Growth Promotion of Yunnan Pine Early Seedlings in Response to Foliar Application of IAA and IBA
Abstract
:1. Introduction
2. Results and Discussion
2.1. Trend of Exogenous Auxin Effect
2.2. Pattern of Exogenous Auxin Effects
2.2.1. Pattern of One-Factor Effects
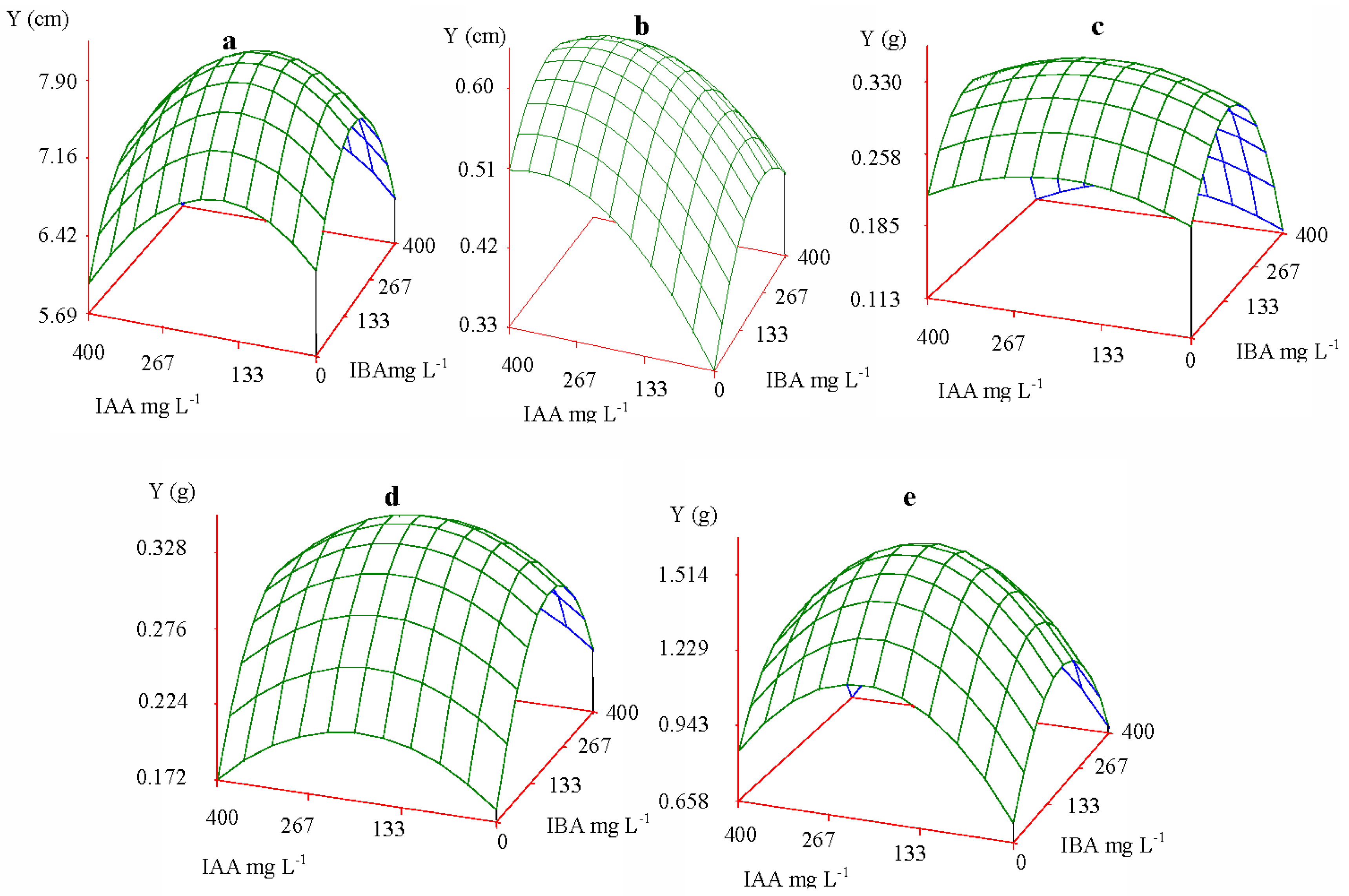
2.2.2. Pattern of Interaction of IAA and IBA
2.2.3. Simulation Test of Whole Factors
2.3. Optimum Concentrations and Ratios of IAA and IBA
3. Experimental Sections
3.1. Plant Material
3.2. Phytohormone Preparation
3.3. Experimental Design
3.4. Data Collection and Statistical Analysis
4. Conclusions
Acknowledgments
References
- Chinese Academy of Sciences, Flora of China; Science Press: Beijing, China, 1978; Volume 7, pp. 255–259.
- Wu, Z.Y. Flora Yunnanica (Tomus 4): Spermatophyta; Science Press: Beijing, China, 1986; pp. 54–57. [Google Scholar]
- Jin, Z.Z.; Peng, J. Yunnan Pine (Pinus yunnanensis Franch.); Yunnan Science and Technology Press: Kunming, China, 2004; pp. 1–66. [Google Scholar]
- Teale, W.D.; Paponov, I.A.; Palme, K. Auxin in action: Signalling, transport and the control of plant growth and development. Nat. Rev. Mol. Cell Biol 2006, 7, 84–759. [Google Scholar]
- Ishii, T.; Soeno, K.; Asami, T.; Fujioka, S.; Shimada, Y. Arabidopsis seedlings over-accumulated indole-3-acetic acid in response to aminooxyacetic acid. Biosci. Biotechnol. Biochem 2010, 74, 2345–2347. [Google Scholar]
- Woodward, A.W.; Bartel, B. Auxin: Regulation, action, and interaction. Ann. Bot. (Lond. ) 2005, 95, 707–735. [Google Scholar]
- Strader, L.C.; Culler, A.H.; Cohen, J.D.; Bartel, B. Conversion of endogenous indole-3-butyric acid to indole-3-acetic acid drives cell expansion in arabidopsis seedlings. Plant Physiol 2010, 153, 1577–1586. [Google Scholar]
- Little, C.H.A.; Savidge, R.A. The role of plant growth regulators in forest tree cambial growth. Plant Growth. Regul 1987, 6, 137–169. [Google Scholar]
- Hoad, G.V.; Lenton, J.R.; Jackson, M.B.; Atkin, R.K. Hormone Action in Plant Development: A Critical Appraisal; Butterworths: London, UK, 1987; p. 315. [Google Scholar]
- Petersson, S.V.; Johansson, A.I.; Kowalczyk, M.; Makoveychuk, A.; Wang, J.Y.; Moritz, T.; Grebe, M.; Benfey, P.N.; Sandberg, G.; Ljung, K. Gradient and maximum in the arabidopsis root apex shown by high-resolution cell-specific analysis of IAA distribution and synthesis. Plant Cell 2009, 21, 1659–1668. [Google Scholar]
- Ahmed, M.G.U.; Lee, C.H. Induction of protonemal gemmae and gametophyte of Cratoneuron decipien (Brid.) G. Roth using IAA and kinetin. Plant Omics 2010, 3, 52–56. [Google Scholar]
- Liao, Y.K.; Liao, C.K.; Ho, Y.L. Maturation of somatic embryos in two embryogenic cultures of Picea morrisonicola Hayata as affected by alternation of endogenous IAA content. Plant Cell Tiss. Org 2008, 93, 257–268. [Google Scholar]
- Sachdev, D.P.; Chaudhari, H.G.; Kasture, V.M.; Dhavale, D.D.; Chopade, B.A. Isolation and characterization of indole acetic acid (IAA) producing Klebsiella pneumoniae strains from rhizosphere of wheat (Triticum aestivum) and their effect on plant growth. Indian J. Exp. Biol 2009, 47, 993–1000. [Google Scholar]
- Wang, Y.; Du, S.T.; Li, L.L.; Huang, L.D.; Fang, P.; Lin, X.Y.; Zhang, Y.S.; Wang, H.L. Effect of CO(2) elevation on root growth and its relationship with indole acetic acid and ethylene in tomato seedlings. Pedosphere 2009, 19, 570–576. [Google Scholar]
- Camolesi, M.R.; Unemoto, L.K.; Sachs, P.J.D.; Roberto, S.R.; Sato, A.J.; Faria, A.P.; Rodrigues, E.B.; da Silva, J.V. Semi-hardwood cuttings rooting of peach tree “Okinawa” under effect of lesion and indolebutyric acid. Cienc. Rural 2007, 37, 1805–1808. [Google Scholar]
- Qaddoury, A.; Amssa, M. Effect of exogenous indole butyric acid on root formation and peroxidase and indole-3-acetic acid oxidase activities and phenolic contents in date Palm offshoots. Bot. Bull. Acad. Sin 2004, 45, 127–131. [Google Scholar]
- Singh, A.; Khan, M.A. Comparative effect of IAA, IBA and NAA on rooting of hardwood stem cuttings of Celtis australis Linn. Range Manag. Agrofor 2009, 30, 78–80. [Google Scholar]
- Li, X.J.; Li, S.Y.; Lin, J.X. Effect of GA3 spraying on lignin and auxin contents and the correlated enzyme activities in bayberry (Myrica rubra Bieb.) during flower-bud induction. Plant Sci 2003, 164, 549–556. [Google Scholar]
- Kurahashi, Y.; Terashima, A.; Takumi, S. Variation in dehydration tolerance, ABA sensitivity and related gene expression patterns in D-Genome progenitor and synthetic hexaploid wheat lines. Int. J. Mol. Sci 2009, 10, 2733–2751. [Google Scholar]
- Fukuda, A.; Tanaka, Y. Effects of ABA, auxin, and gibberellin on the expression of genes for vacuolar H+-inorganic pyrophosphatase, H+-ATPase subunit A, and Na+/H+ antiporter in barley. Plant Physiol. Biochem 2006, 44, 351–358. [Google Scholar]
- Zhang, R.; Zhang, X.; Wang, J.; Letham, D.S.; McKinney, S.A.; Higgins, T.J.V. The effect of auxin on cytokinin levels and metabolism in transgenic tobacco tissue expressing an ipt gene. Planta 1995, 196, 84–94. [Google Scholar]
- Hadi, F.; Bano, A.; Fuller, M.P. The improved phytoextraction of lead (Pb) and the growth of maize (Zea mays L.): The role of plant growth regulators (GA(3) and IAA) and EDTA alone and in combinations. Chemosphere 2010, 80, 457–462. [Google Scholar]
- Li, Y.F.; Zhang, Y.M.; Liu, D.Y.; Zhao, M.C.; Xu, Y.L. The seedling growth of Pinus yunnanensis in response to the seeds soaked by exogenous phytohormone. J. Yunnan Univ 2011, 3, 350–359. [Google Scholar]
- Zhao, M.C.; Li, L.F.; Li, G.Q.; Liu, D.Y.; Zhang, Y.M. Effects of IAA and IBA on rooting of Pinus yunnanensis cuttings. J. West China For. Sci 2008, 3, 13–17. [Google Scholar]
- Shelford, V.E. Law of Toleration. In Animal Communities in Temperate America; University of Chicago Press: Chicago, IL, USA, 1913; pp. 302–303. [Google Scholar]
- Odum, E.P. Foundation of Ecology; People Education Press: Beijing, China, 1981; pp. 104–106. Sun, R.Y.; Qian, G.Z.; Lin, H.R.; Zhu, J.; Li, Z.Y.; Lu, H.J., Translators. [Google Scholar]
- Odum, E.P.; Barrett, G.W. Foundation of Ecology, 4th ed; Higher Education Press: Beijing, China, 2009; pp. 161–163. Lu, J.J.; Wang, W.; Wang, T.H.; He, W.S.; Li, X.Z., Translators. [Google Scholar]
- Tong, M.; Zhang, L.; Wang, Y.; Jiang, H.; Ren, Y. Fe-Chlorophyllin promotes the growth of wheat roots associated with nitric oxide generation. Int. J. Mol. Sci 2010, 11, 5246–5255. [Google Scholar]
- Fässler, E.; Evangelou, M.W.; Robinson, B.H.; Schulin, R. Effects of indole-3-acetic acid (IAA) on sunflower growth and heavy metal uptake in combination with ethylene diamine disuccinic acid (EDDS). Chemosphere 2010, 80, 901–907. [Google Scholar]
- Rahman, A.; Amakawa, T.; Goto, N.; Tsurumi, S. Auxin is a positive regulator for ethylene-mediated response in the growth of Arabidopsis roots. Plant Cell Phys 2001, 42, 301–307. [Google Scholar]
- Ivanchenko, M.G.; Napsucialy-Mendivil, S.; Dubrovsky, J.G. Auxin-induced inhibition of lateral root initiation contributes to root system shaping in Arabidopsis thaliana. Plant J 2010, 64, 740–752. [Google Scholar]
- Ryu, C.-M.; Hu, C.-H.; Locy, R.D.; Kloepper, J.W. Study of mechanisms for plant growth promotion elicited by rhizobacteria in Arabidopsis thaliana. Plant Soil 2005, 268, 285–292. [Google Scholar]
- Liu, K.; Xu, S.; Xuan, W.; Ling, T.F.; Cao, Z.Y.; Huang, B.K.; Sun, Y.G.; Fang, L.; Liu, Z.Y.; Zhao, N.; et al. Carbon monoxide counteracts the inhibition of seed germination and alleviates oxidative damage caused by salt stress in Oryza sativa. Plant Sci 2007, 172, 544–555. [Google Scholar]
- Fässler, E.; Evangelou, M.W.; Robinson, B.H.; Schulin, R. Effects of indole-3-acetic acid (IAA) on sunflower growth and heavy metal uptake in combination with ethylene diamine disuccinic acid (EDDS). Chemosphere 2010, 80, 901–907. [Google Scholar]
- Odum, E.P. Principles Pertaining to Limiting Factors. In Fundamentals of Ecology, 3rd ed; Saunders: Philadelphia, PA, USA, 1971; pp. 107–109. [Google Scholar]
- Pianka, E.R.; Limiting, Factors. Tolerance Limits and the Principle of Allocation. In Evolutionary Ecology; Harper & Row: New York, NY, USA, 1974; pp. 14–17. [Google Scholar]
- Gangwar, S.; Singh, V.P. Indole acetic acid differently changes growth and nitrogen metabolism in Pisum sativum L. seedlings under chromium(VI) phototoxicity: Implication of oxidative stress. Sci. Hortic 2011, 129, 321–328. [Google Scholar]
- Gangwar, S.; Singh, V.P.; Prasad, S.M.; Maurya, J.N. Differential responses of pea seedlings to indole acetic acid under manganese toxicity. Acta Physiol. Plant 2011, 33, 451–462. [Google Scholar]
- Sen, B.R. Statistic of Crop Response to Fertilizer; FAO: Rome, Italy, 1996; pp. 95–103. [Google Scholar]
- Kimura, M. Measuring Methods of Production in Terrestrial Plant Communities; Science Press: Beijing, China, 1976; Shu, J., Translator. [Google Scholar]
- Ovington, J.D.; Heitkamp, D.; Lawrence, D.B. Plant biomass and productivity of prairie, savanna, oak wood and maize field ecosystems in Central Minnesota. Ecology 1963, 44, 52–63. [Google Scholar]
- Li, G.Q.; Tang, D.R. A preliminary study of the fertilization effects by using nitrogen mixed with phosphorus on Eucomia ulmoides young trees. Shanxi For. Sci. Technol 1994, 3, 55–59. [Google Scholar]
- Gwaze, D.P.; Bridgwater, F.E.; Williams, C.G. Genetic analysis of growth curves for a woody perennial species, Pinus taeda L. Theor. Appl. Genet 2002, 105, 526–531. [Google Scholar]

| Growth index(Y) | Auxin effect equations | Correlation coefficient (R) | F value | Significance level (P) | |
|---|---|---|---|---|---|
| Increment | Height | 0.987 | 22.619 | 0.014 | |
| Collar diameter | 0.975 | 11.541 | 0.036 | ||
| Biomass | Root | 0.991 | 32.891 | 0.008 | |
| Stem | 0.970 | 9.532 | 0.046 | ||
| Leaf | 0.822 | 7.476 | 0.001 | ||
| Growth index(Y) | One-factor equation | Maximum yield | One-factor equation | Maximum yield | |
|---|---|---|---|---|---|
| Increment | Height | 6.97 cm | 7.40 cm | ||
| Collar diameter | 0.51 cm | 0.46 cm | |||
| Biomass | Rroot | 0.251 g | 0.301 g | ||
| Stem | 0.218 g | 0.290 g | |||
| Leaf | 1.161 g | 1.120 g | |||
| Growth index (Y) | Optimum concentration | Optimum ratio of IAA and IBA IAA:IBA | Theoretical maximum yield | ||
|---|---|---|---|---|---|
| IAA mg·L−1 | IBA mg·L−1 | ||||
| Increment | Height | 166.75 | 185.66 | 1:1.11 | 7.90 cm |
| Collar diameter | 310.06 | 217.14 | 1:0.70 | 0.60 cm | |
| Biomass | Root | 192.84 | 158.61 | 1:0.82 | 0.330 g |
| Stem | 191.13 | 220.53 | 1:1.15 | 0.328 g | |
| Leaf | 206.47 | 185.55 | 1:0.90 | 1.516 g | |
| Treatment number. | IAA mg·L−1 | IBA mg·L−1 | Increment | Biomass | |||
|---|---|---|---|---|---|---|---|
| Height (cm) | Diameter (cm) | Root (g) | Stem (g) | Leaf (g) | |||
| 1 | IAA0 (0) | IBA0 (0) | 6.47 | 0.33 | 0.222 | 0.180 | 0.732 |
| 2 | IAA0 (0) | IBA1 (200) | 7.39 | 0.46 | 0.296 | 0.289 | 1.120 |
| 3 | IAA0 (0) | IBA2 (400) | 6.14 | 0.43 | 0.116 | 0.217 | 0.684 |
| 4 | IAA1 (200) | IBA0 (0) | 6.95 | 0.47 | 0.251 | 0.218 | 1.160 |
| 5 | IAA1 (200) | IBA1 (200) | 7.88 | 0.58 | 0.325 | 0.327 | 1.514 |
| 6 | IAA1 (200) | IBA2 (400) | 6.64 | 0.53 | 0.146 | 0.266 | 1.043 |
| 7 | IAA2 (400) | IBA0 (0) | 5.96 | 0.50 | 0.217 | 0.172 | 0.844 |
| 8 | IAA2 (400) | IBA1 (200) | 6.91 | 0.58 | 0.291 | 0.282 | 1.163 |
| 9 | IAA2 (400) | IBA2 (400) | 5.69 | 0.51 | 0.113 | 0.210 | 0.658 |
| Growth index(Y) | Tangency point of ridgeline mg·L−1 (Point A, B) | OP line equation | Highest yield (Point O) | ||
|---|---|---|---|---|---|
| OA | OB | ||||
| Increment | Height | IAA = A = 164.84 | IBA = B = 184.50 | 7.90 cm | |
| Collar Diameter | IAA = A = 352.11 | IBA = B = 261.78 | 0.60 cm | ||
| Biomass | Root | IAA = A = 191.57 | IBA = B = 158.23 | 0.330 g | |
| Stem | IAA = A = 190.48 | IBA = B = 220.26 | 0.328 g | ||
| Leaf | IAA = A = 215.05 | IBA = B = 194.17 | 1.516 g | ||
| Treat number | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
|---|---|---|---|---|---|---|---|---|---|
| Concentration of IAA mg·L−1 | 0 | 0 | 0 | 200 | 200 | 200 | 400 | 400 | 400 |
| Concentration of IBA mg·L−1 | 0 | 200 | 400 | 0 | 200 | 400 | 0 | 200 | 400 |
| Item | Field test arrangement | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Replication I | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
| Replication II | 4 | 5 | 6 | 7 | 8 | 9 | 1 | 2 | 3 |
| Replication III | 7 | 8 | 9 | 1 | 2 | 3 | 4 | 5 | 6 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, Y.; Zhang, Y.; Li, Y.; Li, G.; Liu, D.; Zhao, M.; Cai, N. Growth Promotion of Yunnan Pine Early Seedlings in Response to Foliar Application of IAA and IBA. Int. J. Mol. Sci. 2012, 13, 6507-6520. https://doi.org/10.3390/ijms13056507
Xu Y, Zhang Y, Li Y, Li G, Liu D, Zhao M, Cai N. Growth Promotion of Yunnan Pine Early Seedlings in Response to Foliar Application of IAA and IBA. International Journal of Molecular Sciences. 2012; 13(5):6507-6520. https://doi.org/10.3390/ijms13056507
Chicago/Turabian StyleXu, Yulan, Yuemin Zhang, Yunfei Li, Genqian Li, Daiyi Liu, Minchong Zhao, and Nianhui Cai. 2012. "Growth Promotion of Yunnan Pine Early Seedlings in Response to Foliar Application of IAA and IBA" International Journal of Molecular Sciences 13, no. 5: 6507-6520. https://doi.org/10.3390/ijms13056507




