Functional Expression of an Orchid Fragrance Gene in Lactococcus lactis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Plasmid Construction and Stability of Recombinant Strains
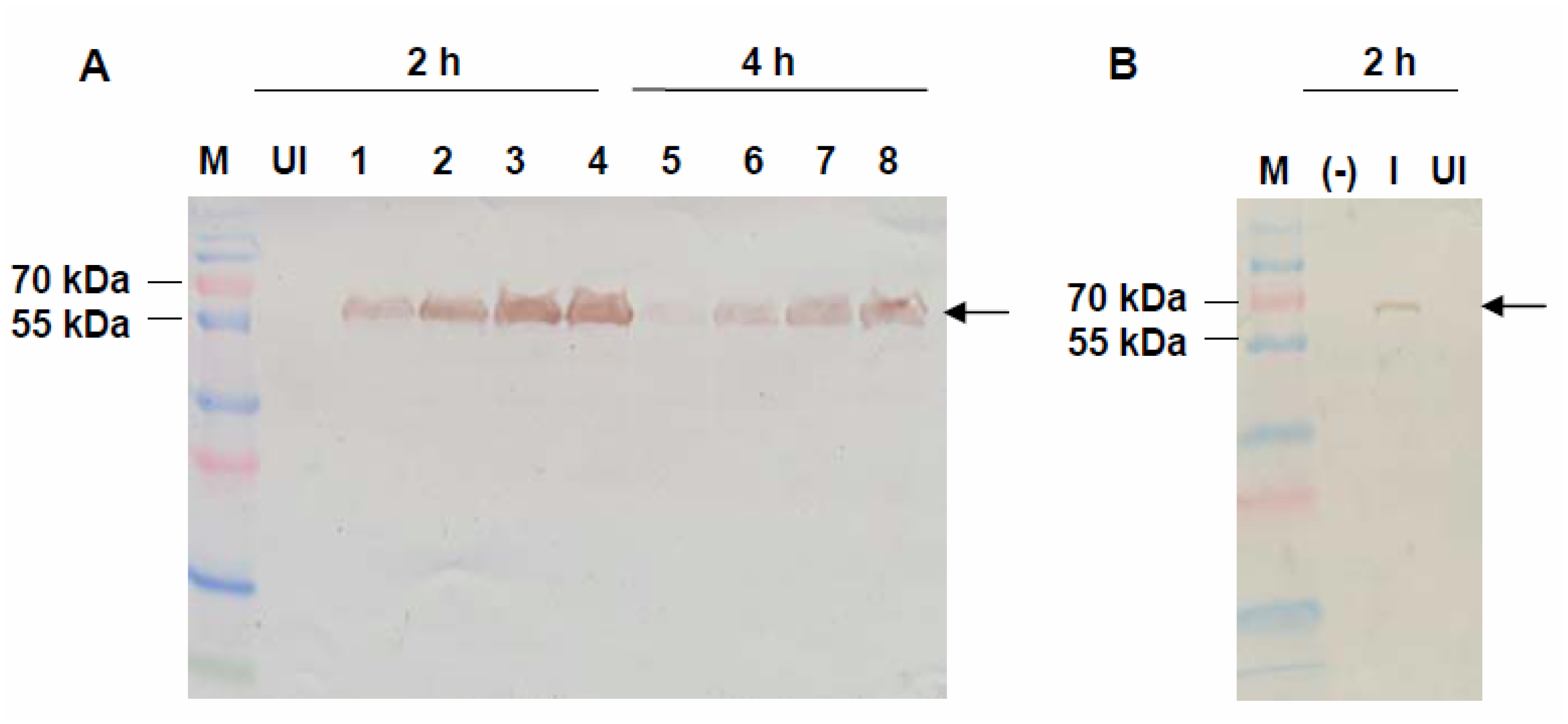
2.2. Expression of VMPSTS in L. lactis
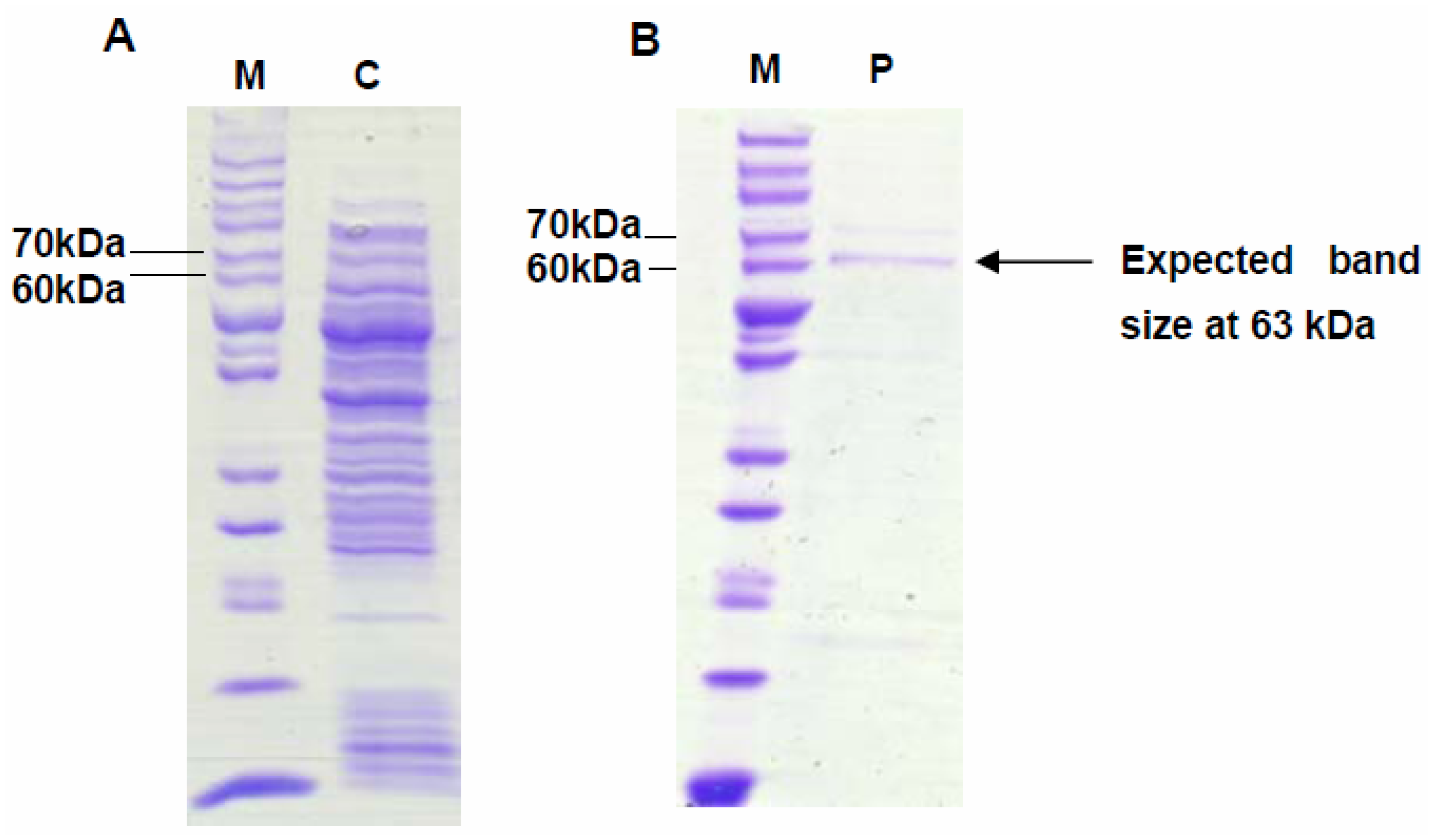
2.3. Enzymatic Assay of Crude VMPSTS
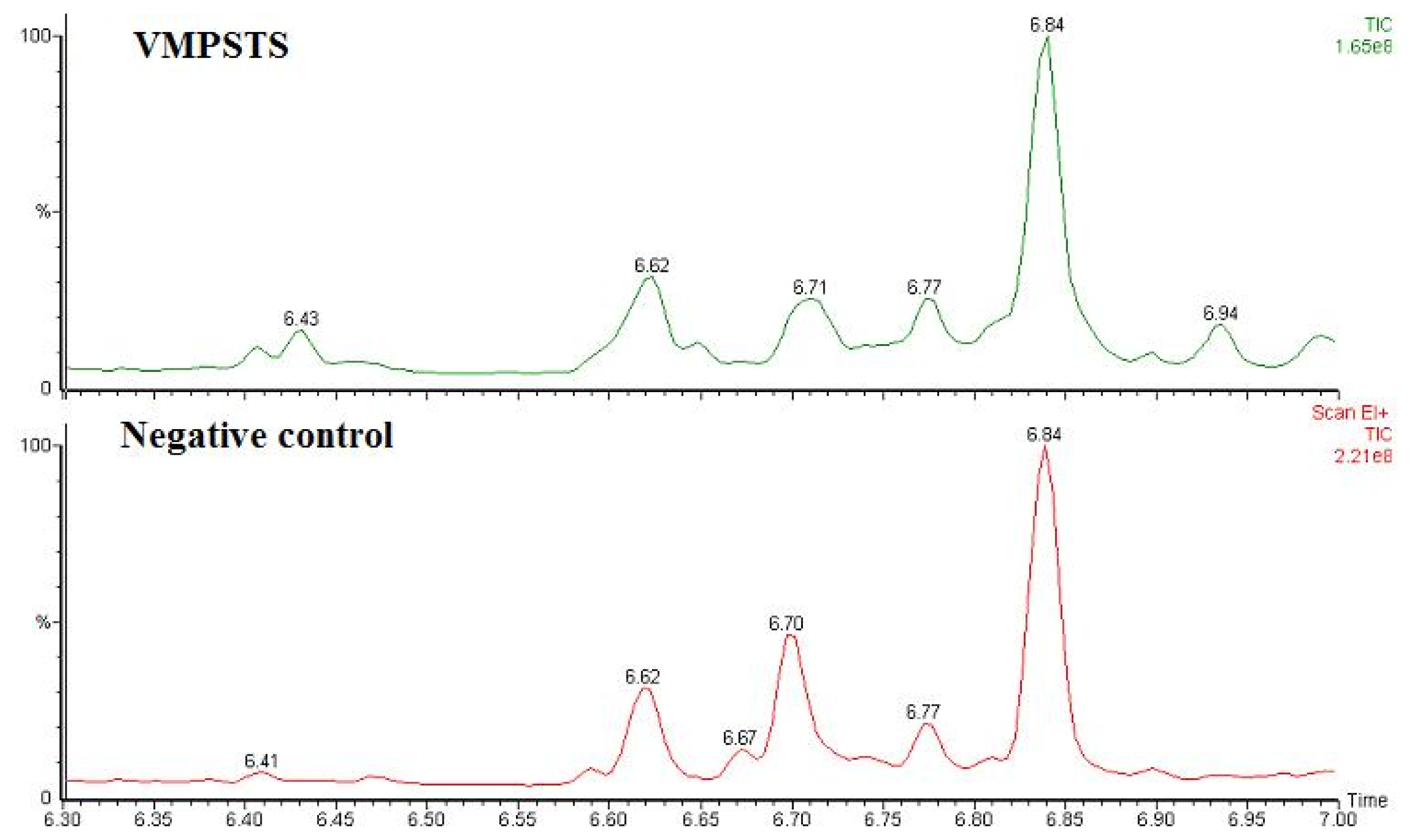
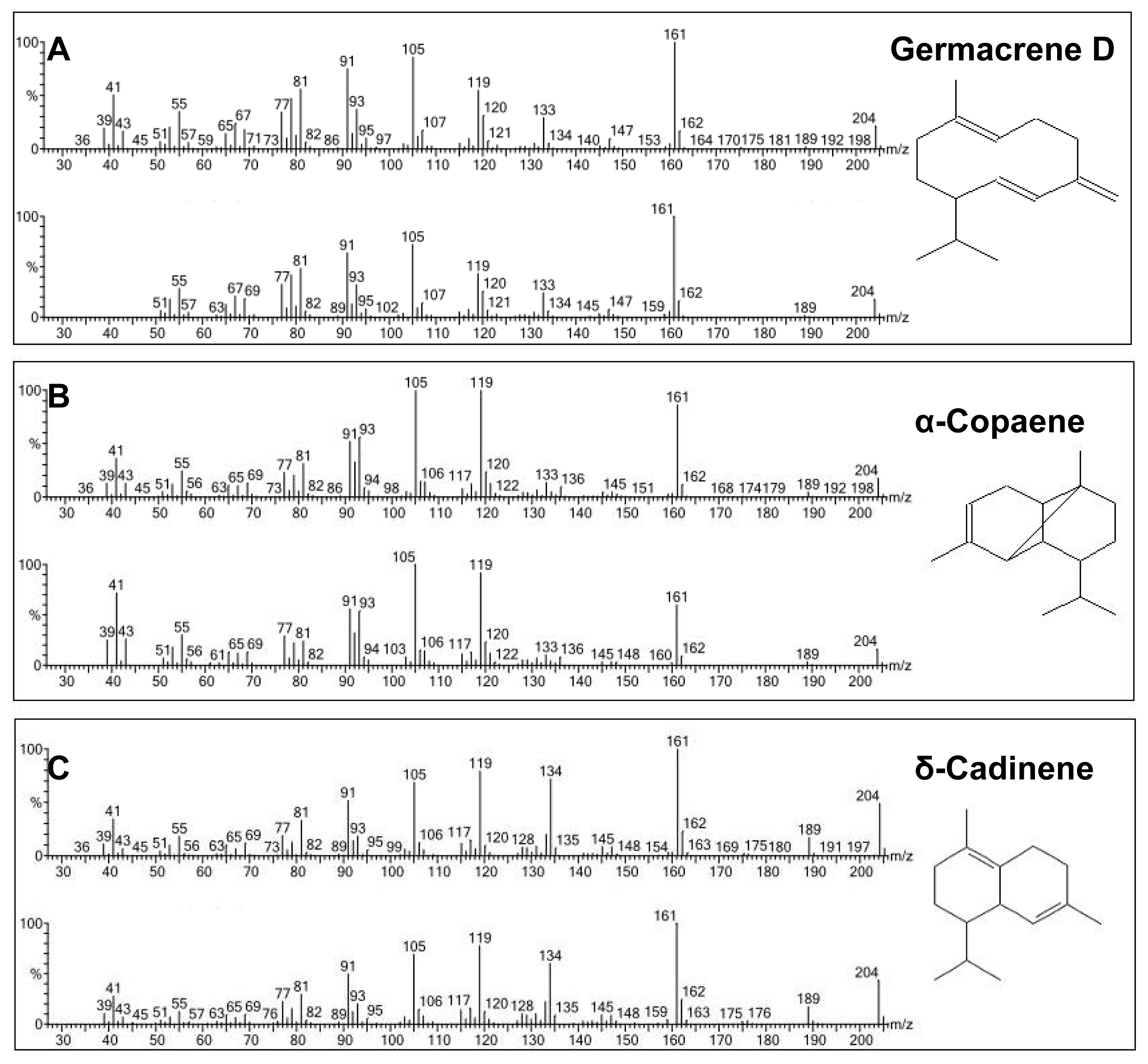
2.4. Enzymatic Assay of His-Tag Purified VMPSTS and End Product Analysis
3. Experimental Section
3.1. Bacteria Strains and Growth Conditions
3.2. VMPSTS Gene Amplification and Plasmid Construction
3.3. Expression of VMPSTS in L. lactis
3.4. Enzymatic Assays
3.5. GC-MS Analysis
4. Conclusions
Acknowledgments
References
- Balibar, C.J.; Shen, X.; Tao, J. The mevalonate pathway of Staphylococcus aureus. J. Bacteriol 2009, 191, 851–861. [Google Scholar]
- Misawa, N. Pathway engineering for functional isoprenoids. Curr. Opin. Biotechnol 2011, 22, 1–7. [Google Scholar]
- Dellomonaco, C.; Fava, F.; Gonzalez, R. The path to next generation biofuels: Successes and challenges in the era of synthetic biology. Microb. Cell Fact 2010, 9. [Google Scholar] [CrossRef]
- Rodríguez-Concepción, M.; Boranat, A. Elucidation of the methylerythhritol phosphate pathway for isoprenoid biosynthesis in bacteria and plastids. A metabolic milestone achieved through genomics. Plant Physiol 2002, 130, 1079–1089. [Google Scholar]
- Barkovich, R.; Liao, J.C. Metabolic engineering of isoprenoids. Metab. Eng 2001, 3, 27–39. [Google Scholar]
- Kirby, J.; Keasling, J.D. Metabolic engineering of microorganisms for isoprenoid production. Nat. Prod. Rep 2008, 25, 656–661. [Google Scholar]
- Horwitz, S.B. How to make taxol from scratch. Nature 1994, 367, 593–594. [Google Scholar]
- Huang, Q.; Roessner, C.A.; Croteau, R.; Scott, A.I. Engineering Escherichia coli for the synthesis of taxadiene, a key intermediate in the biosynthesis of Taxol. Bioorg. Med. Chem 2001, 9, 2237–2242. [Google Scholar]
- Martin, V.J.; Pitera, D.J.; Withers, S.T.; Newman, J.D.; Keasling, J.D. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nat. Biotechnol 2003, 21, 796–802. [Google Scholar]
- Asadollahi, M.A.; Maury, J.; Patil, K.R.; Schalk, M.; Clark, A.; Nielsen, J. Enhancing sesquiterpene production in Saccharomyces cerivisiae through in silico driven metabolic engineering. Metab. Eng 2009, 11, 328–334. [Google Scholar]
- Engels, B.; Dahm, P.; Jennewein, S. Metabolic engineering of taxadiene biosynthesis in yeasts as a first step towards Taxol (Paclitaxel) production. Metab. Eng 2008, 10, 201–206. [Google Scholar]
- Miura, Y.; Kondo, K.; Saito, T.; Nakamura, K.; Misawa, N. Production of lycopene by the food yeast Candida utilis that does not naturally synthesize carotenoid. Biotechnol. Bioeng 1998, 58, 306–308. [Google Scholar]
- Anuradha, K.; Foo, H.L.; Mariana, N.S.; Loh, T.C.; Yusoff, K.; Hassan, M.D.; Sasan, H.; Raha, A.R. Live recombinant Lactococcus lactis vaccine expressing aerolysin genes D1 and D4 for protection against Aeromonas hydrophila in tilapia (Oreochromis niloticus). J. Appl. Microbiol 2010, 109, 1632–1642. [Google Scholar]
- de Vos, W.M.; Hugenholtz, J. Engineering metabolic highways in Lactococcus and other lactic acid bacteria. Trends Biotechnol 2004, 22, 72–79. [Google Scholar]
- Mierau, I.; Kleerebezem, M. 10 years of nisin-controlled gene expression system (NICE) in Lactococcus lactis. Appl. Microbiol. Biotechnol 2005, 68, 705–717. [Google Scholar]
- Kuipers, O.P.; Beerthuyzen, M.M.; de Ruyter, P.G.; Luesink, E.J.; de Vos, W.M. Autoregulation of nisin biosynthesis in Lactococcus lactis by signal transduction. J. Biol. Chem 1995, 270, 27299–27304. [Google Scholar]
- Berlec, A.; Jevnikar, Z.; Majhenič, A.C.; Rogelj, I.; Štrukelj, B. Expression of the sweet-tasting plant protein brazzein in Escherichia coli and Lactococcus lactis: A path toward sweet lactic acid bacteria. Appl. Microbiol. Biotechnol 2006, 73, 158–165. [Google Scholar]
- Frelet-Barrand, A.; Boutigny, S.; Movet, L.; Deniaud, A.; Seigneurin-Berny, D.; Salvi, D.; Bernaudat, F.; Richaud, P.; Pebay-Peyroula, E.; Jovard, J.; Rolland, N. Lactococcus lactis, an alternative system for functional expression of peripheral and intrinsic Arabidopsis membrane proteins. PLoS One 2010, 5. [Google Scholar] [CrossRef]
- Martinez-Cuesta, M.C.; Gasson, M.J.; Narbad, A. Heterelogous expression of the plant coumarate: CoA ligase in Lactococcus lactis. Lett. Appl. Microbiol 2005, 40, 44–49. [Google Scholar]
- Hernandez, I.; Molenaar, D.; Beekwilder, J.; Bouwmeester, H.; van Hylckama Vlieg, J.E.T. Expression of plant flavor genes in Lactococcus lactis. Appl. Environ. Microbiol 2007, 73, 1544–1552. [Google Scholar]
- Nair, H.; Arditti, J. Sustaining Orchids for the Future. Proceedings of the 17th World Orchid Conference, Kota Kinabalu, Malaysia, 24 April–2 May 2002; Natural History Publications (Borneo) Sdn Bhd: Kota Kinabalu, Malaysia, 2002. [Google Scholar]
- Mohd-Hairul, A.R.; Namasivayam, P.; Ee, C.L.G.; Abdullah, J.O. Terpenoid, benzonoid and phenylpropanoid compounds in the floral scent of Vanda Mimi Palmer. J. Plant Biol 2010, 53, 358–366. [Google Scholar]
- Chan, W.; Abdullah, J.O.; Namasivayam, P. Isolation, cloning and characterization of fragrance-related transcripts from Vanda Mimi Palmer. Sci. Hortic 2011, 127, 388–397. [Google Scholar]
- Martin, V.J.J.; Yasuo, Y.; Keasling, J.D. The in vivo synthesis of plant sesquiterpenes by Escherichia coli. Biotechnol. Bioeng 2001, 75, 497–503. [Google Scholar]
- Song, L. Detection of farnesyl diphosphate accumulation in yeast ERG9 mutants. Anal. Biochem 2003, 317, 180–185. [Google Scholar]
- George-Nascimento, C.; Pont-Lezica, R.; Cori, O. Non-enzymic formation of nerolidol from farnesyl pyrophosphate in the presence of bivalent cations. Biochem. Biophys. Res. Commun 1971, 45, 119–124. [Google Scholar]
- Adio, A.M. Germacrenes A-E and related compounds: Thermal, photochemical and acid induced transannular cyclizations. Tetrahedron 2009, 65, 1533–1552. [Google Scholar]
- Martin, D.M.; Bohlmann, J. Identification of Vitis vinefera (−)-α-terpineol synthase by in silico screening of full-length cDNA ESTs and functional characterization of recombinant terpene synthase. Phytochemistry 2004, 65, 1223–1229. [Google Scholar]
- Picaud, S.; Olsson, M.E.; Brodelius, M.; Brodelius, P.E. Cloning, expression, purification and characterization of recombinant (+)-germacrene D synthase from Zingiber officinale. Arch. Biochem. Biophys 2006, 452, 17–28. [Google Scholar]
- Setzer, W.N. Germacrene D cyclization: An ab initio investigation. Int. J. Mol. Sci 2008, 9, 89–97. [Google Scholar]
- Mohd-Hairul, A.R. Universiti Putra Malaysia: Selangor, Malaysia, Unpublished work; 2011.
- Lubertozzi, D.; Keasling, J.D. Expression of a synthetic Artemesia annua amorphadiene synthase in Aspergillus nidulans yield altered product distribution. J. Ind. Microbiol. Biotechnol 2008, 35, 1191–1198. [Google Scholar]
- Lücker, J.; Bowen, P.; Bohlmann, J. Vitis vinefera terpenoid cyclases: Fuctional identification of two sesquiterpene synthase cDNA encoding (+)-valencene synthase and (−)-germacrene D synthase and expression of mono- and sesquiterpene synthases in grapevine flowers and berries. Phytochemistry 2004, 65, 2649–2659. [Google Scholar]
- Kiran, S.R.; Devi, P.S. Evaluation of mosquitocidal activity of essential oil and sesquiterpenes from leaves of Chloroxylon swietenia DC. Parasitol. Res 2007, 101, 413–418. [Google Scholar]
- Bruce, T.J.A.; Birkett, M.A.; Blande, J.; Hooper, A.M.; Martin, J.L.; Khambay, B.; Prosser, I.; Smart, L.E.; Wadhams, L.J. Response of economically important aphids to components of Hemizygia petiolata essential oil. Pest Manag. Sci 2005, 61, 1115–1121. [Google Scholar]
- Mozuraitis, R.; Stranden, M.; Ramirez, M.I.; Borg-Karlson, A.K.; Mustaparta, H. (−)-Germacrene D increases attraction oviposition by the tobacco budworm moth Heliothis virescens. Chem. Senses 2002, 27, 505–509. [Google Scholar]
- Nishida, R.; Shelly, T.E.; Whittier, T.S.; Kaneshiro, K.Y. α-Copaene, a potential rendezvous cue for the Mediterranean fruit fly, Ceratitis capitata. J. Chem. Ecol 1999, 26, 87–100. [Google Scholar]
- Zheljazkov, V.D.; Cantrell, C.L.; Tekwani, B.; Khan, S.I. Content, composition and bioactivity of the essential oils of three basil genotypes as a function of harvesting. J. Agric. Food Chem 2008, 56, 380–385. [Google Scholar]
- Gopfert, J.C.; Macnevin, G.; Ro, D.K.; Spring, O. Identification, functional characterization and developmental regulation of sesquiterpene synthases from sunflower capitate glandular trichomes. BMC Plant Biol 2009, 9. [Google Scholar] [CrossRef]
- Fujisawa, M.; Harada, H.; Kenmoku, H.; Mizutani, S.; Misawa, N. Cloning and characterization of a novel gene that encodes (S)-ß-bisabolene synthase from ginger, Zingiber officinale. Planta 2009, 232, 121–130. [Google Scholar]
- Crocoll, C.; Asbach, J.; Novak, J.; Gershenzon, J.; Degenhardt, J. Terpene synthases of oregano (Origanum vulgare L.) and their roles in the pathway and regulation of terpene biosynthesis. Plant Mol. Biol 2010, 73, 587–603. [Google Scholar]
- Kuipers, O.P.; de Ruyter, P.G.; Kleerebezem, M.; de Vos, W.M. Quorum sensing-controlled gene expression in lactic acid bacteria. J. Biotechnol 1998, 64, 15–21. [Google Scholar]
- Terzaghi, B.E.; Sandine, W.E. Improved medium for lactic streptococci and their bacteriophages. Appl. Microbiol 1975, 29, 807–813. [Google Scholar]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual, 3rd ed; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Holo, H.; Nes, I.F. High-frequency transformation by electroporation of Lactococcus lactis subsp. cremoris grown with glycine in osmotically stabilized media. Appl. Environ. Microbiol 1989, 55, 3119–3123. [Google Scholar]
- Chen, X.; Chen, Y.; Heinstein, P.; Davisson, V.J. Cloning, expression and characterization of (+)-δ-cadinene synthase: A catalyst for cotton phytoalexin biosynthesis. Arch. Biochem. Biophys 1995, 324, 255–266. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
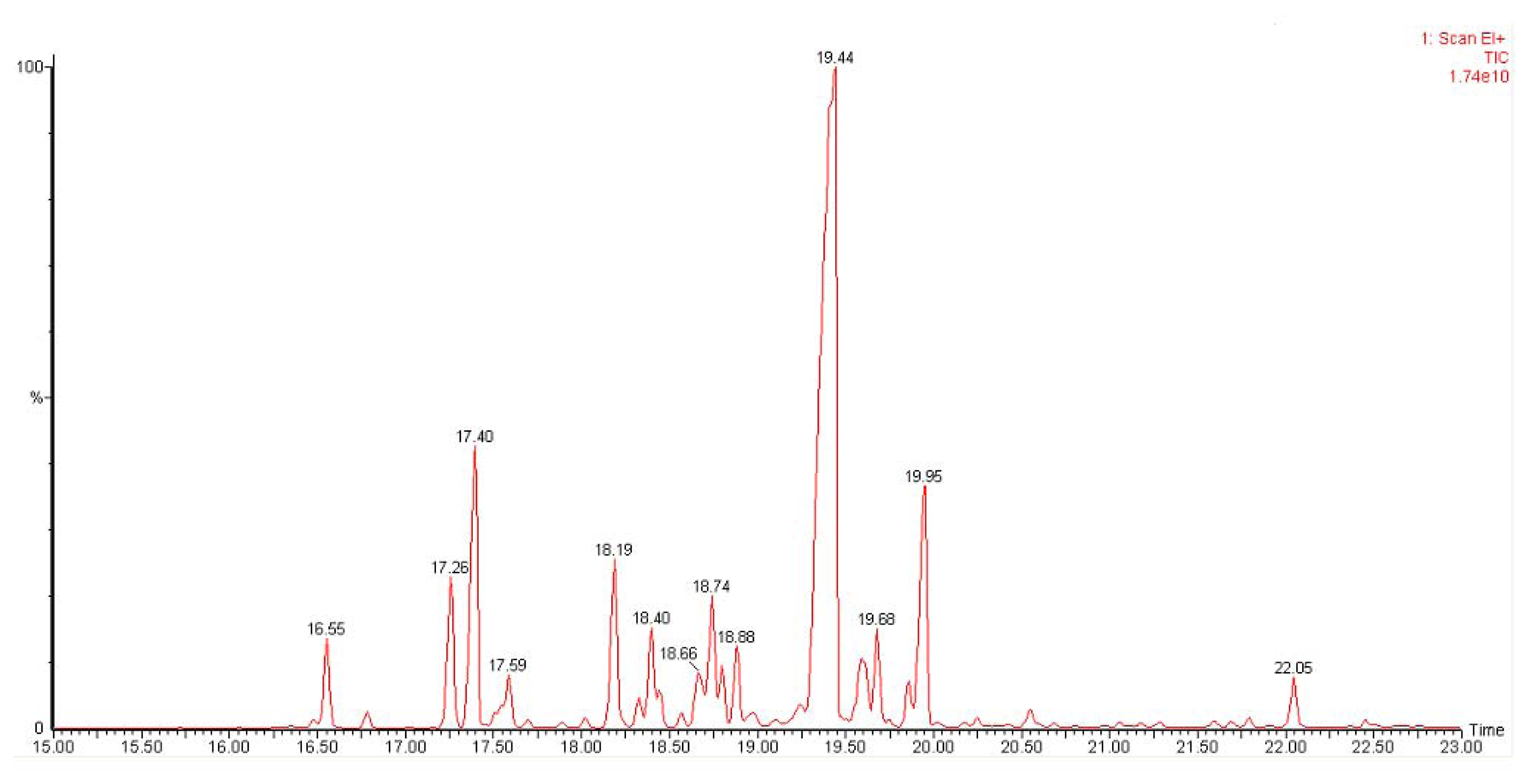
| Sesquiterpenes | Retention Time | Match score | Percentage |
|---|---|---|---|
| δ-Elemene | 16.55 | 950 | 2.4 |
| Ylangene | 17.26 | 955 | 4.5 |
| α-Copaene | 17.40 | 943 | 9.5 |
| ß-Elemene | 17.56 | 922 | 2.2 |
| Germacrene D | 18.19 | 903 | 5.3 |
| Germacrene D | 18.40 | 913 | 3.7 |
| Germacrene D | 18.66 | 890 | 2.1 |
| α-Gurjunene | 18.74 | 905 | 4.2 |
| ɛ-Muurolene | 18.80 | 824 | 1.2 |
| α-Caryophyllene | 18.89 | 946 | 2.1 |
| Germacrene D | 19.44 | 966 | 48.9 |
| γ-Muurolene | 19.68 | 881 | 2.2 |
| γ-Cadinene | 19.86 | 934 | 1.3 |
| δ-Cadinene | 19.95 | 945 | 8.8 |
| .tau.-Muurolol | 22.05 | 927 | 1.6 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Song, A.A.L.; Abdullah, J.O.; Abdullah, M.P.; Shafee, N.; Rahim, R.A. Functional Expression of an Orchid Fragrance Gene in Lactococcus lactis. Int. J. Mol. Sci. 2012, 13, 1582-1597. https://doi.org/10.3390/ijms13021582
Song AAL, Abdullah JO, Abdullah MP, Shafee N, Rahim RA. Functional Expression of an Orchid Fragrance Gene in Lactococcus lactis. International Journal of Molecular Sciences. 2012; 13(2):1582-1597. https://doi.org/10.3390/ijms13021582
Chicago/Turabian StyleSong, Adelene Ai Lian, Janna O. Abdullah, Mohd Puad Abdullah, Norazizah Shafee, and Raha A. Rahim. 2012. "Functional Expression of an Orchid Fragrance Gene in Lactococcus lactis" International Journal of Molecular Sciences 13, no. 2: 1582-1597. https://doi.org/10.3390/ijms13021582




