Voltage Gated Ion Channel Function: Gating, Conduction, and the Role of Water and Protons
Abstract
:1. Introduction
- Gating current consists of protons. In the present form of the model, there are three H+ per domain, leading to a gating current of 12 charges. In Kv1.2, measured gating current is approximately 13 charges. There are multiple ways to get an additional approximately ¼ charge per domain, including partial charge transfer that may be associated with hydrogen bonds, and dipole rotation.
- The path of the protons is along the aqueous pathway that gives rise to the omega current in VSD. This parallels the S4 transmembrane segment. It is similar to the pathway for the protons in the Hv1 proton channel, save that there is no selectivity filter for protons analogous to the D112 [7] in the Hv1 channel.
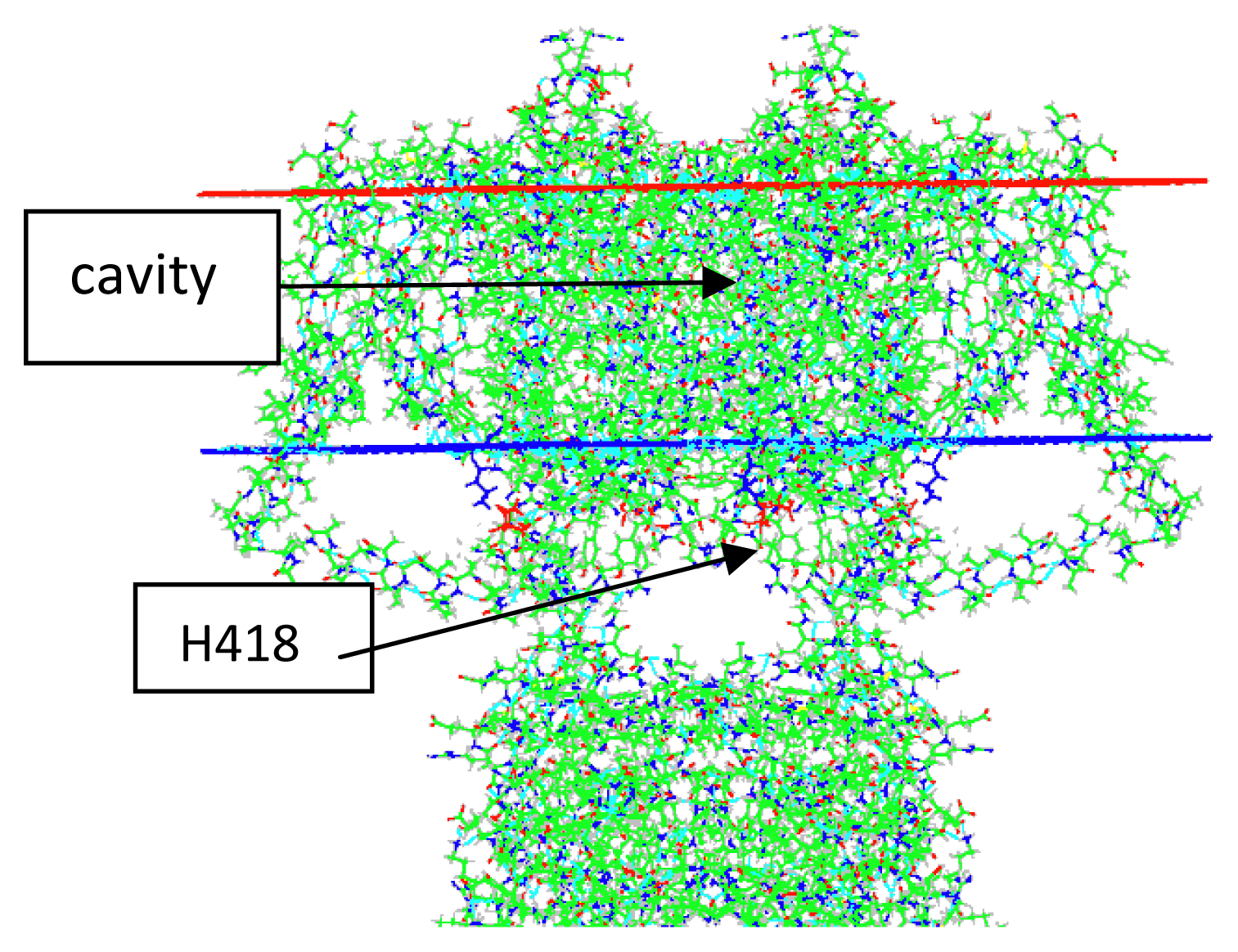
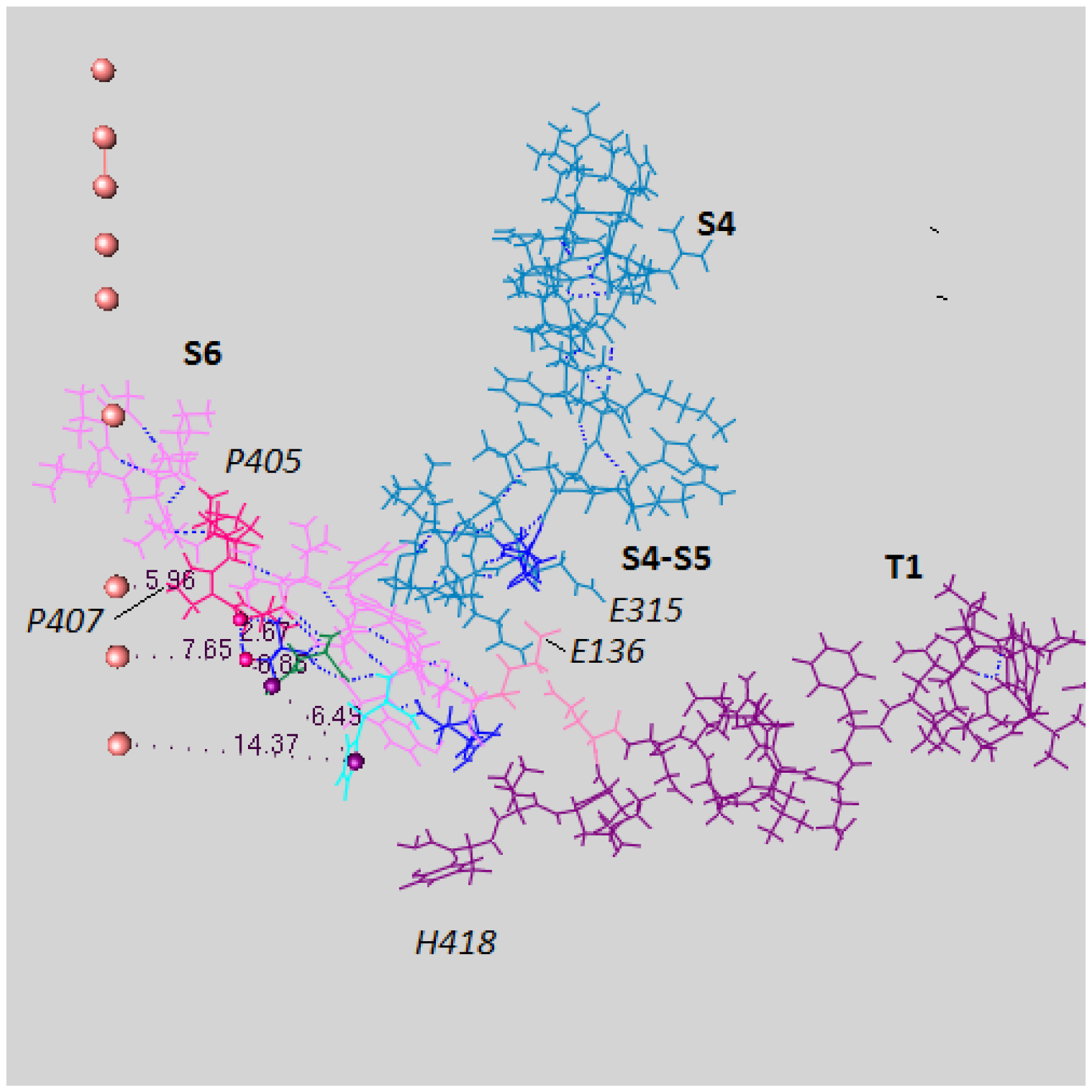
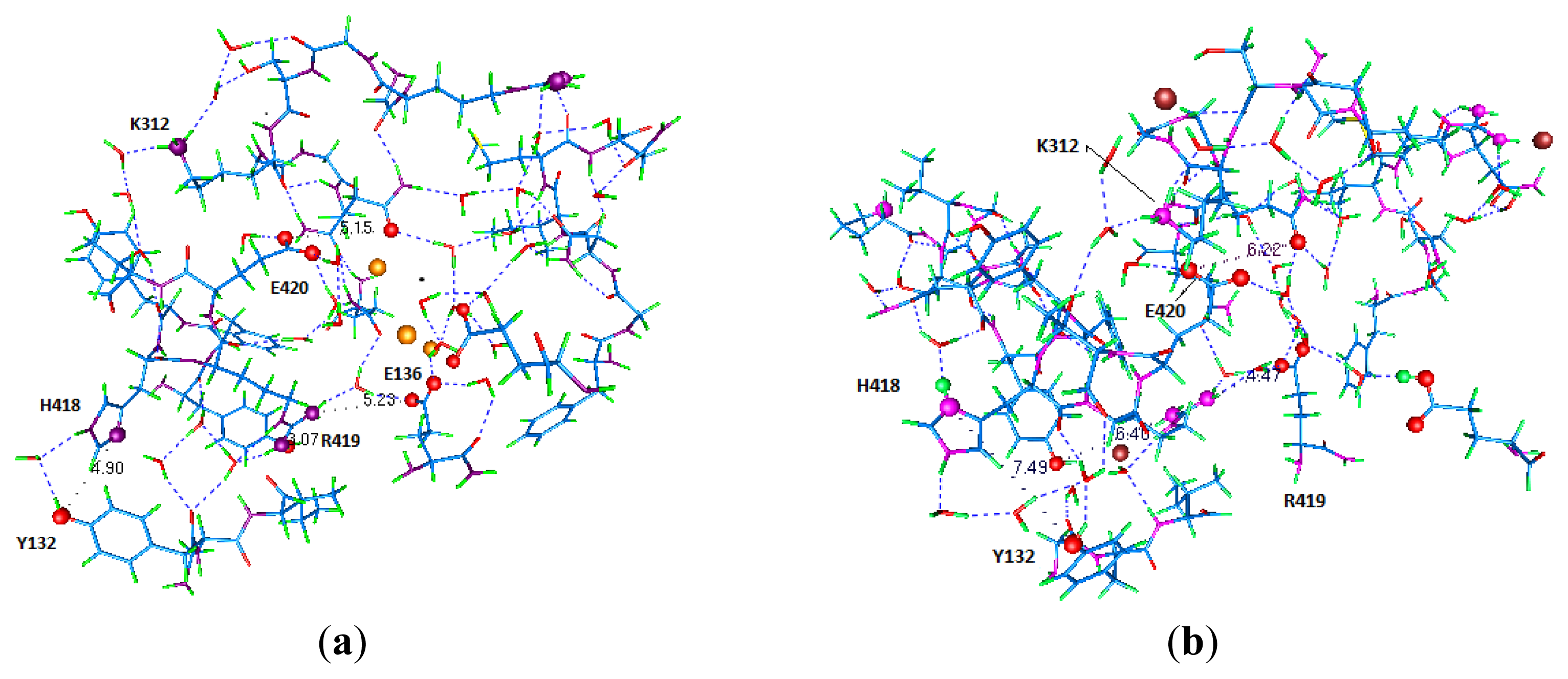
- Key residues that accept the protons at the intracellular end include H418 (S6), E142 (T1), and E136 (T1). The importance of H418 will be discussed below, as it has already been reported to be necessary for channel function [8].
- We have not defined the open state locations for the protons yet. They would have to stop below the outermost arginine, as mutating that to histidine allows proton current through the VSD (which also confirms that protons are capable of moving through the VSD, and that they are near the arginines). It is also possible that lipids help define the extracellular proton reservoir, although the evidence for that is at this point weak.
The Channel
2. Conventional Models
2.1. The Paddle
2.2. The Helical Screw
2.3. A Comment on Both the Paddle and the Helical Screw
2.4. The Alternating Access Model, and Its Descendants
3. The New Model
3.1. Gating Current Derived from Proton Transfer (Proton Current)
3.2. The Oscillating Gate
3.3. There Are Parts of the New Model Taken from Earlier Work by Others
4. Evidence Supporting the New Parts of the Model
4.1. A Key Mutation Cuts Gating Current
4.2. The “Piquito”
4.3. A Switch at the Gate
4.4. D2O Shows a Role for Solvent in the Final Step of the Gating Process
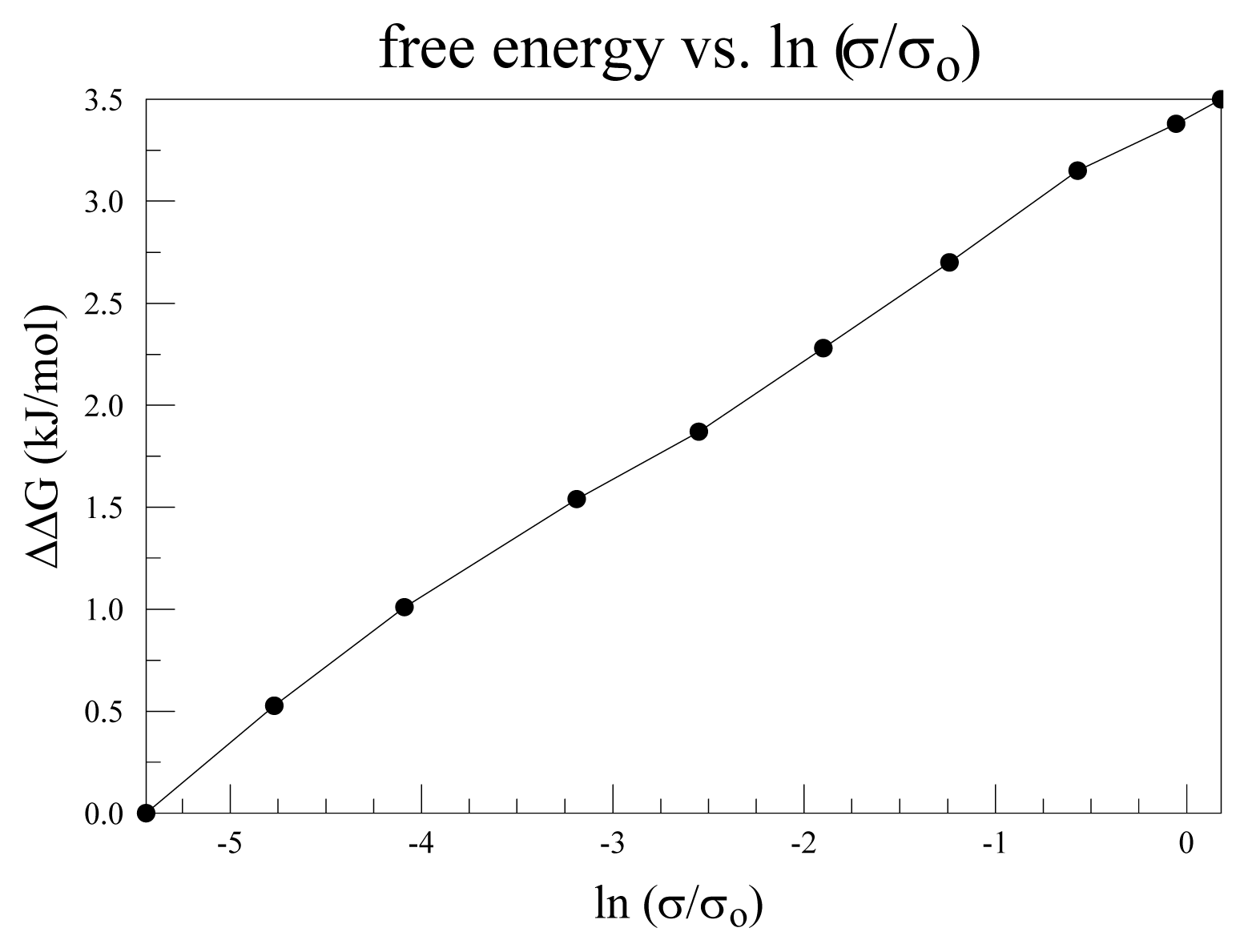
4.5. Pressure: Osmotic vs. Hydrostatic
4.6. Non-Linear Response to Large Gating Voltages
5. The Key Evidence Supporting the Oscillating Gate
5.1. The Gate and the Intracellular Solution
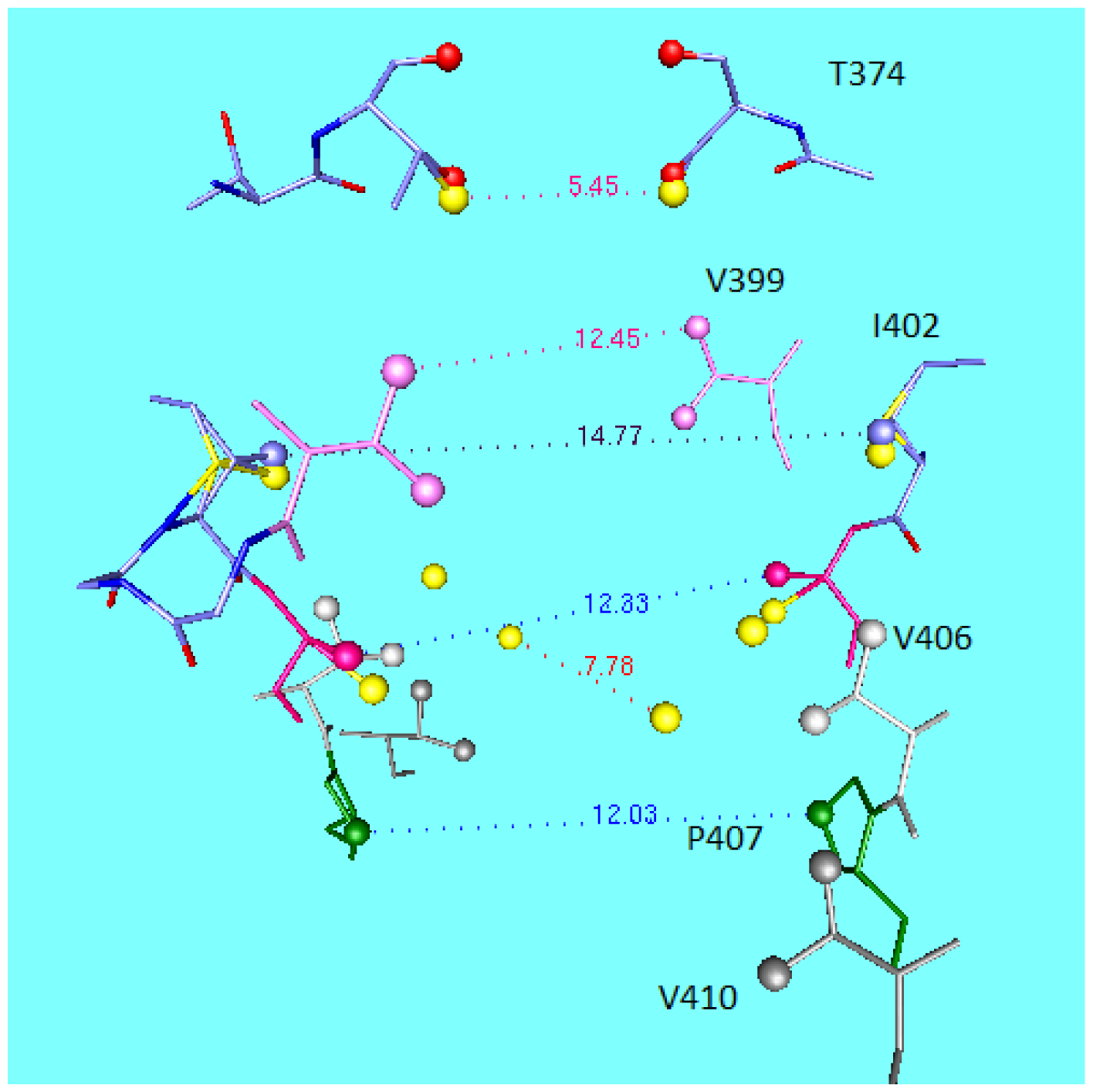
5.2. Minimum at the Gate, Required for the Knock-on Mechanism
5.3. Oscillation of the Gate
5.4. The Proton Reservoirs
5.5. A Role for T1
6. Lipids
7. Slow Inactivation
8. Discussion
8.1. Proposed Experiments
8.2. Osmotic Pressure Combined with Access
8.3. A Possible Means of Detecting an Oscillating Gate
9. Conclusion
Acknowledgements
Appendix A


Appendix B
References
- Hille, B. Ion Channels of Excitable Membranes, 3rd ed; Sinauer Associates: Sunderland, MA, USA, 2001. [Google Scholar]
- Bezanilla, F. How membrane proteins sense voltage. Nat. Rev. Mol. Cell Biol 2008, 9, 323–332. [Google Scholar]
- Roux, B. A brief introduction to voltage-gated K+ channels. Curr. Top. Membr 2008, 60, 369–384. [Google Scholar]
- Boiteux, C.; Kraszewski, S.; Ramseyer, C.; Girardet, C. Ion conductance vs. pore gating and selectivity in KcsA channel: modeling achievements and perspectives. J. Mol. Model 2007, 13, 699–713. [Google Scholar]
- Gagnon, D.G.; Bezanilla, F. A single charged voltage sensor is capable of gating the Shaker K+ channel. J. Gen. Physiol 2009, 133, 467–483. [Google Scholar]
- Sapronova, A.V.; Bystrov, V.S.; Green, M.E. Water, proton transfer, and hydrogen bonding in ion channel gating. Front. Biosci 2003, 8, s1356–s1370. [Google Scholar]
- Musset, B.; Smith, S.M.E.; Rajan, S.; Morgan, D.; Cherny, V.V.; DeCoursey, T.E. Aspartate 112 is the selectivity filter of the human voltage-gated proton channel. Nature 2011, 480, 273–277. [Google Scholar]
- Zhao, L.-L.; Qi, Z.; Zhang, X.-E.; Bi, L.-J.; Jin, G. Regulatory role of the extreme C-terminal end of the S6 inner helix in C-terminal-truncated Kv1.2 channel activation. Cell Biol. Int 2010, 34, 433–439. [Google Scholar]
- Long, S.B.; Campbell, E.B.; MacKinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 2005, 309, 897–903. [Google Scholar]
- Chen, X.; Wang, Q.; Ni, F.; Ma, J. Structure of the full-length Shaker potassium channel Kv1.2 by normal-mode-based X-ray crystallographic refinement. Proc. Natl. Acad. Sci. USA 2010, 107, 11352–11357. [Google Scholar]
- Long, S.B.; Campbell, E.B.; MacKinnon, R. Voltage sensor of Kv1.2: Structural basis of electromechanical coupling. Science 2005, 309, 903–908. [Google Scholar]
- Chanda, B.; Bezanilla, F. A common pathway for charge transport through voltage sensing domains. Neuron 2008, 57, 345–351. [Google Scholar]
- Karlin, A.; Akabas, M.H. Substituted-cysteine accessibility method. Methods Enzymol 1998, 293, 123–145. [Google Scholar]
- Seoh, S.-A.; Sigg, D.; Papazian, D.R.; Bezanilla, F. Voltage-sensing residues in the S2 and S4 segments of the Shaker K+ channel. Neuron 1996, 16, 1159–1167. [Google Scholar]
- Bezanilla, F. The voltage sensor in voltage dependent channels. Physiol. Rev 2000, 80, 555–592. [Google Scholar]
- Ruta, V.; Chen, J.; MacKinnon, R. Calibrated measurement of gating-charge arginine displacement in the KvAP voltage-dependent K+ channel. Cell 2005, 123, 463–475. [Google Scholar]
- Horn, R. Coupled movements in voltage-gated ion channels. J. Gen. Physiol 2002, 120, 449–453. [Google Scholar]
- El-Din, T.M.G.; Heldstab, H.; Lehmann, C.; Greeff, N.G. Double gaps along Shaker S4 demonstrate omega currents at three different closed states. Channels 2010, 4, 93–100. [Google Scholar]
- Broomand, A.; Elinder, F. Large-scale movement within the voltage-sensor paddle of a potassium channel-support for a helical-screw motion. Neuron 2008, 59, 770–777. [Google Scholar]
- Tombola, F.; Pathak, M.M.; Gorostiza, P.; Isacoff, E.Y. The twisted ion-permeation pathway of a resting voltage-sensing domain. Nature 2007, 445, 546–549. [Google Scholar]
- Klassen, T.L.; Spencer, A.N.; Gallin, W.J. A naturally occurring omega current in a Kv3 family potassium channel from a platyhelminth. BMC Neurosci 2008, 9, 52. [Google Scholar]
- Papazian, D.M.; Shao, X.M.; Seoh, S.-A.; Mock, A.F.; Huang, Y.; Wainstock, D.H. Electrostatic interactions of S4 voltage sensor in Shaker K+ channel. Neuron 1995, 14, 1293–1301. [Google Scholar]
- Nguyen, T.P.; Horn, R. Movement and crevices around a sodium channel S3 segment. J. Gen. Physiol 2002, 120, 419–436. [Google Scholar]
- Asamoah, O.K.; Wuskell, J.P.; Loew, L.M.; Bezanilla, F. A fluorometric approach to local electric field measurements in a voltage-gated ion channel. Neuron 2003, 37, 85–97. [Google Scholar]
- Posson, D.J.; Ge, P.; Miller, C.; Bezanilla, F.; Selvin, P.R. Small vertical movement of a K+ channel voltage sensor measured with luminescence energy transfer. Nature 2005, 436, 848–851. [Google Scholar]
- Posson, D.J.; Selvin, P.R. Extent of voltage sensor movement during gating of Shaker K+ channels. Neuron 2008, 59, 98–109. [Google Scholar]
- Chanda, B.; Asamoah, O.K.; Blunck, R.; Roux, B.; Bezanilla, F. Gating charge displacement in voltage-gated ion channels involves limited transmembrane movement. Nature 2005, 436, 852–856. [Google Scholar]
- Ahern, C.A.; Horn, R. Focused electric field across the voltage sensor of potassium channels. Neuron 2005, 48, 25–29. [Google Scholar]
- Gonzalez, C.; Rosenman, E.; Bezanilla, F.; Alvarez, O.; Latorre, R. Periodic perturbations in Shaker K+ channel gating kinetics by deletions in the S3–S4 linker. Proc. Natl. Acad. Sci. USA 2001, 98, 9617–9623. [Google Scholar]
- Starace, D.M.; Bezanilla, F. A proton pore in a potassium channel voltage sensor reveals a focused electric field. Nature 2004, 427, 548–553. [Google Scholar]
- Starace, D.M.; Stefani, E.; Bezanilla, F. Voltage-dependent proton transport by the voltage sensor of the Shaker K+ channel. Neuron 1997, 19, 1319–1327. [Google Scholar]
- DeCoursey, T.E. Four varieties of voltage-gated proton channels. Front. Biosci 1998, 3, D477–D482. [Google Scholar]
- Murphy, R.; Cherny, V.V.; Morgan, D.; DeCoursey, T.E. Voltage-gated proton channels help regulate pHi in rat alveolar epithelium. Am. J. Physiol 2005, 288, L398–L408. [Google Scholar]
- Sasaki, M.; Takagi, M.; Okamura, Y. A voltage-sensor-domain protein is a voltage-gated proton channel. Science 2006, 312, 589–592. [Google Scholar]
- Ramsey, I.S.; Moran, M.M.; Chong, J.A.; Clapham, D.E. A voltage-gated proton-selective channel lacking the pore domain. Nature 2006, 440, 1213–1216. [Google Scholar]
- Ramsey, I.S.; Mokrab, Y.; Carvacho, I.; Sands, Z.A.; Sansom, M.S.P.; Clapham, D.E. An aqueous H+ permeation pathway in the voltage-gated proton channel Hv1. Nat. Struct. Mol. Biol 2010, 17, 869–875. [Google Scholar]
- Tombola, F.; Ulbrich, M.H.; Isacoff, E.Y. Architecture and gating of Hv1 proton channels. J. Physiol 2009, 587, 5325–5329. [Google Scholar]
- Sapronova, A.V.; Bystrov, V.S.; Green, M.E. Ion channel gating and proton transport. J. Mol. Struct (Theochem) 2003, 630, 297–307. [Google Scholar]
- Sapronova, A.V.; Bystrov, V.S.; Green, M.E. Further calculations on proton transfer relevant to the S4 transmembrane segment of Ion Channels, including studies of deuterium motion. Biophys. Soc 2004. abstract 2268. [Google Scholar]
- Gonzalez-Perez, V.; Stack, K.; Boric, K.; Naranjo, D. Reduced voltage sensitivity in a K+-channel voltage sensor by electric field remodeling. Proc. Natl Acad Sci. USA 2010, 107, 5178–5183. [Google Scholar]
- Sigg, D.; Bezanilla, F.; Stefani, E. Fast gating in the Shaker K+ channel and the energy landscape of activation. Proc. Natl Acad. Sci USA 2003, 100, 7611–7615. [Google Scholar]
- Stefani, E.; Ottolia, M.; Noceti, F.; Olcese, R.; Wallner, M.; Latorre, R.; Toro, L. Voltage-controlled gating in a large conductance Ca2+-sensitive K+ channel. Proc. Natl. Acad. Sci. USA 1997, 94, 5427–5431. [Google Scholar]
- Stefani, E.; Sigg, D.; Bezanilla, F. Correlation between the early component of gating current and total gating current in Shaker K channels. Biophys. J 2000, 78, 7A. [Google Scholar]
- Yin, J.; Green, M.E. Intermolecular proton transfer between two methylamine molecules with an external electric field in the gas phase. J. Phys. Chem. A 1998, 102, 7181–7190. [Google Scholar]
- Fatade, A.; Snowhite, J.; Green, M.E. A resonance model gives the response to membrane potential for an ion channel: II. Simplification of the calculation, and prediction of stochastic resonance. J. Theor. Biol 2000, 206, 387–393. [Google Scholar]
- Green, M.E. A resonance model gives the response to membrane potential for an ion channel. J. Theor. Biol 1998, 193, 475–483. [Google Scholar]
- Minor, D.L., Jr; Lin, Y.-F.; Mobley, B.C.; Avelar, A.; Jan, Y.N.; Jan, L.Y. The polar T1 interface is linked to conformational changes that open the voltage-gated potassium channel. Cell 2000, 102, 657–670. [Google Scholar]
- Strang, C.; Cushman, S.J.; DeRubeis, D.; Peterson, D.; Pfaffinger, P.J. A central role for the T1 domain in voltage-gated potassium channel formation and function. J. Biol. Chem 2001, 276, 28493–28502. [Google Scholar]
- Wang, G.; Covarrubias, M. Voltage-dependent gating rearrangements in the intracellular T1-T1 interface of a K+ channel. J.Gen. Physiol 2006, 127, 391–400. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian09, A.1; Gaussian, Inc: Wallingford, CT, USA, 2009. [Google Scholar]
- Schauf, C.L.; Bullock, J.O. Modifications of sodium channel gating in Myxicola giant axons by deuterium oxide, temperature, and internal cations. Biophys. J 1979, 27, 193–208. [Google Scholar]
- Schauf, C.L.; Bullock, J.O. Solvent substitution as a probe of channel gating in Myxicola: Differential effects of D2O on some components of membrane conductance. Biophys. J 1980, 30, 295–306. [Google Scholar]
- Schauf, C.L.; Bullock, J.O. Solvent substitution as a probe of channel gating in Myxicola. Biophys. J 1982, 37, 441–452. [Google Scholar]
- Alicata, D.A.; Rayner, M.A.; Starkus, J.A. Sodium Channel activation mechanisms: Insights from deuterium oxide substitution. Biophys. J 1990, 57, 745–758. [Google Scholar]
- Conti, F. The relationship between electrophysiological data and thermodynamics of ion channel conformations. Neurol. Neurobiol. 1986, 20, 25–41. [Google Scholar]
- Conti, F.; Fioravanti, R.; Segal, J.R.; Stuhmer, W. Pressure dependence of the sodium currents of squid giant axon. J. Membr. Biol 1982, 69, 23–34. [Google Scholar]
- Conti, F.; Inoue, I.; Kukita, F.; Stuhmer, W. Pressure dependence of sodium gating currents in the squid giant axon. Eur. Biophys. J 1984, 11, 137–147. [Google Scholar]
- Zimmerberg, J.; Bezanilla, F.; Parsegian, V.A. Solute inaccessible aqueous volume changes during opening of the potassium channel of the squid giant axon. Biophys. J 1990, 57, 1049–1064. [Google Scholar]
- Starkus, J.G.; Schlief, T.; Rayner, M.D.; Heinemann, S.H. Unilateral exposure of Shaker B potassium channels to hyperosmolar solutions. Biophys. J 1995, 69, 860–872. [Google Scholar]
- Schmidt, D.; MacKinnon, R. Voltage-dependent K+ channel gating and voltage sensor toxin sensitivity depend on the mechanical state of the lipid membrane. Proc. Natl. Acad. Sci. USA 2008, 105, 19276–19281. [Google Scholar]
- Butterwick, J.A.; MacKinnon, R. Solution Structure and Phospholipid Interactions of the Isolated Voltage-Sensor Domain from KvAP. J. Mol. Biol 2010, 403, 591–606. [Google Scholar]
- Fohlmeister, J.F.; Adelman, W.J., Jr. Gating current harmonics: I. Sodium channel activation gating in dynamic steady states. Biophys. J. 1985, 48, 375–390. [Google Scholar]
- Fohlmeister, J.F.; Adelman, W.J., Jr. Gating Current Harmonics II: Model simulations of axonal gating currents. Biophys. J. 1985, 48, 391–400. [Google Scholar]
- LeMasurier, M.; Heginbotham, L.; Miller, C. KcsA: It’s a potassium channel. J. Gen. Physiol 2001, 118, 303–313. [Google Scholar]
- Chung, S.-H.; Allen, T.W.; Kuyucak, S. Conducting-state properties of the KcsA potassium channel from molecular and Browninan dynamics simulations. Biophys. J 2002, 82, 628–645. [Google Scholar]
- Abirami, S.; Wong, C.H.-S.; Tsang, C.-W.; Ma, N.-L.; Goh, N.K. A theoretical study of potassium cation binding to prolylglycine (PG) and glycylproline (GP) dipeptide. J. Mol. Struct (Theochem) 2005, 729, 193–202. [Google Scholar]
- Holmgren, M.; Shin, K.S.; Yellen, G. The activation gate of a voltage-gated K+ channel can be trapped in the open state by an intersubunit bridge. Neuron 1998, 21, 617–621. [Google Scholar]
- Liu, Y.; Holmgren, M.; Jurman, M.E.; Yellen, G. Gated access to the pore of a voltage-dependent K+ channel. Neuron 1997, 19, 175–184. [Google Scholar]
- Lu, Z.; Klem, A.M.; Ramu, Y. Coupling between voltage sensors and activation gate in voltage-gated K+ channels. J. Gen. Physiol 2002, 120, 663–676. [Google Scholar]
- Haddad, G.A.; Blunck, R. Mode shift of the voltage sensors in Shaker K+ channels is caused by energetic coupling to the pore domain. J. Gen. Physiol 2011, 137, 455–472. [Google Scholar]
- Schmidt, D.; Jiang, Q.-X.; MacKinnon, R. Phospholipids and the origin of cationic gating charges in voltage sensors. Nature 2006, 444, 775–779. [Google Scholar]
- Deol, S.S.; Domene, C.; Bond, P.J.; Sansom, M.S.P. Anionic phospholipid interactions with the potassium channel KcsA: simulation studies. Biophys. J 2006, 90, 822–830. [Google Scholar]
- Mokrab, Y.; Sansom, M.S.P. Interaction of diverse voltage sensor homologs with lipid bilayers revealed by self-assembly simulations. Biophys. J 2011, 100, 875–884. [Google Scholar]
- Goforth, R.L.; Chi, A.K.; Greathouse, D.V.; Providence, L.L.; Koeppe, R.E.I.; Andersen, O.S. Hydrophobic coupling of lipid bilayer energetics to channel function. J. Gen. Physiol 2003, 121, 477–493. [Google Scholar]
- Milescu, M.; Bosmans, F.; Lee, S.; Alabi, A.R.A.; Kim, J.I.; Swartz, K.J. Interactions between lipids and voltage sensor paddles detected with tarantula toxins. Nat. Struct. Mol. Biol 2009, 16, 1080–1085. [Google Scholar]
- Borjesson, S.I.; Elinder, F. An electrostatic potassium channel opener targeting the final voltage sensor transition. J. Gen. Physiol 2011, 137, 563–577. [Google Scholar]
- Boerjesson, S.I.; Hammarstroem, S.; Elinder, F. Lipoelectric modification of ion channel voltage gating by polyunsaturated fatty acids. Biophys. J 2008, 95, 2242–2253. [Google Scholar]
- Morris, C.E.; Juranka, P.F. Lipid stress at play: Mechanosensitivity of voltage-gated channels. Curr. Topics Membr 2007, 59, 297–338. [Google Scholar]
- Green, M.E. A possible role for phosphate in complexing the arginines of S4 in voltage gated channels. J. Theor. Biol 2005, 233, 337–341. [Google Scholar]
- Freites, J.A.; Tobias, D.J.; von Heijne, G.; White, S.H. Interface connections of a transmembrane voltage sensor. Proc. Natl. Acad. Sci. USA 2005, 102, 15059–15064. [Google Scholar]
- Panyi, G.; Deutsch, C. Cross talk between activation and slow inactivation gates of Shaker potassium channels. J. Gen. Physiol 2006, 128, 547–559. [Google Scholar]
- Panyi, G.; Deutsch, C. Probing the cavity of the slow inactivated conformation of Shaker potassium channels. J. Gen. Physiol 2007, 129, 403–418. [Google Scholar]
- Ray, E.C.; Deutsch, C. A trapped intracellular cation modulates K+ channel recovery from slow inactivation. J. Gen. Physiol 2006, 128, 203–217. [Google Scholar]
- Ader, C.; Schneider, R.; Hornig, S.; Velisetty, P.; Vardanyan, V.; Giller, K.; Ohmert, I.; Becker, S.; Pongs, O.; Baldus, M. Coupling of activation and inactivation gate in a K+-channel: potassium and ligand sensitivity. EMBO J 2009, 28, 2825–2834. [Google Scholar]
- Cuello, L.G.; Jogini, V.; Cortes, D.M.; Pan, A.C.; Gagnon, D.G.; Dalmas, O.; Cordero-Morales, J.F.; Chakrapani, S.; Roux, B.; Perozo, E. Structural basis for the coupling between activation and inactivation gates in K1 channels. Nature 2010, 466, 272–276. [Google Scholar]
- Imai, S.; Osawa, M.; Takeuchi, K.; Shimada, I. Structural basis underlying the dual gate properties of KcsA. Proc Natl. Acad. Sci. USA 2010, 107, 6216–6221. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kariev, A.M.; Green, M.E. Voltage Gated Ion Channel Function: Gating, Conduction, and the Role of Water and Protons. Int. J. Mol. Sci. 2012, 13, 1680-1709. https://doi.org/10.3390/ijms13021680
Kariev AM, Green ME. Voltage Gated Ion Channel Function: Gating, Conduction, and the Role of Water and Protons. International Journal of Molecular Sciences. 2012; 13(2):1680-1709. https://doi.org/10.3390/ijms13021680
Chicago/Turabian StyleKariev, Alisher M., and Michael E. Green. 2012. "Voltage Gated Ion Channel Function: Gating, Conduction, and the Role of Water and Protons" International Journal of Molecular Sciences 13, no. 2: 1680-1709. https://doi.org/10.3390/ijms13021680



