Role of SDF1/CXCR4 Interaction in Experimental Hemiplegic Models with Neural Cell Transplantation
Abstract
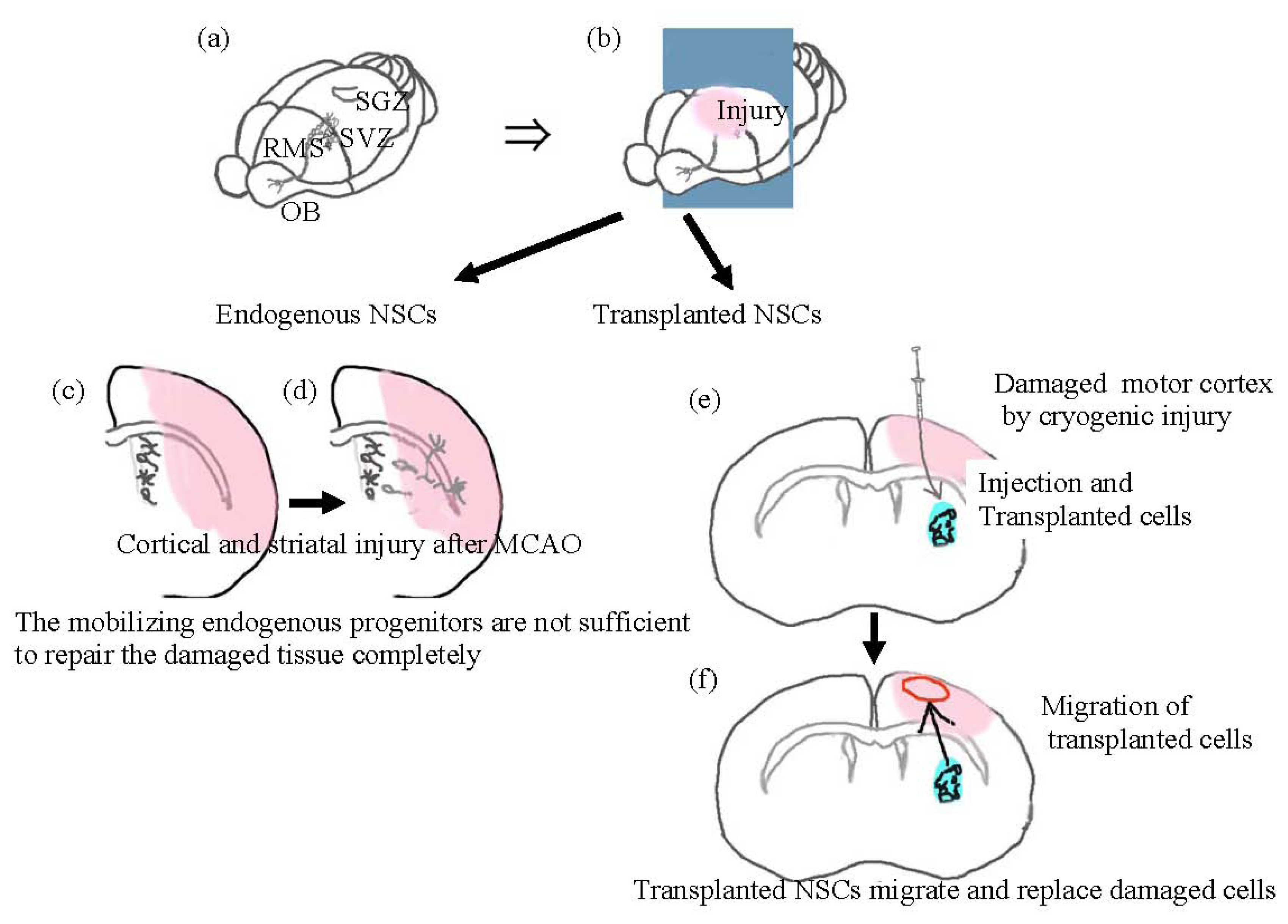
:1. Introduction
2. Chemokine Expressions in the CNS
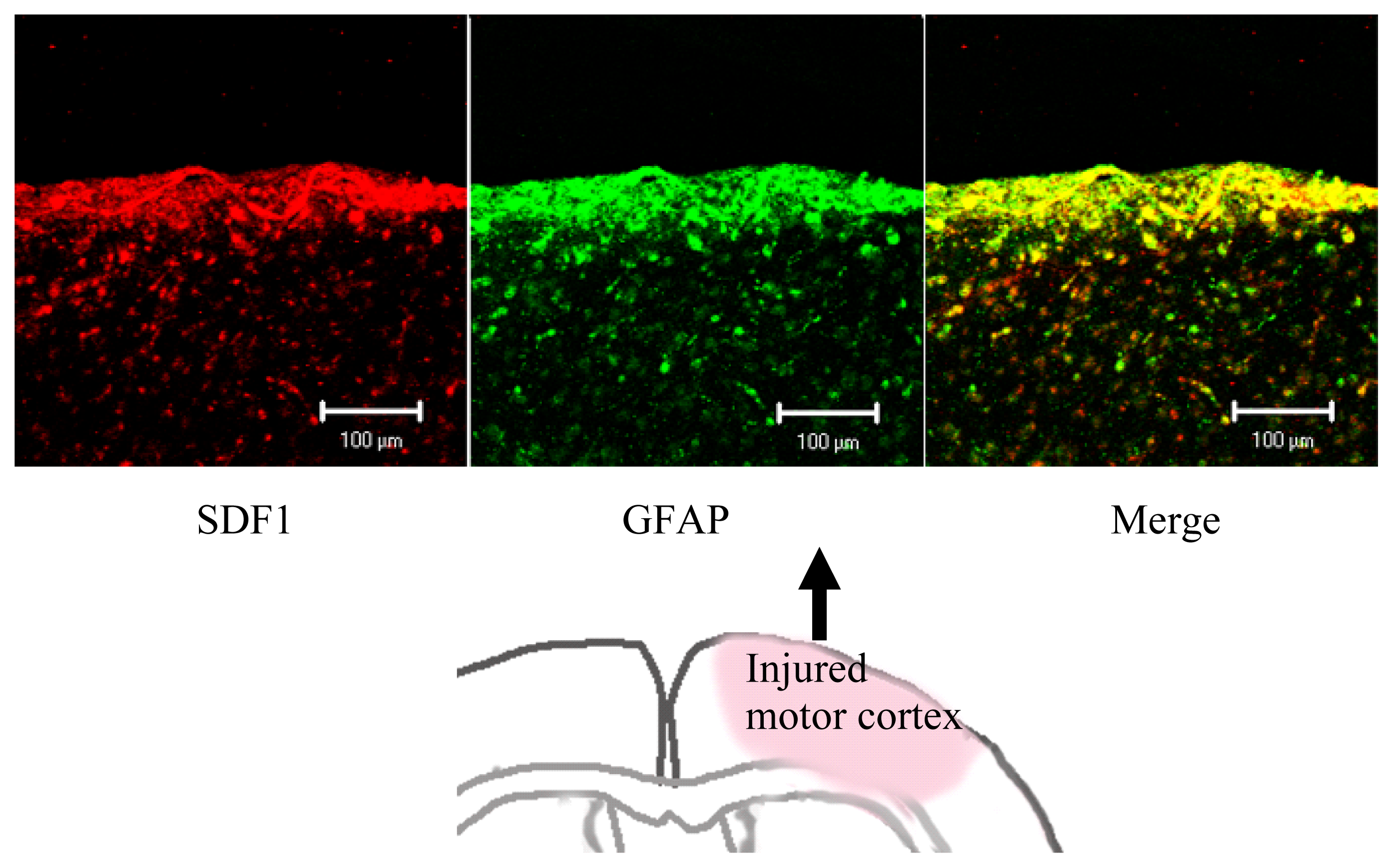
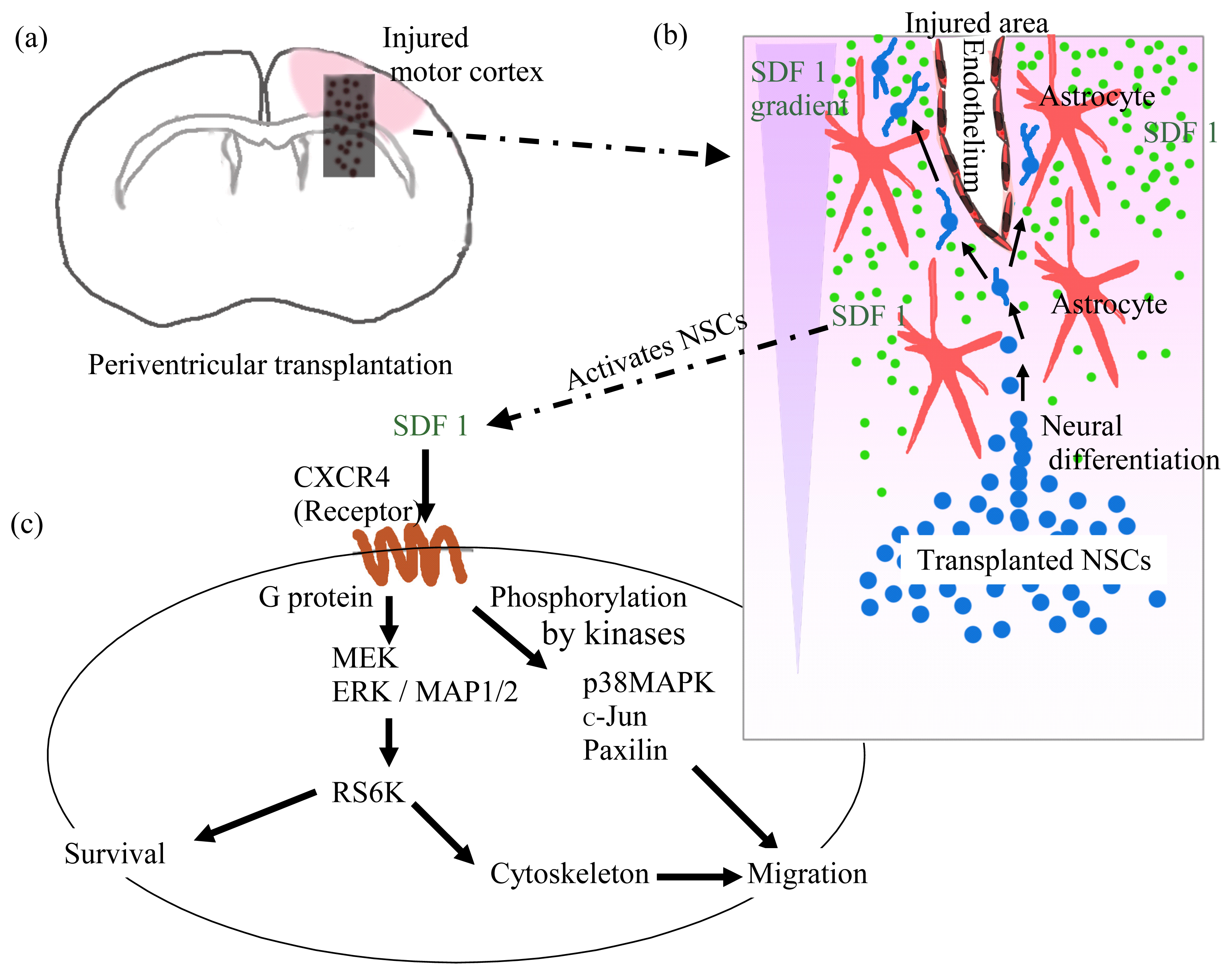
3. Transplanted Neural Cells and Chemokines
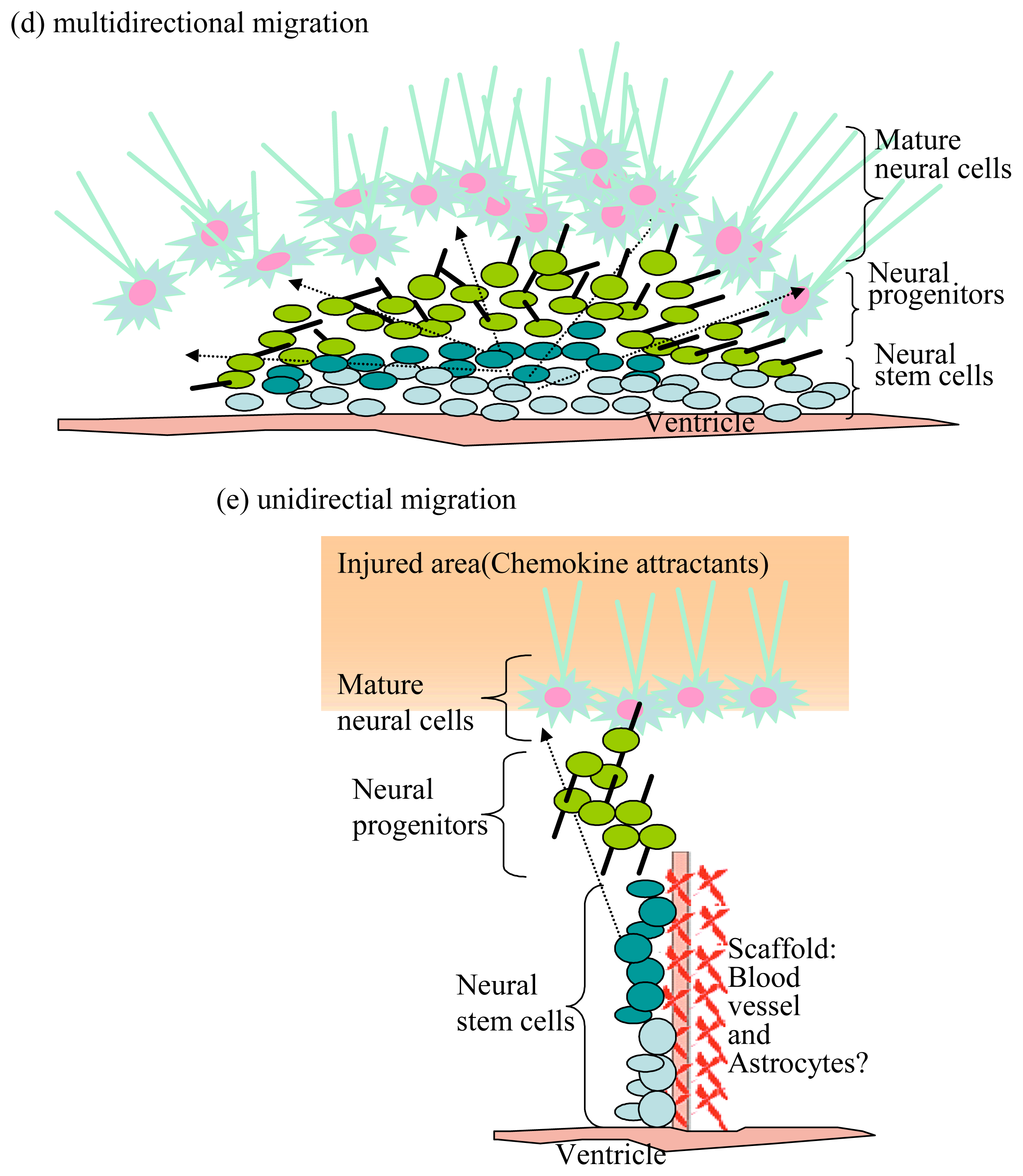
4. Neural Stem/Progenitor Cells and Neural Cell Associated Adhesion Molecules
5. Conclusions
Supplementary Material
ijms-13-02636-s001.pdfReferences
- Lindvall, O.; Kokaia, Z. Stem cells in human neurodegenerative disorders: Time for clinical translation? J. Clin. Invest 2010, 120, 29–40. [Google Scholar]
- Kelly, S.; Bliss, T.M.; Shah, A.K.; Sun, G.H.; Ma, M.; Foo, W.C.; Masel, J.; Yenari, M.A.; Weissman, I.L.; Uchida, N.; et al. Transplanted human fetal neural stem cells survive, migrate, and differentiate in ischemic rat cerebral cortex. Proc. Natl. Acad. Sci. USA 2004, 101, 11839–11844. [Google Scholar]
- Ikeda, R.; Kurokawa, M.S.; Chiba, S.; Yoshikawa, H.; Hashimoto, T.; Tadokoro, M.; Suzuki, N. Transplantation of motoneurons derived from MASH1-transfected mouse ES cells reconstitutes neural networks and improves motor function in hemiplegic mice. Exp. Neurol 2004, 189, 280–292. [Google Scholar]
- Chiba, S.; Ikeda, R.; Kurokawa, M.S.; Yoshikawa, H.; Takeno, M.; Nagafuchi, H.; Tadokoro, M.; Sekino, H.; Hashimoto, T.; Suzuki, N. Anatomical and functional recovery by embryonic stem cell-derived neural tissue of a mouse model of brain damage. J. Neurol. Sci 2004, 219, 107–117. [Google Scholar]
- Hazama, Y.; Kurokawa, M.S.; Chiba, S.; Tadokoro, M.; Imai, T.; Kondo, Y.; Nakatsuji, N.; Suzuki, T.; Hashimoto, T.; Suzuki, N. SDF1/CXCR4 contributes to neural regeneration in hemiplegic mice with a monkey ES-cell-derived neural graft. Inflamm. Regen 2010, 30, 193–205. [Google Scholar]
- Luo, Y. Cell-based therapy for stroke. J. Neural Transm 2011, 118, 61–74. [Google Scholar]
- Doetsch, F.; Caille, I.; Lim, D.A.; Garcia-Verdugo, J.M.; Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 1999, 97, 703–716. [Google Scholar]
- Eriksson, P.S.; Perfilieva, E.; Bjork-Eriksson, T.; Alborn, A.M.; Nordborg, C.; Peterson, D.A.; Gage, F.H. Neurogenesis in the adult human hippocampus. Nat. Med 1998, 4, 1313–1317. [Google Scholar]
- Lois, C.; Alvarez-Buylla, A. Long-distance neuronal migration in the adult mammalian brain. Science 1994, 264, 1145–1148. [Google Scholar]
- McKay, R. Stem cells in the central nervous system. Science 1997, 276, 66–71. [Google Scholar]
- Kameda, M.; Shingo, T.; Takahashi, K.; Muraoka, K.; Kurozumi, K.; Yasuhara, T.; Maruo, T.; Tsuboi, T.; Uozumi, T.; Matsui, T.; et al. Adult neural stem and progenitor cells modified to secrete GDNF can protect, migrate and integrate after intracerebral transplantation in Rats with transient forebrain ischemia. Eur. J. Neurosci 2007, 26, 1462–1478. [Google Scholar]
- Hicks, A.U.; Hewlett, K.; Windle, V.; Chernenko, G.; Ploughman, M.; Jolkkonen, J.; Weiss, S.; Corbett, D. Enriched environment enhances transplanted subventricular zone stem cell migration and functional recovery after stroke. Neuroscience 2007, 146, 31–40. [Google Scholar]
- Arvidsson, A.; Collin, T.; Kirik, D.; Kokaia, Z.; Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat. Med 2002, 8, 963–970. [Google Scholar]
- Zhang, R.; Zhang, Z.; Zhang, C.; Zhang, L.; Robin, A.; Wang, Y.; Lu, M.; Chopp, M. Stroke transiently increases subventricular zone cell division from asymmetric to symmetric and increases neuronal differentiation in the adult rat. J. Neurosci 2004, 24, 5810–5815. [Google Scholar]
- Sanai, N.; Nguyen, T.; Ihrie, R.A.; Mirzadeh, Z.; Tsai, H.H.; Wong, M.; Gupta, N.; Berger, M.S.; Huang, E.; Garcia-Verdugo, J.M.; et al. Corridors of migrating neurons in the human brain and their decline during infancy. Nature 2011, 478, 382–386. [Google Scholar]
- Curtis, M.A.; Faull, R.L.; Eriksson, P.S. The effect of neurodegenerative diseases on the subventricular zone. Nat. Rev. Neurosci 2007, 8, 712–723. [Google Scholar]
- Ikeda, R.; Kurokawa, M.S.; Chiba, S.; Yoshikawa, H.; Ide, M.; Tadokoro, M.; Nito, S.; Nakatsuji, N.; Kondoh, Y.; Nagata, K.; et al. Transplantation of neural cells derived from retinoic acid-treated cynomolgus monkey embryonic stem cells successfully improved motor function of hemiplegic mice with experimental brain injury. Neurobiol. Dis 2005, 20, 38–48. [Google Scholar]
- Rebenko-Moll, N.M.; Liu, L.; Cardona, A.; Ransohoff, R.M. Chemokines, mononuclear cells and the nervous system: Heaven (or hell) is in the details. Curr. Opin. Immunol 2006, 18, 683–689. [Google Scholar]
- Rostene, W.; Guyon, A.; Kular, L.; Godefroy, D.; Barbieri, F.; Bajetto, A.; Banisadr, G.; Callewaere, C.; Conductier, G.; Rovere, C.; et al. Chemokines and chemokine receptors: New actors in neuroendocrine regulations. Front. Neuroendocrinol 2011, 32, 10–24. [Google Scholar]
- Andres, R.H.; Choi, R.; Pendharkar, A.V.; Gaeta, X.; Wang, N.; Nathan, J.K.; Chua, J.Y.; Lee, S.W.; Palmer, T.D.; Steinberg, G.K.; et al. The CCR2/CCL2 interaction mediates the transendothelial recruitment of intravascularly delivered neural stem cells to the ischemic brain. Stroke 2011, 42, 2923–2931. [Google Scholar]
- Meng, S.Z.; Oka, A.; Takashima, S. Developmental expression of monocyte chemoattractant protein-1 in the human cerebellum and brainstem. Brain Dev 1999, 21, 30–35. [Google Scholar]
- Gunsolly, C.; Nicholson, J.D.; Listwak, S.J.; Ledee, D.; Zelenka, P.; Verthelyi, D.; Chapoval, S.; Keegan, A.; Tonelli, L.H. Expression and regulation in the brain of the chemokine CCL27 gene locus. J. Neuroimmunol 2010, 225, 82–90. [Google Scholar]
- Rostasy, K.; Egles, C.; Chauhan, A.; Kneissl, M.; Bahrani, P.; Yiannoutsos, C.; Hunter, D.D.; Nath, A.; Hedreen, J.C.; Navia, B.A. SDF-1α is expressed in astrocytes and neurons in the AIDS dementia complex: An in vivo and in vitro study. J. Neuropathol. Exp. Neurol 2003, 62, 617–626. [Google Scholar]
- Hulshof, S.; van Haastert, E.S.; Kuipers, H.F.; van den Elsen, P.J.; de Groot, C.J.; van der Valk, P.; Ravid, R.; Biber, K. CX3CL1 and CX3CR1 expression in human brain tissue: Noninflammatory control versus multiple sclerosis. J. Neuropathol. Exp. Neurol 2003, 62, 899–907. [Google Scholar]
- Bakhiet, M.; Tjernlund, A.; Mousa, A.; Gad, A.; Stromblad, S.; Kuziel, W.A.; Seiger, A.; Andersson, J. RANTES promotes growth and survival of human first-trimester forebrain astrocytes. Nat. Cell Biol 2001, 3, 150–157. [Google Scholar]
- Hatori, K.; Nagai, A.; Heisel, R.; Ryu, J.K.; Kim, S.U. Fractalkine and fractalkine receptors in human neurons and glial cells. J. Neurosci. Res 2002, 69, 418–426. [Google Scholar]
- Hahn, Y.K.; Vo, P.; Fitting, S.; Block, M.L.; Hauser, K.F.; Knapp, P.E. Beta-chemokine production by neural and glial progenitor cells is enhanced by HIV-1 Tat: Effects on microglial migration. J. Neurochem 2010, 114, 97–109. [Google Scholar]
- Sui, Y.; Potula, R.; Dhillon, N.; Pinson, D.; Li, S.; Nath, A.; Anderson, C.; Turchan, J.; Kolson, D.; Narayan, O.; et al. Neuronal apoptosis is mediated by CXCL10 overexpression in simian human immunodeficiency virus encephalitis. Am. J. Pathol 2004, 164, 1557–1566. [Google Scholar]
- Tong, N.; Perry, S.W.; Zhang, Q.; James, H.J.; Guo, H.; Brooks, A.; Bal, H.; Kinnear, S.A.; Fine, S.; Epstein, L.G.; et al. Neuronal fractalkine expression in HIV-1 encephalitis: Roles for macrophage recruitment and neuroprotection in the central nervous system. J. Immunol 2000, 164, 1333–1339. [Google Scholar]
- Bolin, L.M.; Murray, R.; Lukacs, N.W.; Strieter, R.M.; Kunkel, S.L.; Schall, T.J.; Bacon, K.B. Primary sensory neurons migrate in response to the chemokine RANTES. J. Neuroimmunol 1998, 81, 49–57. [Google Scholar]
- Zou, Y.R.; Kottmann, A.H.; Kuroda, M.; Taniuchi, I.; Littman, D.R. Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature 1998, 393, 595–599. [Google Scholar]
- Lazarini, F.; Casanova, P.; Tham, T.N.; de Clercq, E.; Arenzana-Seisdedos, F.; Baleux, F.; Dubois-Dalcq, M. Differential signalling of the chemokine receptor CXCR4 by stromal cell-derived factor 1 and the HIV glycoprotein in rat neurons and astrocytes. Eur. J. Neurosci 2000, 12, 117–125. [Google Scholar]
- Bagri, A.; Gurney, T.; He, X.; Zou, Y.R.; Littman, D.R.; Tessier-Lavigne, M.; Pleasure, S.J. The chemokine SDF1 regulates migration of dentate granule cells. Development 2002, 129, 4249–4260. [Google Scholar]
- Rezaie, P.; Trillo-Pazos, G.; Everall, I.P.; Male, D.K. Expression of β-chemokines and chemokine receptors in human fetal astrocyte and microglial co-cultures: Potential role of chemokines in the developing CNS. Glia 2002, 37, 64–75. [Google Scholar]
- Robinson, S.; Tani, M.; Strieter, R.M.; Ransohoff, R.M.; Miller, R.H. The Chemokine growth-regulated oncogene-α promotes spinal cord oligodendrocyte precursor proliferation. J. Neurosci 1998, 18, 10457–10463. [Google Scholar]
- Bajetto, A.; Barbero, S.; Bonavia, R.; Piccioli, P.; Pirani, P.; Florio, T.; Schettini, G. Stromal cell-derived factor-1α induces astrocyte proliferation through the activation of extracellular signal-regulated kinases 1/2 pathway. J. Neurochem 2001, 77, 1226–1236. [Google Scholar]
- Bonavia, R.; Bajetto, A.; Barbero, S.; Pirani, P.; Florio, T.; Schettini, G. Chemokines and their receptors in the CNS: Expression of CXCL12/SDF-1 and CXCR4 and their role in astrocyte proliferation. Toxicol. Lett 2003, 139, 181–189. [Google Scholar]
- Losy, J.; Zaremba, J.; Skrobanski, P. CXCL1 (GRO-Alpha) chemokine in acute ischaemic stroke patients. Folia Neuropathol 2005, 43, 97–102. [Google Scholar]
- Zaremba, J.; Ilkowski, J.; Losy, J. Serial measurements of levels of the chemokines CCL2, CCL3 and CCL5 in serum of patients with acute ischaemic stroke. Folia Neuropathol 2006, 44, 282–289. [Google Scholar]
- Cowell, R.M.; Xu, H.; Galasso, J.M.; Silverstein, F.S. Hypoxic-ischemic injury induces macrophage inflammatory protein-1α expression in immature rat brain. Stroke 2002, 33, 795–801. [Google Scholar]
- Schonemeier, B.; Schulz, S.; Hoellt, V.; Stumm, R. Enhanced expression of the CXCL12/SDF-1 chemokine receptor CXCR7 after cerebral ischemia in the rat brain. J. Neuroimmunol 2008, 198, 39–45. [Google Scholar]
- Imitola, J.; Raddassi, K.; Park, K.I.; Mueller, F.J.; Nieto, M.; Teng, Y.D.; Frenkel, D.; Li, J.; Sidman, R.L.; Walsh, C.A.; et al. Directed migration of neural stem cells to sites of CNS injury by the stromal cell-derived factor 1α/CXC chemokine receptor 4 pathway. Proc. Natl. Acad. Sci. USA 2004, 101, 18117–18122. [Google Scholar]
- Hughes, P.M.; Allegrini, P.R.; Rudin, M.; Perry, V.H.; Mir, A.K.; Wiessner, C. Monocyte chemoattractant protein-1 deficiency is protective in a murine stroke model. J. Cereb. Blood Flow Metab 2002, 22, 308–317. [Google Scholar]
- Yan, Y.P.; Sailor, K.A.; Lang, B.T.; Park, S.W.; Vemuganti, R.; Dempsey, R.J. Monocyte chemoattractant protein-1 plays a critical role in neuroblast migration after focal cerebral ischemia. J. Cereb. Blood Flow Metab 2007, 27, 1213–1224. [Google Scholar]
- Tran, P.B.; Ren, D.; Veldhouse, T.J.; Miller, R.J. Chemokine receptors are expressed widely by embryonic and adult neural progenitor cells. J. Neurosci. Res 2004, 76, 20–34. [Google Scholar]
- Robin, A.M.; Zhang, Z.G.; Wang, L.; Zhang, R.L.; Katakowski, M.; Zhang, L.; Wang, Y.; Zhang, C.; Chopp, M. Stromal cell-derived factor 1α mediates neural progenitor cell motility after focal cerebral ischemia. J. Cereb. Blood Flow Metab 2006, 26, 125–134. [Google Scholar]
- Wong, E.V.; Schaefer, A.W.; Landreth, G.; Lemmon, V. Involvement of p90rsk in neurite outgrowth mediated by the cell adhesion molecule L1. J. Biol. Chem 1996, 271, 18217–18223. [Google Scholar]
- Sharma, G.D.; He, J.; Bazan, H.E. P38 and ERK1/2 coordinate cellular migration and proliferation in epithelial wound healing: Evidence of cross-talk activation between MAP kinase cascades. J. Biol. Chem 2003, 278, 21989–21997. [Google Scholar]
- Li, G.; Gustafson-Brown, C.; Hanks, S.K.; Nason, K.; Arbeit, J.M.; Pogliano, K.; Wisdom, R.M.; Johnson, R.S. C-Jun is essential for organization of the epidermal leading edge. Dev. Cell 2003, 4, 865–877. [Google Scholar]
- Huang, C.; Rajfur, Z.; Borchers, C.; Schaller, M.D.; Jacobson, K. JNK phosphorylates paxillin and regulates cell migration. Nature 2003, 424, 219–223. [Google Scholar]
- Lu, M.; Grove, E.A.; Miller, R.J. Abnormal development of the hippocampal dentate gyrus in mice lacking the CXCR4 chemokine receptor. Proc. Natl. Acad. Sci. USA 2002, 99, 7090–7095. [Google Scholar]
- Miller, R.J.; Banisadr, G.; Bhattacharyya, B.J. CXCR4 signaling in the regulation of stem cell migration and development. J. Neuroimmunol 2008, 198, 31–38. [Google Scholar]
- Liu, X.S.; Chopp, M.; Santra, M.; Hozeska-Solgot, A.; Zhang, R.L.; Wang, L.; Teng, H.; Lu, M.; Zhang, Z.G. Functional response to SDF1α through over-expression of CXCR4 on adult subventricular zone progenitor cells. Brain Res 2008, 1226, 18–26. [Google Scholar]
- Levoye, A.; Balabanian, K.; Baleux, F.; Bachelerie, F.; Lagane, B. CXCR7 heterodimerizes with CXCR4 and regulates CXCL12-mediated G protein signaling. Blood 2009, 113, 6085–6093. [Google Scholar]
- Wang, Y.; Li, G.; Stanco, A.; Long, J.E.; Crawford, D.; Potter, G.B.; Pleasure, S.J.; Behrens, T.; Rubenstein, J.L. CXCR4 and CXCR7 have distinct functions in regulating interneuron migration. Neuron 2011, 69, 61–76. [Google Scholar]
- Rubin, J.B.; Kung, A.L.; Klein, R.S.; Chan, J.A.; Sun, Y.; Schmidt, K.; Kieran, M.W.; Luster, A.D.; Segal, R.A. A small-molecule antagonist of CXCR4 inhibits intracranial growth of primary brain tumors. Proc. Natl. Acad. Sci. USA 2003, 100, 13513–13518. [Google Scholar]
- Iwai, M.; Sato, K.; Kamada, H.; Omori, N.; Nagano, I.; Shoji, M.; Abe, K. Temporal profile of stem cell division, migration, and differentiation from subventricular zone to olfactory bulb after transient forebrain ischemia in gerbils. J. Cereb. Blood Flow Metab 2003, 23, 331–341. [Google Scholar]
- Battista, D.; Rutishauser, U. Removal of polysialic acid triggers dispersion of subventricularly derived neuroblasts into surrounding CNS Tissues. J. Neurosci 2010, 30, 3995–4003. [Google Scholar]
- Yamashita, T.; Ninomiya, M.; Acosta, P.H.; Garcia-Verdugo, J.M.; Sunabori, T.; Sakaguchi, M.; Adachi, K.; Kojima, T.; Hirota, Y.; Kawase, T.; et al. Subventricular zone-derived neuroblasts migrate and differentiate into mature neurons in the post-stroke adult striatum. J. Neurosci 2006, 26, 6627–6636. [Google Scholar]
- Kiryushko, D.; Berezin, V.; Bock, E. Regulators of neurite outgrowth: Role of cell adhesion molecules. Ann. N. Y. Acad. Sci 2004, 1014, 140–154. [Google Scholar]
- Maness, P.F.; Schachner, M. Neural recognition molecules of the immunoglobulin superfamily: Signaling transducers of axon guidance and neuronal migration. Nat. Neurosci 2007, 10, 19–26. [Google Scholar]
- Rathjen, F.G.; Schachner, M. Immunocytological and biochemical characterization of a new neuronal cell surface component (L1 antigen) which is involved in cell adhesion. EMBO J 1984, 3, 1–10. [Google Scholar]
- Takei, K.; Chan, T.A.; Wang, F.S.; Deng, H.; Rutishauser, U.; Jay, D.G. The neural cell adhesion molecules L1 and NCAM-180 act in different steps of neurite outgrowth. J. Neurosci 1999, 19, 9469–9479. [Google Scholar]
- Cohen, N.R.; Taylor, J.S.; Scott, L.B.; Guillery, R.W.; Soriano, P.; Furley, A.J. Errors in corticospinal axon guidance in mice lacking the neural cell adhesion molecule L1. Curr. Biol 1998, 8, 26–33. [Google Scholar]
- Tomasiewicz, H.; Ono, K.; Yee, D.; Thompson, C.; Goridis, C.; Rutishauser, U.; Magnuson, T. Genetic deletion of a neural cell adhesion molecule variant (N-CAM-180) produces distinct defects in the central nervous system. Neuron 1993, 11, 1163–1174. [Google Scholar]
- Kadowaki, M.; Nakamura, S.; Machon, O.; Krauss, S.; Radice, G.L.; Takeichi, M. N-cadherin mediates cortical organization in the mouse brain. Dev. Biol 2007, 304, 22–33. [Google Scholar]
- Amoureux, M.C.; Cunningham, B.A.; Edelman, G.M.; Crossin, K.L. N-CAM binding inhibits the proliferation of hippocampal progenitor cells and promotes their differentiation to a neuronal phenotype. J. Neurosci 2000, 20, 3631–3640. [Google Scholar]
- Chazal, G.; Durbec, P.; Jankovski, A.; Rougon, G.; Cremer, H. Consequences of neural cell adhesion molecule deficiency on cell migration in the rostral migratory stream of the mouse. J. Neurosci 2000, 20, 1446–1457. [Google Scholar]
| Chemokine | Receptor | Distribution |
|---|---|---|
| Physiological | ||
| In vivo | ||
| MCP1/CCL2 | CCR2 | Fetal and adult brain: Neurons [21] |
| CTACK/CCL27 | CCR10 | Adult brain: Neurons [22] |
| SDF1/CXCL12 | CXCR4 | Adult brain: Neurons [23] |
| Fractalkine/CX3CL1 | CX3CR1 | Adult brain: Astrocytes [24] |
| In vitro | ||
| MCP1/CCL2 | CCR2 | Primary astrocyte culture [25] |
| MIP1α/CCL3 | CCR3 | Primary astrocyte culture [25] |
| MIP1β/CCL4 | CCR5 | Primary astrocyte culture [25] |
| RANTES/CCL5 | CCR5 | Primary astrocyte culture [25] |
| SDF1/CXCL12 | CXCR4 | Primary fetal neural cell culture [23] |
| Fractalkine/CX3CL1 | CX3CR1 | Primary neural and glial cell culture [26] |
| Pathological | ||
| MCP1/CCL2 | CCR2 | Astrocytes in HIV encephalitis [27] |
| RANTES/CCL5 | CCR5 | Astrocytes in HIV encephalitis [27] |
| IP10/CXCL10 | CXCR3 | Neurons in HIV encephalitis [28] |
| SDF1/CXCL12 | CXCR4 | Neurons and Astrocytes in AIDS dementia [23] |
| Fractalkine/CX3CL1 | CX3CR1 | Astrocytes in multiple sclerosis [24] Neurons in HIV encephalitis [29] |
| Chemokine | Receptor | Target Cells |
|---|---|---|
| Cell Migration | ||
| RANTES/CCL5 | CCR5 | Neurons [30] |
| SDF1/CXCL12 | CXCR4 | Cerebellar granule neurons [31] Cortical neural progenitors [32] Dentate gyrus granular neurons [33] |
| MCP1/CCL2 | CCR2 | Astrocytes [34] |
| Cell Proliferation | ||
| MIP1α/CCL3 | CCR3 | Astrocytes [34] |
| RANTES/CCL5 | CCR5 | Astrocytes [25] |
| GROα/CXCL1 | CXCR2 | Astrocytes, Oligodendrocyte precursors [35] |
| SDF1/CXCL12 | CXCR4 | Astrocytes [36,37] |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Arimitsu, N.; Shimizu, J.; Fujiwara, N.; Takai, K.; Takada, E.; Kono, T.; Ueda, Y.; Suzuki, T.; Suzuki, N. Role of SDF1/CXCR4 Interaction in Experimental Hemiplegic Models with Neural Cell Transplantation. Int. J. Mol. Sci. 2012, 13, 2636-2649. https://doi.org/10.3390/ijms13032636
Arimitsu N, Shimizu J, Fujiwara N, Takai K, Takada E, Kono T, Ueda Y, Suzuki T, Suzuki N. Role of SDF1/CXCR4 Interaction in Experimental Hemiplegic Models with Neural Cell Transplantation. International Journal of Molecular Sciences. 2012; 13(3):2636-2649. https://doi.org/10.3390/ijms13032636
Chicago/Turabian StyleArimitsu, Nagisa, Jun Shimizu, Naruyoshi Fujiwara, Kenji Takai, Erika Takada, Takao Kono, Yuji Ueda, Tomoko Suzuki, and Noboru Suzuki. 2012. "Role of SDF1/CXCR4 Interaction in Experimental Hemiplegic Models with Neural Cell Transplantation" International Journal of Molecular Sciences 13, no. 3: 2636-2649. https://doi.org/10.3390/ijms13032636




