Expression Analysis of an R3-Type MYB Transcription Factor CPC-LIKE MYB4 (TRICHOMELESS2) and CPL4-Related Transcripts in Arabidopsis
Abstract
:1. Introduction
2. Results and Discussion
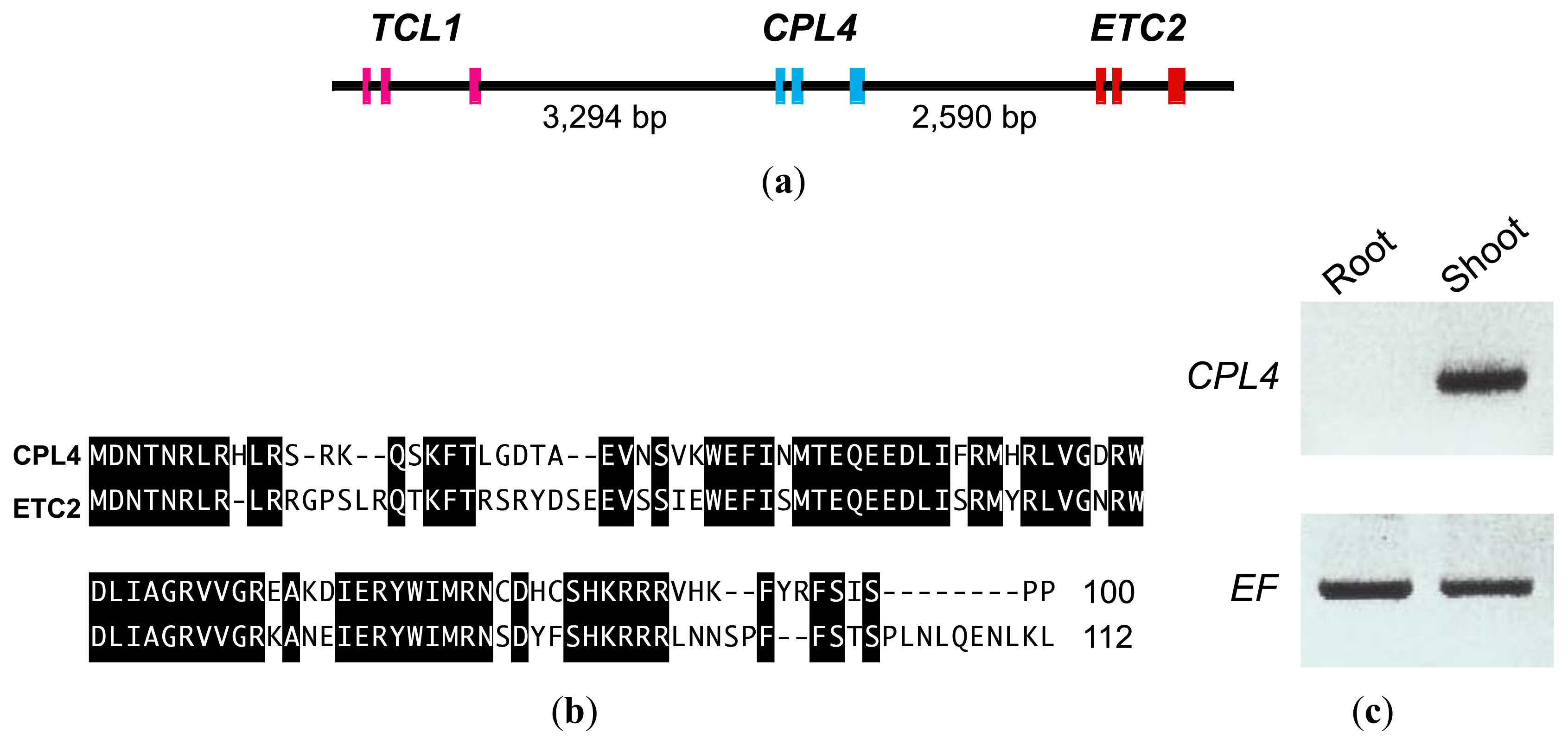
2.1. CPC-LIKE MYB4 (CPL4) Gene in Arabidopsis
2.2. Promoter-GUS Analysis
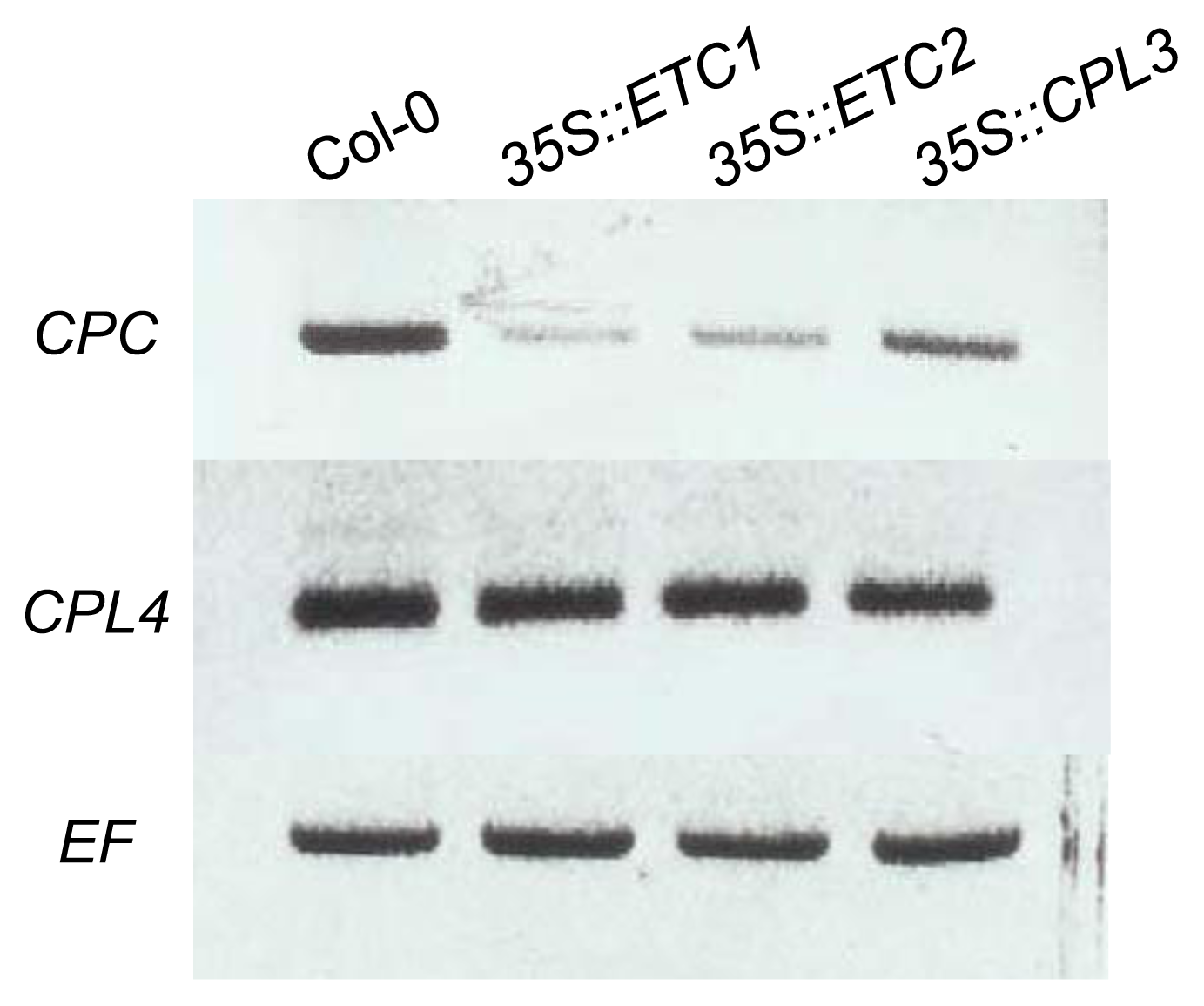
2.3. CPL4 Expression in 35S::ETC1, 35S::ETC2 and 35S::CPL3/ETC3
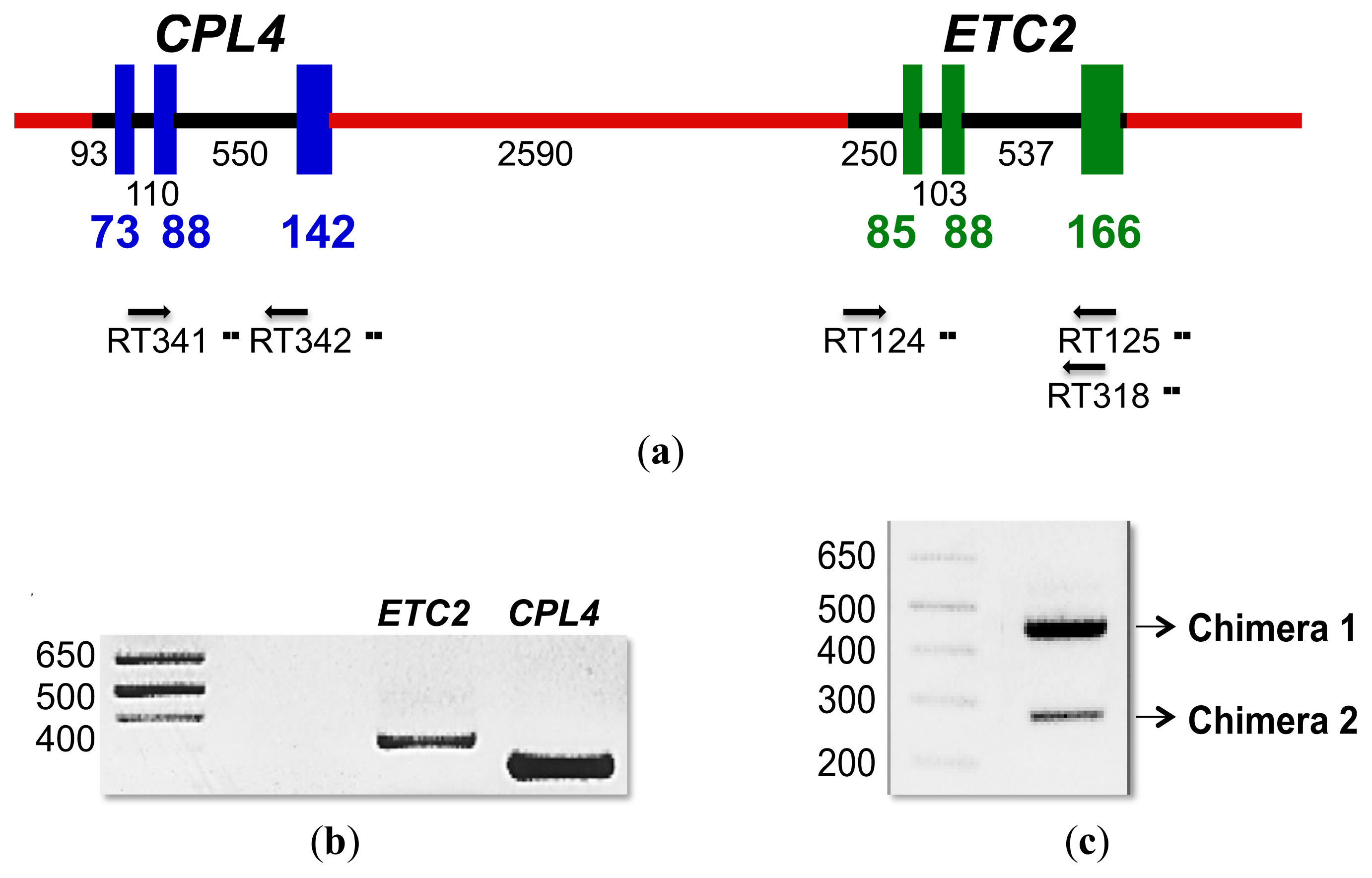
2.4. CPL4-Related Gene
2.4.1. CPL4-Related Gene Expression
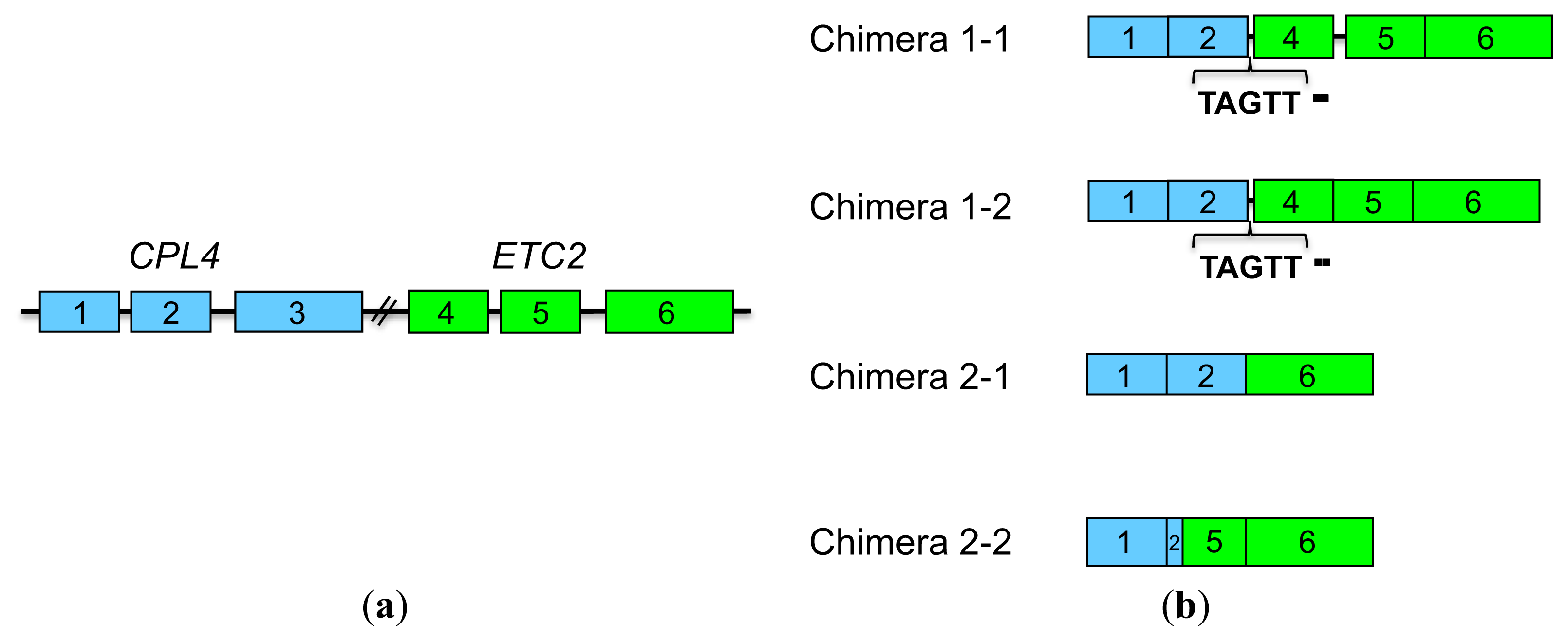
2.4.2. CPL4-Related Chimera Sequences
3. Experimental Section
3.1. Plant Materials and Growth Conditions
3.2. Gene Constructs
3.2.1. Promoter::GUS Constructs
3.2.2. 35S::ETC1 and 35S::ETC2 Constructs
3.3. Transgenic Plants
3.4. RNA Isolation and Semi-Quantitative RT-PCR
3.5. Histology
3.6. Microscopy
3.7. Accession Numbers
4. Conclusions
Acknowledgments
References
- Masucci, J.D.; Rerie, W.G.; Foreman, D.R.; Zhang, M.; Galway, M.E.; Marks, M.D.; Schiefelbein, J.W. The homeobox gene GLABRA2 is required for position-dependent cell differentiation in the root epidermis of Arabidopsis thaliana. Development 1996, 122, 1253–1260. [Google Scholar]
- Lee, M.M.; Schiefelbein, J. WEREWOLF, a MYB-related protein in Arabidopsis, is a position-dependent regulator of epidermal cell patterning. Cell 1999, 99, 473–483. [Google Scholar]
- Rerie, W.G.; Feldmann, K.A.; Marks, M.D. The GLABRA2 gene encodes a homeo domain protein required for normal trichome development in Arabidopsis. Genes Dev 1994, 8, 1388–1399. [Google Scholar]
- di Cristina, M.; Sessa, G.; Dolan, L.; Linstead, P.; Baima, S.; Ruberti, I.; Morelli, G. The Arabidopsis Athb-10 (GLABRA2) is an HD-Zip protein required for regulation of root hair development. Plant J 1996, 10, 393–402. [Google Scholar]
- Oppenheimer, D.G.; Herman, P.L.; Sivakumaran, S.; Esch, J.; Marks, M.D. A myb gene required for leaf trichome differentiation in Arabidopsis is expressed in stipules. Cell 1991, 67, 483–493. [Google Scholar]
- Larkin, J.C.; Oppenheimer, D.G.; Pollock, S.; Marks, M.D. Arabidopsis GLABROUS1 gene requires downstream sequences for function. Plant Cell 1993, 5, 1739–1748. [Google Scholar]
- Kirik, V.; Schnittger, A.; Radchuk, V.; Adler, K.; Hulskamp, M.; Baumlein, H. Ectopic expression of the Arabidopsis AtMYB23 gene induces differentiation of trichome cells. Dev. Biol 2001, 235, 366–377. [Google Scholar]
- Lee, M.M.; Schiefelbein, J. Developmentally distinct MYB genes encode functionally equivalent proteins in Arabidopsis. Development 2001, 128, 1539–1546. [Google Scholar]
- Kang, Y.H.; Kirik, V.; Hulskamp, M.; Nam, K.H.; Hagely, K.; Lee, M.M.; Schiefelbein, J. The MYB23 gene provides a positive feedback loop for cell fate specification in the Arabidopsis root epidermis. Plant Cell 2009, 21, 1080–1094. [Google Scholar]
- Bernhardt, C.; Lee, M.M.; Gonzalez, A.; Zhang, F.; Lloyd, A.; Schiefelbein, J. The bHLH genes GLABRA3 (GL3) and ENHANCER OF GLABRA3 (EGL3) specify epidermal cell fate in the Arabidopsis root. Development 2003, 130, 6431–6439. [Google Scholar]
- Urao, T.; Yamaguchi-Shinozaki, K.; Mitsukawa, N.; Shibata, D.; Shinozaki, K. Molecular cloning and characterization of a gene that encodes a MYC-related protein in Arabidopsis. Plant Mol. Biol 1996, 32, 571–576. [Google Scholar]
- Nesi, N.; Debeaujon, I.; Jond, C.; Pelletier, G.; Caboche, M.; Lepiniec, L. The TT8 gene encodes a basic helix-loop-helix domain protein required for expression of DFR and BAN genes in Arabidopsis siliques. Plant Cell 2000, 12, 1863–1878. [Google Scholar]
- Heim, M.A.; Jakoby, M.; Werber, M.; Martin, C.; Weisshaar, B.; Bailey, P.C. The basic helix-loop-helix transcription factor family in plants: A genome-wide study of protein structure and functional diversity. Mol. Biol. Evol 2003, 20, 735–747. [Google Scholar]
- Galway, M.E.; Masucci, J.D.; Lloyd, A.M.; Walbot, V.; Davis, R.W.; Schiefelbein, J.W. The TTG gene is required to specify epidermal cell fate and cell patterning in the Arabidopsis root. Dev. Biol 1994, 166, 740–754. [Google Scholar]
- Walker, A.R.; Davison, P.A.; Bolognesi-Winfield, A.C.; James, C.M.; Srinivasan, N.; Blundell, T.L.; Esch, J.J.; Marks, M.D.; Gray, J.C. The TRANSPARENT TESTA GLABRA1 locus, which regulates trichome differentiation and anthocyanin biosynthesis in Arabidopsis, encodes a WD40 repeat protein. Plant Cell 1999, 11, 1337–1350. [Google Scholar]
- Payne, C.T.; Zhang, F.; Lloyd, A.M. GL3 encodes a bHLH protein that regulates trichome development in Arabidopsis through interaction with GL1 and TTG1. Genetics 2000, 156, 1349–1362. [Google Scholar]
- Esch, J.J.; Chen, M.; Sanders, M.; Hillestad, M.; Ndkium, S.; Idelkope, B.; Neizer, J.; Marks, M.D. A contradictory GLABRA3 allele helps define gene interactions controlling trichome development in Arabidopsis. Development 2003, 130, 5885–5894. [Google Scholar]
- Zhang, F.; Gonzalez, A.; Zhao, M.; Payne, C.T.; Lloyd, A. A network of redundant bHLH proteins functions in all TTG1-dependent pathways of Arabidopsis. Development 2003, 130, 4859–4869. [Google Scholar]
- Wada, T.; Tachibana, T.; Shimura, Y.; Okada, K. Epidermal cell differentiation in Arabidopsis determined by a Myb homolog, CPC. Science 1997, 277, 1113–1136. [Google Scholar]
- Wada, T.; Okada, K. Development of root-hair and trichome in Arabidopsis. Tanpakushitsu Kakusan Koso 2002, 47, 1599–1604. [Google Scholar]
- Kurata, T.; Ishida, T.; Kawabata-Awai, C.; Noguchi, M.; Hattori, S.; Sano, R.; Nagasaka, R.; Tominaga, R.; Koshino-Kimura, Y.; Kato, T.; et al. Cell-to-cell movement of the CAPRICE protein in Arabidopsis root epidermal cell differentiation. Development 2005, 132, 5387–5398. [Google Scholar]
- Tominaga, R.; Iwata, M.; Okada, K.; Wada, T. Functional analysis of the epidermal-specific MYB genes CAPRICE and WEREWOLF in Arabidopsis. Plant Cell 2007, 19, 2264–2277. [Google Scholar]
- Bernhardt, C.; Zhao, M.; Gonzalez, A.; Lloyd, A.; Schiefelbein, J. The bHLH genes GL3 and EGL3 participate in an intercellular regulatory circuit that controls cell patterning in the Arabidopsis root epidermis. Development 2005, 132, 291–298. [Google Scholar]
- Wada, T.; Kurata, T.; Tominaga, R.; Koshino-Kimura, Y.; Tachibana, T.; Goto, K.; Marks, M.D.; Shimura, Y.; Okada, K. Role of a positive regulator of root hair development, CAPRICE, in Arabidopsis root epidermal cell differentiation. Development 2002, 129, 5409–5419. [Google Scholar]
- Koshino-Kimura, Y.; Wada, T.; Tachibana, T.; Tsugeki, R.; Ishiguro, S.; Okada, K. Regulation of CAPRICE Transcription by MYB Proteins for Root Epidermis Differentiation in Arabidopsis. Plant Cell Physiol 2005, 46, 817–826. [Google Scholar]
- Schellmann, S.; Schnittger, A.; Kirik, V.; Wada, T.; Okada, K.; Beermann, A.; Thumfahrt, J.; Jurgens, G.; Hulskamp, M. TRIPTYCHON and CAPRICE mediate lateral inhibition during trichome and root hair patterning in Arabidopsis. EMBO J 2002, 21, 5036–5046. [Google Scholar]
- Kirik, V.; Simon, M.; Huelskamp, M.; Schiefelbein, J. The enhancer of try and CPC1 gene acts redundantly with triptychon and caprice in trichome and root hair cell patterning in Arabidopsis. Dev. Biol 2004, 268, 506–513. [Google Scholar]
- Kirik, V.; Simon, M.; Wester, K.; Schiefelbein, J.; Hulskamp, M. ENHANCER of TRY and CPC 2 (ETC2) reveals redundancy in the region-specific control of trichome development of Arabidopsis. Plant Mol. Biol 2004, 55, 389–398. [Google Scholar]
- Esch, J.J.; Chen, M.A.; Hillestad, M.; Marks, M.D. Comparison of TRY and the closely related At1g01380 gene in controlling Arabidopsis trichome patterning. Plant J 2004, 40, 860–869. [Google Scholar]
- Simon, M.; Lee, M.M.; Lin, Y.; Gish, L.; Schiefelbein, J. Distinct and overlapping roles of single-repeat MYB genes in root epidermal patterning. Dev. Biol 2007, 311, 566–578. [Google Scholar]
- Tominaga, R.; Iwata, M.; Sano, R.; Inoue, K.; Okada, K.; Wada, T. Arabidopsis CAPRICE-LIKE MYB 3 (CPL3) controls endoreduplication and flowering development in addition to trichome and root hair formation. Development 2008, 135, 1335–1345. [Google Scholar]
- Wang, S.; Kwak, S.H.; Zeng, Q.; Ellis, B.E.; Chen, X.Y.; Schiefelbein, J.; Chen, J.G. TRICHOMELESS1 regulates trichome patterning by suppressing GLABRA1 in Arabidopsis. Development 2007, 134, 3873–3882. [Google Scholar]
- Gan, L.; Xia, K.; Chen, J.G.; Wang, S. Functional characterization of TRICHOMELESS2, a new single-repeat R3 MYB transcription factor in the regulation of trichome patterning in. Arabidopsis. BMC Plant Biol 2011, 11. [Google Scholar] [CrossRef]
- Hulskamp, M.; Misra, S.; Jurgens, G. Genetic dissection of trichome cell development in Arabidopsis. Cell 1994, 76, 555–566. [Google Scholar]
- English, A.C.; Patel, K.S.; Loraine, A.E. Prevalence of alternative splicing choices in. Arabidopsis thaliana. BMC Plant Biol 2010, 10. [Google Scholar] [CrossRef]
- Genetyx Software, version 16.0.2; Genetyx: Tokyo, Japan, 2011.
- Thomas, J.H. Thinking about genetic redundancy. Trends Genet 1993, 9, 395–399. [Google Scholar]
- Cooke, J.; Nowak, M.A.; Boerlijst, M.; Maynard-Smith, J. Evolutionary origins and maintenance of redundant gene expression during metazoan development. Trends Genet 1997, 13, 360–364. [Google Scholar]
- Zimmermann, I.M.; Heim, M.A.; Weisshaar, B.; Uhrig, J.F. Comprehensive identification of Arabidopsis thaliana MYB transcription factors interacting with R/B-like BHLH proteins. Plant J 2004, 40, 22–34. [Google Scholar]
- Eckardt, N.A. Alternative splicing and the control of flowering time. Plant Cell 2002, 14, 743–747. [Google Scholar]
- Okada, K.; Shimura, Y. Reversible root tip rotation in Arabidopsis seedlings induced by obstacle-touching stimulus. Science 1990, 250, 274–276. [Google Scholar]
- Jarvis, P.; Chen, L.J.; Li, H.; Peto, C.A.; Fankhauser, C.; Chory, J. An Arabidopsis mutant defective in the plastid general protein import apparatus. Science 1998, 282, 100–103. [Google Scholar]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 1998, 16, 735–743. [Google Scholar]
- Kurata, T.; Kawabata-Awai, C.; Sakuradani, E.; Shimizu, S.; Okada, K.; Wada, T. The YORE-YORE gene regulates multiple aspects of epidermal cell differentiation in Arabidopsis. Plant J 2003, 36, 55–66. [Google Scholar]


| Primer Name | Sequence |
|---|---|
| RT124 | 5′-GATAATACCAACCGTCTTCGTCTTC-3′ |
| RT125 | 5′-TTCTTGGAGATTAAGAGGAGAAGTAG-3′ |
| RT128 | 5′-CTTCTTGTTTCTCGAGATTTATTCTC-3′ |
| RT129 | 5′-AATAGTAATTCAAGGACAGGTACATTTC-3′ |
| RT318 | 5′-GAATTATTAAGACGTCGTCGTTTGTGAG-3′ |
| RT341 | 5′-AAGCAATCCAAGTTCACTCTAGGG-3′ |
| RT342 | 5′-CGGTAAATTTGTGGACTCGG-3′ |
| NEKO45 | 5′-ATATGTCGACTACCAAAATCACTCCACCATTTTC-3′ |
| NEKO47 | 5′-ATATGGATCCGTTGGTGTTATCCATTGGTATTTG-3′ |
| TW1165 | 5′-ATATGGTACCAATAAAAAATAAATCAC-3′ |
| TW1166 | 5′-TGCTTGTCGACTGTATACACTAA-3′ |
| TW1169 | 5′-ATATGGTACCACTTCATGTTCTTCCCTT-3′ |
| TW1170 | 5′-ATATGTCGACAAGCCAATACATATCCA-3′ |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tominaga-Wada, R.; Nukumizu, Y. Expression Analysis of an R3-Type MYB Transcription Factor CPC-LIKE MYB4 (TRICHOMELESS2) and CPL4-Related Transcripts in Arabidopsis. Int. J. Mol. Sci. 2012, 13, 3478-3491. https://doi.org/10.3390/ijms13033478
Tominaga-Wada R, Nukumizu Y. Expression Analysis of an R3-Type MYB Transcription Factor CPC-LIKE MYB4 (TRICHOMELESS2) and CPL4-Related Transcripts in Arabidopsis. International Journal of Molecular Sciences. 2012; 13(3):3478-3491. https://doi.org/10.3390/ijms13033478
Chicago/Turabian StyleTominaga-Wada, Rumi, and Yuka Nukumizu. 2012. "Expression Analysis of an R3-Type MYB Transcription Factor CPC-LIKE MYB4 (TRICHOMELESS2) and CPL4-Related Transcripts in Arabidopsis" International Journal of Molecular Sciences 13, no. 3: 3478-3491. https://doi.org/10.3390/ijms13033478




