Protective Effect of Caffeic Acid on Paclitaxel Induced Anti-Proliferation and Apoptosis of Lung Cancer Cells Involves NF-κB Pathway
Abstract
:1. Introduction
2. Results and Discussion
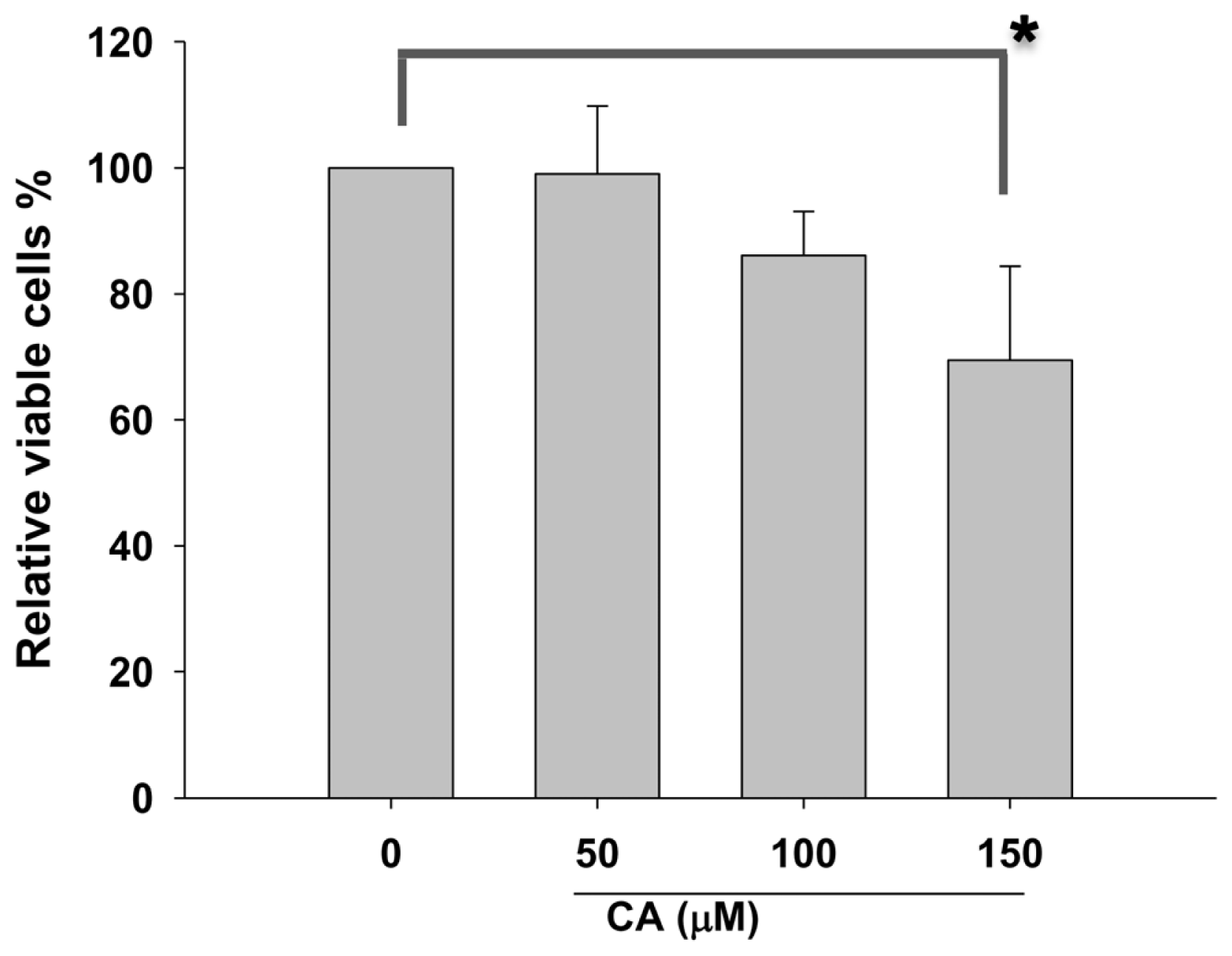
2.1. Effect of CA on Proliferation of Lung Cancer Cells
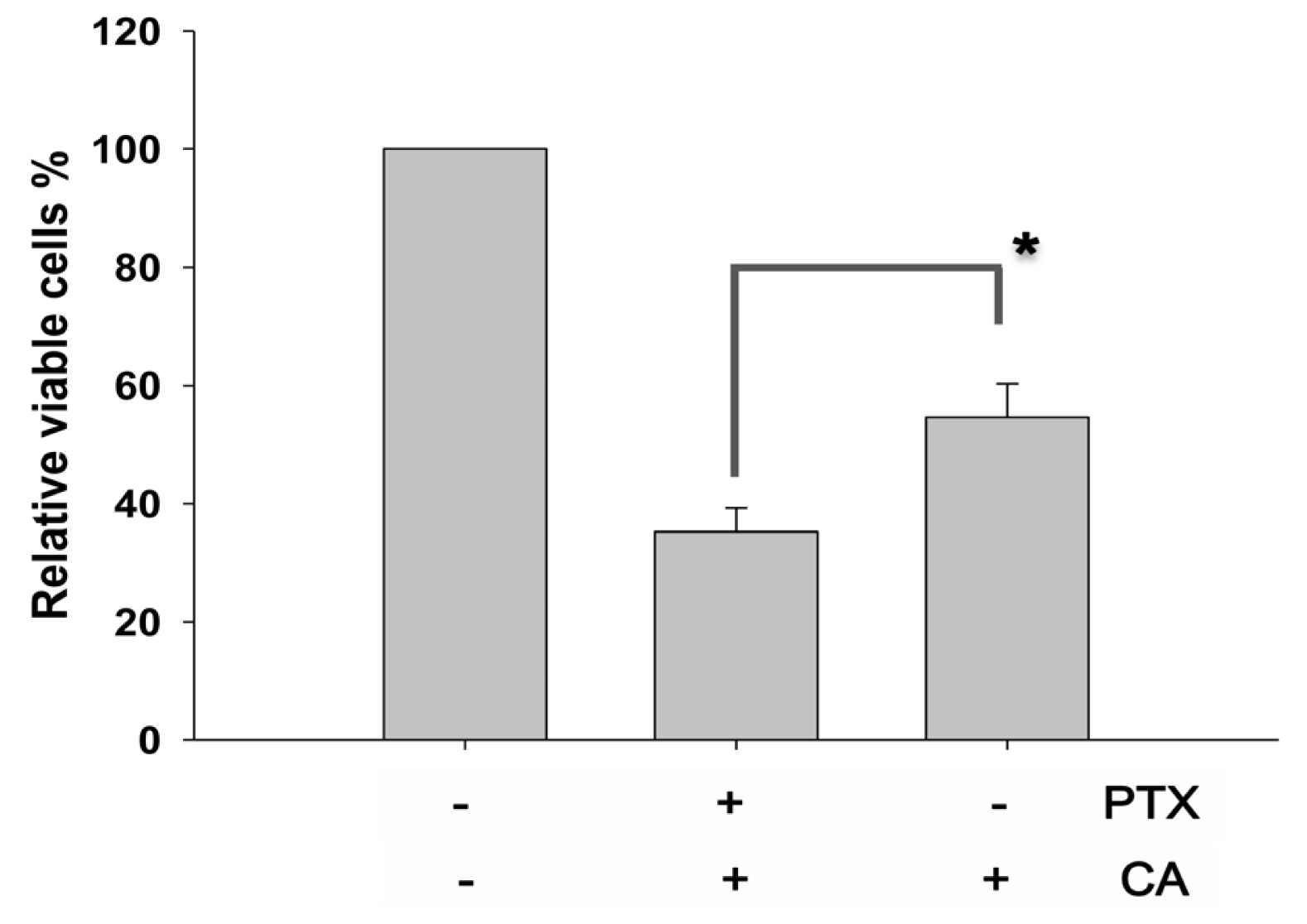
2.2. CA Rescues the PTX-Induced Anti-Proliferation in NSCLC Cells
2.3. CA Reduces the Activation of Pro-Apoptotic in NSCLC Cells
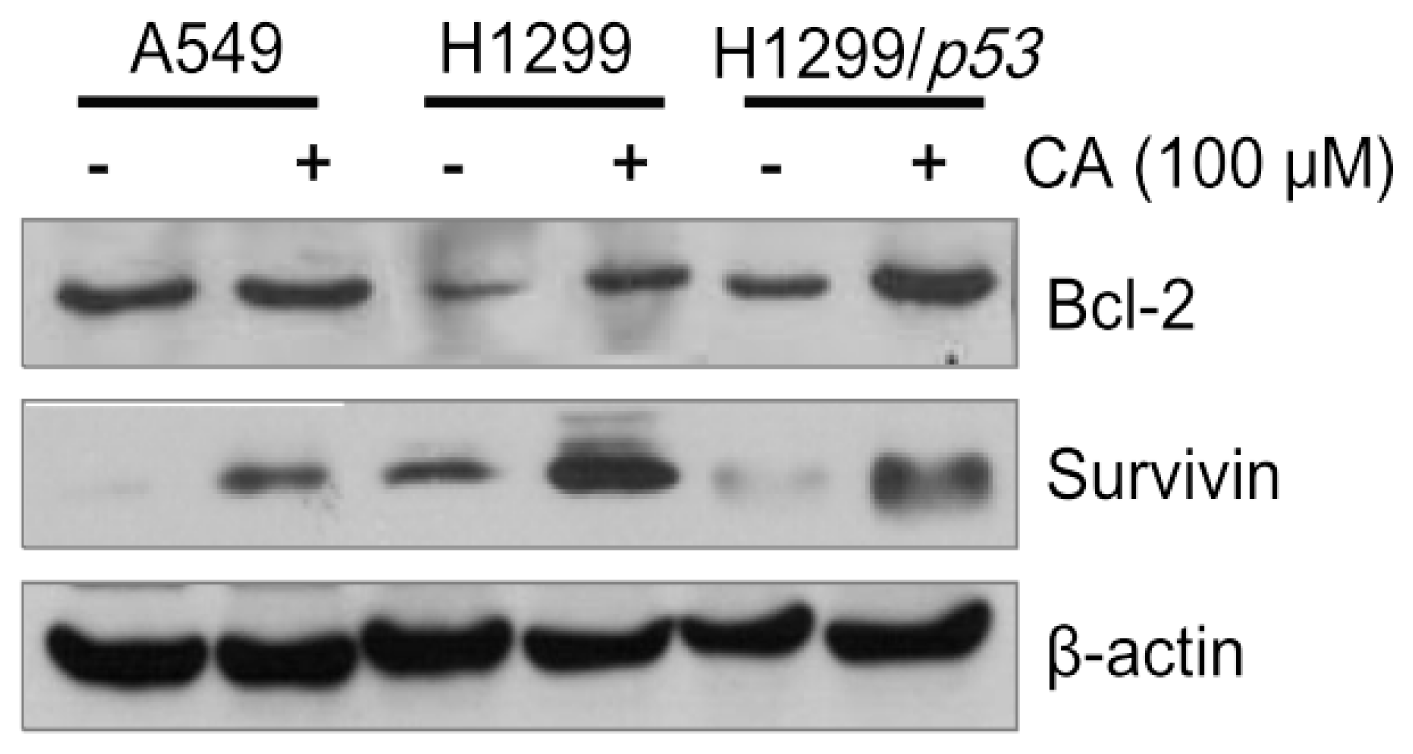
2.4. CA Up-Regulates the Protein Levels of Bcl-2 and Survivin
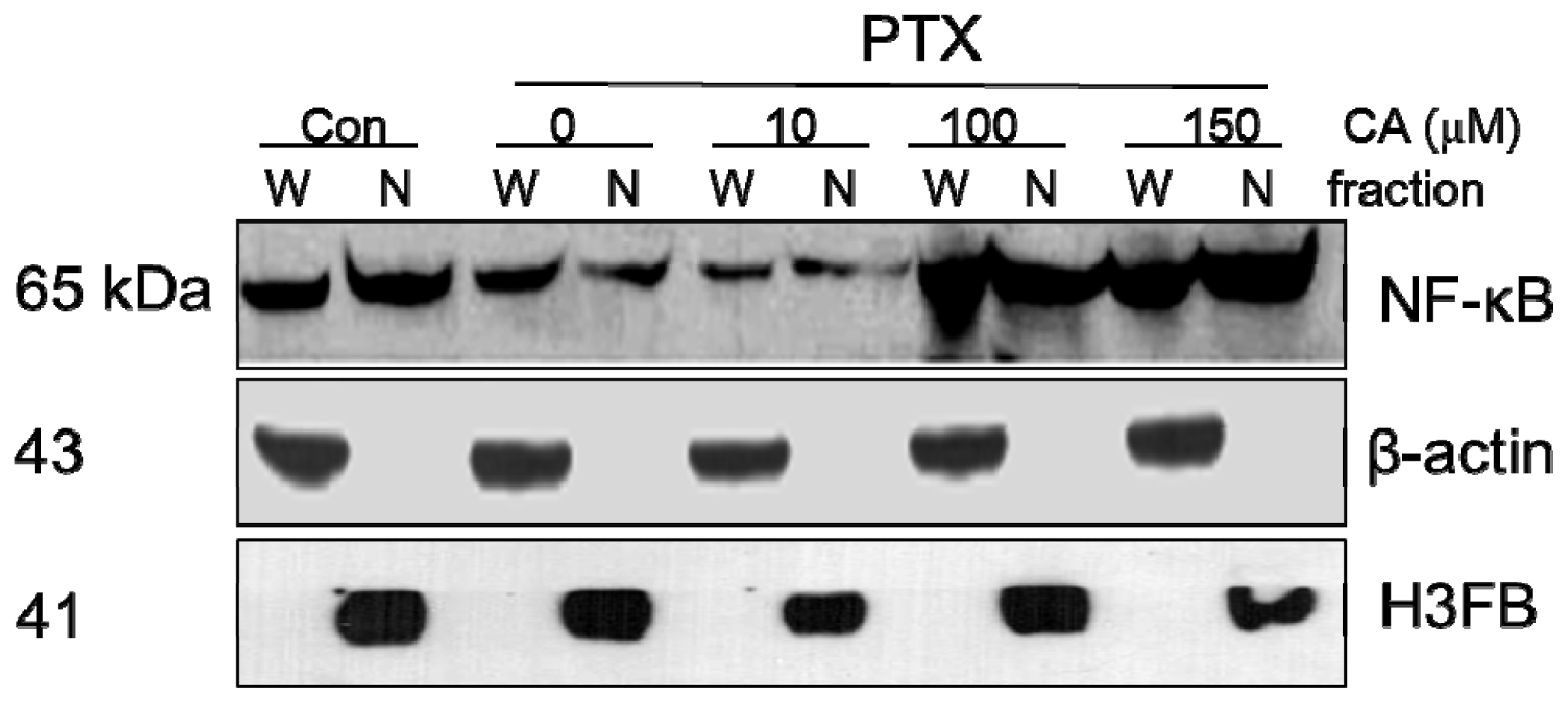
2.5. CA Induces Up-Regulation and Activation of NF-κB
3. Experimental Section
3.1. Cell Cultures
3.2. Reagents and Antibodies
3.3. Growth Proliferation Test
3.4. Western Blot Analysis
3.5. Preparation of Cytosolic and Nuclear Extracts
3.6. Statistical Analysis
4. Conclusions
Acknowledgements
References
- Chiu, C.C.; Chen, J.Y.; Lin, K.L.; Huang, C.J.; Lee, J.C.; Chen, B.H.; Chen, W.Y.; Lo, Y.H.; Chen, Y.L.; Tseng, C.H.; et al. p38 MAPK and NF-κB pathways are involved in naphtho[1,2-b] furan-4,5-dione induced anti-proliferation and apoptosis of human hepatoma cells. Cancer Lett 2010, 295, 92–99. [Google Scholar]
- Li, S.; Bao, P.; Li, Z.; Ouyang, H.; Wu, C.; Qian, G. Inhibition of proliferation and apoptosis induced by a Na+/H+ exchanger-1 (NHE-1) antisense gene on drug-resistant human small cell lung cancer cells. Oncol. Rep 2009, 21, 1243–1249. [Google Scholar]
- Tabuchi, Y.; Matsuoka, J.; Gunduz, M.; Imada, T.; Ono, R.; Ito, M.; Motoki, T.; Yamatsuji, T.; Shirakawa, Y.; Takaoka, M.; et al. Resistance to paclitaxel therapy is related with Bcl-2 expression through an estrogen receptor mediated pathway in breast cancer. Int. J. Oncol 2009, 34, 313–319. [Google Scholar]
- Hsu, F.L.; Chen, Y.C.; Cheng, J.T. Caffeic acid as active principle from the fruit of Xanthium strumarium to lower plasma glucose in diabetic rats. Planta Med 2000, 66, 228–230. [Google Scholar]
- Giovannini, L.; Migliori, M.; Filippi, C.; Origlia, N.; Panichi, V.; Falchi, M.; Bertelli, A.A.; Bertelli, A. Inhibitory activity of the white wine compounds, tyrosol and caffeic acid, on lipopolysaccharide-induced tumor necrosis factor-alpha release in human peripheral blood mononuclear cells. Int. J. Tissue React 2002, 24, 53–56. [Google Scholar]
- Simonetti, P.; Gardana, C.; Pietta, P. Caffeic acid as biomarker of red wine intake. Methods Enzymol 2001, 335, 122–130. [Google Scholar]
- Almeida, A.A.; Farah, A.; Silva, D.A.; Nunan, E.A.; Gloria, M.B. Antibacterial activity of coffee extracts and selected coffee chemical compounds against enterobacteria. J. Agric. Food Chem 2006, 54, 8738–8743. [Google Scholar]
- Chung, M.J.; Walker, P.A.; Hogstrand, C. Dietary phenolic antioxidants, caffeic acid and Trolox, protect rainbow trout gill cells from nitric oxide-induced apoptosis. Aquat. Toxicol 2006, 80, 321–328. [Google Scholar]
- Ahn, C.H.; Choi, W.C.; Kong, J.Y. Chemosensitizing activity of caffeic acid in multidrug-resistant MCF-7/Dox human breast carcinoma cells. Anticancer Res 1997, 17, 1913–1917. [Google Scholar]
- Chang, W.C.; Hsieh, C.H.; Hsiao, M.W.; Lin, W.C.; Hung, Y.C.; Ye, J.C. Caffeic acid induces apoptosis in human cervical cancer cells through the mitochondrial pathway. Taiwan J. Obstet. Gynecol 2010, 49, 419–424. [Google Scholar]
- Onori, P.; DeMorrow, S.; Gaudio, E.; Franchitto, A.; Mancinelli, R.; Venter, J.; Kopriva, S.; Ueno, Y.; Alvaro, D.; Savage, J.; et al. Caffeic acid phenethyl ester decreases cholangiocarcinoma growth by inhibition of NF-κB and induction of apoptosis. Int. J. Cancer 2009, 125, 565–576. [Google Scholar]
- Tian, X.F.; Pu, X.P. Caffeic acid (CA) protects cerebellar granule neurons (CGNs) from apoptosis induced by neurotoxin 1-methyl-4-phenylpyridnium (MPP+). Beijing Da Xue Xue Bao 2004, 36, 27–30. [Google Scholar]
- Khanduja, K.L.; Avti, P.K.; Kumar, S.; Mittal, N.; Sohi, K.K.; Pathak, C.M. Anti-apoptotic activity of caffeic acid, ellagic acid and ferulic acid in normal human peripheral blood mononuclear cells: A Bcl-2 independent mechanism. Biochim. Biophys. Acta 2006, 1760, 283–289. [Google Scholar]
- Lima, C.F.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Phenolic compounds protect HepG2 cells from oxidative damage: Relevance of glutathione levels. Life Sci 2006, 79, 2056–2068. [Google Scholar] [Green Version]
- Kang, K.A.; Lee, K.H.; Zhang, R.; Piao, M.; Chae, S.; Kim, K.N.; Jeon, Y.J.; Park, D.B.; You, H.J.; Kim, J.S.; et al. Caffeic acid protects hydrogen peroxide induced cell damage in WI-38 human lung fibroblast cells. Biol. Pharm. Bull 2006, 29, 1820–1824. [Google Scholar]
- Mirzoeva, O.K.; Calder, P.C. The effect of propolis and its components on eicosanoid production during the inflammatory response. Prostaglandins Leukot. Essent. Fatty Acids 1996, 55, 441–449. [Google Scholar]
- Lutz, U.; Lugli, S.; Bitsch, A.; Schlatter, J.; Lutz, W.K. Dose response for the stimulation of cell division by caffeic acid in forestomach and kidney of the male F344 rat. Fundam. Appl. Toxicol 1997, 39, 131–137. [Google Scholar]
- Hagiwara, A.; Hirose, M.; Takahashi, S.; Ogawa, K.; Shirai, T.; Ito, N. Forestomach and kidney carcinogenicity of caffeic acid in F344 rats and C57BL/6N x C3H/HeN F1 mice. Cancer Res 1991, 51, 5655–5660. [Google Scholar]
- Nardini, M.; Leonardi, F.; Scaccini, C.; Virgili, F. Modulation of ceramide-induced NF-κB binding activity and apoptotic response by caffeic acid in U937 cells: Comparison with other antioxidants. Free Radic. Biol. Med 2001, 30, 722–733. [Google Scholar]
- Jung, J.E.; Kim, H.S.; Lee, C.S.; Park, D.H.; Kim, Y.N.; Lee, M.J.; Lee, J.W.; Park, J.W.; Kim, M.S.; Ye, S.K.; et al. Caffeic acid and its synthetic derivative CADPE suppress tumor angiogenesis by blocking STAT3-mediated VEGF expression in human renal carcinoma cells. Carcinogenesis 2007, 28, 1780–1787. [Google Scholar]
- Cohen, C.; Lohmann, C.M.; Cotsonis, G.; Lawson, D.; Santoianni, R. Survivin expression in ovarian carcinoma: Correlation with apoptotic markers and prognosis. Mod. Pathol 2003, 16, 574–583. [Google Scholar]
- Zaffaroni, N.; Pennati, M.; Colella, G.; Perego, P.; Supino, R.; Gatti, L.; Pilotti, S.; Zunino, F.; Daidone, M.G. Expression of the anti-apoptotic gene survivin correlates with taxol resistance in human ovarian cancer. Cell. Mol. Life Sci 2002, 59, 1406–1412. [Google Scholar]
- Ramnath, V.; Rekha, P.S.; Kuttan, G.; Kuttan, R. Regulation of caspase-3 and Bcl-2 expression in Dalton’s lymphoma ascites cells by Abrin. Evid.-Based Complement. Alternat. Med 2009, 6, 233–238. [Google Scholar]
- Raviv, Z.; Zilberberg, A.; Cohen, S.; Reischer-Pelech, D.; Horrix, C.; Berger, M.R.; Rosin-Arbesfeld, R.; Flescher, E. Methyl jasmonate down-regulates survivin expression and sensitizes colon carcinoma cells towards TRAIL-induced cytotoxicity. Br. J. Pharmacol 2011, 164, 1433–1444. [Google Scholar]
- Ghosh, S.; Karin, M. Missing pieces in the NF-κB puzzle. Cell 2002, 109, S81–S96. [Google Scholar]
- Heckman, C.A.; Mehew, J.W.; Boxer, L.M. NF-κB activates Bcl-2 expression in t(14;18) lymphoma cells. Oncogene 2002, 21, 3898–3908. [Google Scholar]
- Tang, X.; Liu, D.; Shishodia, S.; Ozburn, N.; Behrens, C.; Lee, J.J.; Hong, W.K.; Aggarwal, B.B.; Wistuba, I.I. Nuclear factorκB (NFκB) is frequently expressed in lung cancer and preneoplastic lesions. Cancer 2006, 107, 2637–2646. [Google Scholar]
- Weldon, C.B.; Burow, M.E.; Rolfe, K.W.; Clayton, J.L.; Jaffe, B.M.; Beckman, B.S. NF-κB-mediated chemoresistance in breast cancer cells. Surgery 2001, 130, 143–150. [Google Scholar]
- Karin, M.; Lin, A. NF-κB at the crossroads of life and death. Nat. Immunol 2002, 3, 221–227. [Google Scholar]
- Ding, R.K.; Ding, W.; Wang, C.K.; Lu, H.; He, Z.M. The effect of survivin on multidrug resistance by epidermal growth factor receptor 2 via NF-κB activating in breast cancer cells. Cell Biol. Int 2008, 32. [Google Scholar] [CrossRef]
- Zhang, X.H.; Su, L.D.; Lu, Q.H.; Liang, Y.; Zhao, X.Y. Relationship between drug resistance and the expression of NF-κB induced in leukemic cells. Zhejiang Da Xue Xue Bao Yi Xue Ban 2004, 33, 421–426. [Google Scholar]
- Salvatore, C.; Camarda, G.; Maggi, C.A.; Goso, C.; Manzini, S.; Binaschi, M. NF-κB activation contributes to anthracycline resistance pathway in human ovarian carcinoma cell line A2780. Int. J. Oncol 2005, 27, 799–806. [Google Scholar]
- Chiu, C.C.; Liu, P.L.; Huang, K.J.; Wang, H.M.; Chang, K.F.; Chou, C.K.; Chang, F.R.; Chong, I.W.; Fang, K.; Chen, J.S.; et al. Goniothalamin inhibits growth of human lung cancer cells through DNA damage, apoptosis, and reduced migration ability. J. Agric. Food Chem 2011, 59, 4288–4293. [Google Scholar]


© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, C.-L.; Chen, R.-F.; Chen, J.Y.-F.; Chu, Y.-C.; Wang, H.-M.; Chou, H.-L.; Chang, W.-C.; Fong, Y.; Chang, W.-T.; Wu, C.-Y.; et al. Protective Effect of Caffeic Acid on Paclitaxel Induced Anti-Proliferation and Apoptosis of Lung Cancer Cells Involves NF-κB Pathway. Int. J. Mol. Sci. 2012, 13, 6236-6245. https://doi.org/10.3390/ijms13056236
Lin C-L, Chen R-F, Chen JY-F, Chu Y-C, Wang H-M, Chou H-L, Chang W-C, Fong Y, Chang W-T, Wu C-Y, et al. Protective Effect of Caffeic Acid on Paclitaxel Induced Anti-Proliferation and Apoptosis of Lung Cancer Cells Involves NF-κB Pathway. International Journal of Molecular Sciences. 2012; 13(5):6236-6245. https://doi.org/10.3390/ijms13056236
Chicago/Turabian StyleLin, Chien-Liang, Ruei-Feng Chen, Jeff Yi-Fu Chen, Ying-Chieh Chu, Hui-Min Wang, Han-Lin Chou, Wei-Chiao Chang, Yao Fong, Wen-Tsan Chang, Chang-Yi Wu, and et al. 2012. "Protective Effect of Caffeic Acid on Paclitaxel Induced Anti-Proliferation and Apoptosis of Lung Cancer Cells Involves NF-κB Pathway" International Journal of Molecular Sciences 13, no. 5: 6236-6245. https://doi.org/10.3390/ijms13056236





