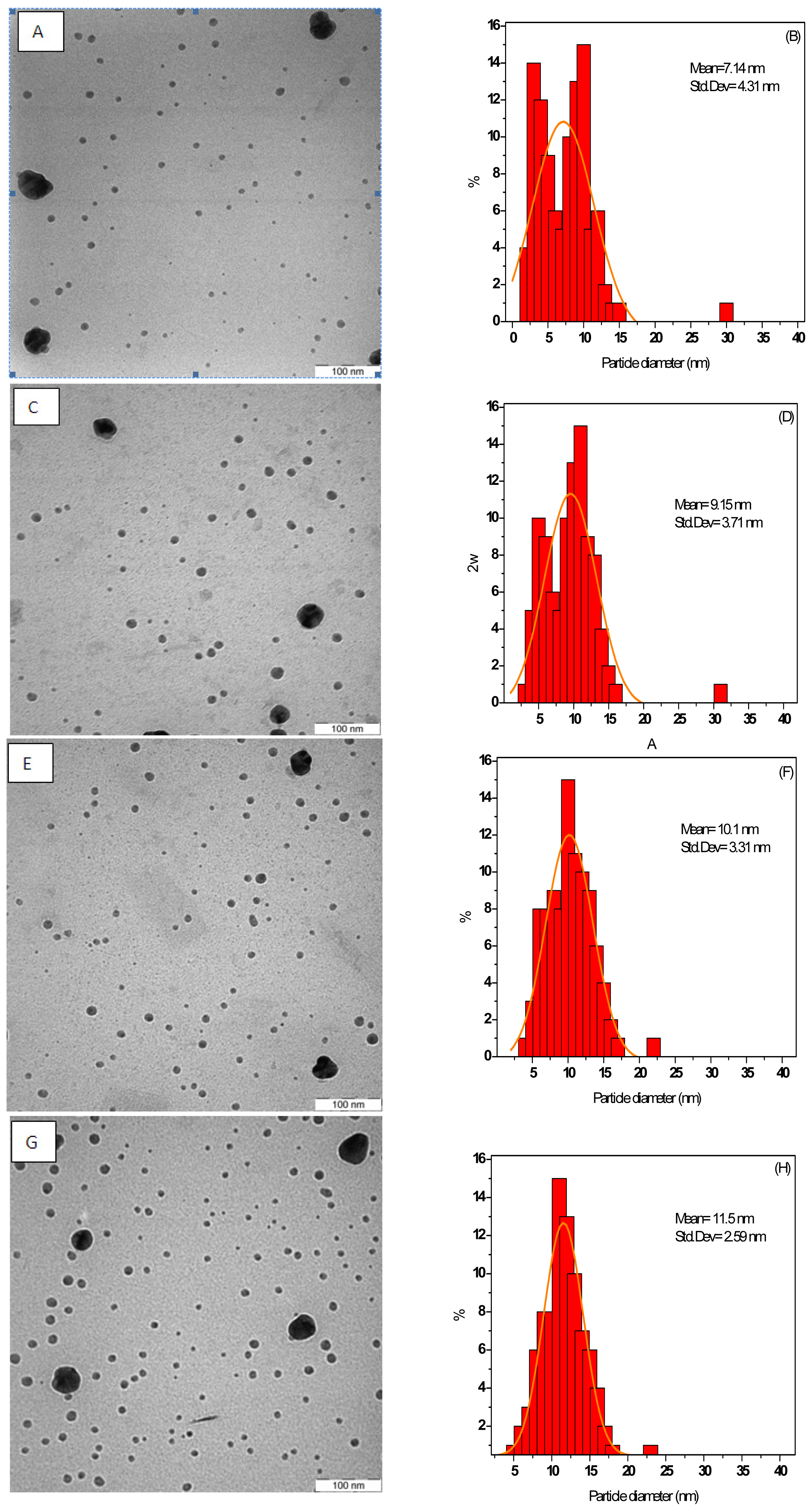


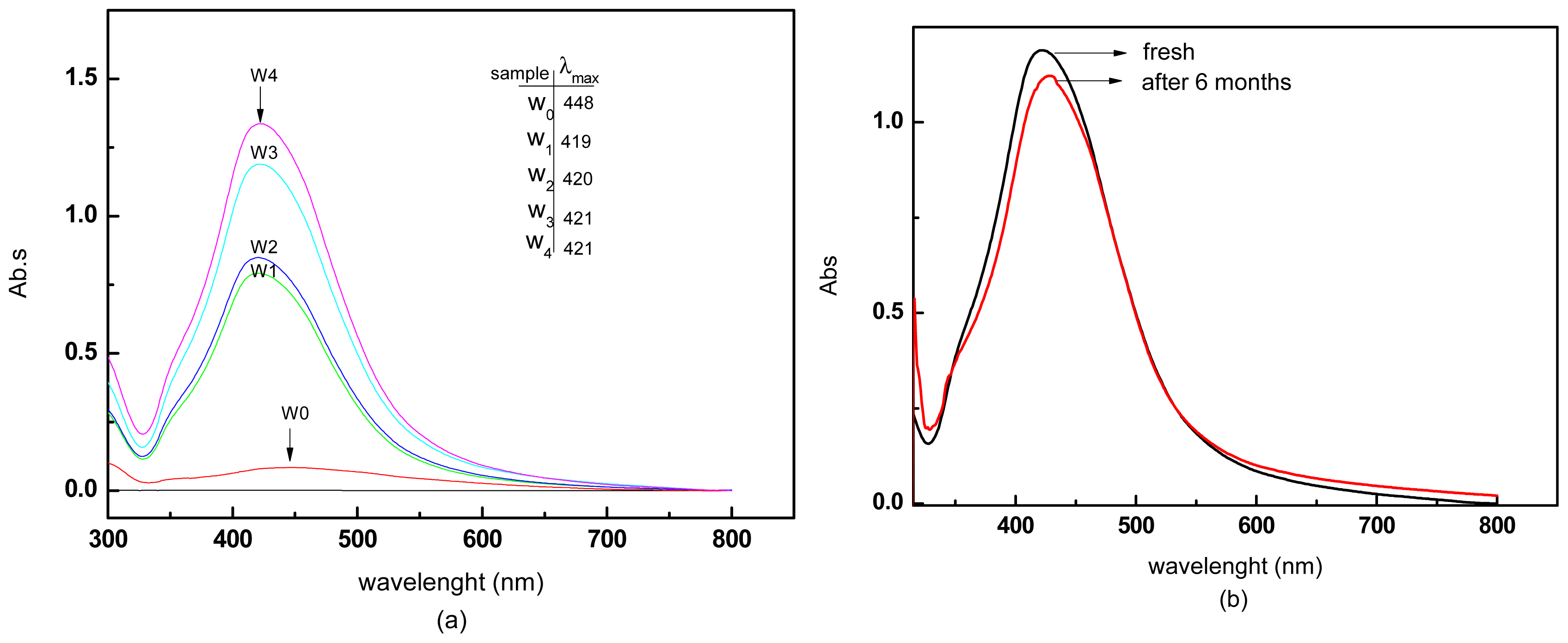

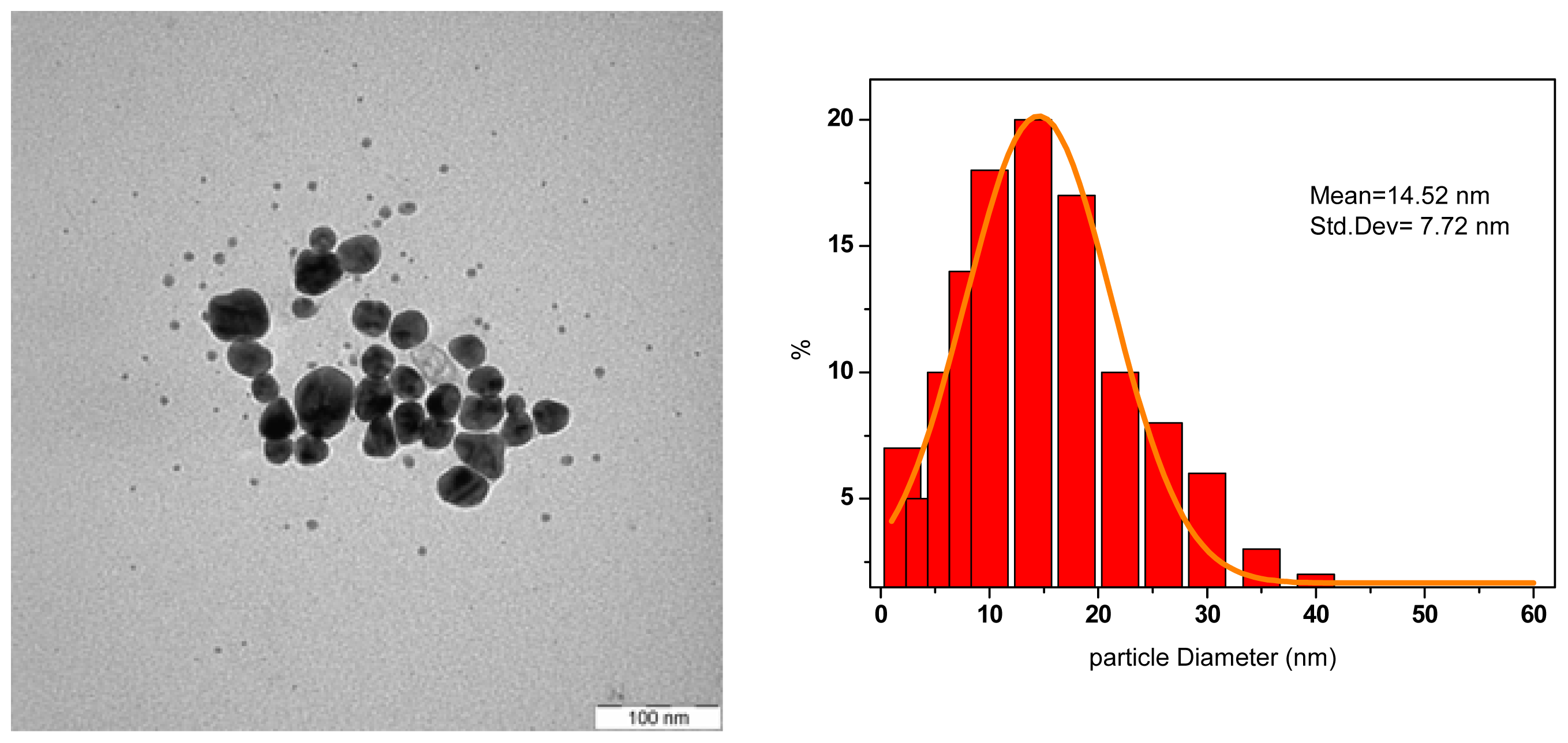
| MW Irradiation Time (s) | Ag-NPs Mean Diameter (nm) | Standard Deviation (nm) |
|---|---|---|
| 20 | 81.51 | 23.86 |
| 40 | 94.49 | 22.97 |
| 60 | 101.25 | 19.45 |
| 90 | 110.66 | 12.25 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Noroozi, M.; Zakaria, A.; Moksin, M.M.; Wahab, Z.A.; Abedini, A. Green Formation of Spherical and Dendritic Silver Nanostructures under Microwave Irradiation without Reducing Agent. Int. J. Mol. Sci. 2012, 13, 8086-8096. https://doi.org/10.3390/ijms13078086
Noroozi M, Zakaria A, Moksin MM, Wahab ZA, Abedini A. Green Formation of Spherical and Dendritic Silver Nanostructures under Microwave Irradiation without Reducing Agent. International Journal of Molecular Sciences. 2012; 13(7):8086-8096. https://doi.org/10.3390/ijms13078086
Chicago/Turabian StyleNoroozi, Monir, Azmi Zakaria, Mohd Maarof Moksin, Zaidan Abd Wahab, and Alam Abedini. 2012. "Green Formation of Spherical and Dendritic Silver Nanostructures under Microwave Irradiation without Reducing Agent" International Journal of Molecular Sciences 13, no. 7: 8086-8096. https://doi.org/10.3390/ijms13078086




