In Vitro Inhibition of Histamine Release Behavior of Cetirizine Intercalated into Zn/Al- and Mg/Al-Layered Double Hydroxides
Abstract
:1. Introduction
2. Results and Discussion
2.1. Powder X-Ray Diffraction
2.2. Molecular Structure and Spatial Orientation of the Intercalated Cetirizine
2.3. Fourier Transforms Infrared Spectroscopy
2.4. Elemental Analysis
2.5. Thermal Study
2.6. Surface Characterization
2.7. Release Behavior of Cetirizine
2.8. Release Kinetics of Cetirizine from CTZAN and CTMAN Nanocomposites
2.9. Cytotoxicity of CTZAN and CTMAN Nanocomposites toward Human Chang Liver Cells Line
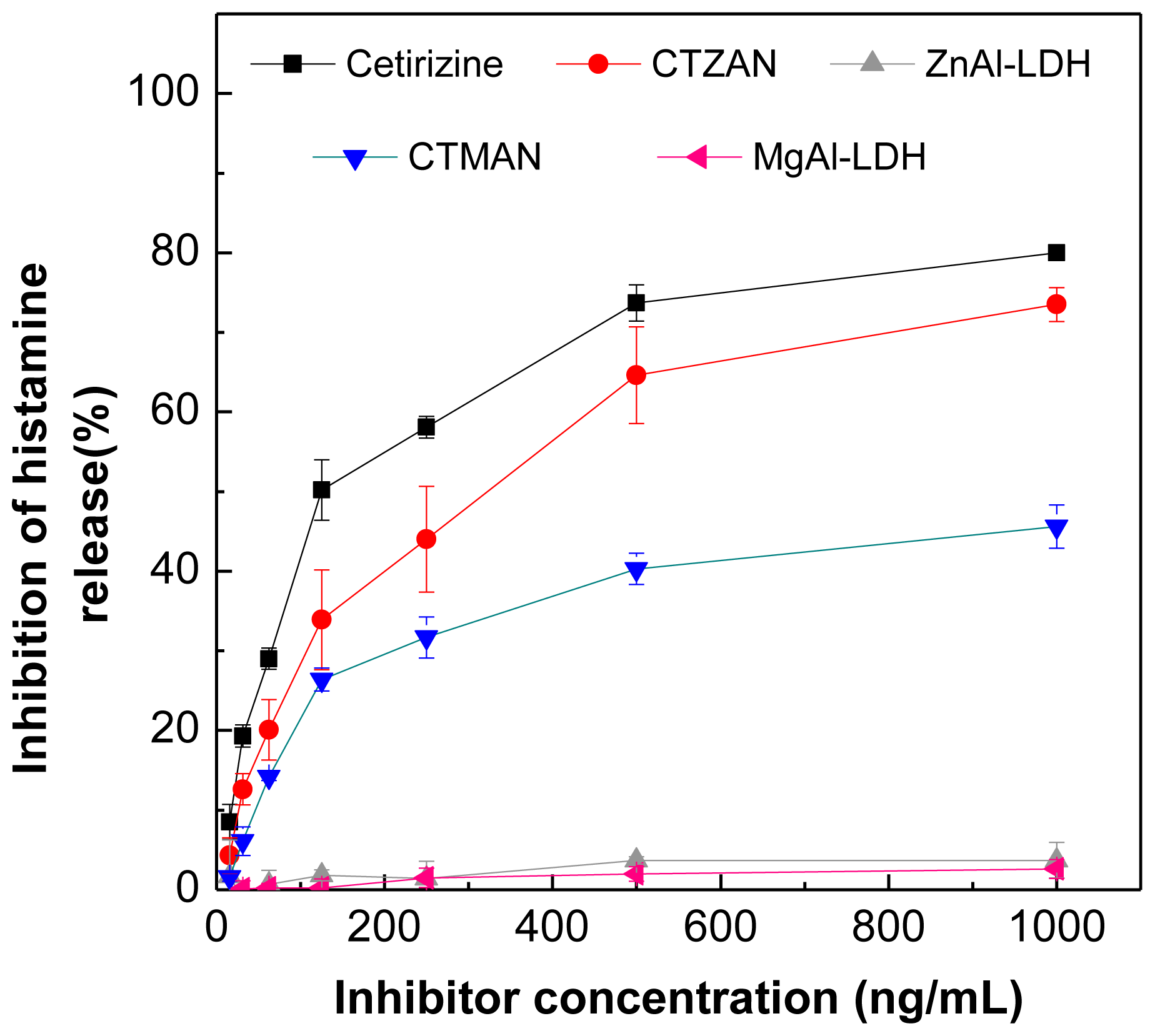
2.10. Effect of Cetirizine, CTZAN, CTMAN, Zn/Al-NO3 and Mg/Al-NO3 on Histamine Release from RBL2H3 Cells
3. Experimental Section
3.1. Materials
3.2. Synthesis of the Cetirizine-MgAl and Cetirizine ZnAl Nanocomposite by the Ion-Exchange Method
3.3. Characterization
3.4. Release Study of Cetirizine from CTZAN and CTMAN Nanocomposites
3.5. Cytotoxicity Assay
3.6. Cell Culture Conditions and Anti IgE-Induced Histamine Release
4. Conclusions
Acknowledgments
References
- Kumar, M. Nano and microparticles as controlled drug delivery devices. J. Pharm. Pharm. Sci 2000, 3, 234–258. [Google Scholar]
- Rives, V. Layered Double Hydroxides: Present and Future; Nova Science Publishers: New York, NY, USA, 2001. [Google Scholar]
- Newman, S.P.; Jones, W. Synthesis, characterization and applications of layered double hydroxides containing organic guests. New J. Chem 1998, 22, 105–115. [Google Scholar]
- Oh, J.-M.; Park, M.; Kim, S.-T.; Jung, J.-Y.; Kang, Y.-G.; Choy, J.-H. Efficient delivery of anticancer drug MTX through MTX-LDH nanohybrid system. J. Phys. Chem. Solids 2006, 67, 1024–1027. [Google Scholar]
- Wang, Z.; Wang, E.; Gao, L.; Xu, L. Synthesis and properties of Mg2Al layered double hydroxides containing 5-fluorouracil. J. Solid State Chem 2005, 178, 736–741. [Google Scholar]
- Qin, L.; Xue, M.; Wang, W.; Zhu, R.; Wang, S.; Sun, J.; Zhang, R.; Sun, X. The in vitro and in vivo anti-tumor effect of layered double hydroxides nanoparticles as delivery for podophyllotoxin. Int. J. Pharm 2010, 388, 223–230. [Google Scholar]
- Li, B.; He, J.; Evans, G.D.; Duan, X. Inorganic layered double hydroxides as a drug delivery system-intercalation and in vitro release of fenbufen. Appl. Clay Sci 2004, 27, 199–207. [Google Scholar]
- Ambrogi, V.; Fardella, G.; Grandolini, G.; Perioli, L.; Tiralti, M.C. Intercalation compounds of hydrotalcite-like anionic clays with anti-inflammatory agents, II: Uptake of Diclofenac for a controlled release formulation. AAPS PharmSciTech 2002, 3, 77–82. [Google Scholar]
- Ambrogi, V.; Fardella, G.; Grandolini, G.; Perioli, L. Intercalation compounds of hydrotalcite-like anionic clays with antiinflammatory agents-I. Intercalation and in vitro release of ibuprofen. Int. J. Pharm 2001, 220, 23–32. [Google Scholar]
- Tyner, K.M.; Schiffman, S.R.; Giannelis, E.P. Nanobiohybrids as delivery vehicles for camptothecin. J. Control. Release 2004, 95, 501–514. [Google Scholar]
- Zhang, H.; Zou, K.; Guo, S.; Duan, X. Nanostructural drug-inorganic clay composites: Structure, thermal property and in vitro release of captopril-intercalated MgAl-layered double hydroxides. J. Solid State Chem 2006, 179, 1792–1801. [Google Scholar]
- Al Ali, S.H.H.; Al-Qubaisi, M.; Hussein, M.Z.; Ismail, M.; Zainal, Z.; Hakim, M.N. Controlled release and angiotensin-converting enzyme inhibition properties of an antihypertensive drug based on a perindopril erbumine-layered double hydroxide nanocomposite. Int. J. Nanomedicine 2012, 7, 2129–2141. [Google Scholar]
- Xia, S.-J.; Ni, Z.-M.; Xu, Q.; Hu, B.-X.; Hu, J. Layered double hydroxides as supports for intercalation and sustained release of antihypertensive drugs. J. Solid State Chem 2008, 181, 2610–2619. [Google Scholar]
- Liu, C.; Hou, W.; Li, L.; Li, Y.; Liu, S. Synthesis and characterization of 5-fluorocytosine intercalated Zn-Al layered double hydroxide. J. Solid State Chem 2008, 181, 1792–1797. [Google Scholar]
- Trikeriotis, M.; Ghanotakis, D.F. Intercalation of hydrophilic and hydrophobic antibiotics in layered double hydroxides. Int. J. Pharm 2007, 332, 176–184. [Google Scholar]
- Brown, V.; Ennis, M. Flow-cytometric analysis of basophil activation: Inhibition by histamine at conventional and homeopathic concentrations. Inflamm. Res 2001, 50, 47–48. [Google Scholar]
- Chao, Y.F.; Chen, P.C.; Wang, S.L. Adsorption of 2,4-D on Mg/Al-NO3 layered double hydroxides with varying layer charge density. Appl. Clay Sci 2008, 40, 193–200. [Google Scholar]
- Wei, M.; Yuan, Q.; Evans, D.G.; Wang, Z.; Duan, X. Layered solids as a “molecular container” for pharmaceutical agents: L-tyrosine-intercalated layered double hydroxides. J. Mater. Chem 2005, 15, 1197–1203. [Google Scholar]
- Testa, B.; Pagliara, A.; Carrupt, P.A. The molecular behaviour of cetirizine. Clin. Exp. Allergy 1997, 27, 13–18. [Google Scholar]
- Cavani, F.; Trifiro, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal Today 1991, 11, 173–301. [Google Scholar]
- Bora, M.M. Adsorption of pigment from annatto seed utilizing Fish Scale as biosorbent. J. Chem. Pharm. Res 2010, 2, 75–83. [Google Scholar]
- Oriakhi, C.O.; Farr, I.V.; Lerner, M.M. Incorporation of poly (acrylic acid), poly (vinylsulfonate) and poly (styrenesulfonate) within layered double hydroxides. J. Mater. Chem 1996, 6, 103–107. [Google Scholar]
- Hussein, M.Z.; Hashim, N.; Yahaya, A.H.; Zainal, Z. Synthesis and characterization of [4-(2,4-dichlorophenoxybutyrate)-zinc layered hydroxide] nanohybrid. Solid State Sci 2010, 12, 770–775. [Google Scholar]
- Kenawi, I.M.; Barsoum, B.N.; Youssef, M.A. Drug-drug interaction between diclofenac, cetirizine and ranitidine. J. Pharm. Biomed. Anal 2005, 37, 655–661. [Google Scholar]
- Tyner, K.M.; Schiffman, S.R.; Giannelis, E.P. Nanobiohybrids as delivery vehicles for camptothecin. J. Control. Release 2004, 95, 501–514. [Google Scholar]
- Dong, L.; Yan, L.; Hou, W.-G.; Liu, S.-J. Synthesis and release behavior of composites of camptothecin and layered double hydroxide. J. Solid State Chem 2010, 183, 1811–1816. [Google Scholar]
- Ho, Y.S.; Ofomaja, A.E. Pseudo-second-order model for lead ion sorption from aqueous solutions onto palm kernel fiber. J. Hazard. Mater 2006, 129, 137–142. [Google Scholar]
- Kong, X.; Shi, S.; Han, J.; Zhu, F.; Wei, M.; Duan, X. Preparation of Glycy-l-Tyrosine intercalated layered double hydroxide film and its in vitro release behavior. Chem. Eng. J 2010, 157, 598–604. [Google Scholar]
- Wood, J.D. Enteric neuroimmuno physiology and pathophysiology. Gastroenterology 2004, 127, 635–657. [Google Scholar]
- Ishiguro, S.; Nishio, A.; Miyao, N.; Morikawa, Y.; Takeno, K.; Yanagiya, I. Effects of magnesium deficiency on dermal mast cells in rats. Folia pharmacol. Jpn 1987, 89, 121–127. [Google Scholar]
- Al Ali, S.H.H.; Al-Qubaisi, M.; Hussein, M.Z.; Ismail, M.; Zainal, Z.; Hakim, M.N. Controlled-release formulation of antihistamine based on cetirizine-zinc layered hydroxide nanocomposites and its effect on histamine release from basophilic leukemia (RBL-2H3) cells. Int. J. Nanomed 2012, in press. [Google Scholar]
- Mohanambe, L.; Vasudevan, S. Anionic clays containing anti-inflammatory drug molecules: Comparison of molecular dynamics simulation and measurements. J. Phys. Chem. B 2005, 109, 15651–15658. [Google Scholar]
- Ribeiro, C.; Arizaga, G.G.C.; Wypych, F.; Sierakowski, M.R. Nanocomposites coated with xyloglucan for drug delivery: In vitro studies. Int. J. Pharm 2009, 367, 204–210. [Google Scholar]
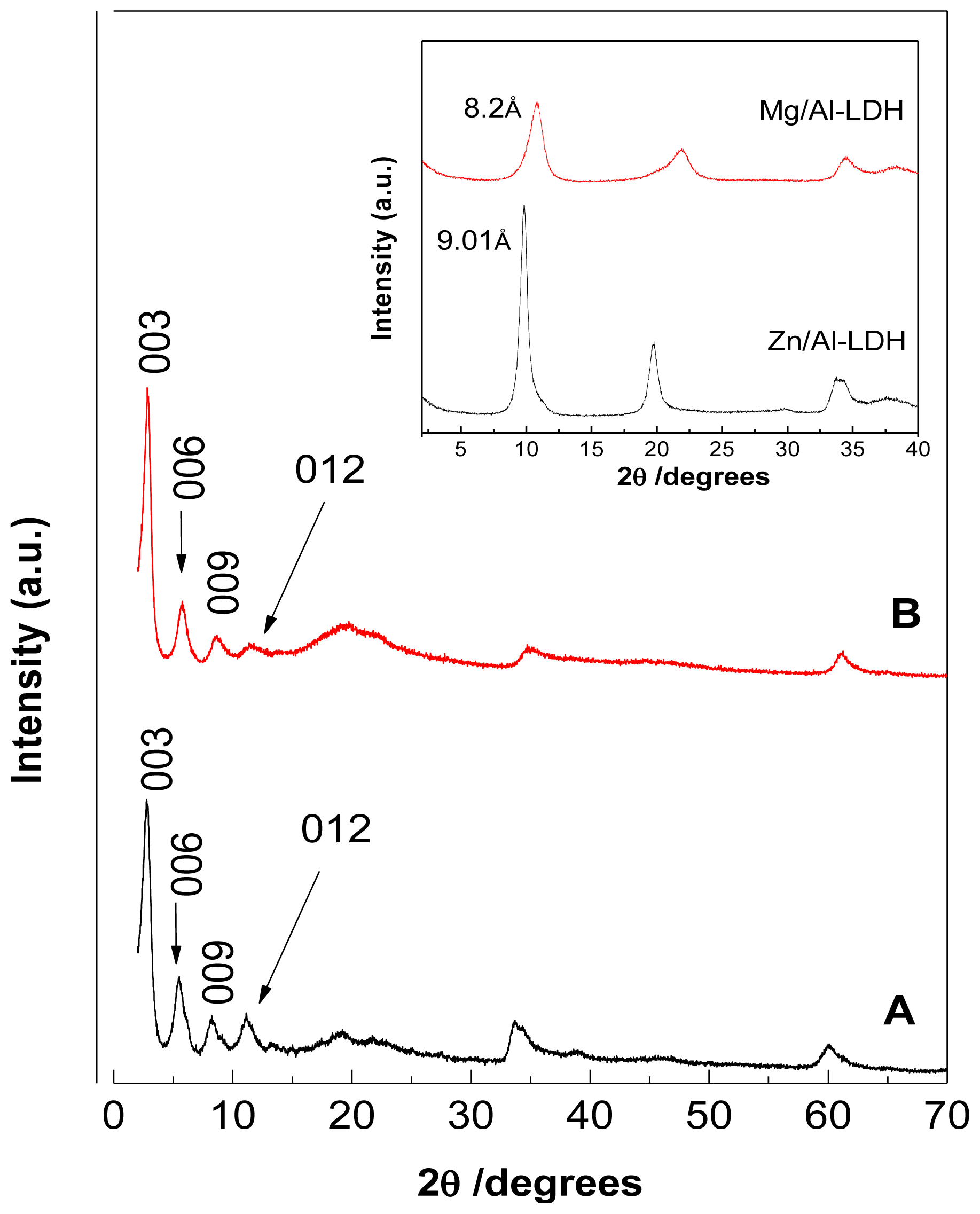


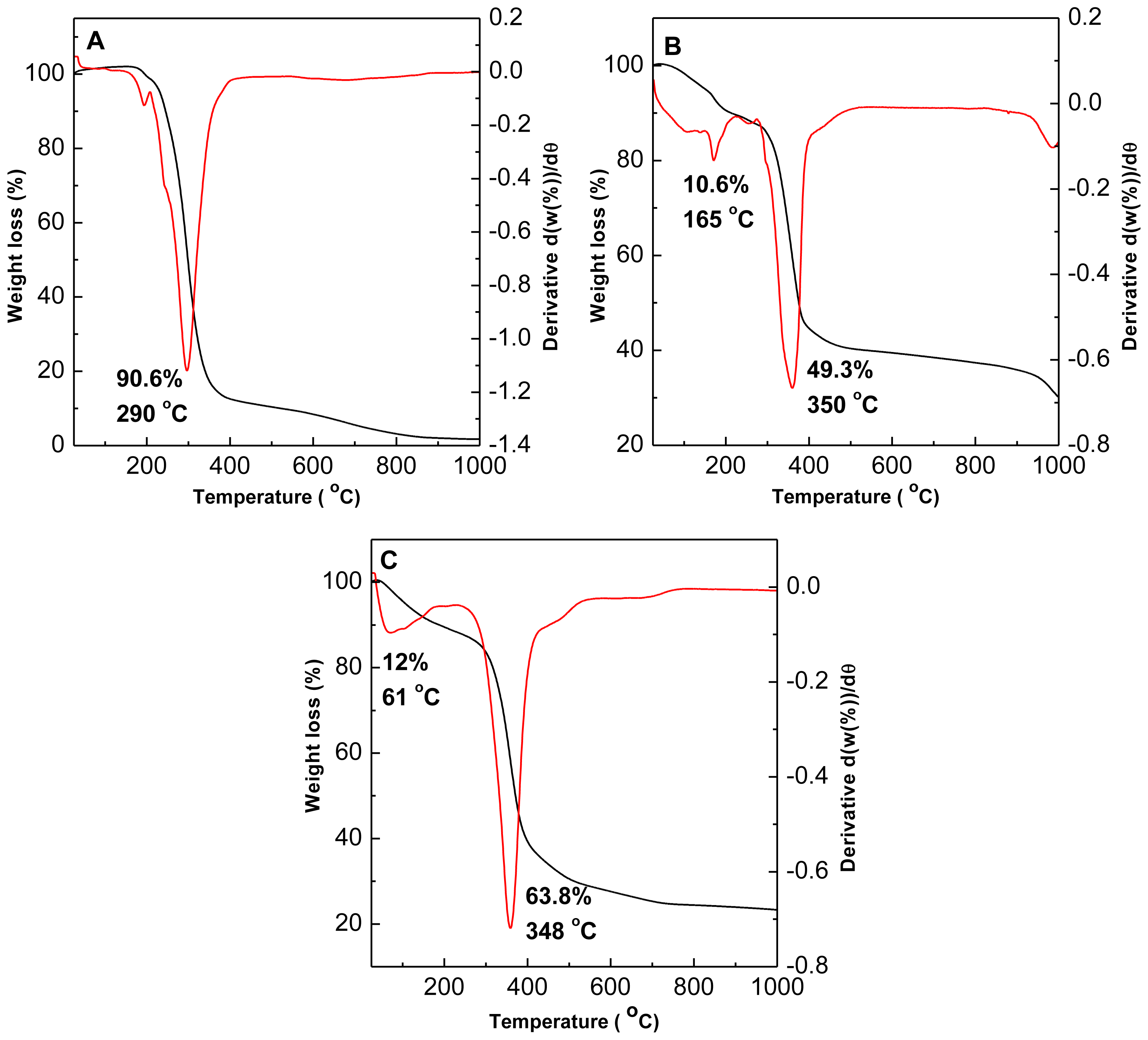

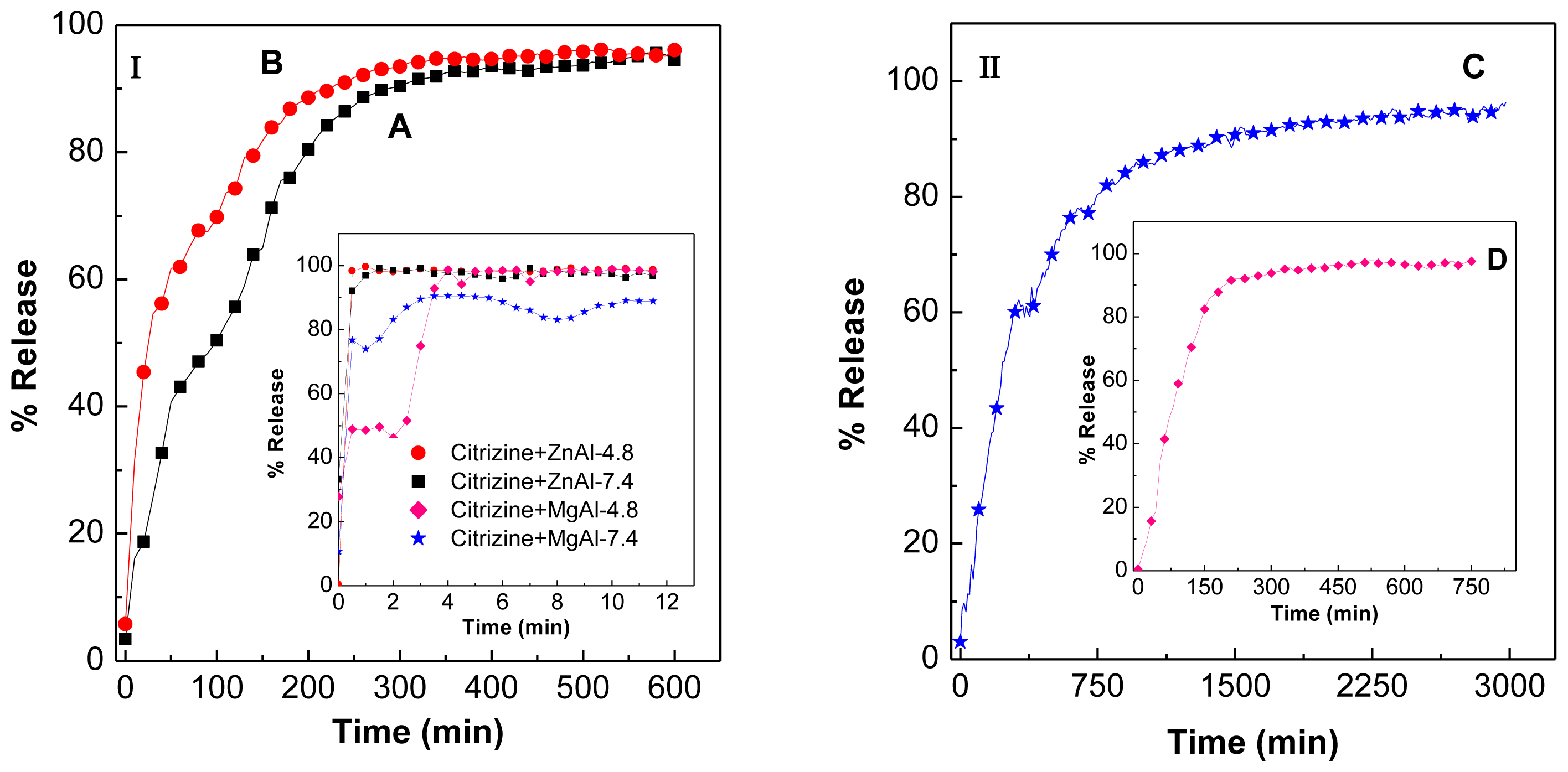



| Samples | d003 (Å) | d006 (Å) | d009 (Å) | d012 (Å) | d110 (Å) | Average d value | a (Å) | c (Å) | M2+/Al3+ | x |
|---|---|---|---|---|---|---|---|---|---|---|
| CTZAN | 31.90 | 15.93 | 10.64 | 7.88 | 1.51 | 31.8 | 3.02 | 95.5 | 3.3 | 0.23 |
| CTMAN | 31.15 | 15.33 | 10.15 | 7.66 | 1.51 | 30.8 | 3.02 | 92.4 | 2.03 | 0.33 |
| Assignments | Cetirizine | CTZAN | CTMAN |
|---|---|---|---|
| v (O–H) | 3432 for O–H in carboxylic group | 3445 in the layer; H2O | 3442 |
| v (CH2) | 2984–2949 | 2958–2817 | 2958–2819 |
| v (COOH) | 1740 | - | - |
| v (ϕ. Para- subst.) | 1601 | 1601 | 1589 |
| v (ϕ. mono- subst.) | 1496, 1077 and 758 | 1487 | 1488 |
| v (C–Cl) | 1457 | 1452 | 1453 |
| v (C–O) in carboxylic group | 1435, 1383, 1356 and 1319 | - | - |
| 2 adj. ϕ | 846 and 809 | 853 and 805 | 853 and 805 |
| v (CH mono- subst.) | 758 | 758 | 758 |
| M-O | - | 428 | 446 |
| vas (COO−) | - | 1601 | 1589 |
| vs (COO−) | - | 1408 | 1408 |
| Samples | M2+% a | Al3+% a | C% b | N% b | C/N | Drug% b | Empirical formula |
|---|---|---|---|---|---|---|---|
| CTZAN | 21.5 | 2.7 | 37.2 | 4.2 | 8.8 | 57.2 | [Zn0.77Al0.23(OH)2](CT)0.22(NO3−)0.01.1.16H2O |
| CTMAN | 8.03 | 4.37 | 39.4 | 4.4 | 9.04 | 60.7 | [Mg0.67Al0.33(OH)2](CT)0.33.1.4H2O |
| Samples | pH | Saturation release (%) | R2 | Pseudo-second order | |||
|---|---|---|---|---|---|---|---|
| Pseudo-first order | Pseudo-second order | Parabolic diffusion model | Rate constant k (mg/min) | t1/2 (min) | |||
| CTZAN | 7.4 | 95.6 | 0.9644 | 0.9885 | 0.8811 | 1.07 × 10−4 | 84.3 |
| CTZAN | 4.8 | 96 | 0.9190 | 0.9985 | 0.8145 | 3.24 × 10−4 | 30.4 |
| CTMAN | 7.4 | 96.3 | 0.9369 | 0.9983 | 0.7781 | 4.14 × 10−5 | 233.3 |
| CTMAN | 4.8 | 97.8 | 0.8056 | 0.9914 | 0.7106 | 1.65 × 10−4 | 56.8 |
| Concentration (ng/mL) | Inhibition (%) | ||||
|---|---|---|---|---|---|
| Cetirizine | CTZAN | CTMAN | Zn/Al-LDH | Mg/Al-LDH | |
| 1000 | 80.0 | 73.5 | 45.6 | 3.7 | 2.6 |
| 500 | 73.7 | 64.6 | 40.3 | 3.7 | 2.0 |
| 250 | 58.1 | 44.0 | 31.7 | 1.4 | 1.5 |
| 125 | 50.2 | 33.9 | 26.4 | 1.8 | 0.2 |
| 62.5 | 29.0 | 20.1 | 14.2 | 0.7 | 0.2 |
| 31.25 | 19.3 | 12.6 | 6.1 | −1.2 | 0.1 |
| 15.63 | 8.5 | 4.3 | 1.6 | 1.7 | −2.9 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hussein-Al-Ali, S.H.; Al-Qubaisi, M.; Hussein, M.Z.; Ismail, M.; Zainal, Z.; Hakim, M.N. In Vitro Inhibition of Histamine Release Behavior of Cetirizine Intercalated into Zn/Al- and Mg/Al-Layered Double Hydroxides. Int. J. Mol. Sci. 2012, 13, 5899-5916. https://doi.org/10.3390/ijms13055899
Hussein-Al-Ali SH, Al-Qubaisi M, Hussein MZ, Ismail M, Zainal Z, Hakim MN. In Vitro Inhibition of Histamine Release Behavior of Cetirizine Intercalated into Zn/Al- and Mg/Al-Layered Double Hydroxides. International Journal of Molecular Sciences. 2012; 13(5):5899-5916. https://doi.org/10.3390/ijms13055899
Chicago/Turabian StyleHussein-Al-Ali, Samer Hasan, Mothanna Al-Qubaisi, Mohd Zobir Hussein, Maznah Ismail, Zulkarnain Zainal, and Muhammad Nazrul Hakim. 2012. "In Vitro Inhibition of Histamine Release Behavior of Cetirizine Intercalated into Zn/Al- and Mg/Al-Layered Double Hydroxides" International Journal of Molecular Sciences 13, no. 5: 5899-5916. https://doi.org/10.3390/ijms13055899




