Lipid Peroxidative Damage on Cisplatin Exposure and Alterations in Antioxidant Defense System in Rat Kidneys: A Possible Protective Effect of Selenium
Abstract
:1. Introduction
2. Experimental Methods
2.1. Chemicals
2.2. Animals and Treatments
- Group 1: Control rats, treated intraperitoneally (i.p.) with isotonic saline.
- Group 2: Cisplatin (CP) (received i.p. a single dose of 7 mg CP/kg b.w.).
- Group 3: Selenium (Se) (received a single i.p. injection of Na2SeO3 in the dose of 6 mg Se/kg b.w.).
- Group 4: Se + CP (treated by Se 1 h before CP injection in the above mentioned amounts).
2.3. Tissue Preparation
2.4. Biochemical Analysis
2.4.1. Lipid Peroxidation Assay
2.4.2. Determination of Antioxidant Enzyme Activity
2.4.3. Reduced Glutathione (GSH) Assay
2.4.4. Protein Concentration Assay
2.5. Statistical Analysis
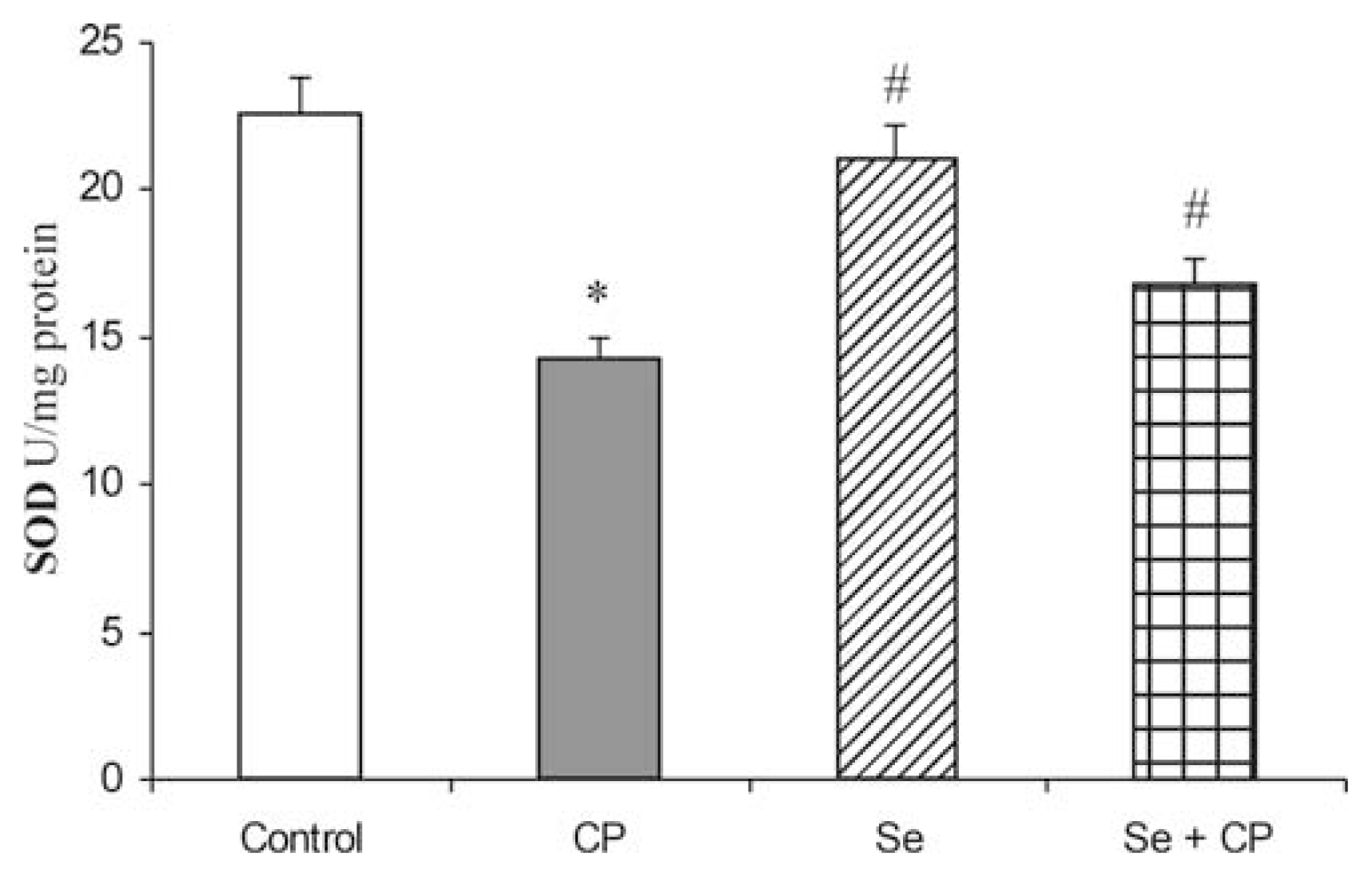
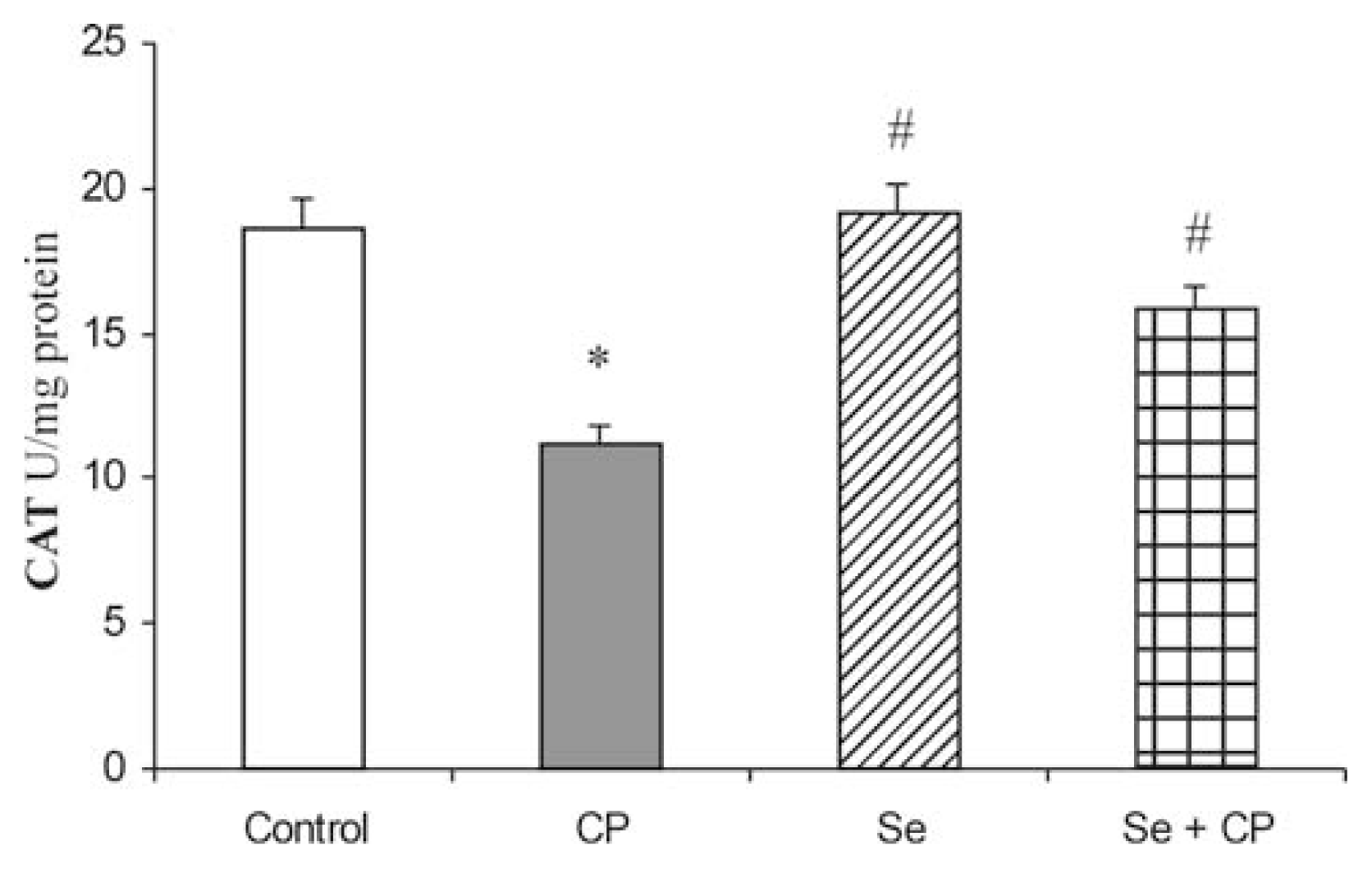
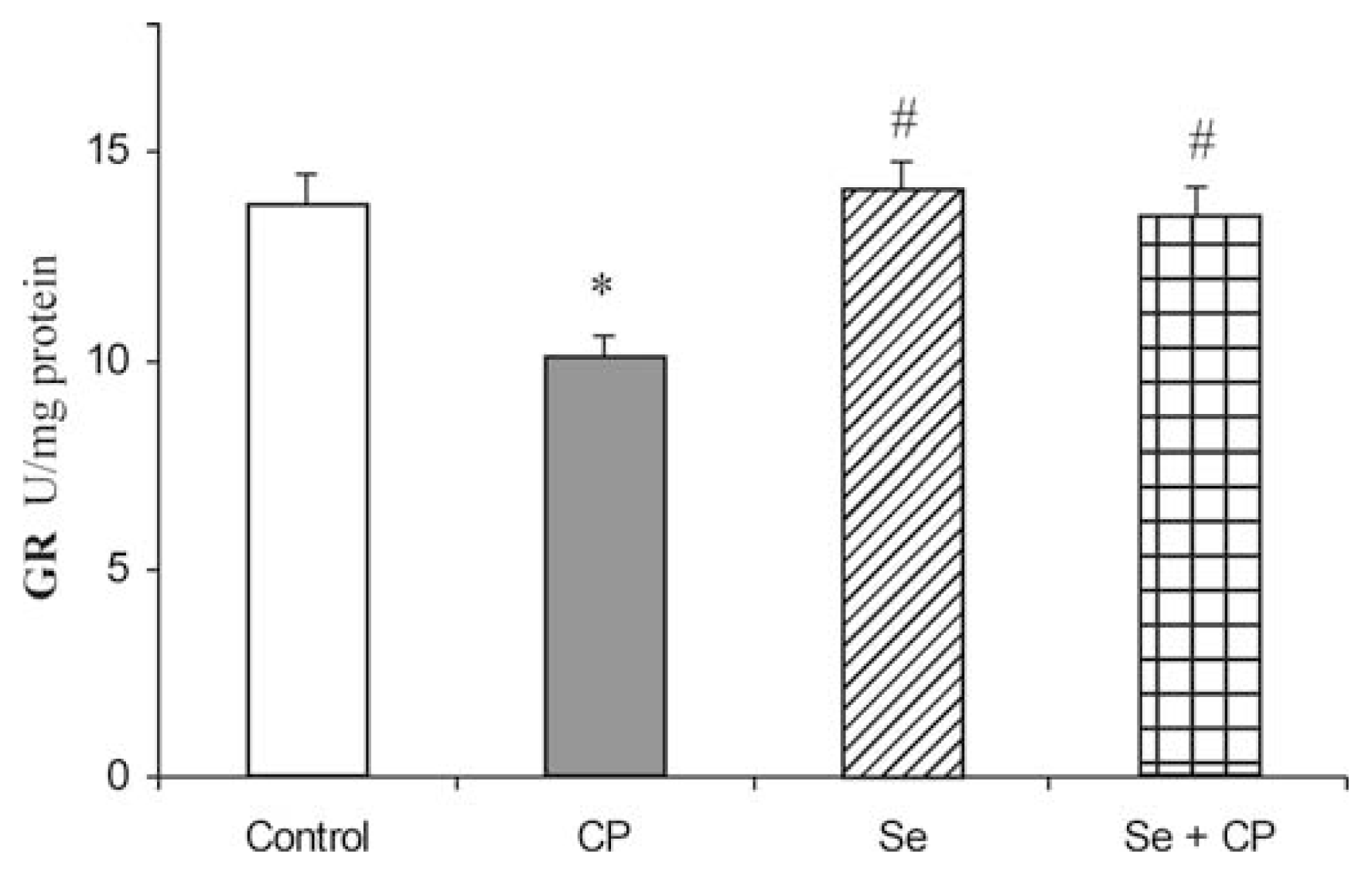
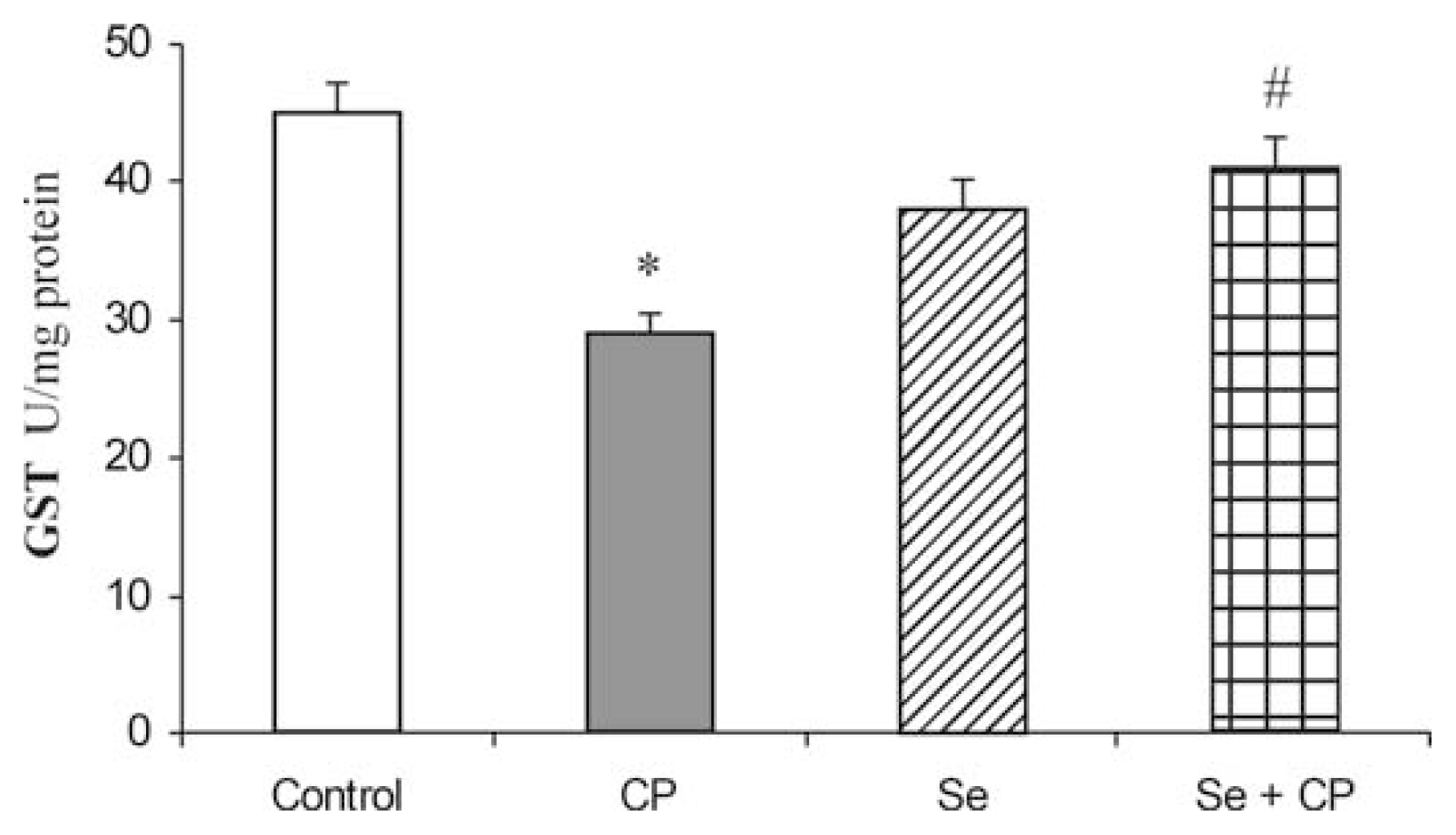
3. Results
4. Discussion
5. Conclusions
Acknowledgements
- Conflict of InterestThe authors declare that there are no conflicts of interest.
References
- Baek, S.M.; Kwon, C.H.; Kim, J.H.; Woo, J.S.; Jung, J.S.; Kim, Y.K. Differential roles of hydrogen peroxide and hydroxyl radical in cisplatin-induced cell death in renal proximal tubular epithelial cells. J. Lab. Clin. Med 2003, 142, 178–186. [Google Scholar]
- Hanigan, M.H.; Devarajan, P. Cisplatin nephrotoxicity: Molecular mechanisms. Cancer Ther 2003, 1, 47–61. [Google Scholar]
- Jordan, P.; Carmo-Fonseca, M. Molecular mechanisms involved in cisplatin cytotoxicity. Cell Mol. Life Sci 2000, 57, 1229–1235. [Google Scholar]
- Kroning, R.; Lichestenstein, A.K.; Nagami, G.T. Sulfur-containing amino acids decrease cisplatin cytotoxicity and uptake in renal tubular epithelial cell lines. Cancer Chemother. Pharmacol 2000, 45, 43–49. [Google Scholar]
- Cohen, S.M.; Lippard, S.J. Cisplatin: From DNA damage to cancer chemotherapy. Prog. Nucleic Acis. Res. Mol. Biol 2001, 67, 93–130. [Google Scholar]
- Sadowitz, P.D.; Hubbard, B.A.; Dabrowiak, J.C.; Goodisman, J.; Tacka, K.A.; Aktas, M.K.; Cunningham, M.J.; Dubowy, R.L.; Souid, A.K. Kinetics of cisplatin binding to cellular DNA and modulations by thiolblocking agents and thiol drugs. Drug Metab. Dispos 2002, 30, 183–190. [Google Scholar]
- Mora, L.O.; Antunes, L.M.G.; Francescato, H.D.C.; Bianchi, M.L.P. The effects of oral glutamine on cisplatin-induced nephrotoxicity in rats. Pharmacol. Res 2003, 47, 517–522. [Google Scholar]
- Matsushima, H.; Yonemura, K.; Ohishi, K.; Hishida, A. The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J. Lab. Clin. Med 1998, 131, 518–526. [Google Scholar]
- Cetin, R.; Devrim, E.; Kilicoglu, B.; Avei, A.; Candir, O.; Durak, I. Cisplatin impairs antioxidant system and causes oxidation in rat kidney tissues: Possible protective roles of natural antioxidant foods. J. Appl. Toxicol 2006, 26, 42–46. [Google Scholar]
- Zhang, J.G.; Lindup, W.E. Role of mitochondria in cisplatin-induced oxidative damage exhibited by rat renal cortical slides. Biochem. Pharmacol 1993, 45, 2215–2222. [Google Scholar]
- El-Daly, E.S. Protective effect of cysteine and vitamin E, Crocus sativus and Nigella sativa extracts on cisplatin-induced toxicity in rats. J. Pharm. Belg 1998, 53, 87–95. [Google Scholar]
- Kim, Y.K.; Jung, J.S.; Lee, S.H.; Kim, Y.W. Effects of antioxidants and Ca2+ in cisplatin-induced cell injury in rabbit renal cortical slices. Toxicol. Appl. Pharmacol 1997, 146, 261–269. [Google Scholar]
- Naziroğlu, M.; Karaoglu, A.; Askoy, A.O. Selenium and high dose vitamin E administration protects cisplatin-induced oxidative damage to renal, liver and lens tissues in rats. Toxicology 2004, 195, 221–230. [Google Scholar]
- Atessahin, A.; Yilmaz, S.; Karahan, I.; Ceribasi, A.O.; Karaoglu, A. Effects of lycopene against cisplatin-induced nephrotoxicity and oxidative stress in rats. Toxicology 2005, 212, 116–123. [Google Scholar]
- Atessahin, A.; Sahna, E.; Türk, G.; Ceribasi, A.O.; Yilmaz, S.; Yüce, A.; Bulmus, O. Chemoprotective effect of melatonin against cisplatin-induced testicular toxicity in rats. J. Pineal. Res 2006, 41, 21–27. [Google Scholar]
- Badary, O.A.; Abdel-Maksoud, S.; Ahmed, W.A.; Owieda, G.H. Naringenin attenuates cisplatin nephrotoxicity in rats. Life Sci 2005, 76, 2125–2135. [Google Scholar]
- Yüce, A.; Atessahin, A.; Ceribasi, A.O.; Aksakal, M. Ellagic acid prevents cisplatin-induced oxidative stress in liver and heart tissue of rats. Basic Clin. Pharmacol. Toxicol 2007, 101, 345–349. [Google Scholar]
- Atasayar, S.; Gürer-Orhan, H.; Orhan, H.; Gürel, B.; Girgin, G.; Özgüneş, H. Preventive effect of aminoguanidine compared to vitamin E and C on cisplatin-induced nephrotoxicity in rats. Exp. Toxicol. Pathol 2009, 61, 23–32. [Google Scholar]
- Combs, G.; Gray, W.P. Chemopreventive agents: Selenium. Pharmacol. Ther 1998, 79, 179–192. [Google Scholar]
- Caffrey, P.B.; Frenkel, G.D. Selenium compounds prevent the induction of drug resistance by cisplatin in human ovarian tumor xenografts in vivo. Cancer Chemother. Pharmacol 2000, 46, 74–78. [Google Scholar]
- Chen, J.; Berry, M.J. Selenium and selenoproteins in the brain and brain diseases. J. Neurochem 2003, 86, 1–12. [Google Scholar]
- Saito, Y.; Yoshida, Y.; Akazawa, T.; Takahashi, K.; Niki, E. Cell death caused by selenium deficiency and protective effect of antioxidants. J. Biol. Chem. 2003, 278, 39428–39434. [Google Scholar]
- Agay, D.; Sandre, C.; Ducros, V.; Faure, H.; Cruz, C.; Alonso, A.; Roussel, A.M.; Chancerelle, Y. Optimization of selenium status by a single intraperitoneal injection of Se in Se-deficient rat: Possible application to burned patient treatment. Free Radic. Biol. Med 2005, 39, 762–768. [Google Scholar]
- Kim, Y.S.; Combs, J.G.F. Effects of dietary selenium and vitamin E on glutathione concentrations and glutathione S-transferase activities in chick liver and plasma. Nutr. Res 1993, 13, 455–463. [Google Scholar]
- Dilsiz, N.; Ölçücü, A.; Çay, M.; Nazıroğlu, M.; Çobanoglu, D. Protective effects of selenium, vitamin C and vitamin E against oxidation stress of cigarette smoke in rats. Cell Biochem. Funct 1999, 17, 1–7. [Google Scholar]
- Ognjanović, B.I.; Marković, S.D.; Pavlović, S.Z.; Žikić, R.V.; Štajn, A.Š.; Saičić, Z.S. Effect of chronic cadmium exposure on antioxidant defense system in some tissues of rats: Protective effect of selenium. Physiol. Res. 2008, 57, 403–411. [Google Scholar]
- Jihen, E.H.; Imed, M.; Fatima, H.; Abdelhamid, K. Protective effects of selenium (Se) and zinc (Zn) on cadmium (Cd) toxicity in the liver of the rat: Effects on the oxidative stress. Ecotoxicol. Environ. Saf 2009, 72, 1559–1564. [Google Scholar]
- Su, L.; Wang, M.; Yin, S.T.; Wang, H.L.; Chen, L.; Sun, L.G.; Ruan, D.Y. The interaction of selenium and mercury in the accumulations and oxidative stress of rat tissues. Ecotoxicol. Environ. Saf 2008, 70, 483–489. [Google Scholar]
- Schrauzer, G.N. Selenium and selenium-antagonistic elements in nutritional cancer prevention. Crit. Rev. Biotechnol 2009, 29, 10–17. [Google Scholar]
- Gregus, Z.; Gyurasics, A.; Csanaky, I. Effects of arsenic-, platinum- and gold-containing drugs on the disposition of exogenous selenium in rats. Toxicol. Sci 2000, 57, 22–31. [Google Scholar]
- Ohkawa, H.; Okishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem 1979, 95, 351–358. [Google Scholar]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and simple assay for superoxide dismutase. J. Biol. Chem 1972, 247, 3170–3175. [Google Scholar]
- Beutler, E. Catalase. In Red cell Metabolism, a Manual of Biochemical Methods; Beutler, E., Ed.; Grune and Stratton: New York, NY, USA, 1982; pp. 105–116. [Google Scholar]
- Tamura, M.; Oschino, N.; Chance, B. Some characteristics of hydrogen and alkylhydroperoxides metabolizing systems in cardiac tissue. J. Biochem 1982, 92, 1019–1031. [Google Scholar]
- Glatzle, D.; Vulliemuier, J.P.; Weber, F.; Decker, K. Glutathione reductase test with whole blood, a convenient procedure for the assesment of the riboflavin status in humans. Experientia 1974, 30, 665–667. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione-S-transferase. J. Biol. Chem 1974, 249, 7130–7139. [Google Scholar]
- Beutler, E. Reduced Glutathione (GSH). In Red Cell Metabolism, a Manual of Biochemical Methods; Beutler, E., Ed.; Grune and Straton: New York, NY, USA, 1975; pp. 112–114. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem 1951, 193, 265–267. [Google Scholar]
- Antunes, L.M.G.; Darin, J.D.C.; Bianchi, M.L.P. Protective effects of vitamin C against cisplatin-induced nephrotoxicity and lipid peroxidation in adult rats: A dose-dependant study. Pharmacol. Res 2000, 41, 405–411. [Google Scholar]
- Mukhopadhyay, P.; Horváth, B.; Zsengellér, Z.; Zielonka, J.; Tanchian, G.; Holovac, E.; Kechrid, M.; Patel, V.; Stillman, I.E.; Parikh, S.M.; et al. Mitochondrial-targeted antioxidants represent a promising approach for prevention of cisplatin-induced nephropathy. Free Radic. Biol. Med 2011. [Google Scholar] [CrossRef]
- Ognjanović, B.I.; Marković, S.D.; Djordjević, N.Z.; Trbojević, I.S.; Štajn, A.Š.; Saičić, Z.S. Cadmium-induced lipid peroxidation and changes in antioxidant defense system in the rat testes: Protective role of coenzyme Q10 and Vitamin E. Reprod. Toxicol. 2010, 29, 191–197. [Google Scholar]
- Matés, M. Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 2000, 153, 83–104. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 4th ed; Oxford University Press: New York, NY, USA, 2007. [Google Scholar]
- Hrubisko, M.; Mc Gown, A.T.; Fox, B.W. The role of metallothionein, glutathione, glutathione S-transferases and DNA repair in resistance to platinum drugs in a series of L1210 cell lines made resistant to anticancer platinum agents. Biochem. Pharmacol 1993, 45, 253–256. [Google Scholar]
- Chen, G.; Hutter, K.J.; Zeller, W.J. Positive correlation between cellular glutathione and acquired cisplatin resistance in human ovarian cancer cells. Cell Biol. Toxicol 1995, 11, 273–281. [Google Scholar]
- Griffith, O.W. Biologic and pharmacologic regulation of mammalian glutathione synthesis. Free Radic. Biol. Med 1999, 27, 922–935. [Google Scholar]
- Baldew, G.S.; Mol, J.G.J.; de Kanter, F.J.J.; Baar, B.V.; de Goeij, J.J.M.; Vermeulen, N.P.E. The mechanism of interaction between cisplatin and selenite. Biochem. Pharmacol 1991, 41, 1429–1437. [Google Scholar]
- Sies, H. Glutathione and its cellular functions. Free Radic. Biol. Med 1993, 27, 916–921. [Google Scholar]
- Yoshida, M.; Itzuka, K.; Hara, M.; Nishijima, H.; Shimada, A.; Nakada, K.; Satoh, Y.; Akama, Y.; Terada, A. Prevention of nephrotoxicity of cisplatin by repeated oral administration of ebselen in rats. Tohoku J. Exp. Med 2000, 191, 209–220. [Google Scholar]
- Sadzuka, Y.; Shimizu, Y.; Takino, Y. Role of glutathione S-transferase isoenzymes in cisplatin-induced nephrotoxicity in the rat. Toxicol. Lett 1994, 70, 211–222. [Google Scholar]
- Olas, B.; Washowicz, B.; Bald, E.; Glowacki, R. The protective effects of resveratrol against changes in blood platelet thiols induced by platinum compounds. J. Physiol. Pharmacol 2004, 55, 467–476. [Google Scholar]
- Feinfeld, D.A.; Fuh, V.L.; Safirstein, R. Urinary glutathione-S-transferase in cisplatin nephrotoxicity in the rat. J. Clin. Chem. Clin. Biochem 1986, 24, 529–532. [Google Scholar]
- Dedon, P.C.; Borch, R.F. Characterization of the reactions of platinum antitumor agents with biologic and nonbiologic sulfur-containing nucleophiles. Biochem. Pharmacol 1987, 36, 1955–1964. [Google Scholar]
- Conklin, K.A. Dietary antioxidans during cancer chemotherapy: Impact on chemotherapeutic effectiveness and development of side effects. Nutr. Cancer 2000, 37, 1–18. [Google Scholar]
- Schweizer, U.; Bräuer, A.U.; Köhrle, J.; Nitsch, R.; Savaskan, N.E. Selenium and brain function: A poorly recognized liaison. Brain Res. Rev 2004, 45, 164–178. [Google Scholar]
- Wu, Q.; Huang, K. Effect of long-term Se deficiency on the antioxidant capacities of rat vascular tissue. Biol. Trace Elem. Res 2004, 98, 73–84. [Google Scholar]

| Parameters | Experimental groups | |||
|---|---|---|---|---|
| Control | CP | Se | Se + CP | |
| LPO (nmol MDA/g tissue) | 21.5 ± 2.8 | 27.3 ± 3.4 * | 18.6 ± 1.5 # | 23.7 ± 2.3 # |
| GSH (μmol/g protein) | 28.9 ± 3.8 | 21.5 ± 3.1 * | 26.7 ± 2.7 # | 24.7 ± 2.7 # |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ognjanović, B.I.; Djordjević, N.Z.; Matić, M.M.; Obradović, J.M.; Mladenović, J.M.; Štajn, A.Š.; Saičić, Z.S. Lipid Peroxidative Damage on Cisplatin Exposure and Alterations in Antioxidant Defense System in Rat Kidneys: A Possible Protective Effect of Selenium. Int. J. Mol. Sci. 2012, 13, 1790-1803. https://doi.org/10.3390/ijms13021790
Ognjanović BI, Djordjević NZ, Matić MM, Obradović JM, Mladenović JM, Štajn AŠ, Saičić ZS. Lipid Peroxidative Damage on Cisplatin Exposure and Alterations in Antioxidant Defense System in Rat Kidneys: A Possible Protective Effect of Selenium. International Journal of Molecular Sciences. 2012; 13(2):1790-1803. https://doi.org/10.3390/ijms13021790
Chicago/Turabian StyleOgnjanović, Branka I., Nataša Z. Djordjević, Miloš M. Matić, Jasmina M. Obradović, Jelena M. Mladenović, Andraš Š. Štajn, and Zorica S. Saičić. 2012. "Lipid Peroxidative Damage on Cisplatin Exposure and Alterations in Antioxidant Defense System in Rat Kidneys: A Possible Protective Effect of Selenium" International Journal of Molecular Sciences 13, no. 2: 1790-1803. https://doi.org/10.3390/ijms13021790




