The Relationship Between Grain Hardness, Dough Mixing Parameters and Bread-Making Quality in Winter Wheat
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Molecular Analysis
2.1.2. Variation in Quality Parameters
2.1.3. Contrast Analysis
2.1.3.1. NIR Parameters
2.1.3.2. Farinograph Parameters
2.1.3.3. Reomixer Parameters
2.1.3.4. Bread-making Properties
2.1.4. Correlations
2.2. Discussion
3. Experimental Section
3.1. Plant Materials
3.2. Grain Hardness Determination
3.3. Molecular Analysis
3.4. Rheological Analysis
3.5. Bread-Making Quality
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Gąsiorowski, H.; Poliszko, S. A wheat endosperm microhardness index. Acta Alim 1977, 6, 113–117. [Google Scholar]
- Greenaway, W.T. A Wheat Hardness Index. Cereal Sci. Today 1969, 14, 4–7. [Google Scholar]
- Stenvert, N.L. Grinding resistance. A simple measure of wheat hardness. Flour Anim. Feed Milling 1974, 7, 24–27. [Google Scholar]
- Chesterfield, R.S. A modified barley pearlier for measuring hardness of Australian wheats. Aust. Inst. Agric. Sci 1971, 6, 148–151. [Google Scholar]
- Noris, K.H.; Hruschka, W.R.; Bean, M.M.; Slaughter, D.C. A definition of wheat hardness using near-infrared spectroscopy. Cereal Foods World 1989, 34, 696–705. [Google Scholar]
- Khalil, I.H.; Carver, B.; Krenzer, E.G.; MacKown, C.T.; Horn, G.W.; Rayas-Duarte, P. Genetic trends in winter wheat grain quality with dual-purpose and grain-only management systems. Crop Sci 2002, 42, 1112–1116. [Google Scholar]
- Pearson, T.C.; Brabec, D.L. Camera attachment for automatic measurement of single-wheat kernel size on a Perten SKCS 4100. Appl. Eng. Agric 2006, 22, 927–933. [Google Scholar]
- Shewry, P.R.; Halford, N.G.; Tatham, A.S. High molecular weight subunits of wheat glutenin. J. Cereal Sci 1992, 15, 105–120. [Google Scholar]
- Autron, J.C.; Hamme, R.J.; Plijter, J.J.; Pogna, N.E. Exploring and improving the industrial use of wheats. Cereal Foods World 1997, 4, 216–227. [Google Scholar]
- Martin, J.M.; Frohberg, R.C.; Morris, C.F.; Talbert, L.E.; Giroux, M.J. Milling and bread baking traits associated with puroindoline sequence type in hard red spring wheat. Crop Sci 2001, 41, 228–234. [Google Scholar]
- Gross, C.; Bervas, E.; Charmet, G. Genetic analysis of grain protein content, grain hardness and dough rheology in a hard×hard bread wheat progeny. J. Cereal Sci 2004, 40, 93–100. [Google Scholar]
- Sourdille, P.; Perretant, M.R.; Charmet, G.; Leroy, P.; Gauteir, M.F.; Joudrier, P.; Nelson, J.C.; Sorrlls, M.E.; Bernard, M. Linkage between RFLP markers and genes affecting kernel hardness in wheat. Theor. Appl. Genet 1996, 93, 580–586. [Google Scholar]
- Appels, R.; Gustafson, J.P.; O’Brien, L. Wheat breeding in the new century: Applying molecular genetic analysis of key quality and agronomic traits. Aust. J. Agric. Res 2001, 52, 1043–1417. [Google Scholar]
- Morris, C.F. Puroindoline: The molecular genetic basis of wheat grain hardness. Plant Mol. Biol 2002, 48, 633–647. [Google Scholar]
- Bettge, A.D.; Morris, C.F.; Greenblatt, G.A. Assessing genotypic softness in single wheat kernels using starch granule associated friabilin as a biochemical marker. Euphytica 1995, 86, 65–72. [Google Scholar]
- Giroux, M.J.; Talbert, L.; Habernicht, D.K.; Lanning, S.; Hemphill, A.; Martin, J.M. Association of puroindoline sequence type and grain hardness in hard red spring wheat. Crop Sci 2000, 40, 370–374. [Google Scholar]
- Pareyt, B.; Finnie, S.M.; Putseys, J.A.; Delcour, J.A. Lipids in bread making: Sources, interactions, and impact on bread quality. J. Cereal Sci 2011, 54, 266–279. [Google Scholar]
- Arbelbide, M.; Bernardo, R. Mixed-model QTL mapping for kernel hardness and dough strength in bread wheat. Theor. Appl. Genet 2006, 112, 885–890. [Google Scholar]
- Röder, M.S.; Korzun, V.; Wendehake, K.; Plaschke, J.; Tixier, M.H.; Leroy, P.; Ganal, M.W. A microsatellite map of wheat. Genetics 1998, 149, 2007–2023. [Google Scholar]
- Galande, A.A.; Tiwari, R.; Ammiraju, J.S.S.; Santra, D.K.; Lagu, M.D.; Rao, V.S.; Gupta, V.S.; Misra, B.K.; Nagarajan, S.; Ranjekar, P.K. Genetic analysis of kernel hardness in bread wheat using PCR-based markers. Theor. Appl. Genet 2001, 10, 601–606. [Google Scholar]
- Turner, A.S.; Bradburne, R.P.; Fish, L.; Snape, J.W. New quantitative trait loci influencing grain texture and protein content in bread wheat. J. Cereal Sci 2004, 40, 51–60. [Google Scholar]
- Somers, D.J.; Isaac, P.; Edwards, K. A high-density microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor. Appl. Genet 2004, 109, 1105–1114. [Google Scholar]
- Kuchel, H.; Langridge, P.; Mosionek, L.; Williams, K.; Jefferies, S.P. The genetic control of milling yield, dough rheology and baking quality of wheat. Theor. Appl. Genet 2006, 112, 1487–1495. [Google Scholar]
- Tsilo, T.J.; Simsek, S.; Ohm, J.B.; Hareland, G.A.; Chao, S.; Anderson, J.A. Quantitative trait loci influencing endosperm texture, dough-mixing strength, and breed-making properties of the hard red spring wheat breeding lines. Genome 2011, 54, 460–470. [Google Scholar]
- Osborne, B.G. Applications of near infrared spectroscopy in quality screening of early-generation material in cereal breeding programmes. J. Near Infrared Spectrosc 2006, 14, 93–101. [Google Scholar]
- Obuchowski, W.; Salmanowicz, B.; Banaszak, Z.; Adamski, T.; Surma, M.; Kaczmarek, Z.; Majcher, M.; Ługowska, B.; Kuczyńska, A.; Krystkowiak, K. Grain hardness of wheat bred in Poland and its relationship to starch damage during milling. Int. Agrophys 2010, 24, 69–74. [Google Scholar]
- Surma, M.; Adamski, T.; Banaszak, Z.; Kaczmarek, Z.; Kuczyńska, A.; Majcher, M.; Ługowska, B.; Obuchowski, W.; Salmanowicz, B.P.; Krystkowiak, K. Effect of genotype, environment and their interaction on quality parameters of wheat breeding lines of diverse grain hardness. Plant Prod. Sci 2012, in press. [Google Scholar]
- Lillemo, M.; Chen, F.; Xi, X.; William, M.; Peña, R.J.; Trethowan, R.; He, Z. Puroindoline grain hardness alleles in CIMMYT bread wheat germplasm. J. Cereal Sci 2006, 44, 86–92. [Google Scholar]
- Li, G.; He, Z.; Lillemo, M.; Sun, Q.; Xia, X. Molecular characterization of allelic variations at Pina and Pinb loci in Shandong wheat landraces; historical and current cultivars. J. Cereal Sci 2008, 47, 510–517. [Google Scholar]
- Hruskova, M.; Svec, I. Wheat hardness in relation to other quality factors. Czech. J. Food Sci. 2009, 27, 240–248. [Google Scholar]
- Bushuk, W.; Szajewska, A.; Haber, T. Mechaniczne uszkodzenie skrobi a wartość wypiekowa mąki. (Mechanical starch damage and flour baking properties). Przegląd Zbożowo-Młynarski 2007, 10, 13–14. [Google Scholar]
- Prasad, M.; Varshney, R.K.; Kumar, A.; Bollon, H.S.; Sharma, P.C.; Edwards, K.J.; Singh, H.; Dhaliwal, H.S.; Roy, J.K.; Gupta, P.K. A microsatellite marker associated with a QTL for grain protein content on chromosome arm 2DL of bread wheat. Theor. Appl. Genet 1999, 99, 341–345. [Google Scholar]
- Moiraghi, M.; Vanzetti, L.; Bainotti, C.; Helguera, M.; Leon, A.; Perez, G. Relationship between soft wheat flour physicochemical composition and cookie-making performance. Cereal Chem 2011, 88, 130–136. [Google Scholar]
- AACC International, Rheological Behavior of Flour by Farinograph: Constant Flour Weight Procedure, Approved January 6, 2011. In Approved Methods of Analysis, 11th ed; Method 54-21.02; AACC International: St. Paul, MN, USA. [CrossRef]
- International Association for Cereal Science and Technology, General Principles of the International Association for Cereal Science and Technology. In ICC Standard Methods; ICC: Vienna, Austria, 1994.
- Bohlin, L. Reomixer Online Software Operation Manual, Beta version 0.9; Reologen i Lund, AB: Lund, Sweden, 2007. [Google Scholar]
- Anderson, C.A. Characterising Wheat Flour Protein Quality from Reomixer; HGCA Report No. 324; HGCA: London, UK, 2003. [Google Scholar]
- Arbeitsgemeinschaft Getreideforschung/AGF, Standard-Methoden fur Getreide, Mehl und Brot, 7th ed; Moritz Schafer Verlag: Detmold, Germany, 1994.
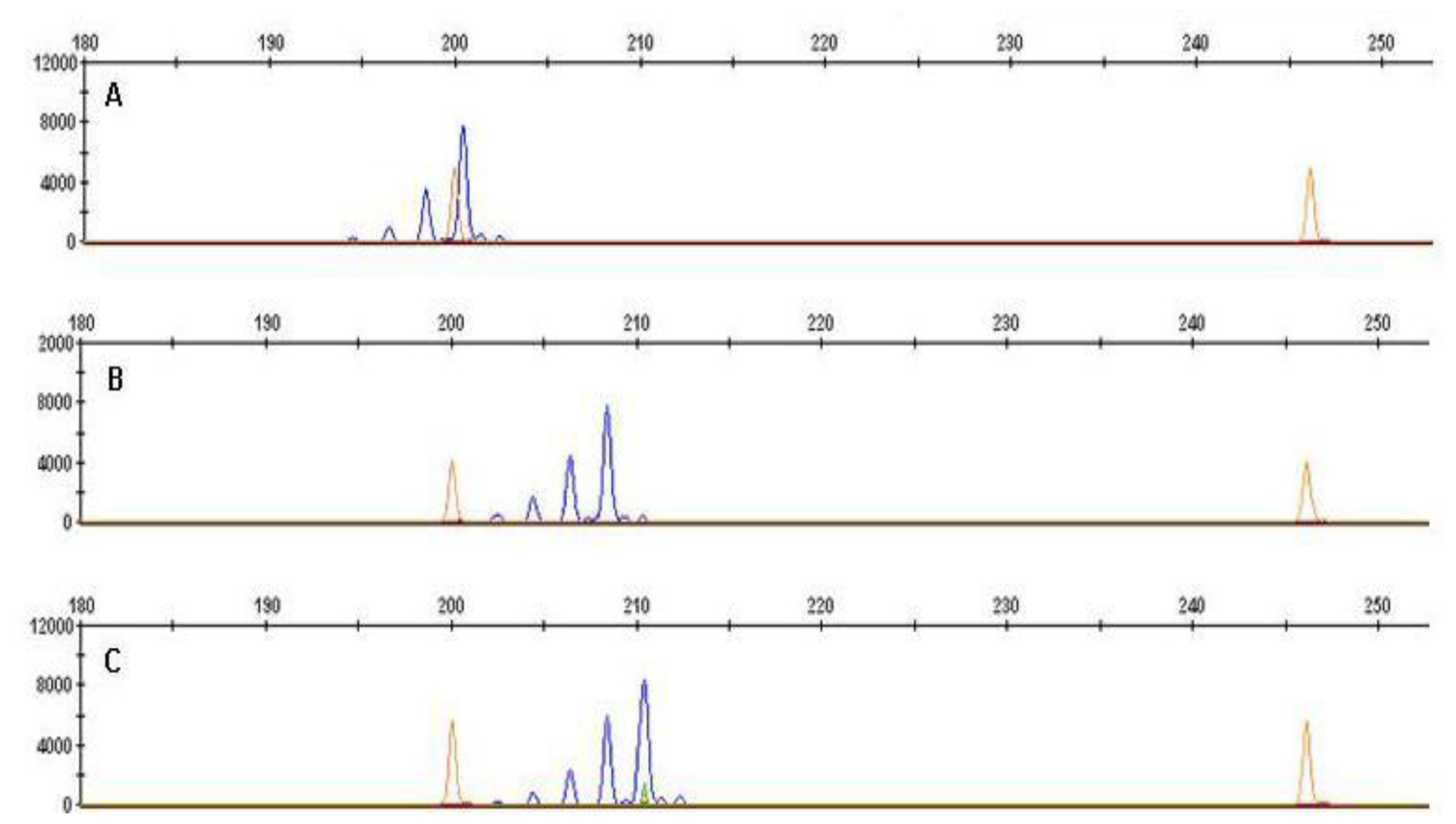
| Line No. | Code | Mean Values at Two Fertilization Levels | Puroindoline Alleles | Microsatellite Markers | |||||
|---|---|---|---|---|---|---|---|---|---|
| Protein Content | Hardness NIR | PSI | Pina [26] | Pinb [26] | Xgwm 190 (bp) | Xgwm 205 (bp) | Xgwm 358 (bp) | ||
| Hard-grained lines * | |||||||||
| 1 | CHD 169/04 | 13.3 | 67.9 | 19.5 | D1a | D1b | 200 | 138 | 162 |
| 9 | DED 4152/03 | 13.3 | 60.4 | 20.6 | D1a | D1b | 200 | 138 | 162 |
| 20 | LAD 252/04 | 12.9 | 62.1 | 19.4 | D1a | D1b | 210 | 138 | 162 |
| 29 | CHD 760/04 | 14.9 | 70.7 | 18.7 | D1a | D1b | 208 | 136 | 162 |
| 30 | CHD 65/04 | 15.6 | 61.4 | 19.2 | D1a | D1a | 210 | 138 | 156 |
| 31 | CHD 114/04 | 13.6 | 62.0 | 18.6 | D1a | D1b | 208 | 138 | 156 |
| 35 | CHD 382/04 | 14.2 | 65.2 | 16.3 | D1b | D1a | 210 | 138 | 156 |
| 36 | CHD 565/04 | 13.7 | 69.1 | 20.1 | D1a | D1a | 208 | 134 | 156 |
| 38 | CHD 737/04 | 14.2 | 65.6 | 18.7 | D1a | D1a | 208 | 138 | 162 |
| 40 | DED 3481/03 | 14.6 | 71.8 | 18.1 | D1a | D1b | 208 | 138 | 162 |
| 46 | LAD 319/04 | 13.9 | 61.3 | 20.8 | D1a | D1a | 210 | 138 | 162 |
| Soft-grained lines * | |||||||||
| 2 | CHD 329/04 | 13.7 | 53.2 | 22.0 | D1a | D1a | 200 | 136 | 158 |
| 7 | DED 5854/03 | 14.6 | 51.8 | 21.4 | D1a | D1a | 200 | 136 | 158 |
| 17 | LAD 180/05 | 13.4 | 48.7 | 22.0 | D1a | D1a | 200 | 136 | 158 |
| 41 | DED 5845/03 | 14.0 | 44.4 | 24.9 | D1a | D1a | 200 | 136 | 158 |
| 42 | DED 5897/03 | 13.8 | 43.8 | 21.2 | D1a | D1a | 200 | 136 | 158 |
| 44 | DED 6986/03 | 13.3 | 40.9 | 24.2 | D1a | D1a | 200 | 136 | 158 |
| 45 | LAD 125/04 | 13.6 | 40.6 | 31.5 | D1a | D1a | 200 | 136 | 158 |
| 48 | LAD 355/04 | 12.7 | 48.2 | 23.1 | D1a | D1a | 200 | 136 | 158 |
| 49 | TONACJA | 13.2 | 53.6 | 21.3 | D1a | D1a | 200 | 136 | 158 |
| Medium-grained lines * | |||||||||
| 8 | DED 6240/03 | 13.5 | 58.9 | 21.3 | D1a | D1b | 200 | 136 | 158 |
| 11 | DED 6658/03 | 13.8 | 54.5 | 22.5 | D1a | D1a | 208 | 138 | 162 |
| 13 | LAD 166/05 | 13.2 | 55.7 | 22.2 | D1a | D1a | 208 | 136 | 162 |
| 19 | LAD 245/04 | 13.7 | 58.6 | 21.0 | D1a | D1b | 210 | 138 | 162 |
| Character | FL1 | FL2 | ||
|---|---|---|---|---|
| Mean | Range | Mean | Range | |
| Grain hardness—PSI | 20.7 | 15.4–30.3 | 21.5 | 14.0–33.0 |
| Grain hardness—NIR | 53.1 | 24.7–71.2 | 61.4 | 46.5–74.5 |
| Protein content (%) | 12.8 | 11.4–14.5 | 14.8 | 13.6–16.7 |
| Starch content (%) | 67.3 | 64.2–69.7 | 64.8 | 62.4–66.9 |
| Wet gluten (%) | 26.6 | 24.6–31.7 | 32.2 | 29.6–36.5 |
| Zeleny sedimentation value (mL) | 37.7 | 25.3–53.8 | 55.0 | 42.2–64.7 |
| Farinograph Parameters | ||||
| Consistency (FU) | 504.6 | 492.0–520.5 | 501.8 | 486.0–520.0 |
| Water absorption (%) | 70.0 | 66.3–74.0 | 70.4 | 64.4–73.6 |
| Development time (min) | 2.25 | 1.45–3.05 | 2.46 | 1.55–3.30 |
| Dough stability (min) | 3.24 | 2.00–5.00 | 3.23 | 1.95–5.30 |
| Degree of softening (FU) | 92.5 | 66.5–133.0 | 85.8 | 62.5–114.0 |
| Farinograph quality number | 35.2 | 20.0–49.0 | 38.0 | 21.0–60.0 |
| Dough Mixing Parameters | ||||
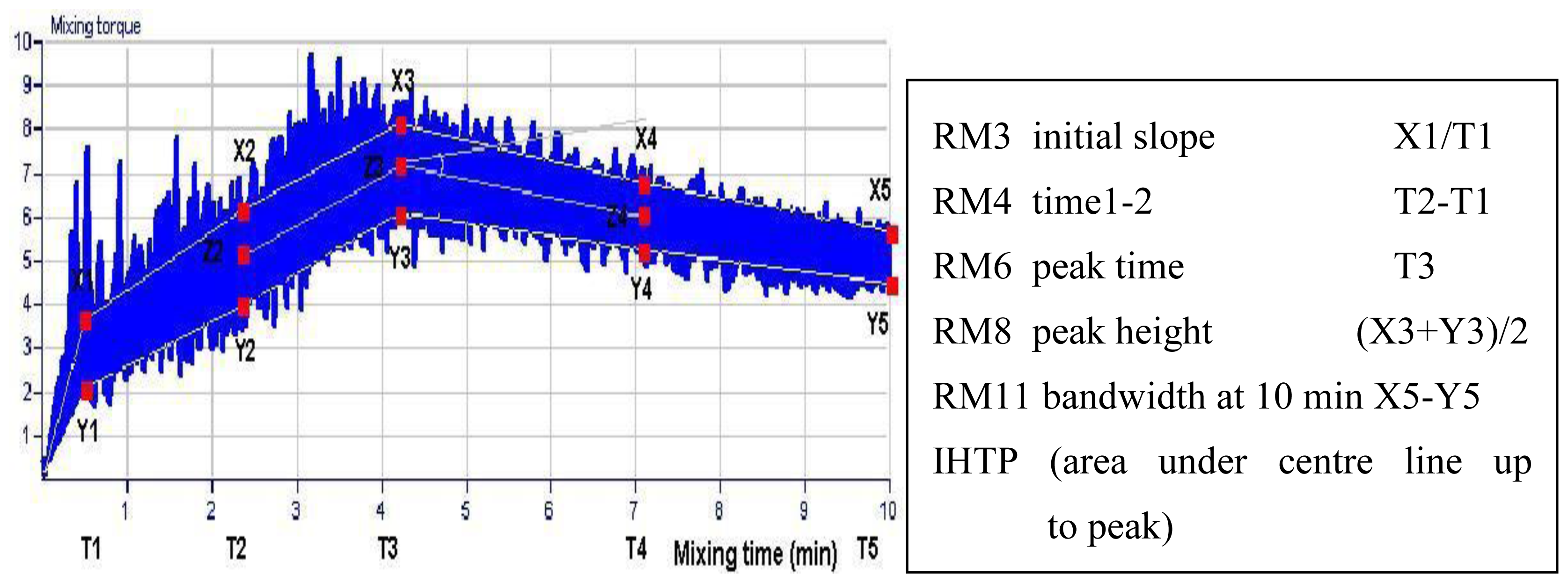
| Area under line (IHTP) | 23.6 | 13.0–42.5 | 20.4 | 11.5–44.2 |
| Initial slope (RM3) | 4.04 | 3.20–5.22 | 5.37 | 4.52–6.04 |
| Time 1–2 (RM4) (min) | 2.67 | 1.17–4.53 | 2.06 | 1.21–3.92 |
| Peak time (RM6) (min) | 5.86 | 3.76–8.55 | 4.10 | 3.03–5.83 |
| Peak height (RM8) | 5.93 | 5.37–7.05 | 5.82 | 5.04–7.67 |
| Bandwidth at 10 min (RM11) | 2.20 | 1.95–2.49 | 2.08 | 1.92–2.36 |
| Bread-Making Parameters | ||||
| Bread yield (%) | 140.5 | 135.2–145.8 | 142.1 | 138.4–146.4 |
| Loaf volume (cm3) | 634.0 | 586.0–687.0 | 615.8 | 557.0–652.0 |
| Bread crumb grain | 5.35 | 3.0–8.0 | 6.75 | 5.0–9.0 |
| Source of Variation | |||
|---|---|---|---|
| Character | Genotype (G); d.f. = 23 | Fertilisation Level (E); d.f. =1 | Interaction GE; d.f. = 23 |
| Grain hardness—PSI | 20.79 ** | 9.40 ** | 3.39 * |
| Grain hardness—NIR | 16.77 ** | 79.23 ** | 1.27 |
| Protein content (%) | 6.02 ** | 32.43 ** | 1.24 |
| Starch content (%) | 11.65 ** | 32.83 ** | 1.03 |
| Wet gluten (%) | 5.43 ** | 32.70 ** | 1.19 |
| Zeleny sedimentation value (mL) | 8.22 ** | 50.01 ** | 1.32 |
| Farinograph Parameters | |||
| Consistency (FU) | 1.77 * | 2.65 | 0.66 |
| Water absorption (%) | 10.34 ** | 2.63 | 4.58 ** |
| Development time (min) | 11.98 ** | 20.48 ** | 2.82 ** |
| Dough stability (min) | 53.11 ** | 0.06 | 7.10 ** |
| Degree of softening (FU) | 17.00 ** | 20.33 ** | 2.70 ** |
| Farinograf quality number | 22.25 ** | 18.93 ** | 5.93 ** |
| Dough Mixing Parameters | |||
| Area under line (IHTP) | 83.13 ** | 67.02 ** | 10.13 ** |
| Initial slope (RM3) | 0.57 | 49.73 ** | 0.41 |
| Time 1–2 (RM4) (min) | 45.96 ** | 162.83 ** | 6.23 ** |
| Peak time (RM6) (min) | 13.89 ** | 153.71 ** | 4.02 ** |
| Peak height (RM8) | 0.60 | 0.21 | 0.23 |
| Bandwidth at 10 min (RM11) | 8.91 ** | 31.72 ** | 0.95 * |
| Bread-Making Parameters | |||
| Bread yield (%) | 2.91 ** | 16.28 ** | 1.09 |
| Loaf volume(cm3) | 1.57 | 17.13 ** | 1.00 |
| Bread crumb grain | 0.015 | 0.470 ** | 0.092 |
| Character | Contrast: FL1–FL2 | Contrast 1: Hard–soft | Contrast 2: Hart–soft | ||
|---|---|---|---|---|---|
| FL1 | FL2 | FL1 | FL2 | ||
| Grain hardness—PSI | −0.84 * | −3.29 ** | −3.01 ** | −1.56 | −2.53 * |
| Grain hardness—NIR | −8.28 ** | 18.82 ** | 11.18 ** | 8.01 ** | 6.07 * |
| Protein content (%) | −2.03 ** | 0.42 ** | 0.41 * | −0.03 | 0.37 |
| Starch content (%) | 2.51 ** | −1.28 ** | −1.10 ** | −0.34 | −0.59 |
| Wet gluten (%) | −5.55 ** | 1.29 ** | 1.04 * | −0.13 | 1.02 * |
| Zeleny sedimentation value (mL) | −17.29 ** | 6.99 ** | 4.76 ** | 1.57 | 3.48 * |
| Farinograph parameters | |||||
| Consistency (FU) | 2.71 | 3.01 | 3.91 | 4.24 | 4.21 |
| Water absorption (%) | −0.40 | 4.00 ** | 1.84 ** | 1.45 * | −0.11 |
| Development time (min) | −0.21 ** | 0.05 | −0.03 | 0.34 ** | 0.02 |
| Dough stability (min) | 0.01 | 0.42 ** | 0.55 ** | −0.40 ** | −0.26 * |
| Degree of softening (FU) | 6.67 ** | −11.62 ** | −4.02 | −4.33 | −0.93 |
| Farinograph quality number | −2.81 | ** 0.33 | −0.22 | 2.42 ** | −4.43 ** |
| Dough mixing parameters | |||||
| Area under centre line (IHTP) | 3.28 ** | 12.47 ** | 7.62 ** | 2.94 * | 0.16 |
| Initial slope (RM3) | −1.32 ** | −0.31 | −0.28 | −0.32 | 0.05 |
| Time 1–2 (RM4) (min) | 0.61 ** | 1.11 ** | 0.63 ** | 0.19 | −0.09 |
| Peak time (RM6) (min) | 1.76 ** | 2.26 ** | 0.74 ** | 0.34 * | 0.06 |
| Peak height (RM8) | 0.12 | 0.21 | −0.20 | 0.13 | 0.12 |
| Bandwidth at 10 min (RM11) | 0.11 ** | 0.19 ** | 0.16 ** | 0.07 | 0.02 |
| Bread-making parameters | |||||
| Bread yield (%) | −1.55 ** | 2.20 ** | 1.64 ** | 0.95 | 1.81 |
| Loaf volume(cm3) | 18.27 ** | −1.06 | 3.92 | 4.66 | −2.17 |
| Bread crumb grain | −1.39 ** | 0.44 | −0.31 | 0.02 | −0.20 |
| Character | Grain Hardness—PSI | Grain Hardness—NIR | ||
|---|---|---|---|---|
| FL1 | FL2 | FL1 | FL2 | |
| Grain hardness—NIR | −0.601 ** | −0.578 ** | – | – |
| Protein content (%) | −0.239 | −0.288 * | 0.445 ** | 0.232 |
| Starch content (%) | 0.385 ** | −0.578 ** | −0.550 ** | −0.200 |
| Wet gluten (%) | −0.270 | −0.182 | 0.527 ** | 0.342 * |
| Zeleny sedimentation value (mL) | −0.446 ** | −0.310 * | 0.642 ** | 0.477 ** |
| Farinograph Parameters | ||||
| Consistency (FU) | −0.159 | −0.323 * | 0.446 ** | 0.279 * |
| Water absorption (%) | −0.597 ** | −0.022 | 0.834 ** | 0.289 * |
| Development time (min) | −0.106 | 0.141 | 0.085 | 0.064 |
| Dough stability (min) | −0.284 * | −0.120 | 0.280 * | 0.334 * |
| Degree of softening (FU) | 0.523 ** | 0.018 | −0.427 ** | −0.183 |
| Farinograph quality number | −0.051 | 0.215 | 0.007 | −0.054 |
| Dough Mixing Parameters | ||||
| Area under line (IHTP) | −0.322 * | −0.349 * | 0.616 ** | 0.535 ** |
| Initial slope (RM3) | 0.216 | 0.023 | −0.154 | −0.142 |
| Time 1–2 (RM4) (min) | −0.254 | −0.341 * | 0.516 ** | 0.485 ** |
| Peak time (RM6) (min) | −0.249 | −0.296 * | 0.510 ** | 0.476 ** |
| Peak height (RM8) | −0.223 | −0.147 | 0.261 | −0.088 |
| Bandwidth at 10 min (RM11) | −0.446 ** | −0.383 ** | 0.650 ** | 0.574 ** |
| Bread-Making Parameters | ||||
| Bread yield (%) | −0.281 * | −0.379 ** | 0.429 ** | 0.328 * |
| Loaf volume (cm3) | −0.142 | −0.165 | 0.134 | 0.051 |
| Bread crumb grain | 0.025 | −0.022 | 0.040 | 0.300 * |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Salmanowicz, B.P.; Adamski, T.; Surma, M.; Kaczmarek, Z.; Karolina, K.; Kuczyńska, A.; Banaszak, Z.; Ługowska, B.; Majcher, M.; Obuchowski, W. The Relationship Between Grain Hardness, Dough Mixing Parameters and Bread-Making Quality in Winter Wheat. Int. J. Mol. Sci. 2012, 13, 4186-4201. https://doi.org/10.3390/ijms13044186
Salmanowicz BP, Adamski T, Surma M, Kaczmarek Z, Karolina K, Kuczyńska A, Banaszak Z, Ługowska B, Majcher M, Obuchowski W. The Relationship Between Grain Hardness, Dough Mixing Parameters and Bread-Making Quality in Winter Wheat. International Journal of Molecular Sciences. 2012; 13(4):4186-4201. https://doi.org/10.3390/ijms13044186
Chicago/Turabian StyleSalmanowicz, Bolesław P., Tadeusz Adamski, Maria Surma, Zygmunt Kaczmarek, Krystkowiak Karolina, Anetta Kuczyńska, Zofia Banaszak, Bogusława Ługowska, Małgorzata Majcher, and Wiktor Obuchowski. 2012. "The Relationship Between Grain Hardness, Dough Mixing Parameters and Bread-Making Quality in Winter Wheat" International Journal of Molecular Sciences 13, no. 4: 4186-4201. https://doi.org/10.3390/ijms13044186




