Arabidopsis Serine Decarboxylase Mutants Implicate the Roles of Ethanolamine in Plant Growth and Development
Abstract
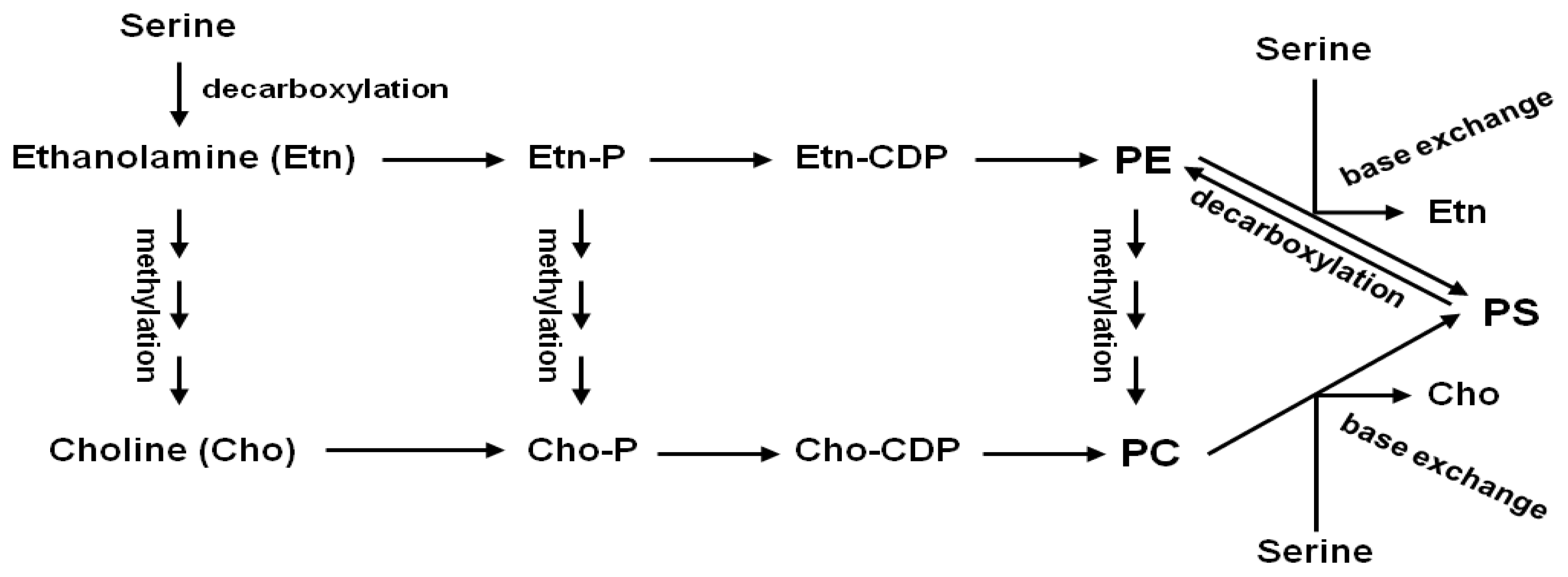
:1. Introduction
2. Results
2.1. Atsdc-1 Developmental Defects and Their Rescue under Short Day Conditions
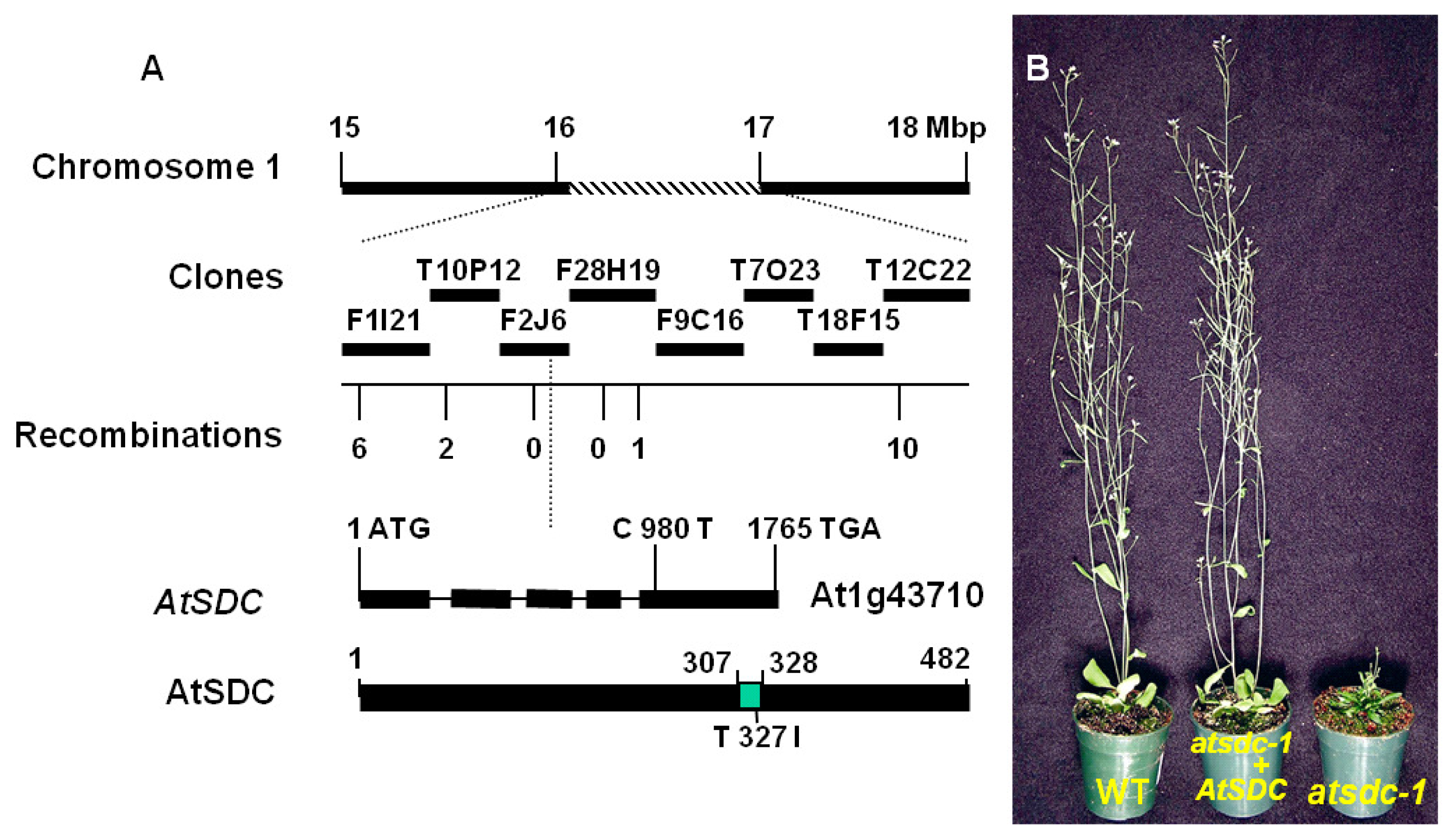
2.2. Molecular Cloning of AtSDC
2.3. AtSDC Encodes a Serine Decarboxylase
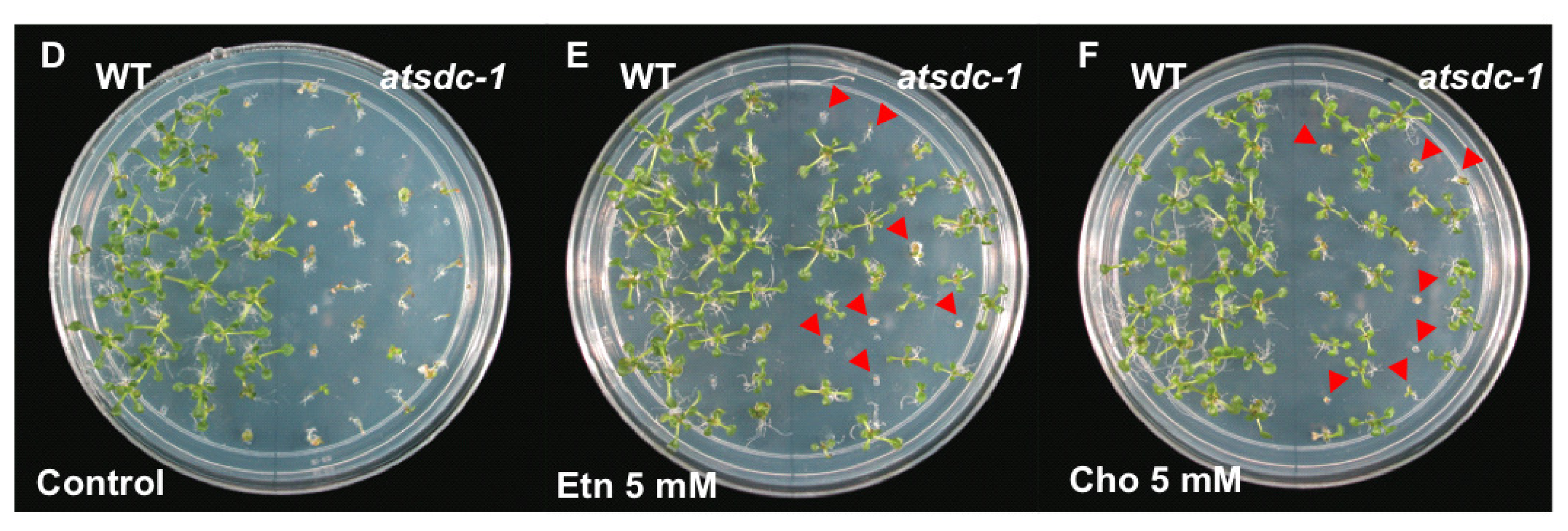
2.4. Ethanolamine Rescues the atsdc-1 Developmental Defects
3. Discussion
4. Experimental Section
4.1. Plant Materials and Growth Conditions
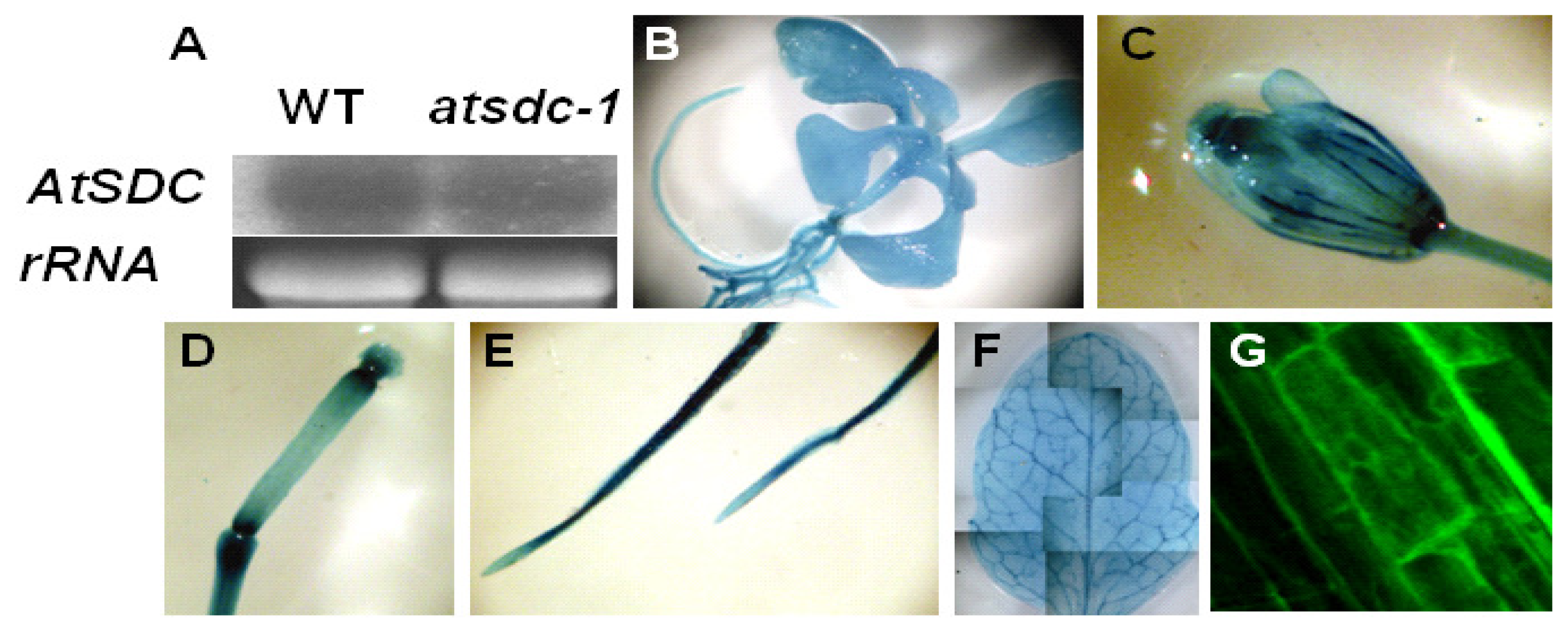
4.2. Gene Expression Analysis
4.3. Positional Cloning
4.4. Plasmid Construction and Plant Transformation
4.5. Microscopic Analysis
5. Conclusions
Acknowledgments
References
- Gibellini, F.; Smith, T.K. The Kennedy pathway-de novo synthesis of phosphatidylethanolamine and phosphatidylcholine. IUBMB Life 2010, 62, 414–428. [Google Scholar]
- Mudd, S.H.; Datko, A.H. Synthesis of ethanolamine and its regulation in Lemna paucicostata. Plant Physiol 1989, 91, 587–597. [Google Scholar]
- Raetz, C.R. Molecular genetics of membrane phospholipid synthesis. Annu. Rev. Genet 1986, 20, 253–295. [Google Scholar]
- Rhodes, D.; Hanson, A.D. Quaternary ammonium and tertiary sulfonium compouns in higher plants. Annu. Rev. Plant Physiol. Plant Mol. Biol 1993, 44, 357–384. [Google Scholar]
- Zinser, E.; Sperkagottlieb, C.D.M.; Fasch, E.V.; Kohlwein, S.D.; Paltauf, F.; Daum, G. Phospholipid synthesis and lipid composition of subcellular membranes in the unicellular membranes in the unicellular eukaryote Saccharomyces cerevisiae. J. Bacteriol 1991, 173, 2026–2034. [Google Scholar]
- Kent, C. Eukaryotic phospholipid biosynthesis. Annu. Rev. Biochem 1995, 64, 315–343. [Google Scholar]
- Rontein, D.; Nishida, I.; Tashiro, G.; Yoshioka, K.; Wu, W.I.; Voelker, D.R.; Basset, G.; Hanson, A.D. Plants synthesize ethanolamine by direct decarboxylation of serine using a pyridoxal phosphate enzyme. J. Biol. Chem 2001, 276, 35523–35529. [Google Scholar]
- Rontein, D.; Rhodes, D.; Hanson, A.D. Evidence from engineering that decarboxylation of free serine is the major source of ethanolamine moieties in plants. Plant Cell Physiol 2003, 44, 1185–1191. [Google Scholar]
- Voelker, D.R. Phosphatidylserine decarboxylase. Biochim. Biophys. Acta-Lipids Lipid Metab 1997, 1348, 236–244. [Google Scholar]
- Vance, J.E.; Steenbergen, R. Metabolism and functions of phosphatidylserine. Prog. Lipid Res 2005, 44, 207–234. [Google Scholar]
- Elabbadi, N.; Ancelin, M.L.; Vial, H.J. Phospholipid metabolism of serine in Plasmodium-infected erythrocytes involves phosphatidylserine and direct serine decarboxylation. Biochem. J 1997, 324, 435–445. [Google Scholar]
- Lykidis, A. Comparative genomics and evolution of eukaryotic phospholipid biosynthesis. Prog. Lipid Res 2007, 46, 171–199. [Google Scholar]
- Kennedy, E.P.; Weiss, S.B. The function of cytidine coenzymes in the biosynthesis of phospholipids. J. Biol. Chem 1956, 222, 193–214. [Google Scholar]
- Wallis, J.G.; Browse, J. Mutants of Arabidopsis reveal many roles for membrane lipids. Prog. Lipid Res 2002, 41, 254–278. [Google Scholar]
- Mou, Z.L.; Wang, X.Q.; Fu, Z.M.; Dai, Y.; Han, C.; Ouyang, J.; Bao, F.; Hu, Y.X.; Li, J.Y. Silencing of phosphoethanolamine N-methyltransferase results in temperature-sensitive male sterility and salt hypersensitivity in Arabidopsis. Plant Cell 2002, 14, 2031–2043. [Google Scholar]
- Yamada, K.; Lim, J.; Dale, J.M.; Chen, H.M.; Shinn, P.; Palm, C.J.; Southwick, A.M.; Wu, H.C.; Kim, C.; Nguyen, M.; et al. Empirical analysis of transcriptional activity in the Arabidopsis genome. Science 2003, 302, 842–846. [Google Scholar]
- Sandmeier, E.; Hale, T.I.; Christen, P. Multiple evolutionary origin of pyridoxal-5′-phosphate-dependent amino acid decarboxylases. Eur. J. Biochem 1994, 221, 997–1002. [Google Scholar]
- The Arabidopsis Information Resource Home Page. Available online: http://www.arabidopsis.org accessed on 14 September 2007.
- Alonso, J.M.; Stepanova, A.N.; Leisse, T.J.; Kim, C.J.; Chen, H.M.; Shinn, P.; Stevenson, D.K.; Zimmerman, J.; Barajas, P.; Cheuk, R.; et al. Genome-wide Insertional mutagenesis of Arabidopsis thaliana. Science 2003, 301, 653–657. [Google Scholar]
- SeedGenes Project Home Page. Available online: http://www.seedgenes.org accessed on 7 November 2008.
- The Arabidopsis Information Resource Germplasm Datapage. Available online: http://www.arabidopsis.org/servlets/TairObject?type=germplasm&id=1005161765 accessed on 10 January 2009.
- Tian, G.W.; Mohanty, A.; Chary, S.N.; Li, S.J.; Paap, B.; Drakakaki, G.; Kopec, C.D.; Li, J.X.; Ehrhardt, D.; Jackson, D.; et al. High-throughput fluorescent tagging of full-length arabidopsis gene products in planta. Plant Physiol 2004, 135, 25–38. [Google Scholar]
- Mizoi, J.; Nakamura, M.; Nishida, I. Defects in CTP: PHOSPHORYLETHANOLAMINE CYTIDYLYLTRANSFERASE affect embryonic and postembryonic development in Arabidopsis. Plant Cell 2006, 18, 3370–3385. [Google Scholar]
- Yamaoka, Y.; Yu, Y.B.; Mizoi, J.; Fujiki, Y.; Saito, K.; Nishijima, M.; Lee, Y.; Nishida, I. Phosphatidylserine Synthase1 is required for microspore development in Arabidopsis thaliana. Plant J 2011, 67, 648–661. [Google Scholar]
- Lightner, J.; James, D.W.; Dooner, H.K.; Browse, J. Altered Body Morphology Is Caused by Increased Stearate Levels in a Mutant of Arabidopsis. Plant J 1994, 6, 401–412. [Google Scholar]
- Masclaux-Daubresse, C.; Valadier, M.H.; Carrayol, E.; Reisdorf-Cren, M.; Hirel, B. Diurnal changes in the expression of glutamate dehydrogenase and nitrate reductase are involved in the C/N balance of tobacco source leaves. Plant Cell Environ 2002, 25, 1451–1462. [Google Scholar]
- Schaffer, R.; Landgraf, J.; Accerbi, M.; Simon, V.; Larson, M.; Wisman, E. Microarray analysis of diurnal and circadian-regulated genes in Arabidopsis. Plant Cell 2001, 13, 113–123. [Google Scholar]
- Matt, P.; Schurr, U.; Klein, D.; Krapp, A.; Stitt, M. Growth of tobacco in short-day conditions leads to high starch, low sugars, altered diurnal changes in the Nia transcript and low nitrate reductase activity, and inhibition of amino acid synthesis. Planta 1998, 207, 27–41. [Google Scholar]
- Liu, J.; Zhu, J.K. Proline accumulation and salt-stress-induced gene expression in a salt-hypersensitive mutant of Arabidopsis. Plant Physiol 1997, 114, 591–596. [Google Scholar]
- Bell, C.J.; Ecker, J.R. Assignment of 30 microsatellite loci to the linkage map of Arabidopsis. Genomics 1994, 19, 137–144. [Google Scholar]
- Monsanto Arabidopsis Polymorphism and Ler Sequence Collections Page. Available online: http://www.arabidopsis.org/Cereon/index.html accessed on 6 March 2007.


© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kwon, Y.; Yu, S.-i.; Lee, H.; Yim, J.H.; Zhu, J.-K.; Lee, B.-h. Arabidopsis Serine Decarboxylase Mutants Implicate the Roles of Ethanolamine in Plant Growth and Development. Int. J. Mol. Sci. 2012, 13, 3176-3188. https://doi.org/10.3390/ijms13033176
Kwon Y, Yu S-i, Lee H, Yim JH, Zhu J-K, Lee B-h. Arabidopsis Serine Decarboxylase Mutants Implicate the Roles of Ethanolamine in Plant Growth and Development. International Journal of Molecular Sciences. 2012; 13(3):3176-3188. https://doi.org/10.3390/ijms13033176
Chicago/Turabian StyleKwon, Yerim, Si-in Yu, Hyoungseok Lee, Joung Han Yim, Jian-Kang Zhu, and Byeong-ha Lee. 2012. "Arabidopsis Serine Decarboxylase Mutants Implicate the Roles of Ethanolamine in Plant Growth and Development" International Journal of Molecular Sciences 13, no. 3: 3176-3188. https://doi.org/10.3390/ijms13033176





