Early Fixation of Cobalt-Chromium Based Alloy Surgical Implants to Bone Using a Tissue-engineering Approach
Abstract
:1. Introduction
2. Results and Discussion
2.1. In Vitro Experiment
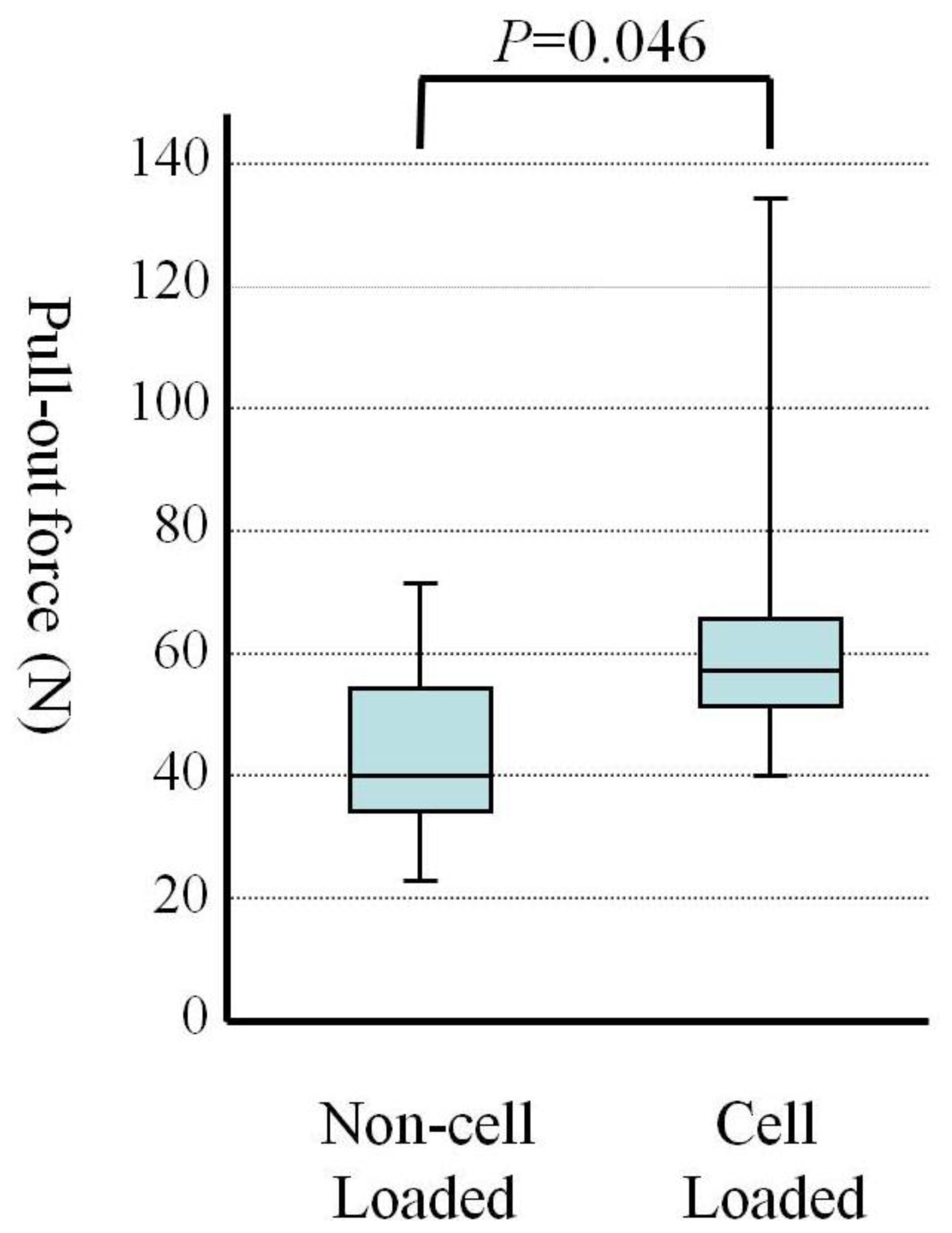
2.2. Mechanical Testing
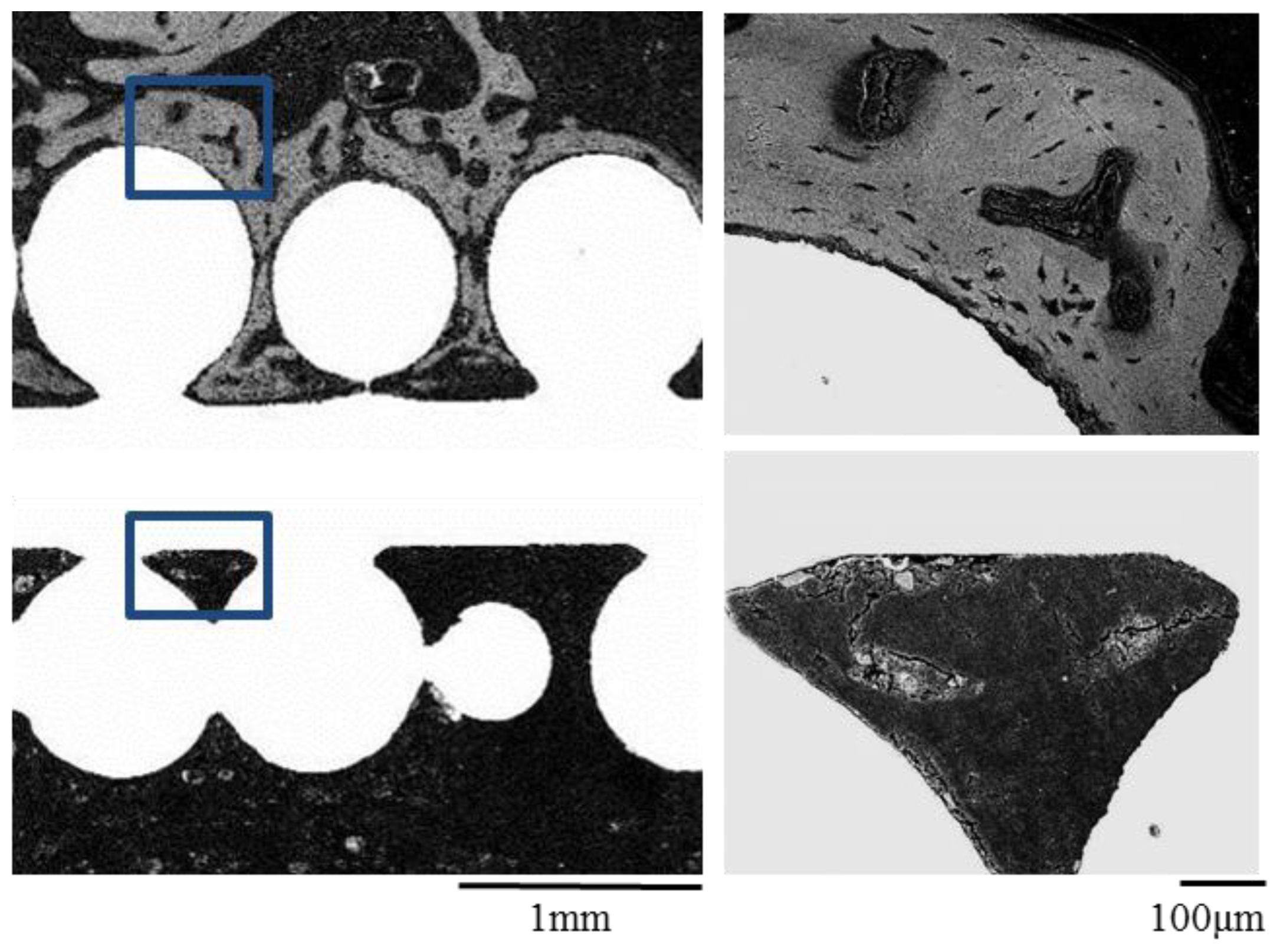
2.3. Histological Examination and Electron Microscope Analysis
2.4. Discussion
3. Materials and Methods
3.1. Preparation of Marrow Mesenchymal Cells and Implants
3.2. Implantation
3.3. Alizarin Red S Staining
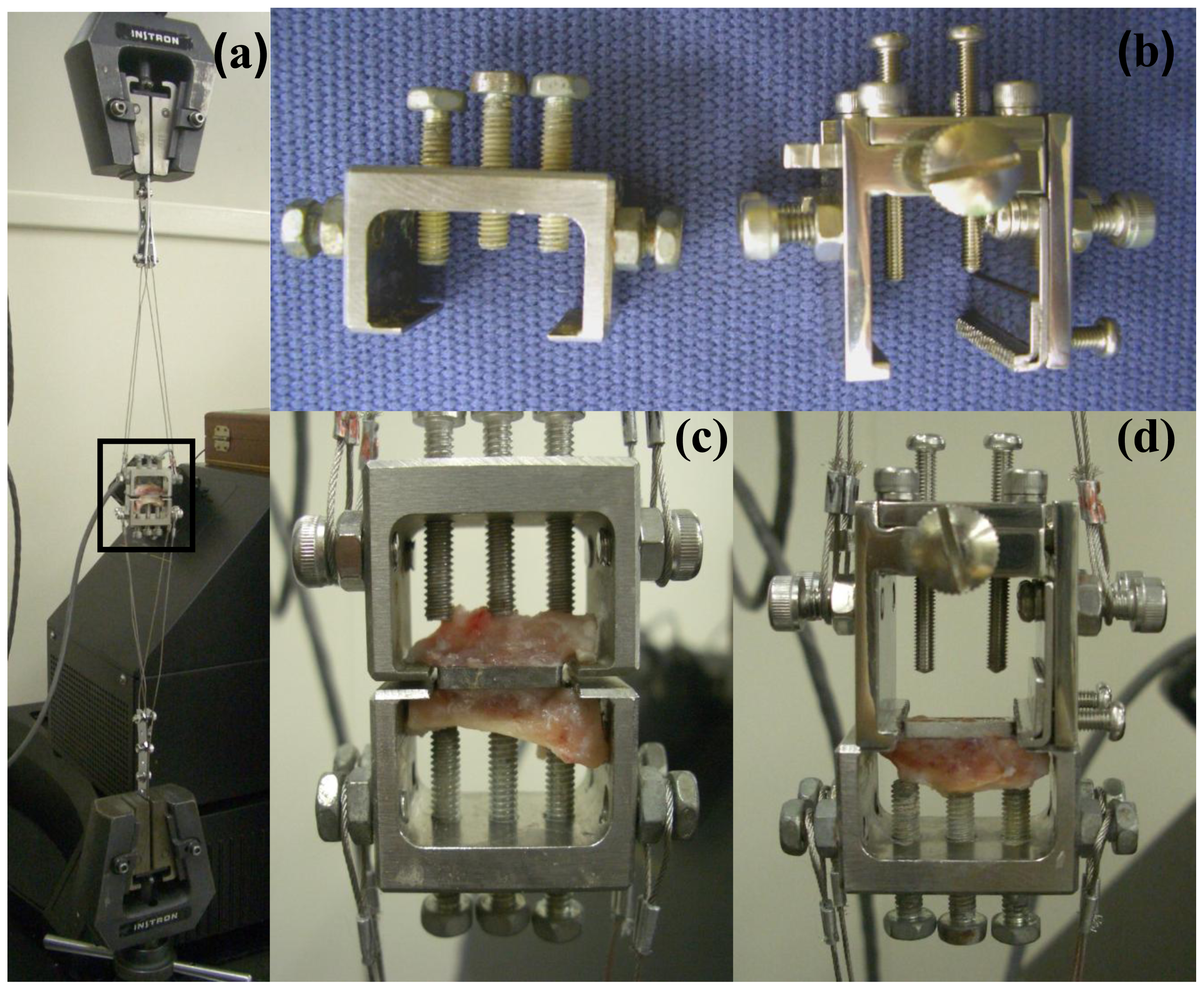
3.4. Measurement of Pull-out Force
3.5. Histological Examination and Electron Microscope Analysis
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Sundfeldt, M.; Carlsson, L.V.; Johansson, C.B.; Thomsen, P.; Gretzer, C. Aseptic loosening, not only a question of wear: A review of different theories. Acta Orthop 2006, 77, 177–197. [Google Scholar]
- Webster, T.J.; Ejiofor, J.U. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials 2004, 25, 4731–4739. [Google Scholar]
- Bal, B.S.; Garino, J.; Ries, M.; Rahaman, M.N. A review of ceramic bearing materials in total joint arthroplasty. Hip Int 2007, 17, 21–30. [Google Scholar]
- Lohmann, C.H.; Schwartz, Z.; Koster, G.; Jahn, U.; Buchhorn, G.H.; MacDougall, M.J.; Casasola, D.; Liu, Y.; Sylvia, V.L.; Dean, D.D.; et al. Phagocytosis of wear debris by osteoblasts affects differentiation and local factor production in a manner dependent on particle composition. Biomaterials 2000, 21, 551–561. [Google Scholar]
- Haynes, D.R.; Crotti, T.N.; Haywood, M.R. Corrosion of and changes in biological effects of cobalt chrome alloy and 316L stainless steel prosthetic particles with age. J. Biomed. Mater. Res 2000, 49, 167–175. [Google Scholar]
- Ohgushi, H.; Kotobuki, N.; Funaoka, H.; Machida, H.; Hirose, M.; Tanaka, Y.; Takakura, Y. Tissue engineered ceramic artificial joint-ex vivo osteogenic differentiation of patient mesenchymal cells on total ankle joints for treatment of osteoarthritis. Biomaterials 2005, 26, 4654–4661. [Google Scholar]
- Ohgushi, H.; Caplan, A.I. Stem cell technology and bioceramics: From cell to gene engineering. J. Biomed. Mater. Res 1999, 48, 913–927. [Google Scholar]
- Caplan, A.I. The mesengenic process. Clin. Plast. Surg 1994, 21, 429–435. [Google Scholar]
- Ohgushi, H.; Dohi, Y.; Katuda, T.; Tamai, S.; Tabata, S.; Suwa, Y. In vitro bone formation by rat marrow cell culture. J. Biomed. Mater. Res 1996, 32, 333–340. [Google Scholar]
- Maniatopoulos, C.; Sodek, J.; Melcher, A.H. Bone formation in vitro by stromal cells obtained from bone marrow of young adult rats. Cell Tissue Res 1988, 254, 317–330. [Google Scholar]
- Tohma, Y.; Tanaka, Y.; Ohgushi, H.; Kawate, K.; Taniguchi, A.; Hayashi, K.; Isomoto, S.; Takakura, Y. Early bone in-growth ability of alumina ceramic implants loaded with tissue-engineered bone. J.Orthop. Res 2006, 24, 595–603. [Google Scholar]
- Sotereanos, N.G.; Engh, C.A.; Glassman, A.H.; Macalino, G.E.; Engh, C.A., Jr. Cementless femoral components should be made from cobalt chrome. Clin. Orthop. Related Res 1995, 146–153. [Google Scholar]
- Heimke, G.; Leyen, S.; Willmann, G. Knee arthoplasty: Recently developed ceramics offer new solutions. Biomaterials 2002, 23, 1539–1551. [Google Scholar]
- Da Rocha, S.S.; Adabo, G.L.; Henriques, G.E.; Nobilo, M.A. Vickers hardness of cast commercially pure titanium and ti-6al-4v alloy submitted to heat treatments. J. Brazilian Dent 2006, 17, 126–129. [Google Scholar]
- Nguyen, N.C.; Maloney, W.J.; Dauskardt, R.H. Reliability of pmma bone cement fixation: Fracture and fatigue crack-growth behaviour. J. Mater. Sci. Mater. Med 1997, 8, 473–483. [Google Scholar]
- Navarro, M.; Michiardi, A.; Castano, O.; Planell, J.A. Biomaterials in orthopaedics. J. R. Soc. Interface/R. Soc 2008, 5, 1137–1158. [Google Scholar]
- Patel, B.; Inam, F.; Reece, M.; Edirisinghe, M.; Bonfield, W.; Huang, J.; Angadji, A. A novel route for processing cobalt-chromium-molybdenum orthopaedic alloys. J. R. Soc. Interface/R. Soc 2010, 7, 1641–1645. [Google Scholar]
- Patntirapong, S.; Habibovic, P.; Hauschka, P.V. Effects of soluble cobalt and cobalt incorporated into calcium phosphate layers on osteoclast differentiation and activation. Biomaterials 2009, 30, 548–555. [Google Scholar]
- Tomas, H.; Carvalho, G.S.; Fernandes, M.H.; Freire, A.P.; Abrantes, L.M. The use of rat, rabbit or human bone marrow derived cells for cytocompatibility evaluation of metallic elements. J. Mater. Sci. Mater. Med 1997, 8, 233–238. [Google Scholar]
- Endres, S.; Bartsch, I.; Sturz, S.; Kratz, M.; Wilke, A. Polyethylene and cobalt-chromium molybdenium particles elicit a different immune response in vitro. J. Mater. Sci. Mater. Med 2008, 19, 1209–1214. [Google Scholar]
- Hallab, N.J.; Vermes, C.; Messina, C.; Roebuck, K.A.; Glant, T.T.; Jacobs, J.J. Concentration- and composition-dependent effects of metal ions on human MG-63 osteoblasts. J. Biomed. Mater. Res 2002, 60, 420–433. [Google Scholar]
- Hallab, N.J.; Anderson, S.; Caicedo, M.; Brasher, A.; Mikecz, K.; Jacobs, J.J. Effects of soluble metals on human peri-implant cells. J. Biomed. Mater. Res. Part A 2005, 74, 124–140. [Google Scholar]
- Wang, J.Y.; Wicklund, B.H.; Gustilo, R.B.; Tsukayama, D.T. Titanium, chromium and cobalt ions modulate the release of bone-associated cytokines by human monocytes/macrophages in vitro. Biomaterials 1996, 17, 2233–2240. [Google Scholar]
- Niki, Y.; Matsumoto, H.; Suda, Y.; Otani, T.; Fujikawa, K.; Toyama, Y.; Hisamori, N.; Nozue, A. Metal ions induce bone-resorbing cytokine production through the redox pathway in synoviocytes and bone marrow macrophages. Biomaterials 2003, 24, 1447–1457. [Google Scholar]
- Kihara, T.; Oshima, A.; Hirose, M.; Ohgushi, H. Three-dimensional visualization analysis of in vitro cultured bone fabricated by rat marrow mesenchymal stem cells. Biochem. Biophys. Res. Commun 2004, 316, 943–948. [Google Scholar]
- Tadokoro, M.; Kanai, R.; Taketani, T.; Uchio, Y.; Yamaguchi, S.; Ohgushi, H. New bone formation by allogeneic mesenchymal stem cell transplantation in a patient with perinatal hypophosphatasia. J. Pediat 2009, 154, 924–930. [Google Scholar]
- Jones, L.C.; Hungerford, D.S. Cement disease. Clin. Orthop. Related Res 1987, 192–206. [Google Scholar]
- Nilsson, K.G.; Karrholm, J.; Carlsson, L.; Dalen, T. Hydroxyapatite coating versus cemented fixation of the tibial component in total knee arthroplasty: Prospective randomized comparison of hydroxyapatite-coated and cemented tibial components with 5-year follow-up using radiostereometry. J. Arthroplast 1999, 14, 9–20. [Google Scholar]
- Hofmann, A.A.; Heithoff, S.M.; Camargo, M. Cementless total knee arthroplasty in patients 50 years or younger. Clin. Orthop. Related Res 2002, 102–107. [Google Scholar]
- Hungerford, D.S.; Krackow, K.A.; Kenna, R.V. Cementless total knee replacement in patients 50 years old and under. Orthop. Clin. North Am 1989, 20, 131–145. [Google Scholar]
- Gao, F.; Henricson, A.; Nilsson, K.G. Cemented versus uncemented fixation of the femoral component of the NexGen CR total knee replacement in patients younger than 60 years: A prospective randomised controlled RSA study. The Knee 2009, 16, 200–206. [Google Scholar]
- Bojescul, J.A.; Xenos, J.S.; Callaghan, J.J.; Savory, C.G. Results of porous-coated anatomic total hip arthroplasty without cement at fifteen years: A concise follow-up of a previous report. J. Bone Joint Surg. Am 2003, 85-A, 1079–1083. [Google Scholar]
- Haddad, R.J., Jr; Cook, S.D.; Thomas, K.A. Biological fixation of porous-coated implants. J. Bone Joint Surg. Am 1987, 69, 1459–1466. [Google Scholar]
- Kavolus, C.H.; Ritter, M.A.; Keating, E.M.; Faris, P.M. Survivorship of cementless total knee arthroplasty without tibial plateau screw fixation. Clin. Orthop. Related Res 1991, 170–176. [Google Scholar]
- Ryd, L.; Toksvig-Larsen, S. Early postoperative fixation of tibial components: An in vivo roentgen stereophotogrammetric analysis. J. Orthopaedic Res. 1993, 11, 142–148. [Google Scholar]
- Grandfield, K.; Palmquist, A.; Goncalves, S.; Taylor, A.; Taylor, M.; Emanuelsson, L.; Thomsen, P.; Engqvist, H. Free form fabricated features on CoCr implants with and without hydroxyapatite coating in vivo: A comparative study of bone contact and bone growth induction. J. Mater. Sci.: Mater. Med 2011, 22, 899–906. [Google Scholar]
- Nakamura, T.; Yamamuro, T.; Higashi, S.; Kokubo, T.; Itoo, S. A new glass-ceramic for bone replacement: Evaluation of its bonding to bone tissue. J. Biomed. Mater. Res 1985, 19, 685–698. [Google Scholar]
- Kitsugi, T.; Yamamuro, T.; Nakamura, T.; Higashi, S.; Kakutani, Y.; Hyakuna, K.; Ito, S.; Kokubo, T.; Takagi, M.; Shibuya, T. Bone bonding behavior of three kinds of apatite containing glass ceramics. J. Biomed. Mater. Res 1986, 20, 1295–1307. [Google Scholar]




© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ogawa, M.; Tohma, Y.; Ohgushi, H.; Takakura, Y.; Tanaka, Y. Early Fixation of Cobalt-Chromium Based Alloy Surgical Implants to Bone Using a Tissue-engineering Approach. Int. J. Mol. Sci. 2012, 13, 5528-5541. https://doi.org/10.3390/ijms13055528
Ogawa M, Tohma Y, Ohgushi H, Takakura Y, Tanaka Y. Early Fixation of Cobalt-Chromium Based Alloy Surgical Implants to Bone Using a Tissue-engineering Approach. International Journal of Molecular Sciences. 2012; 13(5):5528-5541. https://doi.org/10.3390/ijms13055528
Chicago/Turabian StyleOgawa, Munehiro, Yasuaki Tohma, Hajime Ohgushi, Yoshinori Takakura, and Yasuhito Tanaka. 2012. "Early Fixation of Cobalt-Chromium Based Alloy Surgical Implants to Bone Using a Tissue-engineering Approach" International Journal of Molecular Sciences 13, no. 5: 5528-5541. https://doi.org/10.3390/ijms13055528





