Gastroprotective Effect of Selenium on Ethanol-Induced Gastric Damage in Rats
Abstract
:1. Introduction
2. Results and Discussion
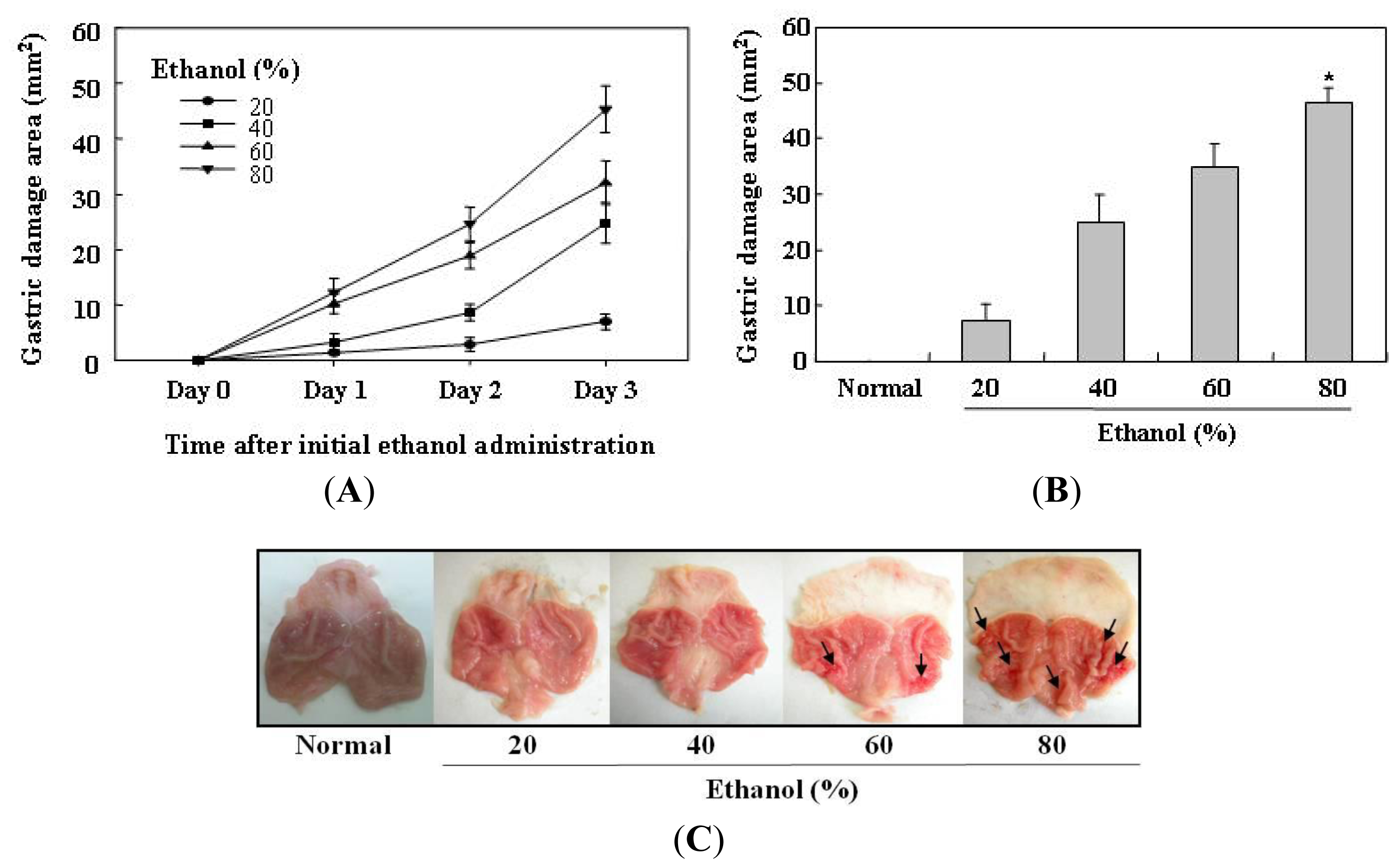
2.1. Determination of the Optimal Condition for Induction of Ethanol-Induced Gastric Mucosal Lesions in Rats
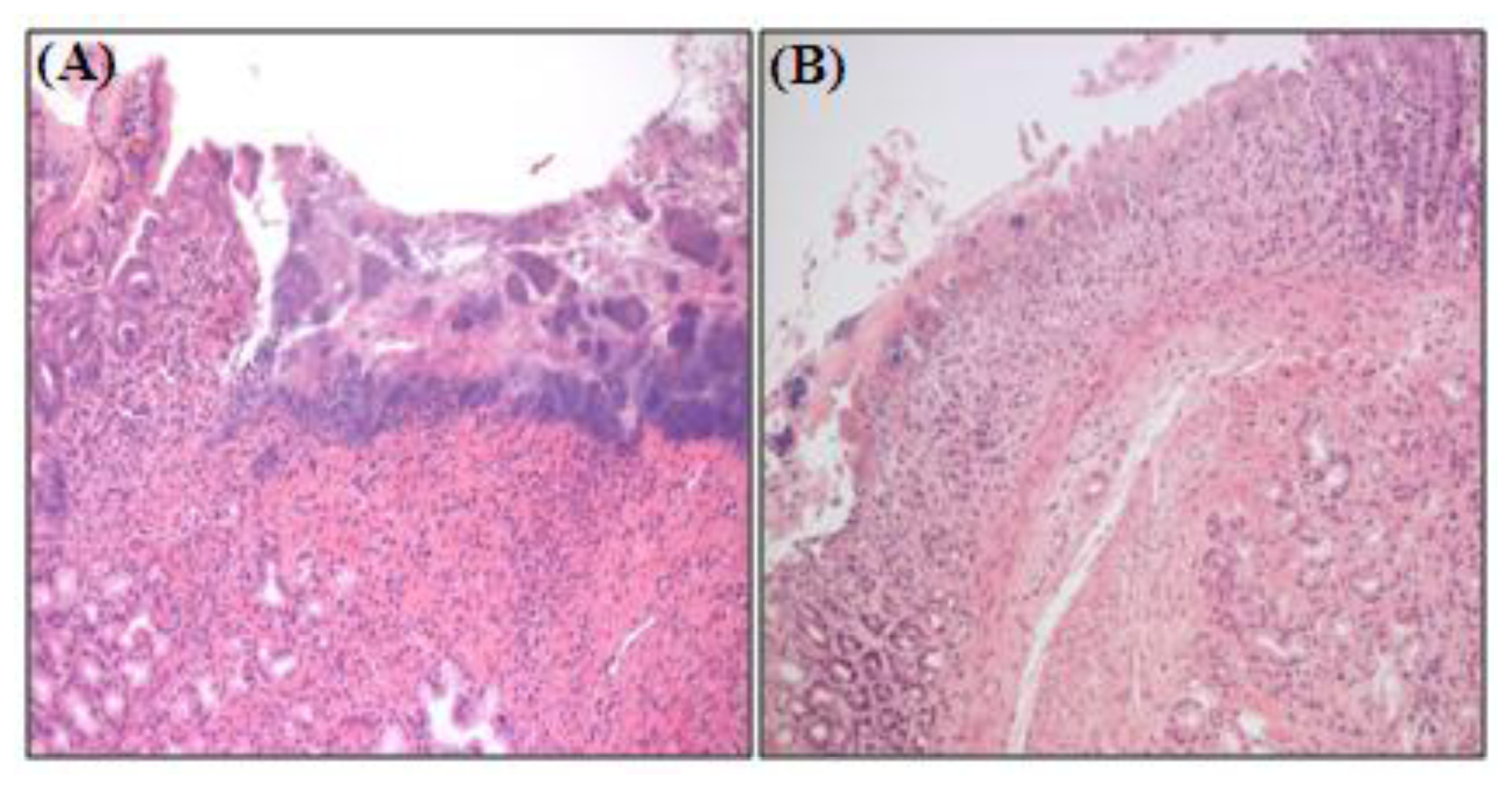
2.2. Selenium Inhibits Ethanol-Induced Gastric Mucosal Lesions in Rats
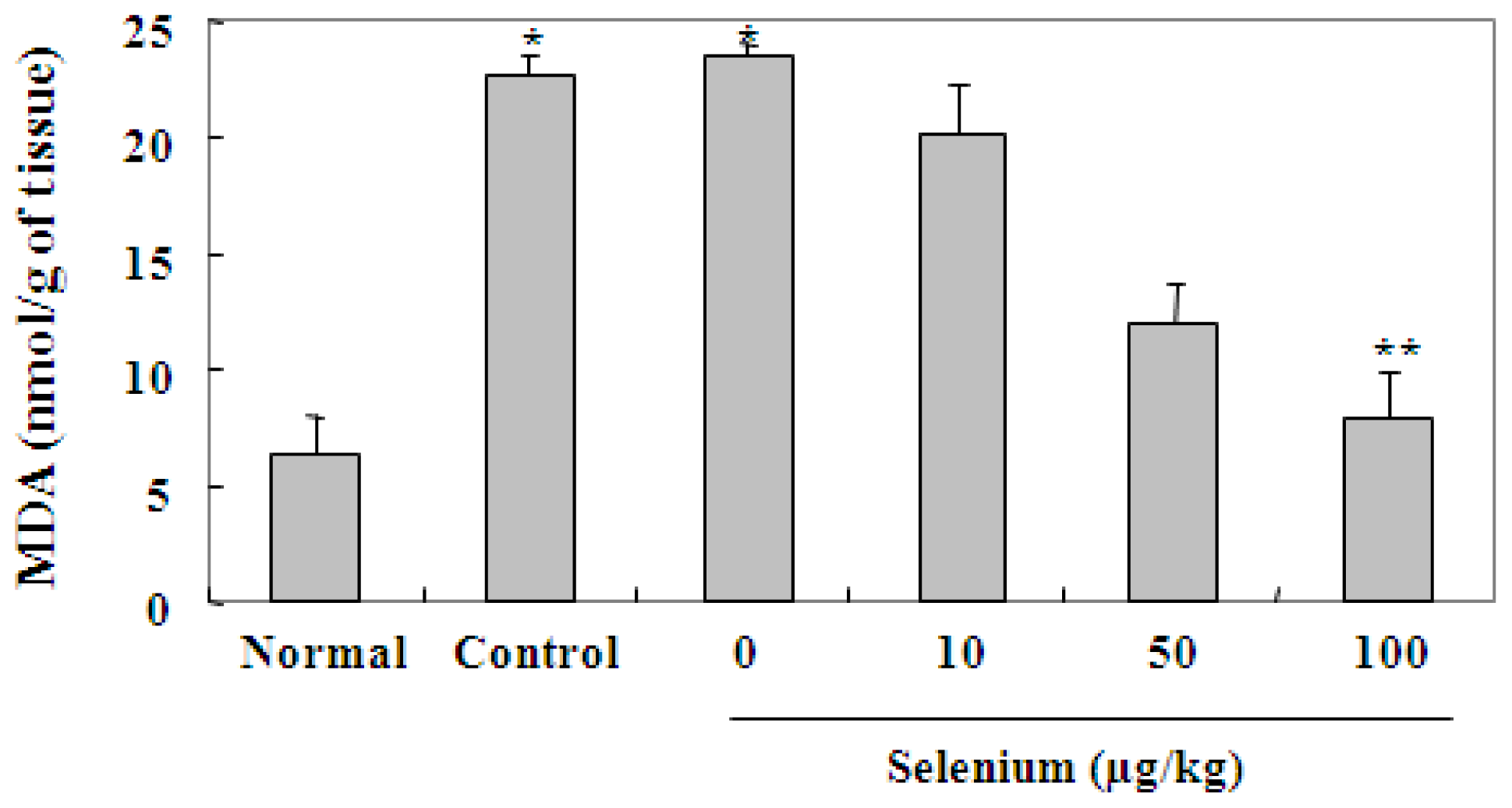
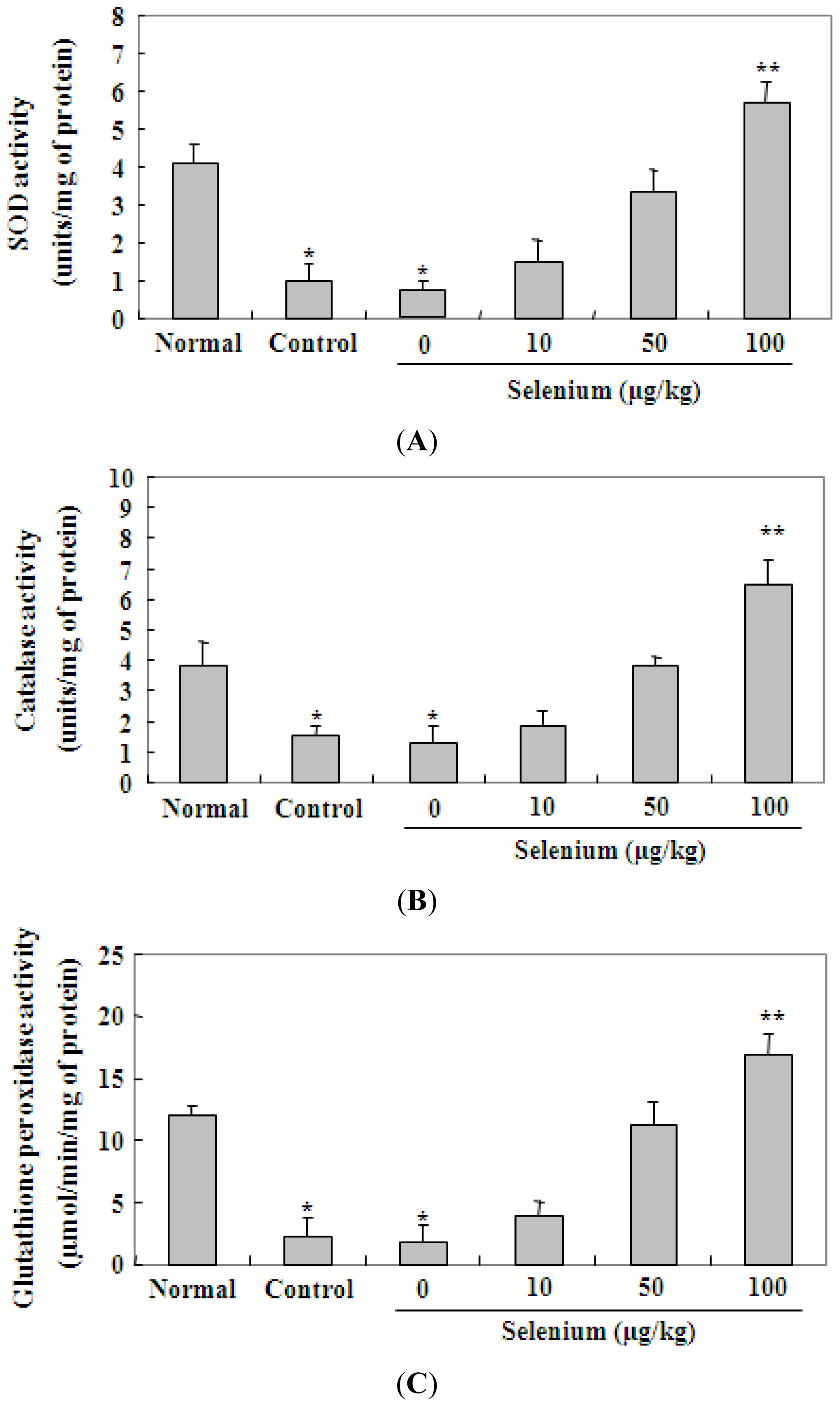
2.3. Selenium Inhibits Ethanol-Induced Gastric Mucosal Lesions through Prevention of Lipid Peroxidation and Activation of Radical Scavenging Enzymes
3. Experimental Section
3.1. Chemicals
3.2. Animals
3.3. Induction and Evaluation of Ethanol-Induced Gastric Lesions
3.4. Experimental Protocol
3.5. Malondialdehyde Levels
3.6. SOD Assay
3.7. Catalase Assay
3.8. Glutathione Peroxidase Assay
3.9. Histopathology
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Wallace, J.L.; Granger, D.N. The cellular and molecular basis of gastric mucosal defense. FASEB J 1996, 10, 731–740. [Google Scholar]
- Laine, L.; Takeuchi, K.; Tarnawski, A. Gastric mucosal defense and cytoprotection: Bench to bedside. Gastroenterology 2008, 135, 41–60. [Google Scholar]
- Eastwood, G.L.; Kirchner, J.P. Changes in the fine structure of mouse gastric epithelium produced by ethanol and urea. Gastroenterology 1974, 67, 71–84. [Google Scholar]
- Brzozowski, T.; Konturek, P.C.; Konturek, S.J.; Brzozowska, I.; Pawlik, T. Role of prostaglandins in gastroprotection and gastric adaptation. J. Physiol. Pharmacol 2005, 56, 33–55. [Google Scholar]
- Lulu, D.J.; Dragstedt, L.R. Massive bleeding due to acute hemorrhagic gastritis. Arch. Surg 1970, 101, 550–554. [Google Scholar]
- Tarnawski, A.; Stachura, J.; Ivey, K.J.; Mach, T.; Bogdal, J.; Klimczyk, B. Ethanol induced duodenal lesions in man. Protective effect of prostaglandin. Prostaglandins 1981, 21, 147–153. [Google Scholar]
- Laine, L. Endoscopic therapy for peptic ulcer hemorrhage: Heater probe and alcohol injection. Gastroenterology 1991, 100, 575–577. [Google Scholar]
- Szabo, S. Mechanisms of mucosal injury in the stomach and duodenum: Time-sequence analysis of morphologic, functional, biochemical and histochemical studies. Scand. J. Gastroenterol 1987, 127, S21–S28. [Google Scholar]
- Gottfried, E.B.; Korsten, M.A.; Lieber, C.S. Alcohol-induced gastric and duodenal lesions in man. Am. J. Gastroenterol 1978, 70, 587–592. [Google Scholar]
- Szabo, S.; Trier, J.S.; Brown, A.; Schnoor, J. Early vascular injury and increased vascular permeability in gastric mucosal injury caused by ethanol in the rat. Gastroenterology 1985, 88, 228–236. [Google Scholar]
- Lutnicki, K.; Wrobel, J.; Ledwozyw, A.; Trebas-Pietras, E. The effect of calcium ions on the intensity of peroxidation processes and the severity of ethanol-induced injury to the rat gastric mucosa. Arch. Vet. Pol 1992, 32, 125–132. [Google Scholar]
- Park, J.G.; Oh, G.T. The role of peroxidases in the pathogenesis of atherosclerosis. BMB Rep 2011, 44, 497–505. [Google Scholar]
- Pihan, G.; Regillo, C.; Szabo, S. Free radicals and lipid peroxidation in ethanol- or aspirin-induced gastric mucosal injury. Dig. Dis. Sci 1987, 32, 1395–1401. [Google Scholar]
- Szelenyi, I.; Brune, K. Possible role of oxygen free radicals in ethanol-induced gastric mucosal damage in rats. Dig. Dis. Sci 1988, 33, 865–871. [Google Scholar]
- Shaw, S.; Herbert, V.; Colman, N.; Jayatilleke, E. Effect of ethanol-generated free radicals on gastric intrinsic factor and glutathione. Alcohol 1990, 7, 153–157. [Google Scholar]
- Ito, M.; Suzuki, Y.; Ishihara, M.; Suzuki, Y. Anti-ulcer effects of antioxidants: Effect of probucol. Eur. J. Pharmacol 1998, 7, 189–196. [Google Scholar]
- Alvarez-Suarez, J.M.; Dekanski, D.; Ristić, S.; Radonjić, N.V.; Petronijević, N.D.; Giampieri, F.; Astolfi, P.; González-Paramás, A.M.; Santos-Buelga, C.; Tulipani, S.; et al. Strawberry polyphenols attenuate ethanol-induced gastric lesions in rats by activation of antioxidant enzymes and attenuation of MDA increase. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Ibrahim, I.A.; Qader, S.W.; Abdulla, M.A.; Nimir, A.R.; Abdelwahab, S.I.; Al-Bayaty, F.H. Effects of Pithecellobium jiringa ethanol extract against ethanol-induced gastric mucosal injuries in Sprague-Dawley rats. Molecules 2012, 17, 2796–2811. [Google Scholar]
- Mei, X.; Xu, D.; Xu, S.; Zheng, Y.; Xu, S. Novel role of Zn(II)-curcumin in enhancing cell proliferation and adjusting proinflammatory cytokine-mediated oxidative damage of ethanol-induced acute gastric ulcers. Chem. Biol. Interact 2012, 15, 31–39. [Google Scholar]
- Rotruck, J.T.; Pope, A.L.; Ganther, H.E.; Swanson, A.B.; Hafeman, D.G.; Hoekstra, W.G. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973, 179, 588–590. [Google Scholar]
- Marcocci, L.; Floche, L.; Packer, L. Evidence for a functional role of the selenocysteine residue in mammalian thioredoxin reductase. Biofactors 1997, 6, 351–358. [Google Scholar]
- Lee, S.R.; Bar-Noy, S.; Kwon, S.; Levine, R.L.; Stadtman, T.C.; Rhee, S.G. Mammalian thioredoxin reductase: Oxidation of the C-terminal cysteine/selenocysteine active site forms a thioselenide, and replacement of selenium with sulfur markedly reduces catalytic activity. Proc. Natl. Acad. Sci. USA 2000, 97, 2521–2526. [Google Scholar]
- Imam, S.Z.; Newport, G.D.; Islam, F.; Slikker, W.; Ali, S.F., Jr. Selenium, an antioxidant, protects against methamphetamine-induced dopaminergic neurotoxicity. Brain Res 1999, 818, 575–578. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Biologically relevant metal ion-dependent hydroxyl radical generation: An update. Fed. Eur. Biochem. Soc. Lett 1992, 307, 108–112. [Google Scholar]
- Sanmartin, C.; Plano, D.; Font, M.; Palop, J.A. Selenium and clinical trials: New therapeutic evidence for multiple diseases. Curr. Med. Chem 2011, 18, 4635–4650. [Google Scholar]
- Loef, M.; Schrauzer, G.N.; Walach, H. Selenium and Alzheimer’s disease: A systematic review. J. Alzheimers Dis 2011, 26, 81–104. [Google Scholar]
- Hogeboom, G.H. Methods in Enzymology; Colowick, S.P., Kaplan, N.O., Eds.; Academic Press: New York, NY, USA, 1955; pp. 16–19. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Mihara, M.; Uchiyama, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem 1978, 86, 271–278. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem 1979, 95, 351–358. [Google Scholar]
- McCord, J.M.; Fridovich, I. Superoxide dismutase, an enzymatic function for erythrocuprein (hemocuprein). J. Biol. Chem 1967, 244, 6049–6055. [Google Scholar]
- Aebi, H. Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Academic Press: New York, NY, USA, 1974; pp. 674–678. [Google Scholar]
- Lawrence, R.A.; Burk, R.F. Glutathione peroxidase activity in selenium-deficient rat liver. Biochem. Biophys. Res. Commun 1976, 71, 952–958. [Google Scholar]
- Kim, H.; Lee, S.W.; Baek, K.M.; Park, J.S.; Min, J.H. Continuous hypoxia attenuates paraquat-induced cytotoxicity in the human A549 lung carcinoma cell line. Exp. Mol. Med 2011, 43, 494–500. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, J.-H.; Park, S.-H.; Nam, S.-W.; Choi, Y.-H. Gastroprotective Effect of Selenium on Ethanol-Induced Gastric Damage in Rats. Int. J. Mol. Sci. 2012, 13, 5740-5750. https://doi.org/10.3390/ijms13055740
Kim J-H, Park S-H, Nam S-W, Choi Y-H. Gastroprotective Effect of Selenium on Ethanol-Induced Gastric Damage in Rats. International Journal of Molecular Sciences. 2012; 13(5):5740-5750. https://doi.org/10.3390/ijms13055740
Chicago/Turabian StyleKim, Jeong-Hwan, Shin-Hyung Park, Soo-Wan Nam, and Yung-Hyun Choi. 2012. "Gastroprotective Effect of Selenium on Ethanol-Induced Gastric Damage in Rats" International Journal of Molecular Sciences 13, no. 5: 5740-5750. https://doi.org/10.3390/ijms13055740




