Valuable Nutrients and Functional Bioactives in Different Parts of Olive (Olea europaea L.)—A Review
Abstract
:1. Introduction
2. Distribution, Cultivation and Production of Table Olives and Olive Oil
- Spanish-style (pickled) green olives in brine
- Californian-style (pickled) black olives in brine
- Greek-style natural black olives in brine
3. Composition of High-Value Nutrients and Functional Bioactives in Different Parts of Olive
3.1. Olive Fruit
3.2. Olive Oil
3.2.1. Fatty Acid Composition of Olive Oil
3.2.2. Olive Oil Phenolics
3.2.2.1. Biological Activities and Potential Health Benefits of Olive Oil Biophenols
3.2.3. Volatile and Aromatic Compounds
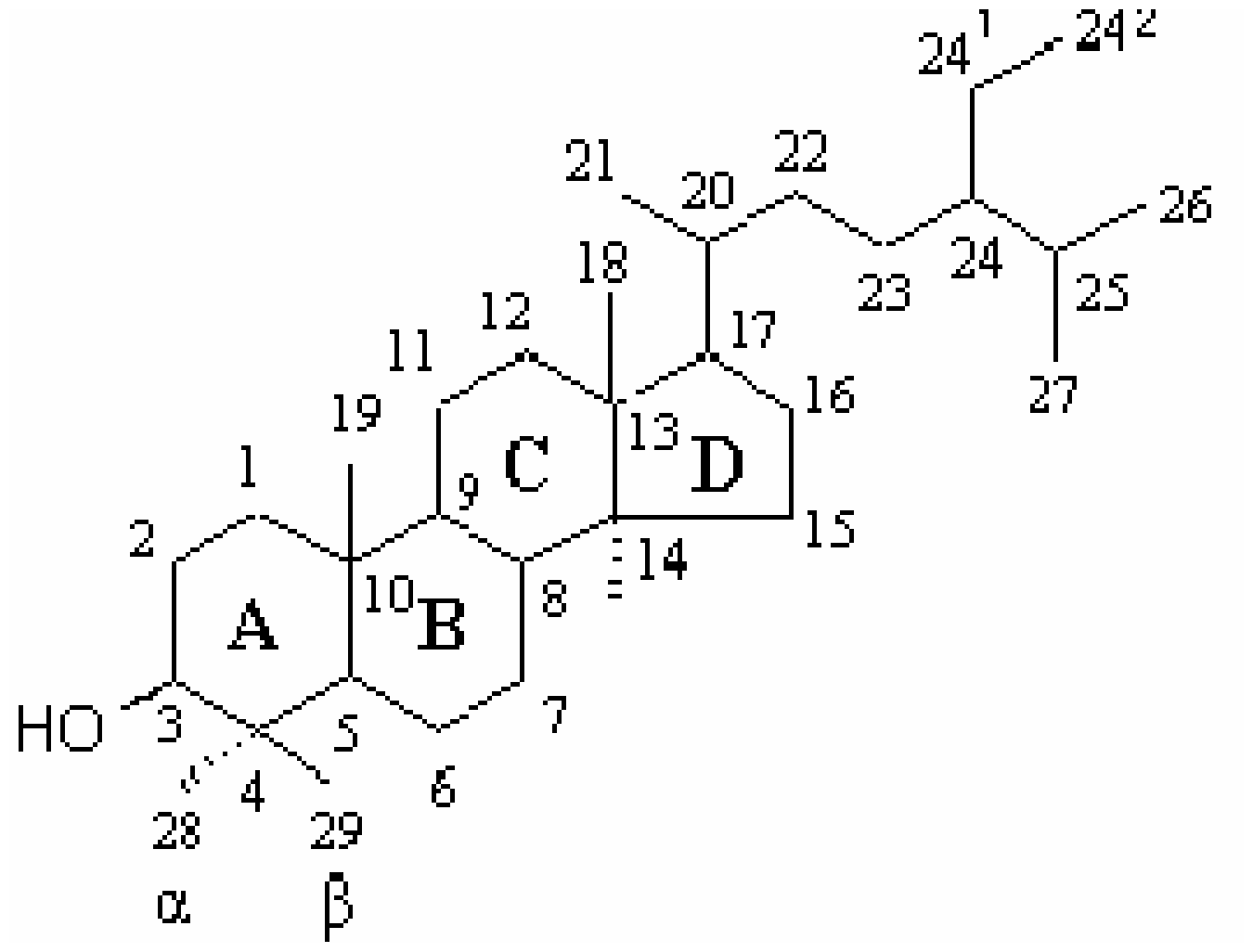
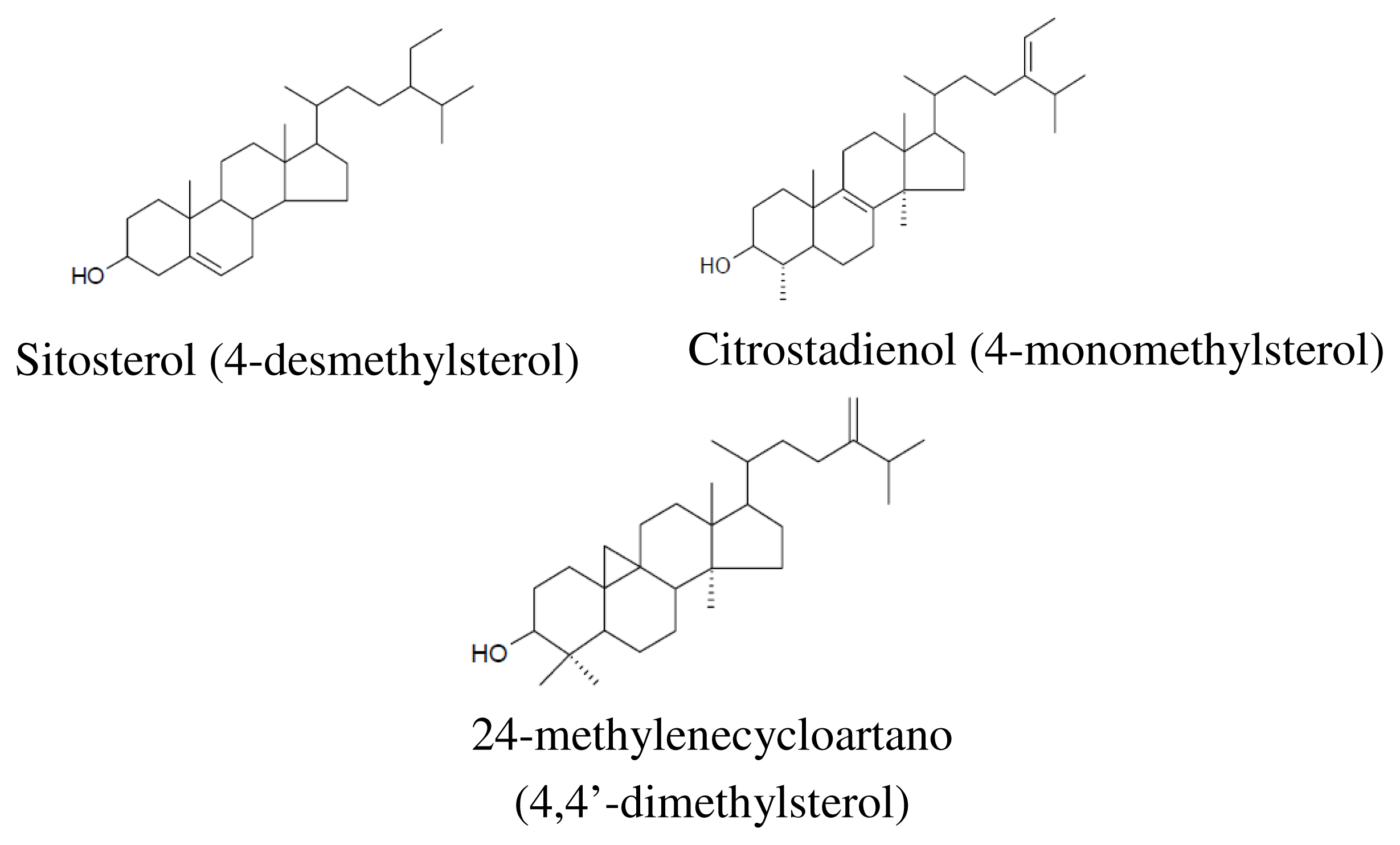
3.2.4. Phytostrols
3.2.5. Tocopherols
3.2.6. Colouring Pigments
3.2.7. Squalene
3.2.8. Triterpene Dialcohols
3.3. Olive Leaf
3.4. Olive by-Products
3.4.1. Olive Cake
3.4.2. Olive Oil Mill Waste Water
3.4.3. Olive Stone
3.4.4. Olive Wood
4. Factors Affecting Chemical Composition of Olives
4.1. Cultivars
4.2. Fruits Maturity
4.3. Irrigation
4.4. Technological Aspects in Olive Oil Extraction
4.4.1. Crushing
4.4.2. Malaxation
4.4.3. Decantation
4.4.4. Filtration
5. Potential for Recovery of Valuable Olive Natural Constituents
6. Conclusions
7. Future Prospects
References
- Boskou, D. History and characteristics of the olive tree. In Olive Oil Chemistry and Technology; Boskou, D., Ed.; Am. Oil Chem. Soc. Press: Champaign, IL, USA, 1996. [Google Scholar]
- Ryan, D.; Robards, K. Phenolic compounds in olives. Analyst 1998, 123, 31R–44R. [Google Scholar]
- Gooch, E. Ten plus one things you may not know about olive. Epikouria Magazine. 2005. Available online: http://www.epikouria.com/issue1/10+1-things-olives.php accessed on 5 November 2011.
- Soler-Rivas, C.; Epsin, J.C.; Wichers, H.J. Oleuropein and related compounds. J. Sci. Food Agric 2000, 80, 1013–1023. [Google Scholar]
- Ribarova, F.; Zanev, R.; Shishkov, S.; Rizov, N. α-Tocopherol, fatty acids and their correlations in Bulgarian foodstuffs. J. Food Compos. Anal 2003, 16, 659–667. [Google Scholar]
- Vinha, A.F.; Ferreres, F.; Silva, B.M.; Valentão, P.; Gonçalves, A.; Pereira, J.A.; Oliveira, M.B.; Seabra, R.M.; Andrade, P.B. Phenolic profiles of Portuguese olive fruits (Olea europaea L.): Influences of cultivar and geographical origin. Food Chem 2005, 89, 561–568. [Google Scholar]
- Knoops, K.T.; de Groot, L.C.; Kromhout, D. Mediteranean diet, lifestyle factors, and 10-year mortality in elderly European men and women. J. Am. Med. Assoc 2004, 292, 1433–1439. [Google Scholar]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a mediterranean diet and survival in a Greek population. N. Engl. J. Med 2003, 348, 2599–2608. [Google Scholar]
- Covas, M.I.; Nyyssonen, K.; Poulsen, H.E. The effect of polyphenols in olive oil on heart disease risk factors. Ann. Int. Med 2006, 145, 333–431. [Google Scholar]
- Covas, M.I. Bioactive effects of olive oil phenolic compounds in humans: Reduction of heart disease factors and oxidative damage. Inflammopharmacology 2008, 16, 216–218. [Google Scholar]
- IOOC Home Page (International Olive Oil Council Activities: World Olive Oil Figures: World Olive Oil Consumption). Available online: www.internationaloliveoil.org accessed on 25 November 2011.
- Fernández, A.G.; Díez, M.J.F.; Adams, M.R. Table Olives: Production and Processing; Chapman & Hall: London, UK, 1997; p. 478. [Google Scholar]
- Fernandez Diez, M.J. Olives. In Biotechnology; Rehm, H.J., Reed, J., Eds.; Verlag Chemie: Weinheim, Germany, 1983; Volume 5, pp. 379–397. [Google Scholar]
- FAO Home Page. Available online: www.fao.org accessed on 18 May 2009.
- FAOSTAT Crops Processed Data for Olive Oil; FAO: Rome, Italy, 2009. Available online: http://faostat.fao.org/site/636/DesktopDefault.aspx?PageID=636#ancor accessed on 5 October 2011.
- Trade Standard Applying to Olive Oils and Olive-Pomace Oils; COI/ T.15/NC no. 3/Rev. 6. Internnationa Olive Council: Madrid, Spain, 2011. Available online: http://www.internationaloliveoil.org accessed on 20 December 2011.
- Luchetti, F. Introduction. In Handbook of Olive Oil: Analysis and Properties; Harwood, J.A., Aparicio, R., Eds.; Aspen Publishers, Inc: Gaithersburg, MD, USA, 2000; pp. 1–16. [Google Scholar]
- Boskou, D.; Blekas, G.; Tsimidou, M. Olive oil composition. In Olive Oil: Chemistry and Technology; Boskou, D., Ed.; Am. Oil Chem. Soc. Press: Champaign, IL, USA, 2006; pp. 1–33. [Google Scholar]
- Niaounakis, M.; Halvadakis, C.P. Characterization of Olive Processing Waste. In Waste Management Series, 2nd ed; Elsevier: Amsterdam, the Netherlands, 2006; Volume 5, Chapter 2; pp. 23–64. [Google Scholar]
- Cunha, S.; Ferreira Isabel, M.P.L.V.O.; Fernandes, J.O.; Faria, M.A.; Beatriz, M.; Oliveira, P.P. Determination of lactic, acetic, succinic and citric acids in table olive by HPLC/UV. J. Liq. Chromatogr. Relat. Technol 2001, 24, 1029–1038. [Google Scholar]
- Bianchi, G. Lipids and phenols in table olives. Eur. J. Lipid Sci. Technol 2003, 105, 229–242. [Google Scholar]
- Andreasen, M.F.; Christensen, L.P.; Meyer, A.S.; Hansen, Å. Content of phenolic acids and ferulic acid dehydrodimers in 17 rye (Secale cereale L.) varieties. J. Agric. Food Chem 2000, 48, 2837–2842. [Google Scholar]
- Prim, N.; Pastor, F.I.J.; Diaz, P. Biochemical studies on cloned Bacillus sp. BP-7 phenolic acid decarboxylase PadA. Appl. Microbiol. Biotechnol 2003, 63, 51–56. [Google Scholar]
- Kountouri, A.M.; Mylona, A.; Kaliora, A.C.; Andrikopoulos, N.K. Bioavailability of the phenolic compounds of the fruits (drupes) of Olea europaea (olives): Impact on plasma antioxidant status in humans. Phytomedicine 2007, 14, 659–667. [Google Scholar]
- Viola, P.; Viola, M. Virgin olive oil as a fundamental nutritional component and skin protector. Clin. Dermatol 2009, 27, 159–165. [Google Scholar]
- Servili, M.; Baldioli, M.; Selvaggini, R.; Macchioni, A.; Montedor, G. Phenolic compounds of olive fruit: One- and two-dimensional nuclear magnetic resonance characterization of nüzhenide and its distribution in the constitutive parts of fruit. J. Agric. Food Chem 1999, 47, 12–18. [Google Scholar]
- Andary, C.; Wylde, R.; Laffite, C.; Privat, G.; Winternitz, F. Structure of verbascoside and orobancoside, caffeic acid, suger esters from orobanche rapum-genistae. Phytochemistry 1982, 21, 1123–1127. [Google Scholar]
- Obied, H.K.; Allen, M.S.; Bedgood, D.R.; Prenzler, P.D.; Robards, K.; Stockmann, R. Bioactivity and analysis of biophenols recovered from olive mill waste. J. Agric. Food Chem 2005, 53, 823–837. [Google Scholar]
- Romero, C.; Brenes, M.; Garcia, P.; Garrido, A. Hydroxytyrosol 4-a-Dglucoside, an important phenolic compound in olive fruits and derived products. J. Agric. Food Chem 2002, 50, 3835–3839. [Google Scholar]
- Amiot, M.J.; Fleuriet, A.; Macheix, J. Importance and evolution of phenolic compounds in olive during growth and maturation. J. Agric. Food Chem 1986, 34, 823–826. [Google Scholar]
- Romani, A.; Mulinacci, N.; Pinelli, P.; Vincieri, F.F.; Cimato, A. Polyphenolic content in five tuscany cultivars of Olea europaea L. J. Agric. Food Chem 1999, 47, 964–967. [Google Scholar]
- Juan, M.; Planas, J.; Ruiz-Gutierrez, V.; Daniel, H.; Wenzel, U. Antiproliferative and apoptosis-inducing effects of maslinic and oleanolic acids, two pentacyclic triterpenes from olives, on HT-29 colon cancer cells. Br. J. Nutr 2008, 100, 36–43. [Google Scholar]
- Fernández-Bolaños, J.; Rodríguez, G.; Rodríguez, R.; Guillén, R.; Jiménez, A. Extraction of interesting organic compounds from olive oil waste. Grasas Aceites 2006, 57, 95–106. [Google Scholar]
- Ragazzi, E.; Veronese, G.; Guitto, A. The demethyloleuropein, a new glucoside extracted from ripe olives. Ann. Chim 1973, 63, 13–20. [Google Scholar]
- Kubo, I.; Matsumoto, A. Molluscicides from olives Olea europaea and their efficient isolation by counter current chromatography. J. Agric. Food Chem 1984, 32, 687–688. [Google Scholar]
- Vasquez, R.A.; Costante, E.G.; Duran, R.M. Components fenolicos de la aceituna. Polifenoles de la pulpa. Grasas Aceites 1974, 25, 269–279. [Google Scholar]
- Mannino, S.; Cosio, M.S.; Bertuccioli, M. High performance liquid chromatography of phenolic compounds in virgin olive oil using amperometryc detector. Ital. J. Food Sci 1993, 4, 363–370. [Google Scholar]
- Panizzi, L.; Scarpati, M.L.; Oriente, G. Chemical structure of Oleuropein, bitter glucoside of olive with hypotensive activity. Gazz. Chim. Ital 1960, 90, 1449–1458. [Google Scholar]
- Bourquelot, E.; Vintilesco, J.C.R. Sur l oleuropein, nouveau principle de nature glucosidique reter de lolivier (Olea europaea L.). Compt. Rend. Hebd. Acad. Sci 1908, 147, 533–535. [Google Scholar]
- Huang, C.L.; Sumpio, B.E. Olive oil, the Mediterranean diet, and cardiovascular health. J. Am. Coll. Surg 2008, 207, 407–416. [Google Scholar]
- Juan, M.E.; Wenzel, U.; Ruiz-Gutierrez, V.; Daniel, H.; Planas, J.M. Olive fruit extracts inhibit proliferation and induce apoptosis in HT-29 human colon cancer cells. J. Nutr 2006, 136, 2553–2557. [Google Scholar]
- Gilani, A.H.; Khan, A.U.; Shah, A.J.; Connor, J.; Jabeen, Q. Blood pressure lowering effect of olives is mediated through calcium channel blockade. Int. J. Food Sci. Nutr 2005, 56, 613–620. [Google Scholar]
- Gilani, A.H.; Khan, A.U.; Shah, A.J. Calcium antagonist and cholinomimetic activities explain the medicinal uses of olives in gut disorders. Nutr. Res 2006, 26, 277–283. [Google Scholar]
- Gilani, A.H.; Khan, A.U. Medicinal value of novel combination of cholinergic and calcium antagonist constituents in olive. In Olives and Olive Oil in Health and Disease Prevention; Preedy, V.R., Watson, R.R., Eds.; Academic Press Elsevier: Amsterdam, the Netherlands, 2009; pp. 835–843. [Google Scholar]
- Gavriilidou, V.; Boskou, D. Chemical interesterification of olive oil-tristearin blends for margarines. Int. J. Food Sci. Technol 1991, 26, 451–456. [Google Scholar]
- Aparicio, R.; Aparicio-Ruíz, R. Authentication of vegetable oils by chromatographic techniques. J. Chromatogr. A 2000, 881, 93–104. [Google Scholar]
- Artajo, L.S.; Romero, M.P.; Morelloa, J.R.; Motilva, M.J. Enrichment of refined olive oil with phenolic compounds; evaluation of their antioxidant activity and their effect on the bitter index. J. Agric. Food Chem 2006, 54, 6079–6088. [Google Scholar]
- Tuck, K.L.; Hayball, P.J. Major phenolic compounds in olive oil: Metabolism and health effects. J. Nutr. Biochem 2002, 13, 636–644. [Google Scholar]
- Kochhar, S.P. The composition of frying oils. In Frying Improving Quality; Rossel, J.B., Ed.; Woodhead Publishing Ltd: Cambridge, UK, 2001; pp. 87–114. [Google Scholar]
- Monounsaturated fatty acids from olive oil and coronary heart disease. In Health Claim Petition Docket No. 2003Q-0559; FDA: Rome, Italy, 2004.
- Grundy, S.M. Comparison of monounsaturated fatty acids and carbohydrates for lowering plasma cholesterol. N. Engl. J. Med 1986, 314, 745–748. [Google Scholar]
- Ranalli, A.; Angerosa, F. Integral centrifuges for olive oil extraction. The qualitative characteristics of products. J. Am. Oil Chem. Soc 1996, 73, 417–422. [Google Scholar]
- Covas, M.I. Olive oil and the cardiovascular system-Review. Pharm. Res 2007, 55, 175–186. [Google Scholar]
- Allalout, A.; Krichene, D.; Methenni, K.; Taamalli, A.; Oueslati, I.; Daoud, D. Characterization of virgin olive oil from super intensive Spanish and Greek varieties grown in northern Tunisia. Sci. Hort 2009, 120, 77–83. [Google Scholar]
- Paz Aguilera, M.; Beltran, G.; Ortega, D.; Fernandez, A.; Jimenez, A.; Uceda, M. Characterisation of virgin olive oil of Italian olive cultivars: ‘Frantoio’ and ‘Leccino’, grown in Andalusia. Food Chem 2005, 89, 387–391. [Google Scholar]
- Skevin, D.; Rade, D.; Strucelj, D.; Mokrovãak, Z.; Nederal, S.; Benãiç, D. The influence of variety and harvest time on the bitterness and phenolic compounds of olive oil. Eur. J. Lipid Sci. Technol 2003, 105, 536–541. [Google Scholar]
- Boskou, D. Polar phenolic compounds. In Olive Oil: Chemistry and Technology; Boskou, D., Ed.; Am. Oil Chem. Soc. Press: Champaign, IL, USA, 2006; pp. 73–92. [Google Scholar]
- Naczk, M.; Shahidib, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar]
- Boskou, D.; Blekas, G.; Tsimidou, M. Phenolic compounds in olive oil and olives. Curr. Top. Nutraceutical Res 2005, 3, 125–136. [Google Scholar]
- Baldioli, M.; Servili, M.; Perretti, G.G.F. Montedoro antioxidant activity of tocopherols and phenolic compounds of virgin olive oil. J. Am. Oil Chem. Soc 1996, 73, 1583–1593. [Google Scholar]
- Bianco, A.; Coccioli, F.; Guiso, M.; Marra, C. The occurrence in olive oil of a new class of phenolic compounds: Hydroxy-isochromans. Food Chem 2001, 77, 405–411. [Google Scholar]
- Montedoro, G.F. Phenolic constituents of virgin olive oils. I. Identification of some phenolic acids and their antioxidant capacity. Sci. Technol. Alimenti 1972, 3, 177–186. [Google Scholar]
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic molecules in virgin olive oils: A survey of their sensory properties, health effects, antioxidant activity and analytical methods—An overview of the last decade. Molecules 2007, 12, 1679–1719. [Google Scholar]
- Owen, R.W.; Mier, W.; Giacosa, A.; Hull, W.E.; Spiegelhalder, B.; Bartsch, H. Phenolic compounds and sequalene in olive oils: the concentration and antioxidant potential of total phenols, simple phenols, secoiridoids, lignans and sequalene. Food Chem. Toxicol 2000, 38, 647–659. [Google Scholar]
- Rovellini, P.; Cortesi, N. Liquid chromatography-mass spectrometry in the study of oleuropein and ligstroside aglycons in virgin olive oils: Aldehydic, dialdehydic forms and their oxidized products. Riv. Ital. Sostanze Grasse 2002, 79, 1–14. [Google Scholar]
- Montedoro, G.F.; Servili, M. Tradizone ed Innovazioni Tecnologiche nell Estrazione degli Olii Extravergini Di Oliva. Proceedings of International Congress “Olive Oil Quality”, Firenze, Italy, 1–3 December, 1992; pp. 97–108.
- Brenes, M.; García, A.; García, P.; Garrido, A. Acid hydrolysis of secoiridoid aglycons during storage of virgin olive oil. J. Agric. Food Chem 2001, 49, 5609–5614. [Google Scholar]
- Rovellini, P.; Cortesi, N.; Fedeli, E. Analysis of flavonoids from olea europaea by HPLC-UV and HPLC-electrospray-MS. Riv. Ital. Sostanze Grasse 1997, 74, 273–279. [Google Scholar]
- Brenes, M.; García, A.; Rios, J.J.; García, P.; Garrido, A. Use of 1-acetoxypinoresinol to authenticate Picual olive oils. Int. J. Food Sci. Technol 2002, 37, 615–625. [Google Scholar]
- Bianco, A.; Buiarelli, F.; Cartoni, G.; Coccioli, F.; Muzzalupo, I.; Polidori, A.; Uccella, N. Anlaysis by HPLC-MS/MS of biophenolic components in olives and oils. Anal. Lett 2001, 34, 1033–1051. [Google Scholar]
- Angerosa, F.; Alessandro, N.D.; Corana, F.; Mellerio, G. Characterisation of phenolic and secoiridoid aglycons present in virgin olive oil by gas chromatography-chemical ionization mass spectrometry. J. Agric. Food Chem 1995, 43, 1802–1807. [Google Scholar]
- Perri, E.; Raffaelli, A.; Sindona, G. Quantitation of oleuropein in virgin olive oil by ionspray mass spectrometry-selected reaction monitoring. J. Agric. Food Chem 1999, 47, 4156–4160. [Google Scholar]
- Montedoro, G.; Servili, M.; Baldioli, M.; Miniati, E. Simple and hydrolyzable phenolic compounds in virgin olive oil. 1. Their extraction, separation, and quantitative and semiquantitative evaluation by HPLC. J. Agric. Food Chem 1992, 40, 1571–1576. [Google Scholar]
- Jenner, P.; Olanow, C.W. Oxidative stress and the pathogenesis of Parkinson’s disease. Neurology 1996, 47, 161S–170S. [Google Scholar]
- Visioli, F.; Galli, C.; Bornet, F.; Mattei, A.; Patelli, R.; Galli, G.; Caruso, D. Olive oil phenolics are dose-dependently absorbed in humans. FEBS Lett 2000, 486, 159–160. [Google Scholar]
- Aviram, M. Interaction of oxidized low density lipoprotein with macrophages in atherosclerosis, and the antiatherogenicity of antioxidants. Eur. J. Clin. Chem. Clin. Biochem 1996, 34, 599–608. [Google Scholar]
- Marrugat, J.; Covas, M.-I.; Fitó, M.; Schröder, H.; Miró-Casas, E.; Gimeno, E.; López-Sabater, M.; Torre, R.; Farré, M. Effects of differing phenolic content in dietary olive oils on lipids and LDL oxidation.arandomized controlled trial. Eur. J. Nutr 2004, 43, 140–147. [Google Scholar]
- Caruso, D.; Visioli, F.; Patelli, R.; Galli, C.; Galli, G. Urinary excretion of olive oil phenols and their metabolites in humans. Metabolism 2001, 50, 1426–1428. [Google Scholar]
- Miro-Casas, E.; Covas, M.-I.; Farre, M.; Fito, M.; Ortuño, J.; Weinbrenner, T.; Roset, P.; Torre, R.D.L. Hydroxytyrosol disposition in humans. Clin. Chem 2003, 49, 945–952. [Google Scholar]
- Mensink, R.P.; Janssen, M.C.; Katan, M.B. Effect on blood pressure of two diets differing in total fat but not in saturated and polyunsaturated fatty acids in healthy volunteers. Am. J. Clin. Nutr 1988, 47, 976–980. [Google Scholar]
- Rasmussen, O.; Thomsen, C.; Hansen, K.W.; Vesterlund, M.; Winther, E.; Hermansen, K. Effects on blood pressure, glucose, and lipid levels of a high-monounsaturated fat diet compared with a high-carbohydrate diet in NIDDM subjects. Diabetes Care 1993, 16, 1565–1571. [Google Scholar]
- Ruız-Gutierrez, V.; Muriana, F.J.; Guerrero, A.; Cert, A.M.; Villar, J. Plasma lipids, erythrocyte membrane lipids and blood pressure of hypertensive women after ingestion of dietary oleic acid from two different sources. J. Hypertens 1996, 14, 1483–1490. [Google Scholar]
- Fitó, M.; Cladellas, M.; Torre, R.D.L.; Martí, J.; Alcántara, M.; Pujadas-Bastardes, M.; Marrugat, J.; Bruguera, J.; López-Sabater, M.C.; Vila, J. Antioxidant effect of virgin olive oil in patients with stable coronary heart disease: A randomised, crossover, controlled, clinical trial. Atherosclerosis 2005, 181, 149–158. [Google Scholar]
- Martin-Moreno, J.M.; Willet, W.C.; Gorgoio, L.; Banegas, J.R.; Rodriguez-Artalejo, F.; Fernandez-Rodriguez, J.C.; Maisonneuve, P.; Boyle, P. Dietary fat, olive oil intake and breast cancer risk. Int. J. Cancer 1994, 58, 774–780. [Google Scholar]
- Trichopoulou, A.; Katsouyanni, K.; Stuver, S.; Tzala, L.; Gnardellis, C.; Rimm, E.; Trichopoulo, D. Consumption of olive oil and specific food groups in relation to breast cancer risk in Greece. J. Natl. Cancer Inst 1995, 87, 110–116. [Google Scholar]
- Vecchia, C.L.; Negri, E.; Franceschi, S.; Favero, A.; Nanni, O.; Filiberti, R.; Conti, E.; Montella, M.; Veronesi, A.; Ferraroni, M. Hormone replacement treatment and breast cancer risk: A cooperative Italian study. Br. J. Cancer 1995, 72, 244–248. [Google Scholar]
- Soler, M.; Chatenaud, L.; Vecchia, C.L.; Franceschi, S.; Negri, S. Diet, alcohol, coffee and pancreatic cancer: Final results from an Italian study. Eur. J. Cancer Prev 1998, 7, 455–460. [Google Scholar]
- Franceschi, S.; Favero, A.; Conti, E.; Salamini, R.; Volpe, R.; Negri, E.; Barman, L.; Vecchia, C.L. Food groups, oils and butter, and cancer of the oral cavity and pharynx. Br. J. Cancer 1999, 80, 614–620. [Google Scholar]
- Bosetti, C.; Trichopoulou, A.; Franceschi, S.; Negri, E.; Vecchia, C.L. Influence of the Mediterranean diet on the risk of cancers of the upper aerodigestive. Tract. Cancer Epidemiol. Biomarkers Prev 2003, 12, 1091–1094. [Google Scholar]
- Stoneham, M.; Goldacre, M.; Seagroatt, V.; Gill, L.; Epidemiol, J. Olive oil, diet and colorectal cancer: An ecological study and a hypothesis. J. Epidemiol. Community Health 2000, 54, 756–760. [Google Scholar]
- Hodge, E.; English, D.R.; McCredie, M.R.E.; Severi, G.; Boyle, P.; Hopper, J.L.; Giles, G.G. Foods, nutrients and prostate cancer. Cancer Causes Control 2004, 15, 11–20. [Google Scholar]
- Fortes, C.; Forestiere, F.; Farchi, S.; Mallone, S.; Trequattrini, T.; Anatra, F.; Schmid, G.; Peducci, C.A. the protective effect of the mediterranean diet on lung cancer. Nutr. Cancer 2003, 46, 30–37. [Google Scholar]
- Ichihashi, M.; Ueda, M.; Budiyanto, A.; Bito, T.; Oka, M.; Fukunaga, M.; Tsuru, K.; Horikawa, T. UV-Induced skin Damage. Toxicology 2003, 189, 21–39. [Google Scholar]
- Kohyama, N.; Nagata, T.; Fujimoto, S. Inhibition of arachidonate lipoxygenase activities by 2-(3,4-dihydroxyphenyl) ethanol, a phenolic compound from olives. Biosci. Biotechnol. Biochem 1997, 61, 347–350. [Google Scholar]
- Petroni, A.; Blasevich, M.; Salami, M.; Papini, N.; Montedoro, G.F.; Galli, C. Inhibition of platelet aggregation and eicosanoid production by phenolic components of olive oil. Thromb. Res 1995, 78, 151–160. [Google Scholar]
- Puerta, R.D.L; Gutierrez, V.R.; Hoult, J.R. Inhibition of leukocyte 5-lipoxygenase by phenolics from virgin olive oil. Biochem. Pharmacol 1999, 57, 445–449. [Google Scholar]
- Turner, R.; Etienne, N.; Alonso, M.G. Antioxidant and anti-atherogenic activities of olive oil phenolics. Int. J. Vitam. Nutr. Res 2005, 75, 61–70. [Google Scholar]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Covas, M.I. Effects of a Mediterranean- style diet on cardiovascular risk factors: A randomized trial. Ann. Intern. Med 2006, 145, 1–11. [Google Scholar]
- Visioli, F.; Bellomo, G.; Montedoro, G.F.; Galli, C. Low density lipoprotein oxidation is inhibited in vitro by olive oil constituents. Atherosclerosis 1995, 117, 25. [Google Scholar]
- Ruano, J.; Lopez-Miranda, J.; Fuentes, F.; Moreno, J.A.; Bellido, C.; Perez-Martinez, P.; Lozano, A.; Gómez, P.; Jiménez, Y.; Jiménez, F.P. Phenolic content of virgin olive oil improves ischemic reactive hyperemia in hypercholesterolemic patients. J. Am. Coll. Cardiol 2005, 46, 1864–1868. [Google Scholar]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol 2003, 23, 622–629. [Google Scholar]
- Dejana, M.; Trauma, O.I.; Bianchi, M.P.; Spencer, J.P.E.; Harparkash, K.; Halliwell, B.; Haeschbach, R.; Banni, S.; Dessi, M.A.; Corongiu, F. Inhibition of peroxynitrite dependent DNA base modification and tyrosine nitration by the extra virgin olive oil-derived antioxidant hydroxytyrosol. Free Radic. Biol. Med 1999, 26, 762–769. [Google Scholar]
- Machowetz, A.; Poulsen, H.E.; Gruendel, S.; Weimann, A.; Fitó, M.; Marrugat, J.; Torre, R.D.L.; Salonen, J.T.; Nyyssönen, K.; Mursu, J.; et al. Effect of olive oils on biomarkers of oxidative DNA stress in Northern and Southern Europeans. FASEB J 2007, 21, 45–52. [Google Scholar]
- Perona, J.S.; Cabello-Moruno, R.; Ruiz-Gutierrez, V. The role of virgin olive oil components in the modulation of endothelial function. J. Nutr. Biochem 2006, 17, 429–445. [Google Scholar] [Green Version]
- Martinez-Dominguez, E.; de la Puerta, R.; Ruiz-Gutierrez, V. Protective effects upon experimental inflammation models of a polyphenol-supplemented virgin olive oil diet. Inflamm. Res 2001, 50, 102–106. [Google Scholar]
- Aziz, N.H.; Farag, S.E.; Mousa, L.A.A.; Abo-Zaid, M.A. Comparative antibacterial and antifungal effects of some phenolic compounds. Microbios 1998, 93, 43–54. [Google Scholar]
- Gourama, H.; Bullerman, L.B. Effects of oleuropein on growth and aflatoxin production by Aspergillus parasiticus. Lebensm. Wiss. Technol 1987, 20, 226–228. [Google Scholar]
- Deiana, M.; Rosa, A.; Cao, C.F.; Pirisi, F.M.; Bandino, G.; Dessi, A. Novel approach to study oxidative stability of extra virgin olive oils: Importance of α-tocopherol concentration. J. Agric. Food Chem 2002, 50, 4342–4346. [Google Scholar]
- Lavelli, V. Comparison of the antioxidant activities of extra virgin olive oils. J. Agric. Food Chem 2002, 50, 7704–7708. [Google Scholar]
- Leenen, R.; Roodenburg, A.J.; Vissers, M.N. Supplementation of plasma with olive oil phenols and extracts: Influence on LDL oxidation. J. Agric. Food Chem 2002, 50, 1290–1297. [Google Scholar]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol 2003, 23, 622–629. [Google Scholar]
- Servili, M.; Esposto, S.; Fabiani, R.; Urbani, S.; Taticchi, A.; Mariucci, F.; Selvaggini, R.; Montedoro, G.F. Phenolic compounds in olive oil: Antioxidant, health and sensory activities according to their chemical structure. Inflammopharmacology 2009, 17, 76–84. [Google Scholar]
- Visioli, F.; Galli, C. Olive oil phenols and their potential effects on human health. J. Agric. Food Chem 1998, 46, 4292–4296. [Google Scholar]
- Fitó, M.; Covas, M.I.; Lamuela-Raventós, R.M.; Vila, J.; Torrents, J.; Torre, C.D.L.; Marrugat, J. Protective effect of olive oil and its phenolic compounds against low density lipoprotein oxidation. Lipids 2000, 35, 633–638. [Google Scholar]
- Pellegrini, N.; Visioli, F.; Buratti, S.; Brighenti, F. Direct analysis of total antioxidant activity of olive oil and studies on the influence of heating. J. Agric. Food Chem 2001, 49, 2532–2538. [Google Scholar]
- Manna, C.; D’Angelo, S.; Migliardi, V.; Loffredi, E.; Mazzoni, O.; Morrica, P.; Galletti, P.; Zappia, V. Protective effect of the phenolic fraction from virgin olive oils against oxidative stress in human cells. J. Agric. Food Chem 2002, 50, 6521–6526. [Google Scholar]
- Stupans, I.; Kirlich, A.; Tuck, K.L.; Hayball, P.J. Comparison of radical scavenging effect, inhibition of microsomal oxygen free radical generation, and serum lipoprotein oxidation of several natural antioxidants. J. Agric. Food Chem 2002, 50, 2464–2469. [Google Scholar]
- Gorinstein, S.; Martin-Belloso, O.; Katrich, E.; Lojek, A.; Èíž, M.; Gligelmo-Miguel, N.; Haruenkit, R.; Park, Y.-S.; Jung, S.-T.; Trakhtenberg, S. Comparison of the contents of the main biochemical compounds and the antioxidant activity of some Spanish olive oils as determined by four different radical scavenging tests. J. Nutr. Biochem 2003, 14, 154–159. [Google Scholar]
- Somova, L.I.; Shode, F.O.; Ramnanan, P.; Nadar, A. Antihypertensive, antiatherosclerotic and antioxidant activity of triterpenoids isolated from Olea europaea, subspecies africana leaves. J. Ethnopharmacol 2003, 84, 299–305. [Google Scholar]
- Masella, R.; Varì, R.; D’Archivio, M.; Benedetto, R.D.; Matarrese, P.; Malorni, W.; Scazzocchio, B.; Giovannini, C. Extra virgin olive oil biophenols inhibit cell-mediated oxidation of LDL by increasing the mRNA transcription of glutathione-related enzymes. J. Nutr 2004, 134, 785–791. [Google Scholar]
- Bouaziz, M.; Grayer, R.J.; Simmonds, M.S.J.; Damak, M.; Sayadi, S. Identification and antioxidant potential of flavonoids and low molecular weight phenols in olive cultivar chemlali growing in Tunisia. J. Agric. Food Chem 2005, 53, 236–241. [Google Scholar]
- Škergeta, M.; Kotnika, P.; Hadolinb, M.; Hrašb, A.R.; Simonièa, M.; Kneza, Ž. Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities. Food Chem 2005, 89, 191–198. [Google Scholar]
- Dabbou, S.; Issaoui, M.; Servili, M.; Taticchi, A.; Sifi, S.; Montedoro, G.F.; Hammami, M. Characterisation of virgin olive oils from European olive cultivars introduced in Tunisia. Eur. J. Lipid Sci. Technol 2009, 111, 392–401. [Google Scholar]
- Lee, O.; Lee, B. Antioxidant and antimicrobial activities of individual and combined phenolics in Olea europaea leaf extract. Bioresour. Technol 2010, 101, 3751–3754. [Google Scholar]
- Hashimoto, T.; Ibi, M.; Matsuno, K.; Nakashima, S.; Tanigawa, T.; Yoshikawa, T.; Yabe-Nishimura, C. An endogenous metabolite of dopamine, 3,4-dihydroxyphenylethanol, acts as a unique cytoprotective agent against oxidative stress-induced injury. Free Radic. Biol. Med 2004, 36, 555–564. [Google Scholar]
- Trichopoulou, A.; Lagiou, P. Healthy traditional Mediterranean diet: An expression of culture, history and lifestyle. Nutr. Rev 1997, 55, 383–389. [Google Scholar]
- Tapiero, H.; Ba, G.N.; Couvreur, P.; Tew, K.D. Polyunsaturated fatty acids (PUFA) and eicosanoids in human health and pathologies. Biomed. Pharmacother 2002, 56, 215–222. [Google Scholar]
- Tassou, C.C.; Nychas, G.J. Inhibition of Staphylococous aureus by olive phenolics in broth and in a model food system. J. Food Protect 1994, 57, 120–124. [Google Scholar]
- Bisignano, G.; Tomaino, A.; Lo Cascio, R.C.G.; Uccella, N.; Saija, A. On the in-vitro antimicrobial activity of oleuropein and hydroxytyrosol. J. Pharm. Pharmacol 1999, 51, 971–974. [Google Scholar]
- Tranter, H.S.; Tassou, S.C.; Nychas, G.J. The effect of the olive phenolic compound, oleuropein, on growth and enterotoxin B production by staphylococcus aureus. J. Appl. Bacteriol 1993, 74, 253–259. [Google Scholar]
- Sudjana, A.N.; Orazio, C.D.; Ryan, V.; Rasool, N.; Ng, J.; Islam, N.; Rileyae, T.V.; Hammer, K.A. Antimicrobial activity of commercial Olea europaea (olive) leaf extract. Int. J. Antimicrob. Agents 2009, 33, 461–463. [Google Scholar]
- Gerber, M. Olive oil and cancer. In Epidemiology of Diet and Cancer; Hill, M.J., Giacosa, A., Caygill, C.P.G., Eds.; Ellis Horwood: Chichester, UK, 1994; pp. 263–275. [Google Scholar]
- Simopoulos, A.P. The Mediterranean diets: What is so special about the diet of Greece? J. Nutr 2001, 131, 3065S–3073S. [Google Scholar]
- Giovannini, C.; Scazzocchio, B.; Mattarrese, P.; Vari, R.; Archivio, M.D.; Benedetto, R.D.; Casciani, S.; Dessic, M.R.; Struface, E.; Malorni, W.; et al. Apoptosis induced by oxidized lipids is associated with upregulation of p66Shc in intestinal Caco-2 cells: Protective effects of phenolic compounds. J. Nutr. Biochem 2008, 19, 118–128. [Google Scholar]
- García-Villalba, R.; Carrasco-Pancorbo, A.; Oliveras-Ferraros, C.; Vázquez-Martín, A.; Menéndez, J.A.; Segura-Carretero, A.; Fernández-Gutiérre, A. Characterization and quantification of phenolic compounds of extra-virgin olive oils with anticancer properties by a rapid and resolutive LC-ESI-TOF MS method. J. Pharmaceut. Biomed. Anal 2010, 51, 416–429. [Google Scholar]
- Andrikopoulos, N.K.; Antonopoulou, S.; Kaliora, A.C. Oleuropein inhibits LDL oxidation induced by cooking oil frying by-products and platelet aggregation induced by platelet-activating factor. LWT-Food Sci. Technol 2002, 35, 479–484. [Google Scholar]
- Driss, F.; Duranthon, V.; Viard, V. Biological activity of olive tree polyphenolic compounds. OCL-Ol. Corps Gras Lipides 1996, 3, 448–451. [Google Scholar]
- Khayyal, M.T.; El-Ghazaly, M.A.; Abdallah, D.M.; Nassar, N.N.; Okpanyi, S.N.; Kreuter, M.H. Blood pressure lowering effect of an olive leaf extract (Olea europaea) in L-NAME induced hypertension in rats. Arzneimittelforschung 2002, 52, 797–802. [Google Scholar]
- Binukumar, B.; Mathew, A. Dietary fat and risk of breast cancer. World J. Surg. Oncol 2005, 3, 45. [Google Scholar] [CrossRef] [Green Version]
- Angerosa, F. Influence of volatile compounds on virgin olive oil quality evaluated by analytical approaches and sensor panels. Eur. J. Lipid Sci.Technol 2002, 104, 639–660. [Google Scholar]
- Lorenzo, I.M.; Pavon, J.L.P.; Laespada, M.E.F.; Pinto, C.G.; Cordero, B.M. Detection of adulterants in olive oil by headspace-mass spectrometry. J. Chromatogr. A 2002, 945, 221–230. [Google Scholar]
- Morales, M.T.; Rios, J.J.; Aparicio, R. Changes in the volatile composition of virgin olive oil during oxidation: Flavors and off-flavors. J. Agric. Food Chem 1997, 45, 2666–2673. [Google Scholar]
- Baccouri, O.; Bendini, A.; Cerretani, L.; Guerfel, M.; Baccouri, B.; Lercker, G.; Zarrouk, M.; Miled, D.D.B. Comparative study on volatile compounds from Tunisian and Sicilian monovarietal virgin olive oils. Food Chem 2008, 111, 322–328. [Google Scholar]
- Kiritsakis, A.K.; Nanos, G.D.; Polymenoupoulos, Z.; Thomai, T.; Sfakiotakis, E.Y. Effect of fruit storage conditions on olive oil quality. J. Am. Oil Chem. Soc 1998, 75, 721–724. [Google Scholar]
- Kalua, C.M.; Allen, M.S.; Bedgood, D.R.; Bishop, A.G.; Prenzler, P.D.; Robards, K. Olive oil volatile compounds, flavour development and quality: A critical review. Food Chem 2007, 100, 273–286. [Google Scholar]
- Vichi, S.; Castellote, A.I.; Pizzale, L.; Conte, L.S.; Buxaderas, S.; Lopez-Tamames, E. Solid-phase microextraction in the analysis of virgin olive oil volatile fraction: Characterization of virgin olive oils from two distinct geographical areas of northern Italy. J. Agric. Food Chem 2003, 51, 6572–6577. [Google Scholar]
- Angerosa, F.; Basti, C. Olive oil volatile compounds from the lipoxygenase pathway in relation to fruit ripeness. Ital. J. Food Sci 2001, 13, 421–428. [Google Scholar]
- Morales, M.T.; Alonso, M.V.; Rios, J.J.; Aparicio, R. Virgin olive oil aroma: Relationship between volatile compounds and sensory attributes by chemometrics. J. Agric. Food Chem 1995, 43, 2925–2931. [Google Scholar]
- Morales, M.T.; Luna, G.; Aparicio, R. Comparative study of virgin olive oil sensory defects. Food Chem 2005, 91, 293–301. [Google Scholar]
- Goodwin, T.W. Biosynthesis of sterols. In The Biochemistry of Plants. Lipids: Structure and Function, 4th ed; Stumpf, P.K., Conn, E.E., Eds.; Academic Press: London, UK, 1980; pp. 485–507. [Google Scholar]
- Benitez-Sánchez, P.L.; Camacho, L.M.; Aparicio, R. A comprehensive study of hazelnut oil composition with comparisons to other vegetable oils, particularly olive oil. Eur. Food Res. Technol 2003, 218, 13–19. [Google Scholar]
- Morchio, G.; De Anreis, R.; Fedeli, E. Investigations of total sterols content in the olive oil and their variation during refining process. Riv. Ital. Sostanze Grasse 1987, 64, 185–192. [Google Scholar]
- Sivakumar, G.; Bati, C.B.; Perri, E.; Uccella, N.U. Gas chromatography screening of bioactive phytosterols from mono-cultivar olive oils. Food Chem 2006, 95, 525–528. [Google Scholar]
- Canabate-Díaz, B.; Carretero, A.S.; Fernández-Gutiérrez, A.; Belmonte Vega, A.; Garrido Frenich, A.; Martínez Vidal, J.L.; Duran Martos, J. Separation and determination of sterols in olive oil by HPLC-MS. Food Chem 2007, 102, 593–598. [Google Scholar]
- Moss, G.P. The nomenclature of steroids: Recommendations by the IUPAC-IUB joint commission on biochemical nomenclature. Eur. J. Biochem 1989, 186, 429–458. [Google Scholar]
- Akihisa, T.; Kokke, W.; Tamura, T. Naturally occurring sterols and related compounds from plants. In Physiology and Biochemistry of Sterols; Patterson, G.W., Nes, W.D., Eds.; American Oil Chemists’ Society: Champaign, IL, USA, 1991; pp. 172–228. [Google Scholar]
- Moreau, R.A.; Whitakerb, B.D.; Hicksa, K.B. Phytosterols, phytostanols, and their conjugates in foods: Structural diversity, quantitative analysis, and health-promoting uses. Prog. Lipid Res 2002, 41, 457–500. [Google Scholar]
- Heupel, R.C. Isolation and primary characterization of sterols. In Analysis of Sterols and Other Biologically Significant Steroids; Nes, D.W., Parish, E.J., Eds.; Academic Press Inc.: San Diego, CA, USA, 1989; pp. 1–32. [Google Scholar]
- Hartmann, M.A. Plant sterols and the membrane environment. Trends Plant Sci 1998, 3, 170–175. [Google Scholar]
- Quillez, J.; Garcila-Lorda, P.; Salas-Salvadol, J. Potential uses and benefits of phytosterols in diet: Present situation and future directions. Clin. Nutr 2003, 22, 343–351. [Google Scholar]
- Awad, A.B.; Fink, C.S. Phytosterols as anticancer dietary components: Evidence and mechanism of action. J. Nutr 2000, 130, 2127–2130. [Google Scholar]
- Wilt, T.J.; Mac Donald, R.; Ishani, A. Beta-sitosterol for the treatment of benign prostatic hyperplasia: A systematic review. Br. J. Urol. Int 1999, 83, 976–983. [Google Scholar]
- Wolfreys, A.M.; Hepburn, P.A. Safety evaluation of phytosterol esters. Part 7. Assessment of mutagenic activity of phytosterols, phytosterol esters and the cholesterol derivative, 4-cholesten-3-one. Food Chem. Toxicol 2002, 40, 461–470. [Google Scholar]
- Hepburn, P.A.; Horner, S.A.; Smith, M. Safety evaluation of phytosterol esters. Part 2. Subchronic 90-day oral toxicity study on phytosterol esters a novel functional food. Food Chem. Toxicol 1999, 37, 521–532. [Google Scholar]
- Ranalli, A.; Pollastri, L.; Contento, S.; di Loreto, G.; Iannucci, E.; Lucera, L.; Russi, F. Sterol and alcohol components of seed, pulp and whole olive fruit oils. Their use to characterise olive fruit variety by multivariates. J. Sci. Food Agric 2002, 82, 854–859. [Google Scholar]
- Esterbauer, H.; Dieber-Rotheneder, M.; Striegl, G.; Waeg, G. Role of vitamin E in preventing the oxidation of low-density lipoprotein. Am. J. Clin. Nutr 1991, 53, 314S–321S. [Google Scholar]
- Blekas, G.; Psomiadou, E.; Tsimidou, M. On the importance of total polar phenols to monitor the stability of Greek virgin olive oil. Eur. J. Lipid Sci. Technol 2002, 104, 340–346. [Google Scholar]
- Dionisi, F.; Prodolliet, J.; Tagliaferri, E. Assessment of olive oil adulteration by reversed-phase high-performance liquid chromatography/amperometric detection of tocopherols and tocoterienols. J. Am. Oil Chem. Soc 1995, 72, 1505–1511. [Google Scholar]
- Cunha, S.C.; Amaral, J.S.; Fernandes, J.O.; Oliveira, M.B.P.P. Quantification of tocopherols and tocotrienols in Portuguese olive oils using HPLC with three different detection systems. J. Agric. Food Chem 2006, 54, 3351–3356. [Google Scholar]
- Kamal-Eldin, A.; Appelqvist, L.A. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 1996, 31, 671–701. [Google Scholar]
- Yamauchi, R.; Matsushita, S. Quenching effect of tocopherols on methyl linoleate photoxidation and their oxidation products. Agric. Biol. Chem 1977, 41, 1425–1430. [Google Scholar]
- Cheeseeman, K.H.; Slater, T.F. An introduction to free radical biochemistry. Br. Med. Bull 1993, 49, 481–493. [Google Scholar]
- Kamal-Eldin, A.; Andersson, R.A. Multivariate study of the correlation between tocopherol content and fatty acid composition in vegetable oils. J. Am. Oil Chem. Soc 1997, 74, 375–380. [Google Scholar]
- Doelman, C.J. Antioxidant Therapy and Preventive Medicine; Plenum Press: New York NY, USA, 1989; Volume 9. [Google Scholar]
- Armstrong, N.; Paganga, G.; Brunev, E.; Miller, N.; Nanchahal, K. Reference values for α-tocopherol and β-carotene in the Whitehall II study. Free Radic. Res 1997, 27, 207–219. [Google Scholar]
- Caruso, D.; Berra, B.; Giovanini, F.; Cortesi, N.; Fedeli, E.; Galli, G. Effect of virgin olive oil phenolic compounds on in vitro oxidation of human low density lipoproteins. Nutr. Metab. Cardiovasc. Dis 1997, 99, 102–107. [Google Scholar]
- Nicolaiew, N.; Lemort, N.; Adorni, L.; Berra, B.; Montorfano, G.; Rapelli, S. Comparison between extra virgin olive oil and oleic acid rich sunflower oil: Effects on postprandial lipemia and LDL susceptibility to oxidation. Ann. Nutr. Metab 1998, 42, 251–260. [Google Scholar]
- Hudson, B.; Lewis, J. Polyhydroxy flavonoid antioxidants for edible oils. Structural criteria for activity. Food Chem 1983, 10, 47–55. [Google Scholar]
- Psomiadou, P.; Tsimidou, M. Simultaneous HPLC determination of tocopherols, carotenoids, and chlorophylls for monitoring their effect on virgin olive oil oxidation. J. Agric. Food Chem 1998, 46, 5132–5138. [Google Scholar]
- Lanzón, A.; Albi, T.; Cert, A. The hydrocarbon fraction of virgin olive oil and changes resulting from refining. J. Am. Oil Chem. Soc 1994, 3, 285–291. [Google Scholar]
- Gandul-Rojas, B.; Minguez-Mosquera, M.I. Chlorophyll and carotenoid composition in virgin olive oils from various Spanish olive varieties. J. Sci. Food Agric 1996, 72, 31–39. [Google Scholar]
- Kupper, H.; Dedic, R.; Svoboda, A.; Hala, J.; Kroneck, P.M. Kinetics and efficency of excitation energy transfer from chlorophylls, their heavy metal-substituted derivatives, and pheophytins to singlet oxygen. Biochim. Biophys. Acta 2002, 1572, 107–113. [Google Scholar]
- Foote, C.S.; Denny, R.W. Chemistry of singlet oxygen VII. quenching by β-carotene. J. Am. Chem. Soc 1968, 90, 6233–6234. [Google Scholar]
- Hansen, E.; Skibsted, L.H. Light-induced oxidative changes in a model dairy spread. Wavelength dependence of quantum yields. J. Agric. Food Chem 2000, 48, 3090–3094. [Google Scholar]
- Burton, G.; Ingold, K. β-Carotene: An unusual type of lipid antioxidant. J. Sci. Food Agric 1984, 224, 569–573. [Google Scholar]
- Yanishlieva, N.V.; Aitzetmüller, K.; Raneva, V. β-Carotene and lipid oxidation. Lipid/Fett 1998, 100, 444–462. [Google Scholar]
- Matyas, G.R.; Wassef, N.M.; Rao, M.; Alving, C.R. Induction and detection of antibodies to squalene. J. Immunol. Methods 2000, 245, 1–14. [Google Scholar]
- Newmark, H.L. Squalene, olive oil, and cancer risk: A review and hypothesis, Cancer Epidemiol. Biomark. Prev 1997, 6, 1101–1103. [Google Scholar]
- Owen, R.W.; Haubner, R.; Würtele, G.W.; Hull, W.E.; Spiegelhalder, B.; Bartsch, H. Olives and olive oil in cancer prevention. Eur. J. Cancer Prev 2004, 13, 319–326. [Google Scholar]
- Koivisto, P.V.I. Miettinen, Increased amount of cholesterol precursors in lipoproteins after ileal exclusion. Lipids 1988, 23, 993–996. [Google Scholar]
- Stewart, M.E. Sebaceous gland lipids. Semin. Dermatol 1992, 11, 100–105. [Google Scholar]
- Newmark, H.L. Squalene, olive oil, and cancer risk: Review and hypothesis. Ann. N. Y. Acad. J. Sci. Food Agric 1999, 889, 193–203. [Google Scholar]
- Dennis, K.; Shimamoto, T. Production of malonyldialdehyde from squalene, a major skin surface lipid, during UV irradiation. Photochem. Photobiol 1989, 49, 711–719. [Google Scholar]
- Reddy, L.H.; Couvreur, P. Squalene: A natural triterpene for use in disease management and therapy. Adv. Drug Deliv. Rev 2009, 61, 1412–1426. [Google Scholar]
- Kelly, G. Squalene and its potential clinical uses. Altern. Med. Rev 1999, 4, 29–36. [Google Scholar]
- Kalogeropoulos, N.; Chiou, A.; Gavala, E.; Christea, M.; Andrikopoulos, K.N. Nutritional evaluation and bioactive microconstituents (carotenoids, tocopherols, sterols and squalene) of raw and roasted chicken fed on DHA-rich microalgae. Food Res. Int 2010, 43, 2006–2013. [Google Scholar]
- Whittenton, J.; Harendra, S.; Pitchumani, R.; Mohanty, K.; Vipulanandan, C.; Thevananther, S. Evaluation of asymmetric liposomal nanoparticles for encapsulation of polynucleotides. Langmuir 2008, 24, 8533–8540. [Google Scholar]
- Fox, C.B.; Anderson, R.C.; Dutill, T.S.; Goto, Y.; Reed, S.G.; Vedvick, T.S. Monitoring the effects of component structure and source on formulation stability and adjuvant activity of oil-in-water emulsions. Colloids Surf. B 2008, 65, 98–105. [Google Scholar]
- Kamimara, H.; Koga, N.; Oguri, K.; Yoshimura, H. Enhanced elimination of theophylline, phenobarbital and strychnine from the bodies of rats and mice by squalene treatment. J. Pharmacobiodyn 1992, 15, 215–221. [Google Scholar]
- Kiritsakis, Markakis. Olive oil: A review. In Advances in Food Research; Elsevier Inc: New York, NY, USA, 1988; Volume 31, pp. 453–482. [Google Scholar]
- Aparicio, R.; Luna, G. Characterisation of monovarietal virgin olive oil. Eur. J. Lipid Sci. Technol 2002, 104, 614–627. [Google Scholar]
- Grob, K.; Lanfranchi, M.; Mariani, C. Evaluation of olive oils through the fatty alcohols, the sterols and their esters by coupled LC-GC. J. Am. Oil Chem. Soc 1990, 67, 626–634. [Google Scholar]
- Gucci, R.; Lombardini, L.; Tattini, M. Analysis of leaf water relations in leaves of two olive (Olea europaea) cultivars differing in tolerance to salinity. Tree Physiol 1997, 17, 13–21. [Google Scholar]
- Fernandez-Escobar, R.; Moreno, R.; Garcia-Creus, M. Seasonal changes of mineral nutrients in olive leaves during the alternate-bearing cycle. Sci. Hort. -Amsterdam 1999, 82, 25–45. [Google Scholar]
- Ciafardini, G.; Zullo, B.A. Microbiological activity in stored olive oil. Int. J. Food Microbiol 2002, 75, 111–118. [Google Scholar]
- karakaya, S.E.S. Studies of olive tree leaf extract indicate seveal potential health benefits. Nutr. Rev 2009, 67, 632–639. [Google Scholar]
- Zarzuelo, A. Vasodilator effect of olive leaf. Planta Med 1991, 57, 417–419. [Google Scholar]
- Samuelsson, G. The blood pressure lowering factor in leaves of Olea europaea. Farmacevtisk Revy 1951, 15, 229–239. [Google Scholar]
- Pereira, A.P.; Ferreira, I.C.F.R.; Marcelino, F.; Valentão, P.; Andrade, B.P.; Seabra, R.; Estevinho, L.; Bento, A.; Pereira, J.A. Phenolic compounds and antimicrobial activity of olive (Olea europaea L. Cv. Cobrançosa) leaves. Molecules 2007, 12, 1153–1162. [Google Scholar]
- Benavente-Garcia, O.; Castillo, J.; Lorente, J.; Ortuno, A.; Del Rio, J.A. Antioxidant activity of phenolics extracted from Olea europaea L. leaves. Food Chem 2000, 68, 457–462. [Google Scholar]
- Furneri, P.M.; Marino, A.; Saija, A.; Uccella, N.; Bisignano, G. In vitro antimycoplasmal activity of oleuropein. Int. J. Antimicrob. Agents 2002, 20, 293–296. [Google Scholar]
- Briante, R.; Febbraio, F.; Nucci, R. Antioxidant properties of low molecular weight phenols present in the Mediterranean diet. J. Agric. Food Chem 2003, 51, 6975–6981. [Google Scholar]
- Skerget, M.; Kotnik, P.; Hadolin, M.; Hradolin, A.R.; Simoni, M.; Knez, Z. Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities. Food Chem 2005, 89, 191–198. [Google Scholar]
- Ryan, D.; Prenzler, P.D.; Lavee, S.; Antolovich, M.; Robards, K. Quantitative changes in phenolic content during physiological development of the olive (Olea europaea) cultivar Hardy’s Mammoth. J. Agric. Food Chem 2003, 51, 2532–2538. [Google Scholar]
- Bianco, A.; Uccella, N. Biophenolic components of olives. Food Res. Int 2000, 33, 475–485. [Google Scholar]
- Tasioula-Margari, M.; Ologeri, O. Isolation and characterization of virgin olive oil phenolic compounds by HPLC/UV and GC/MS. J. Food Sci 2001, 66, 530–534. [Google Scholar]
- Delgado-Pertinez, M.; Gomez-Cabrera, A.; Garrido, A. Predicting the nutritive value of the olive leaf (Olea europaea): Digestibility and chemistry composition and in vitro studies. Anim. Feed Sci. Technol 2000, 87, 187–201. [Google Scholar]
- Lesage-Meessen, L.; Navarro, D.; Maunier, S.; Sigoillot, J.C.; Lorquin, J.; Delattre, M. Simple phenolic content in olive oil residues as a function of extraction systems. Food Chem 2001, 75, 501–507. [Google Scholar]
- Sabbah, I.; Marsook, T.; Basheer, S. The effect of pretreatment on anaerobic activity of olive mill wastewater using batch and continuous systems. Process Biochem 2004, 39, 1947–1951. [Google Scholar]
- Manios, T. The composting potential of different organic solid wastes, experience from the island of Crete. Environ. Int 2004, 29, 1079–1089. [Google Scholar]
- Ranalli, A.; Lucera, L.; Contento, S. Antioxidizing potency of phenol compounds in olive oil mill wastewater. J. Agric. Food Chem 2003, 51, 7636–7641. [Google Scholar]
- Shahidi, F.; Naczk, M. Phenolic in Food and Nutraceutical; CRC Press: Boca Raton, FL USA, 2004. [Google Scholar]
- Amro, B.; Aburjai, T.; Al-Khalil, S. Antioxidative and radical scavenging effects of olive cake extract. Fitoterapia 2002, 73, 456–461. [Google Scholar]
- Fernandez-Bolanos, J.; Rodriguez, G.; Rodriguez, R.; Heredia, A.; Guillen, R.; Jimenez, A. Production in large quantities of highly purified hydroxytyrosol from liquid-solid waste of two phase olive oil processing or”Alperujo”. J. Agric. Food Chem 2002, 50, 6804–6811. [Google Scholar]
- Mulinacci, N.; Romani, A.; Galardi, C.; Pinelli, P.; Giaccherini, C.; Vincieri, F.F. Polyphenolic content in olive oil wastewaters and related olive samples. J. Agric. Food Chem 2001, 49, 3509–3514. [Google Scholar]
- Allouche, N.; Fki, I.; Sayadi, S. Toward a high yield recovery of antioxidants and purified hydroxytyrosol from olive mill wastewaters. J. Agric. Food Chem 2004, 52, 267–273. [Google Scholar]
- Bianco, A.; Buiarelli, F.; Cartoni, G.; Coccioli, F.; Jasionowska, R.; Margherita, P. Analysis by liquid chromatography-tandem mass spectrometry of biophenolic compounds in olives and vegetation waters, Part I. J. Sep. Sci 2003, 26, 409–416. [Google Scholar]
- Fiorentino, A.; Gentili, A.; Isidori, M.; Monaco, P.; Nardelli, A.; Parrella, A.E.; et al. Environmental effects caused by olive mill wastewaters: Toxicity comparison of low-molecular weight phenol components. J. Agric. Food Chem 2003, 51, 1005–1009. [Google Scholar]
- Rodis, P.S.; Karathanos, V.T.; Mantzavinou, A. Partitioning of olive oil antioxidants between oil and water phases. J. Agric. Food Chem 2002, 50, 596–601. [Google Scholar]
- Aludatt, M.H.; Alli, I.; Ereifej, K.; Alhamad, M.; Al-Tawaha, A.R.; Rababah, T. Optimisation, characterisation and quantification of phenolic compounds in olive cake. Food Chem 2010, 123, 117–122. [Google Scholar]
- Parades, C.; Cegarra, J.; Roig, A.; Sanchez-Monedero, M.A.; Bernal, M.P.; Brenes, M. Characterization of olive mill wastewater (alpechin) audits sludge for agricultural purposes. Bioresour. Technol 1999, 67, 111–115. [Google Scholar]
- Rice-Evans, C.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci 1997, 2, 152–159. [Google Scholar]
- Khoufi, S.; Aloui, F.; Sayadi, S. Extraction of antioxidants from olive mill wastewater and electro-coagulation of exhausted fraction to reduce its toxicity on anaerobic digestion. J. Hazard. Mater 2008, 151, 531–539. [Google Scholar]
- Manna, C.; Galletti, P.; Cucciolla, V.; Moltedo, O.; Leone, A.; Zappia, V. The protective effect of the olive oil polyphenols (3,4-dyhydroxyphenyl)-ethanol counteracts reactive oxygen metabolite induced cytoxicity in caco-2-cells. J. Nutr 1997, 127, 286–292. [Google Scholar]
- Kubo, I.; Hanke, F.J. Chemistry based resistence in plants. In Chemically Mediated Interaction Between Plants and Other Organisims; Plenum: New York, NY, USA, 1985. [Google Scholar]
- Legar, C.L.; Kadiri-Hassani, N.; Descomps, B. Decreased superoxide anion production in cultured human promonocyte cells (THP-l) due to polyphenol mixtures from olive oil processing wastewaters. J.Agric. Food Chem 2000, 48, 5061–5067. [Google Scholar]
- González, M.D.; Moreno, E.; Quevedo-Sarmiento, J.; Ramos-Cormenzana, A. Studies on antibacterial activity of wastewaters from olive oil mills (alpechin): Inhibitory activity of phenolic and fatty acids. Chemosphere 1990, 20, 423–432. [Google Scholar]
- Moreno, R.; Benitez, E.; Melgar, R.; Polo, A.; Gomez, M.; Nogales, R. Vermicomposting as an alternative for reusing by-products from the olive oil industry. Fresen. Environ. Bull 2000, 9, 1–8. [Google Scholar]
- Perez, J.; Delarubia, T.; Moreno, J.; Martinez, J. phenolic content and antibacterial activity of olive oil wastewaters. Environ. Toxicol. Chem 1992, 11, 489–495. [Google Scholar]
- Capasso, R.; Evidente, A; Schivo, L.; Orru, G.; Marcialis, M.A.; Cristinizo, G. Antibacterial polyphenols from olive oil mill wastewaters. J. Appl. Bacteriol 1995, 79, 393–398. [Google Scholar]
- Fki, I.; Allouche, N.; Sayadi, S. The use of polyphenolic extract, purified hydroxytyrosol and 3,4-dihydroxyphenyl acetic acid from olive mill wastewater for the stabilization of refined oils: A potential alternative to synthetic antioxidants. Food Chem 2005, 93, 197–204. [Google Scholar]
- Heredia, A.; Guillén, R.; Fernández-Bolaños, J.; Rivas, M. Olives stone as a source of fermentable sugars. Biomass 1987, 14, 143–148. [Google Scholar]
- Najar-Souissi, S.; Ouedereni, A.; Ratel, A. Adsorption of dyes onto activated carbon prepared from olive stones. J. Environ. Sci. China 2005, 17, 998–1003. [Google Scholar]
- Budinova, T.; Petrov, N.; Razvigorova, M.; Parra, J.; Galiatsatou, P. Removal of arsenic(III) from aqueous solution by activated carbons prepared from solvent extracted olive pulp and olive stones. Ind. Eng. Chem. Res 2006, 45, 1896–1901. [Google Scholar]
- Ghazy, S.E.; Samra, S.E.; May, A.E.M.; El-Morsy, S.M. Removal of aluminium from some water samples by sorptive-flotation using powdered modified activated carbon as a sorbent and oleic acid as a surfactant. Anal. Sci 2006, 22, 377–382. [Google Scholar]
- Montane, D.; Salvado, J.; Torras, C.; Farriol, X. High-temperature dilute acid hydrolysis of olive stone for furfural production. Biomass Bioenerg 2001, 22, 295–304. [Google Scholar]
- Siracusa, G.; La Rosa, A.D.; Siracusa, V.; Trovato, M. Eco Compatible use of olive huso as filler in thermoplastic composites. J. Polym. Environ 2001, 9, 157–161. [Google Scholar]
- Rodríguez, G.; Lama, A.; Rodríguez, R.; Jiménez, A.; Guillén, R.; Pages, J.F.B. Olive stone an attractive source of bioactive and valuable compounds. Bioresour. Technol 2008, 99, 5261–5269. [Google Scholar]
- Carraro, L.; Trocino, A.; Xiccato, G. Dietary supplementation with olive stone meal in growing rabbits. Ital. J. Anim. Sci 2005, 4, 88–90. [Google Scholar]
- Tejeda-Ricardez, J.; Vaca-Garcia, C.; Borredon, M.E. Design of a batch solvolytic liquefaction reactor for the vaporization of residues from the agricultural foodstuff. Chem. Eng. Res 2003, 81, 1066–1070. [Google Scholar]
- Pérez-Bonilla, M.; Salido, S.; van Beek, T.A.; Linares-Palomino, P.J.; Altarejos, J.; Nogueras, M. Isolation and identification of radical scavengers in olive tree (Olea europaea) wood. J. Chromatogr. A 2006, 111, 311–318. [Google Scholar]
- Zbidi, H.; Salido, S.; Altarejos, J.; Pérez-Bonilla, M.; Bartegi, A.; Rosado, J.A. Olive tree wood phenolic compounds with human platelet antiaggregant properties. Blood Cell Mol. Dis 2009, 42, 279–285. [Google Scholar]
- Pérez-Bonilla, M.; Salido, S.; Teris, A.V.B.; Waard, P.D.; Linares-Palomino, P.J.; Sánchez, A.; Altarejos, J. Isolation of antioxidative secoiridoids from olive wood (Olea europaea L.) guided by on-line HPLC-DAD-radical scavenging detection. Food Chem 2011, 124, 36–41. [Google Scholar]
- Patumi, M.; D’andria, R.; Marsilio, V.; Fontanazza, G.; Morelli, G.; Lanza, B. Olive and olive oil quality after intensive monocone olive growing (Olea europaea L., cv. Kalmata) in different irrigation regimes. Food Chem 2002, 77, 27–34. [Google Scholar]
- Tovar, M.J.; Romero, M.P.; Alegre, S.; Girona, J.; Motilva, M.J. Composition and organoleptic characteristics of oil from Arbequina olive (Olea europaea L.) trees under deficit irrigation. J. Sci. Food Agric 2002, 82, 1755–1763. [Google Scholar]
- Morelló, J.R.; Vuorela, S.; Romero, M.P.; Motilva, M.J.; Heinonen, M. Antioxidant activity of olive pulp and olive oil phenolic compounds of the Arbequina cultivar. J. Agric. Food Chem 2005, 53, 2002–2008. [Google Scholar]
- Kevin, D.; Rade, D.; Trucelj, D.; Mokrovãak, Î.; Nederal, S.; Benãiç, D. The influence of variety and harvest time on the bitterness and phenolic compounds of olive oil. Eur. J. Lipid Sci. Technol 2003, 105, 536–541. [Google Scholar]
- Garcia, A.; Brenes, M.; Garcia, P.; Romero, C.; Garrido, A. Phenolic content of commercial olive oils. Eur. Food Res. Technol 2003, 216, 520–525. [Google Scholar]
- Gómez-Rico, A.; Fregapane, G.; Salvador, M.D. Effect of cultivar and ripening on minor components in Spanish olive fruits and their corresponding virgin olive oils. Food Res. Int 2008, 41, 433–440. [Google Scholar]
- Esti, M.; Cinquanta, L.; La Notte, E. Phenolic compounds in different olive varieties. J. Agric. Food Chem 1998, 46, 32–35. [Google Scholar]
- Gutiérrez, F.; Jímenez, B.; Ruíz, A.; Albi, M.A. Effect of olive ripeness on the oxidative stability of virgin olive oil extracted from the varieties Picual and Hojiblanca and on the different components involved. J. Agric. Food Chem 1999, 47, 121–127. [Google Scholar]
- Ryan, D.; Robards, K.; Lavee, S. Changes in phenolic content of olive during maturation. J. Food Sci. Technol 1999, 34, 265–274. [Google Scholar]
- Cimato, A.; Mattei, A.; Osti, M. Variation of polyphenol composition with harvesting period. Acta Hortic 1990, 286, 453–456. [Google Scholar]
- Mraicha, F.; Ksantini, M.; Zouch, O.; Ayadi, M.; Sayadi, S.; Bouaziz, M. Effect of olive fruit fly infestation on the quality of olive oil from Chemlali 3 cultivar during ripening. Food Res. Int 2008, 41, 433–440. [Google Scholar]
- Amiot, M.J.; Fleuriet, A.; Macheix, J.J. Accumulation of oleuropein derivatives during olive maturation. Phytochemistry 1989, 28, 67–69. [Google Scholar]
- Beltrán, G.; Sanchez, S.; Martinez, L. Influence of fruit ripening process on the natural antioxidant content of Hojiblanca virgin olive oils. Food Chem 2005, 89, 207–215. [Google Scholar]
- Bouaziz, M.; Chamkha, M.; Sayadi, S. Comparative study on phenolic content and antioxidant activity during maturation of the olive cultivar chemlali from Tunisia. J. Agric. Food Chem 2004, 52, 5476–5481. [Google Scholar]
- Garcia, A.; Brenes, M.; Romero, C.; Garcia, P.; Garrido, A. Study of phenolic compounds in virgin olive oils of the Picual variety. Eur. Food Res. Technol 2002, 5, 407–412. [Google Scholar]
- Kotsifaki, A.K.F.; Stefanoudaki, E. Effect of extraction system, stage of ripeness and kneading temperature on the sterol composition of virgin olive oil. J. Am. Oil Chem. Soc 1999, 76, 1477–1481. [Google Scholar]
- Morelloa, J.R.; Romero, M.P.; Motilva, M.J. Effect of the maturation process of the olive fruit on the phenolic fraction of drupes and oils from arbequina, farga, and morrut cultivars. J. Agric. Food Chem 2004, 52, 6002–6009. [Google Scholar]
- Rotondi, A.; Bendni, A.; Cerretani, L.; Mari, M.; Lercker, G.; Toschi, T.G. Effect of olive ripening degree on the oxidative stability and organoleptic properties of Cv. Nostrana di Brisighella extra virgin olive oil. Agric. Food Chem 2004, 52, 3649–3654. [Google Scholar]
- Rovellini, P.; Cortesi, N. Determination of phenolic compounds in different cultivars during olive drupe ripening by liquid chromatography-mass spectrometry. Olive 2003, 95, 32–38. [Google Scholar]
- Salvador, M.; Aranda, F.; Fregapane, G. Influence of fruit ripening on “Cornicabra” virgin olive oil quality. A study of four successive crop seasons. Food Chem 2001, 73, 45–53. [Google Scholar]
- Yousfi, K.; Cert, R.M.; Garcıa, J.M. Changes in quality and phenolic compounds of virgin olive oils during objectively described fruit maturation. Eur. Food Res. Technol 2006, 223, 117–124. [Google Scholar]
- Moriana, A.; Orgaz, F.; Fereres, E; Pastor, M. Yield responses of a mature olive orchard to water deficits. J. Am. Soc. Hortic. Sci 2003, 128, 425–431. [Google Scholar]
- Gomez-Rico, A.; Desamparados Salvador, M.; Moriana, A.; Perez, D.; Olmedilla, N.; Ribas, F.; Fregapane, G. Influence of different irrigation strategies in a traditional Cornicabra cv. olive orchard on virgin olive oil composition and quality. Food Chem 2007, 100, 568–578. [Google Scholar]
- Gómez-Rico, A.; Salvador, M.D.; La Greca, M.; Fregapane, G. Phenolic and volatile compounds of extra virgin olive oil (Olea europaea L. Cv. Cornicabra) with regard to fruit ripening and irrigation management. J. Agric. Food Chem 2006, 54, 7130–7136. [Google Scholar]
- Artajo, L.S.; Romero, M.P.; Tovar, M.J.; Motilva, M.J. Effect of irrigation applied to olive trees (Olea europaea L.) on phenolic compound transfer during olive oil extraction. Eur. J. Lipid Sci. Technol 2006, 108, 19–27. [Google Scholar]
- Berenguer, M.J.; Vossen, P.M.; Grattan, R.S.; Connell, J.H.; Polito, V.S. Tree irrigation levels for optimum chemical and sensory properties of olive oil. HortScience 2006, 41, 427–432. [Google Scholar]
- Wiesman, Z.; Itzhak, D.; Dom, N.B. Optimization of saline water level for sustainable Barnea olive and oil production in desert conditions. Sci. Hort 2004, 100, 257–266. [Google Scholar]
- Gharsallaoui, M.; Ben Amar, F.; Khabou, W.; Ayadi, M. Valorisation des ressources en eau non conventionnelles au Sud Tunisien par la culture de l’olivier (Olea europaea L.). Meeting International: Gestion des Ressources et Application Biotechnologiques en Aridoculture et Culture Sahariennes: Perspectives pour la valorisation des potentialités du Sahara, Djerba, Tunisia, 25–28 December 2006; Institue des Régions Arides: Djerba, Tunisia, 2006. [Google Scholar]
- Palese, A.M.; Celano, G.; Masi, S.; Xiloyannis, C. Treated wastewater for irrigation of olive trees: Effects on yield and oil quality. In Olivebioteq; Mazara del Vallo: Marsala, Italy, 2006; pp. 123–129. [Google Scholar]
- European Community. Council Regulation (EC) No. 1513/2001 of 23 July 2001 Amending Regulations No. 136/66/EEC and (EC) No 1638/98 as regards the extension of the period of validity of the aid scheme and the quality strategy for olive oil. Off. J. Eur. Commun. 2001, L201, 4–7.
- Capella, P.; Fedeli, E.; Bonaga, G.; Lerker, G. Manuale degli Oli e dei Grassi; Tecniche Nuove: Milano, Italy, 1997. [Google Scholar]
- Servili, M.; Baldioli, M.; Montedoro, G.F. Phenolic composition of virgin olive oil in relationship to some chemical and physical aspects of malaxation. Acta Hortic 1994, 356, 331–336. [Google Scholar]
- Veillet, S.; Tomao, V.; Bornard, I.; Ruiz, K.; Chemat, F. Chemical changes in virgin olive oils as a function of crushing systems: Stone mill and hammer crusher. C.R. Chim 2009, 12, 895–904. [Google Scholar]
- Caponio, F.; Gomes, T. Influence of olive crushing temperature on phenols in olive oils. Eur. Food Res. Technol 2001, 212, 156–159. [Google Scholar]
- Campeol, E.; Flamini, G.; Chericoni, S.; Catalano, S.; Cremonini, R. Volatile compounds from three cultivars of Olea europaea from Italy. J. Agric. Food Chem 2001, 49, 5409–5411. [Google Scholar]
- Servili, M.; Selvaggini, R.; Taticchi, A.; Esposto, S.; Montedoro, G.F. Volatile compounds and phenolic composition of virgin olive oil: Optimization of temperature and time of exposure of olive paste to air contact during the mechanical extraction process. J. Agric. Food Chem 2003, 51, 7980–7988. [Google Scholar]
- Kachouri, F.; Hamdi, M. Use of Lactobacillus plantarum in olive oil process and improvement of phenolic compounds content. J. Food Eng 2006, 77, 746–752. [Google Scholar]
- Vierhuis, E.; Servili, M.; Baldioli, M.; Schols, H.A.; Voragen, A.G.J.; Montedoro, G. Effect of enzyme treatment during mechanical extraction of olive oil on phenolic compounds and polysaccharides. J. Agric. Food Chem 2001, 49, 1218–1223. [Google Scholar]
- Faveri, D.D.; Aliakbariana, B.; Avogadroa, M.; Peregoa, P.; Converti, A. Improvement of olive oil phenolics content by means of enzyme formulations: Effect of different enzyme activities and levels. Chem. Eng. J 2008, 41, 149–156. [Google Scholar]
- Aliakbarian, B.; Faveri, D.D.; Converti, A.; Perego, P. Optimisation of olive oil extraction by means of enzyme processing aids using response surface methodology. Chem. Eng. J 2008, 42, 34–40. [Google Scholar]
- Morello, J.R.; Motilva, M.J.; Tovar, M.J.; Romero, M.P. Changes in commercial virgin olive oil (cv. Arbequina) during storage, with special emphasis on the phenolic fraction. Food Chem 2004, 85, 357–364. [Google Scholar]
- Stefanoudaki, E.; Koutsaftakis, A.; Kotsifaki, F.; Angerosa, F.; DiGirolamo, M. Quality characteristics of olive oils of dual- and three-phase decanters and laboratory mill. Acta Hortic 1999, 474, 705–708. [Google Scholar]
- Giovacchino, L.D.; Solinas, M.; Miccoli, M. Effect of extraction systems on the quality of virgin olive oil. J. Am. Oil Chem. Soc 1994, 71, 1189–1194. [Google Scholar]
- Cert, A.; Alba, J.; León-Camacho, M.; Moreda, W.; Pérez-Camino, M.C. Effects of talc addition and operating mode on the quality and oxidative stability of virgin olive oils obtained by centrifugation. J. Agric. Food Chem 1996, 44, 3930–3934. [Google Scholar]
- Galli, C.; Visioli, F. Antioxidant and other properties of phenolics in olives/olive oil, typical compounds of the mediterranean diet. Lipids 1999, 34, S23–S26. [Google Scholar]
- Manna, C.; Galletti, P.; Cucciolla, V.; Montedoro, G.; Zappia, V. Olive oil hydroxytyrosol protects human erythrocytes against oxidative damages. J. Nutr. Biochem 1999, 10, 159–165. [Google Scholar]
- Lozano-Sánchez, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Characterisation of the phenolic compounds retained in different organic and inorganic filter aids used for filtration of extra virgin olive oil. Food Chem 2011, 124, 1146–1150. [Google Scholar]
- Fregapane, G.; Lavelli, V.; León, S.; Kapuralin, J.; Salvador, M.D. Effect of filtration on virgin olive oil stability during storage. Eur. J. Lipid Sci. Technol 2006, 108, 134–142. [Google Scholar]
- Agalias, A.; Magiatis, P.; Skaltsounis, A.; Mikros, E.; Tsarbopoulos, A.; Gikas, E. A new process for the management of olive oil mill waste water and recovery of natural antioxidants. J. Agirc. Food Chem 2007, 55, 2671–2676. [Google Scholar]
- Gortzi, O.; Lalas, S.; Chatzilazarou, A.; Katsoyannos, E.; Papaconstandinou, S.; Dourtoglou, E. Recovery of natural antioxidants from olive mill wastewater using Genapol-X080. J. Am. Oil Chem. Soc 2008, 85, 133–140. [Google Scholar]
- Paraskeva, C.A.; Papadakis, V.G.; Kanellopoulou, D.G.; Koutsoukos, P.G.; Angelopoulos, K.C. Membrane filtration of olive mill wastewater and exploitation of its fractions. Water Environ. Res 2007, 79, 421–429. [Google Scholar]
- Roig, A.; Cayuela, M.L.; Sanchez-Monedero, M.A. An overview on olive mill wastes and their valorisation methods. Waste Manag 2006, 26, 960–969. [Google Scholar]
- Arsuaga, J.M.; López-Muñoz, M.J.; Sotto, A. Correlation between retention and adsorption of phenolic compounds in nanofiltration membranes. Desalination 2010, 250, 829–832. [Google Scholar]
- Lafka, T.-I.; Lazou, A.E.; Sinanoglou, V.J.; Lazos, E.S. Phenolic and antioxidant potential of olive oil mill wastes. Food Chem 2011, 125, 92–98. [Google Scholar]
- Schieber, A.; Stintzing, F.C.; Carle, R. By-products of plant food processing as a source of functional compounds—Recent developments. Trends Food Sci. Technol 2001, 12, 401–413. [Google Scholar]
| Origin | Cultivar |
|---|---|
| Greece | Adramitini, Amigdalolia, Amphissis, Chalkidikis (Chondrolia), Daphnoelia Doppia, Frantoio, Gordal, Koroneiki, Karidolia, Lianolia, Patrini, Chondrolia (aka Throumbolia), Tsounati, Valanolia |
| Italy | Biancolilla, Bosana, Canino, Casaliva, Cellina di Nardo, Coratina Dolce Agogio, Dritta, Moraiolo, Rosciola, Pisciottana, Grignan, Ottobratica |
| Spain | Alfafara, Arbequina, Bical, Blanqueta, Empeltre, Farga, Gordal, Lechin, Hojiblanca, Manzanilla de Jaén, Morrut Palomar, Picual, Sevillenca Verdiell, Vilallonga |
| France | Aglandau, Amellau, Cayon, Germaine, Picholine, Lucques, Sabine, Salonenque Picholine, Zinzala |
| Portugal | Cobrancosa, Galega |
| Croatia | Oblica and Leccino |
| Tunisia | Chemlali, Chetoui, Gerboui, Meski, Oueslati |
| Cultivar | %Oil yield | Ratio flesh to pit | Olive size (Fruits/kg) | Purpose | Shape of the fruits | Origin |
|---|---|---|---|---|---|---|
| Hojiblanca | 23–29 | 4.9 and 6.6:1 | 230–700 | Oil extraction and table olives | Regular | Spain |
| Verdial | 22–30 | 6:1 | 220–800 | Oil extraction and table olives | Ellipsoidal | Spain |
| Picual | 23–27 | 3.8 and 6.1:1 | 270–470 | Oil extraction and table olives | Prominent tip at the button end | Spain |
| Domat | 22 | - | 180–190 | Green table olive | Regular | Turkey |
| Gemlik | 27 | 6:1 | 270–280 | Black table olives | Pronounced tip at the end | Turkey |
| Memecik | 22 | 6:1 | 205–215 | Oil extraction | Pronounced tip at the end | Turkey |
| Memeli | 25 | 7 : 1 | 200–210 | Green-type table olives | Small tip at the end | Turkey |
| Conservolea | 22–25 | 8:1 | 180–200 | Table olive | Round to oval | Greece |
| Nychati Kalamon | 25 | 8:1 | 220–240 | Black table olive | Cylindro-conical, Curved | Greece |
| Chalkidiki | 19 | - | 120–140 | Green table olive, oil extraction | Pronounced tip at the end | Greece |
| Sevillano | 14 | 7.3:1 | 70–80 | Table olive | - | Spain |
| Ascolano | 19 | 8.2:1 | 110–120 | Table olive | - | Italy |
| Ascolana | 17 | - | 100–180 | Table olive | Spherica | Italy |
| Barouni | 17 | 6.8:1 | 130–140 | Green & black - ripe olives | - | Tunisia |
| Picholine marocaine | 17 | 5:1 | 300–500 | Table olive | - | Morocco |
| Arauco | 22–24 | 7:1 | 125–300 | Table olive and oil extraction | Pronounced tip | Argentina |
| Galega vulga | - | 4:1 | 430 | Black table olive | - | Portugal |
| Oblitza | 22 | 6.5:1 | 200 | Table olive | Apple and heart shape | Yugoslavia |
| Ladoelia | 18–21 | 4.6:1 | 330 | Table olive | - | Cyprus |
| Country | Production (tonnes) | Cultivated area (hectares) | Yield (quintal/hectar) |
|---|---|---|---|
| World | 18,241,809 | 9,922,836 | 18.383 |
| Spain | 6,204,700 | 2,500,000 | 24.818 |
| Italy | 3,600,500 | 1,159,000 | 31.065 |
| Greece* | 2,444,230 | 765,000 | 31.4 |
| Turkey | 1,290,654 | 727,513 | 17.740 |
| Syria | 885,942 | 635,691 | 13.936 |
| Morocco | 770,000 | 550,000 | 14.000 |
| Tunisia | 750,000 | 2,300,000 | 3.260 |
| Egypt | 500,000 | 110,000 | 45.454 |
| Algeria | 475,182 | 288,442 | 16.474 |
| Portugal | 362,600 | 380,700 | 9.524 |
| Libya | 180,000 | Na | Na |
| Argentina | 160,000 | 52,000 | 30.769 |
| Country | Production in tons (2009)a | Production % (2009) | Consumption (2005)b | Consumption per person annually (liters/kg)c |
|---|---|---|---|---|
| World | 2,907,985 | 100% | 100% | 0.43 |
| Spain | 1,199,200 | 41.2% | 20% | 13.62 |
| Italy | 587,700 | 20.2% | 30% | 12.35 |
| Greece | 332,600 | 11.4% | 9% | 23.7 |
| Syria | 168,163 | 5.8% | 3% | 7.0 |
| Tunisia | 150,000 | 5.2% | 2% | 11.1 |
| Turkey | 143,600 | 4.9% | 2% | 1.2 |
| Morocco | 95,300 | 3.3% | 2% | 1.8 |
| Portugal | 53,300 | 1.8% | 2% | 7.1 |
| France | 6,300 | 0.2% | 4% | 1.34 |
| United States | 2,700 | 0.1% | 8% | 0.56 |
| Others | 169,122 | 5.8% | 18% | 1.18 |
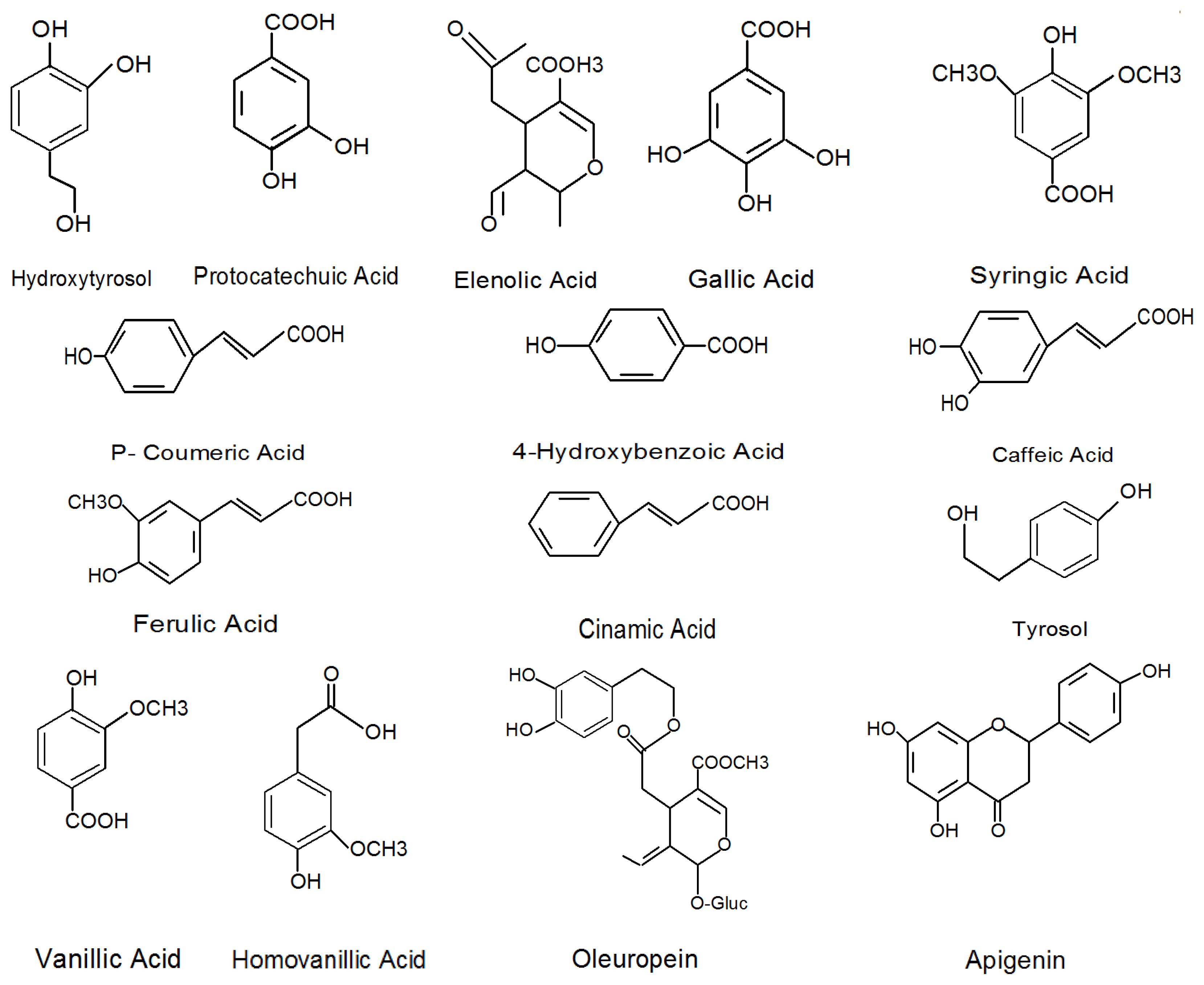
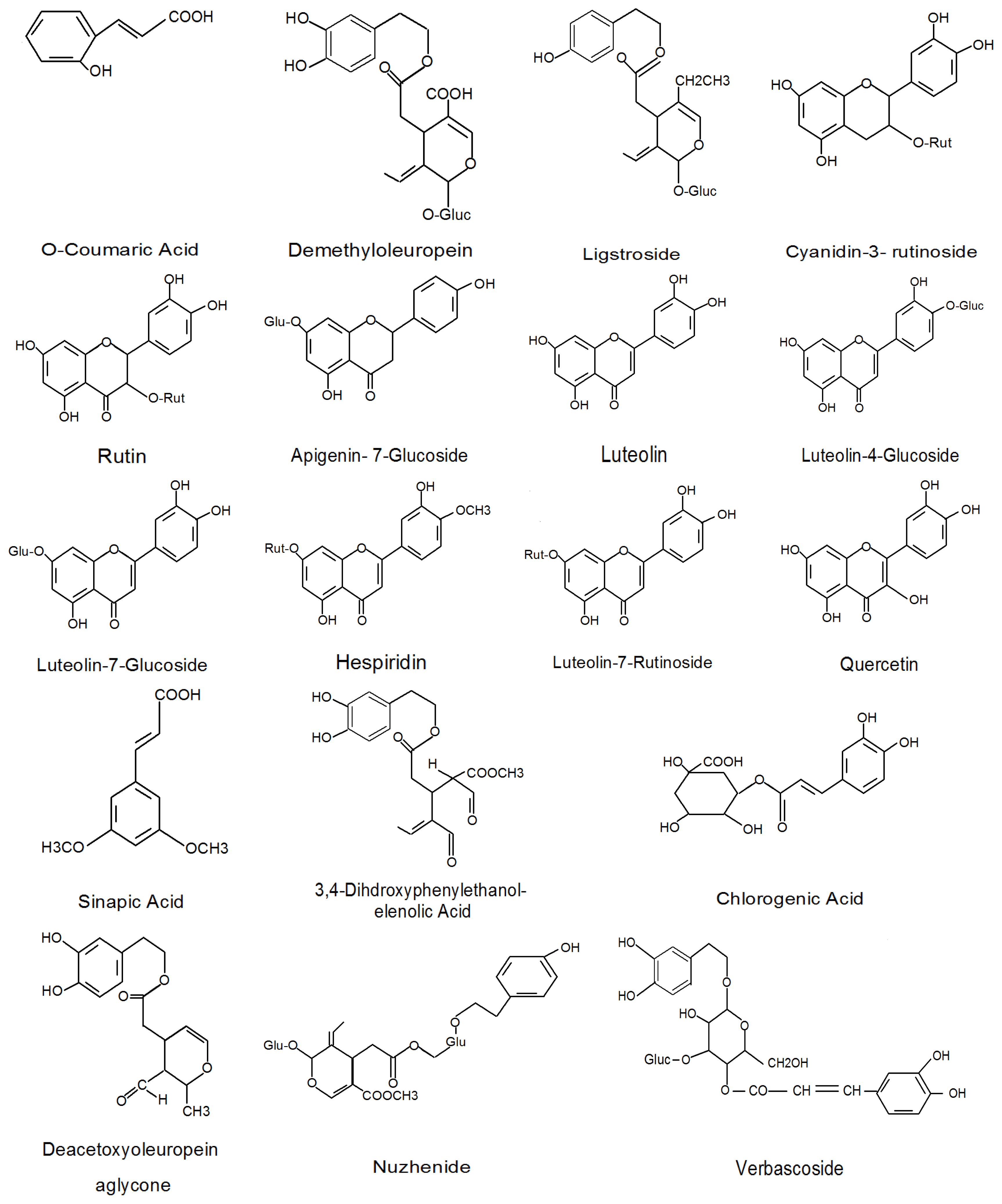
| Phenolic compounds | Reference |
|---|---|
| Flavonols Quercetin-3-rutinoside, Luteolin-7-glucoside, Luteolin-5-glucoside, Apigenin-7-glucoside | [2,36] |
| Phenolic acids Chlorogenic acid, Caffeic acid, p-Hydroxybenzoic acid, Protocatechuic acid, Vanilic acid, Syringic acid, p-Coumaric acid, o-Coumaric acid, Ferulic acid, Sinapic acid, Benzoic acid, Cinnamic acid, Gallic acid | [2,12,36,37] |
| Phenolic alcohols (3,4-Dihydroxyphenyl) ethanol (3,4-DHPEA), (p-Hydroxyphenyl) ethanol (p-HPEA) | [2,31,38] |
| Secoiridoids Oleuropein, Demethyloteuropein, Ligstroside, Nuzhenide | [26,34,35,38,39] |
| Hydroxycinnamic acid derivatives Verbascoside | [26,27] |
| Fatty Acid | IOOC (a) | Arbequina (b) | Abosasana (b) | Koroneiki (b) | Frantoio (c) | Leccino (d) | Busa (d) |
|---|---|---|---|---|---|---|---|
| Myristic acid | < 0.05 | ND | ND | ND | ND | ND | ND |
| Palmitic acid | 7.5–20.0 | 17.57 | 17.78 | 11.65 | 10.9 | 13.7 | 12.07 |
| Palmitoleic acid | 0.3–3.5 | 2.41 | 2.12 | 1.07 | 0.89 | 1.32 | 1.02 |
| Heptadecanoic acid | < 0.3 | ND | ND | ND | 0.07 | ND | ND |
| Stearic acid | 0.5–5.0 | 1.88 | 2.07 | 2.15 | 1.53 | 1.9 | 1.97 |
| Oleic acid | 55.0–83.0 | 58.82 | 64.79 | 75.53 | 78.3 | 75.69 | 74.54 |
| Linoleic acid | 3.5–21.0 | 12.93 | 12.09 | 8.56 | 6.79 | 5.65 | 8.36 |
| Linolenic acid | < 1.0 | 0.63 | 0.54 | 0.26 | 0.49 | 0.161 | 0.66 |
| Arachidic acid | < 0.6 | 0.40 | 0.33 | 0.42 | 0.33 | 0.3 | 0.33 |
| Gadoleic acid (eicosenoic) | < 0.4 | ND | ND | ND | 0.27 | ND | ND |
| Behenic acid | < 0.2 | ND | ND | ND | 0.18 | ND | ND |
| Lignoceric acid | < 0.2 | ND | ND | ND | ND | ND | ND |
| Saturates (%) | Monounsaturates (%) | ω-6 (%) | ω-3(%) | |
|---|---|---|---|---|
| Butter | 45–55 | 35–55 | 1.5–2.5 | 0.5 |
| Lard | 40–46 | 42–44 | 6–8 | 0.5–0.9 |
| Olive oil | 8–14 | 65–83 | 6–15 | 0.2–1.5 |
| Peanut oil | 17–21 | 40–70 | 13–28 | - |
| Maize oil | 12–28 | 32–35 | 40–62 | 0.1–0.5 |
| Soyabean oil | 10–18 | 18–30 | 35–52 | 6.5–9 |
| Sunflower oil | 5–13 | 21–35 | 56–66 | - |
| Biological Activity | Potential Clinical Target | References |
|---|---|---|
| Antioxidant activity | Cardiovascular and degenerative diseases | [101,104,108–125] |
| Anti-inflammatory activity | Inhibition of pro-inflammatory enzymes | [94–96,126,127] |
| Antimicrobial activity | Infectious diseases | [124,128–131] |
| Anti-atherogenic activity | Coronary heart diseases, stroke | [25,95,101,119] |
| Anti tumor activity | Various cancers | [84–86,95,96,132–135] |
| Anti platelet aggregation | Coronary heart diseases, stroke | [95,136,137] |
| Anti-hypertensive activity | Hypertension | [40,83,98,119,138,139] |
| Increased vitamin A and β-carotene activity | Antiaging/skin protection | [25] |
| Increased immune activity | Infectious diseases; various cancers | [25] |
| Anti-allergic activity | [25] | |
| Reduction in the levels of plasma cholesterol and oxidized LDL | Coronary heart diseases | [25,98–100] |
| Attribute/Aroma | Correlated compounds |
|---|---|
| Green | methyl acetate, 1,3-hexadien-5-yne,4-methyl pentan-2-one, 2-methyl-1-propanol, (Z)-3-hexenal, hexyl acetate, 3-hexenyl acetate, (Z)-2-penten-1-ol, (E)-2-hexen-1-ol, (Z)-3-hexen-1-ol |
| Sweet | ethyl furan, ethyl propanoate, 1-penten-3-one, butyl acetate, hexanal, Ethyl butanoate |
| bitter and pungent | ethyl benzene, (E)-2-hexenal, (Z)-2-hexenal, 6-methyl-5-hepten-2-one, quinine, caffeine, alkaloids. tridecene,1-penten-3-one, 1-penten-3-one |
| Undesirable | 1-penten-3-ol, 3-methyl butanol, 2-octanone, 1-hexanol, acetic acid |
| Fruity | 2-butanone, 3-methyl butanal, 2-methyl butyl propanoate, ethenyl benzene, 2-nonanone |
| Musty-humid | 2-heptanone and 2-nonanonetrans |
| Metallic | 1-penten-3-one |
| Rancid | unsaturated aldehydes |
| Bound phenolic compounds | Full-fat olive cakeb | Defatted olive cakeb |
|---|---|---|
| Gallic acid | ND | ND |
| Protocatechuic acid | 21.2 ± 0.24 | 13.8 ± 0.41 |
| Hydroxybenzoic acid | 5.8 ± 0.17 | 7.1 ± 0.17 |
| Vanillic acid | 4.9 ± 0.12 | 4.9 ± 0.43 |
| Caffeic acid | 13.7 ± 0.28 | 11.1 ± 0.25 |
| Syringic acid | 22.4 ± 0.38 | 22.7 ± 0.36 |
| Sinapic acid | 13.1 ± 0.29 | 16.6 ± 0.59 |
| Ferulic acid | 7.2 ± 0.08 | 7.9 ± 0.35 |
| p-Coumaric acid | ND | ND |
| Rutin | 11.7 ± 0.39 | 8.2 ± 0.24 |
| Hesperidin | ND | 4.3 ± 0.13 |
| Quercetin | ND | 3.4 ± 0.19 |
| Cinnamic acid | ND | ND |
| Components | Whole stone (%, w/w) | Seed (%, w/w) |
|---|---|---|
| Ash content | 0.01–0.68 | 0.03–0.13 |
| Moisture content | 9.79 | 9.98 |
| Fat | 5.53 | 1.01 |
| Protein | 3.20 | 1.29 |
| Free suger | 0.48 | 0.36 |
| Phenolics | 0.1 | 0.5–1 |
| Cultivar | Total phenols (mg/kg) | Reference |
|---|---|---|
| Arbequina | 108.27 | [54] |
| Arbosana | 137.84 | [54] |
| Koroneiki | 236.48 | [54] |
| Picual | 400 | [258] |
| Arbequina | 334 | [258] |
| Hojiblanca | 355 | [258] |
| Ornicabra | 495 | [258] |
| Leccino | 130 | [56] |
| Bianchera | 305 | [56] |
| Busa | 125 | [56] |
| Arbequina | 243.8 | [259] |
| Picolimon | 159.9 | [259] |
| Morisca | 435.4 | [259] |
| Compounds | Stone (ppm) | Hammer (ppm) |
|---|---|---|
| Gallic acid | 1.60 ± 0.20 | 1.32 ± 0.14 |
| Tyrosol | 2.99 ± 0.17 | 3.00 ± 0.54 |
| Vanilic acid | 1.83 ± 0.12 | 1.27 ± 0.12 |
| p-Coumaric acid | 2.25 ± 0.16 | 1.97 ± 0.42 |
| Ferulic acid | 1.95 ± 0.12 | 1.62 ± 0.22 |
| Luteolin | 4.66 ± 0.25 | 4.20 ± 0.31 |
| trans-Cinnamic acid | 0.12 ± 0.01 | 0.11 ± 0.01 |
| Apigenin | 1.64 ± 0.17 | 1.61 ± 0.14 |
| Phenolic composition | Two phases (ppm) | Three phases (ppm) |
|---|---|---|
| (3,4-DHPEA) Hydroxytyrosol | 0.87 ± 0.02 | 0.58 ± 0.08 |
| (p-HPEA) Tyrosol | 3.74 ± 0.07 | 2.34 ± 0.08 |
| Vanillic acid | 0.41 ± 0.01 | 0.19 ± 0.01 |
| Caffeic acid | 0.16 ± 0.01 | 0.12 ± 0.02 |
| (3,4-DHPEA-EDA) 3,4-dihydroxyphenyl-ethanol linked to elenolic acid | 522.2 ± 13.5 | 427.2 ± 13.8 |
| (p-HPEA-EDA p-hydroxyphenylethanol linked to dialdehydic form of elenolic acid | 78.16 ± 0.52 | 67.26 ± 2.55 |
| p-HPEA-ester | 38.41 ± 0.10 | 35.62 ± 1.11 |
| (3,4-DHPEA-EA)3,4-dihydroxyphenyl-ethanol linked to elenolic acid | 351.71 ± 11.0 | 244.9 ± 13.6 |
| Total polyphenols | 673 ± 4 | 585 ± 7 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ghanbari, R.; Anwar, F.; Alkharfy, K.M.; Gilani, A.-H.; Saari, N. Valuable Nutrients and Functional Bioactives in Different Parts of Olive (Olea europaea L.)—A Review. Int. J. Mol. Sci. 2012, 13, 3291-3340. https://doi.org/10.3390/ijms13033291
Ghanbari R, Anwar F, Alkharfy KM, Gilani A-H, Saari N. Valuable Nutrients and Functional Bioactives in Different Parts of Olive (Olea europaea L.)—A Review. International Journal of Molecular Sciences. 2012; 13(3):3291-3340. https://doi.org/10.3390/ijms13033291
Chicago/Turabian StyleGhanbari, Rahele, Farooq Anwar, Khalid M. Alkharfy, Anwarul-Hassan Gilani, and Nazamid Saari. 2012. "Valuable Nutrients and Functional Bioactives in Different Parts of Olive (Olea europaea L.)—A Review" International Journal of Molecular Sciences 13, no. 3: 3291-3340. https://doi.org/10.3390/ijms13033291