Identification of kakusei, a Nuclear Non-Coding RNA, as an Immediate Early Gene from the Honeybee, and Its Application for Neuroethological Study
Abstract
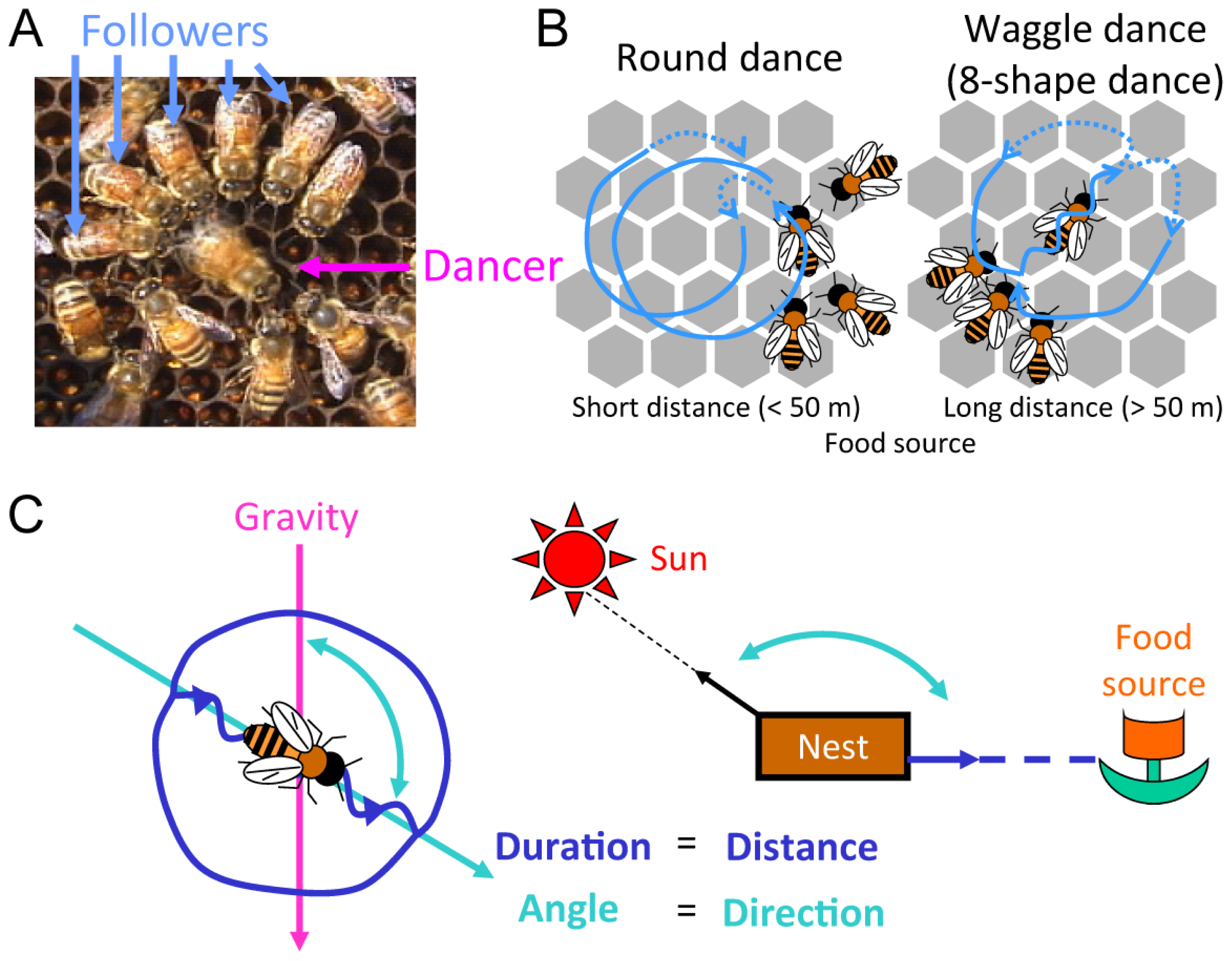
:1. Introduction
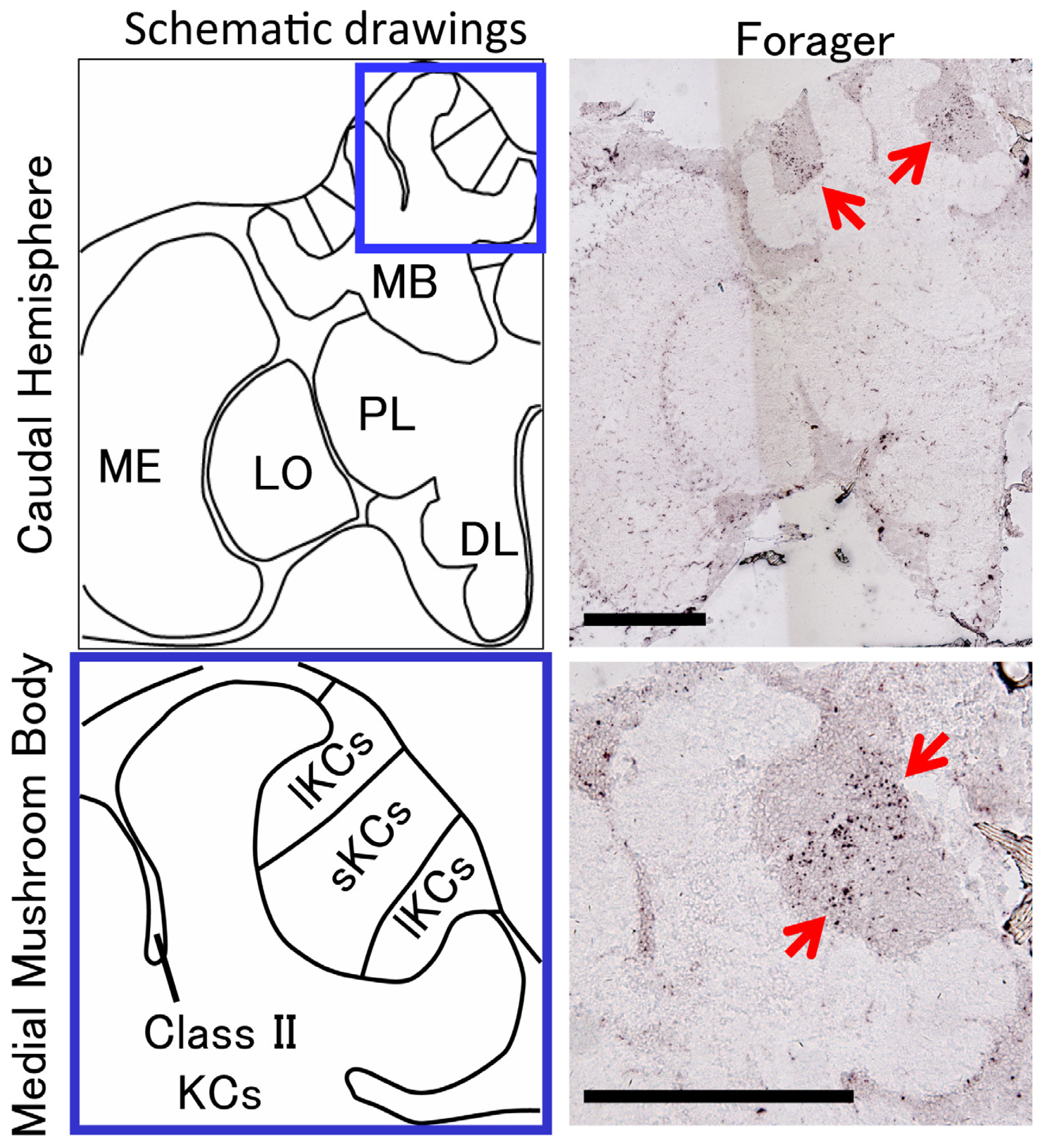
2. Identification of a Novel Non-Coding IEG, kakusei, which Can Be Used as a Marker to Visualize Neural Activity in the Honeybee Brain
3. Identification of Constitutive-Type Transcript Variants Implicates Cellular Function of kakusei
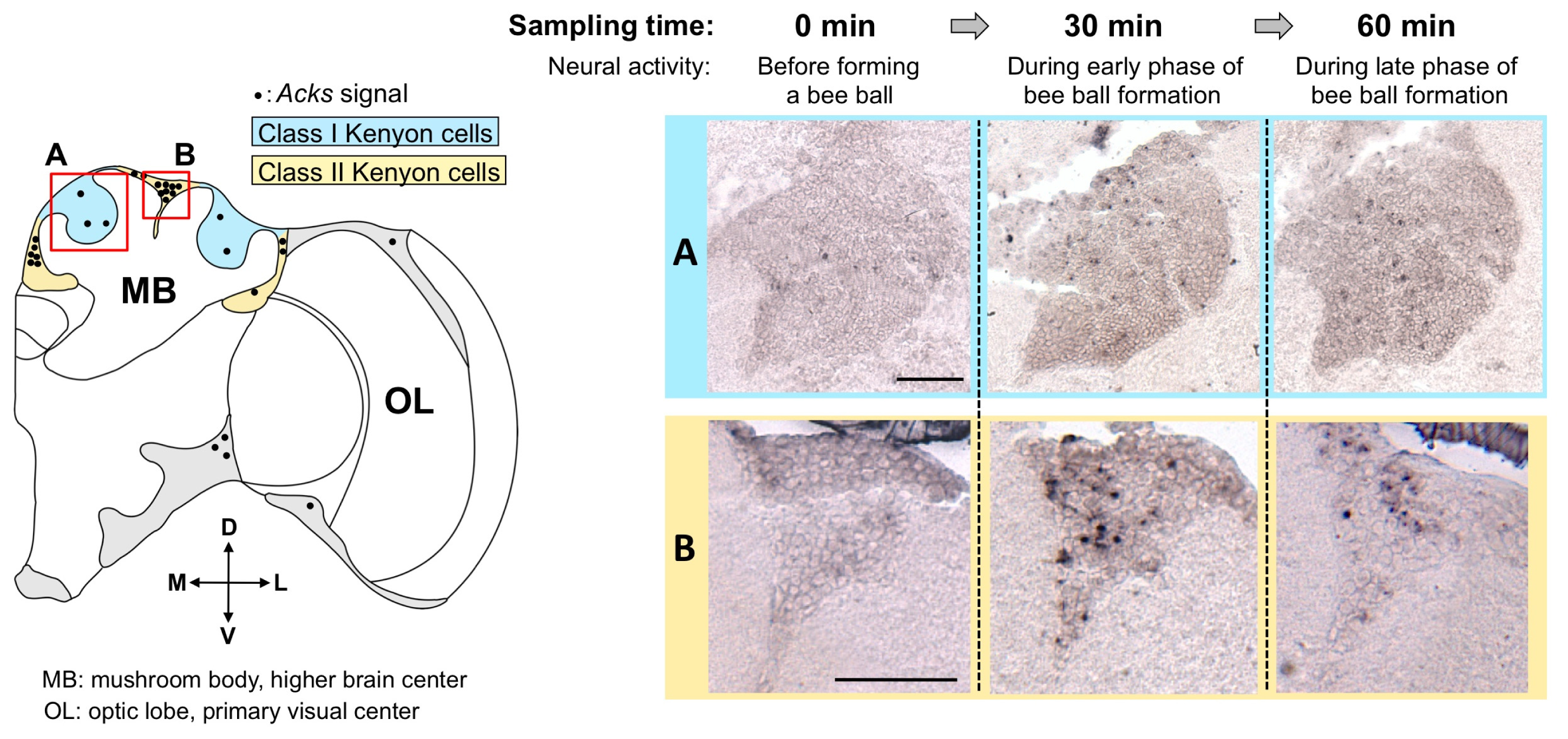
4. Isolation of Acks, a Homologue of kakusei from the Japanese Honeybee, and Detection of Active Neurons in the Workers Fighting with the Giant Hornet
5. Conclusions and Perspectives
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Seeley, T.D. The Wisdom of the Hive: The Social Physiology of Honey Bee Colonies; Harvard University Press: Cambridge, MA, USA, 1995. [Google Scholar]
- Winston, M.L. The Biology of the Honey Bee; Harvard University Press: Cambridge, MA, USA, 1987. [Google Scholar]
- Nijhout, H.F. Insect Hormones; Princeton University Press: Princeton, NJ, USA; p. 1998.
- Whitfield, C.W.; Band, M.R.; Bonaldo, M.F.; Kumar, C.G.; Liu, L.; Pardinas, J.R.; Robertson, H.M.; Soares, M.B.; Robinson, G.E. Annotated expressed sequence tags and cDNA microarrays for studies of brain and behavior in the honey bee. Genome Res 2002, 12, 555–566. [Google Scholar]
- Whitfield, C.W.; Cziko, A.M.; Robinson, G.E. Gene expression profiles in the brain predict behavior in individual honey bees. Science 2003, 302, 296–299. [Google Scholar]
- Whitfield, C.W.; Ben-Shahar, Y.; Brillet, C.; Leoncini, I.; Crauser, D.; Leconte, Y.; Rodriguez-Zas, S.; Robinson, G.E. Genomic dissection of behavioral maturation in the honey bee. Proc. Natl. Acad. Sci. USA 2006, 103, 16068–16075. [Google Scholar]
- Von Frisch, K. The Dance Language and Orientation of Bees; Harvard University Press: Cambridge, MA, USA, 1993. [Google Scholar]
- Dyer, F.C. The biology of the dance language. Annu. Rev. Entomol 2002, 47, 917–949. [Google Scholar]
- Riley, J.R.; Greggers, U.; Smith, A.D.; Reynolds, D.R.; Menzel, R. The flight paths of honeybees recruited by the waggle dance. Nature 2005, 435, 205–207. [Google Scholar]
- Srinivasan, M.V.; Zhang, S.; Altwein, M.; Tautz, J. Honeybee navigation: Nature and calibration of the “odometer”. Science 2000, 287, 851–853. [Google Scholar]
- Esch, H.E.; Zhang, S.; Srinivasan, M.V.; Tautz, J. Honeybee dances communicate distances measured by optic flow. Nature 2001, 411, 581–583. [Google Scholar]
- Nieh, J.C.; Tautz, J. Behaviour-locked signal analysis reveals weak 200–300 Hz comb vibrations during the honeybee waggle dance. J. Exp. Biol 2000, 203, 1573–1579. [Google Scholar]
- Sandeman, D.; Tautz, J.; Lindauer, M. Transmission of vibration across honeycombs and its detection by bee leg receptors. J. Exp. Biol 1996, 199, 2585–2594. [Google Scholar]
- Kiya, T.; Kunieda, T.; Kubo, T. Increased neural activity of a mushroom body neuron subtype in the brains of forager honeybees. PLoS One 2007, 2, e371. [Google Scholar]
- Kiya, T.; Kubo, T. Dance type and flight parameters are associated with different mushroom body neural activities in worker honeybee brains. PLoS One 2011, 6, e19301. [Google Scholar]
- Kiya, T.; Kubo, T. Analysis of GABAergic and non-GABAergic neuron activity in the optic lobes of the forager and re-orienting worker honeybee (Apis mellifera L.). PLoS One 2010, 5, e8833. [Google Scholar]
- Kiya, T.; Kunieda, T.; Kubo, T. Inducible- and constitutive-type transcript variants of kakusei, a novel non-coding immediate early gene, in the honeybee brain. Insect. Mol. Biol 2008, 17, 531–536. [Google Scholar]
- Ugajin, A.; Kiya, T.; Kunieda, T.; Ono, M.; Yoshida, T.; Kubo, T. Detection of neural activity in the brains of Japanese honeybee workers during the formation of a “hot defensive bee ball”. PLoS One 2012, 7, e32902. [Google Scholar]
- Guzowski, J.F.; McNaughton, B.L.; Barnes, C.A.; Worley, P.F. Environment-specific expression of the immediate-early gene Arc in hippocampal neuronal ensembles. Nat. Neurosci 1999, 2, 1120–1124. [Google Scholar]
- Guzowski, J.F.; Setlow, B.; Wagner, E.K.; McGaugh, J.L. Experience-dependent gene expression in the rat hippocampus after spatial learning: A comparison of the immediate-early genes Arc, c-fos, and zif268. J. Neurosci 2001, 21, 5089–5098. [Google Scholar]
- Sakata, S.; Kitsukawa, T.; Kaneko, T.; Yamamori, T.; Sakurai, Y. Task-dependent and cell-type-specific Fos enhancement in rat sensory cortices during audio-visual discrimination. Eur. J. Neurosci 2002, 15, 735–743. [Google Scholar]
- Mello, C.V.; Vicario, D.S.; Clayton, D.F. Song presentation induces gene expression in the songbird forebrain. Proc. Natl. Acad. Sci. USA 1992, 89, 6818–6822. [Google Scholar]
- Okuyama, T.; Suehiro, Y.; Imada, H.; Shimada, A.; Naruse, K.; Takeda, H.; Kubo, T.; Takeuchi, H. Induction of c-fos transcription in the medaka brain (Oryzias latipes) in response to mating stimuli. Biochem. Biophys. Res. Commun 2011, 404, 453–457. [Google Scholar]
- Wada, K.; Howard, J.T.; McConnell, P.; Whitney, O.; Lints, T.; Rivas, M.V.; Horita, H.; Patterson, M.A.; White, S.A.; Scharff, C.; et al. A molecular neuroethological approach for identifying and characterizing a cascade of behaviorally regulated genes. Proc. Natl. Acad. Sci. USA 2006, 103, 15212–15217. [Google Scholar]
- Vo, N.; Klein, M.E.; Varlamova, O.; Keller, D.M.; Yamamoto, T.; Goodman, R.H.; Impey, S. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc. Natl. Acad. Sci. USA 2005, 102, 16426–16431. [Google Scholar]
- Schratt, G.M.; Tuebing, F.; Nigh, E.A.; Kane, C.G.; Sabatini, M.E.; Kiebler, M.; Greenberg, M.E. A brain-specific microRNA regulates dendritic spine development. Nature 2006, 439, 283–289. [Google Scholar]
- Mobbs, P. The brain of the honeybee Apis mellifera. I. The connections and spatial organization of the mushroom bodies. Phil. Trans. R. Soc. Lond. B 1982, 298, 309–354. [Google Scholar]
- Strausfeld, N.J. Organization of the honey bee mushroom body: Representation of the calyx within the vertical and gamma lobes. J. Comp. Neurol 2002, 450, 4–33. [Google Scholar]
- Takeuchi, H.; Kage, E.; Sawata, M.; Kamikouchi, A.; Ohashi, K.; Ohara, M.; Fujiyuki, T.; Kunieda, T.; Sekimizu, K.; Natori, S.; et al. Identification of a novel gene, Mblk-1, that encodes a putative transcription factor expressed preferentially in the large-type Kenyon cells of the honeybee brain. Insect. Mol. Biol 2001, 10, 487–494. [Google Scholar]
- Sawata, M.; Yoshino, D.; Takeuchi, H.; Kamikouchi, A.; Ohashi, K.; Kubo, T. Identification and punctate nuclear localization of a novel noncoding RNA, Ks-1, from the honeybee brain. RNA 2002, 8, 772–785. [Google Scholar]
- Menzel, R. Searching for the memory trace in a mini-brain, the honeybee. Learn. Mem 2001, 8, 53–62. [Google Scholar]
- Heisenberg, M. Mushroom body memoir: From maps to models. Nat. Rev. Neurosci 2003, 4, 266–275. [Google Scholar]
- Keene, A.C.; Waddell, S. Drosophila olfactory memory: Single genes to complex neural circuits. Nat. Rev. Neurosci 2007, 8, 341–354. [Google Scholar]
- Ehmer, B.; Gronenberg, W. Segregation of visual input to the mushroom bodies in the honeybee (Apis mellifera). J. Comp. Neurol 2002, 451, 362–373. [Google Scholar]
- Gronenberg, W. Subdivisions of hymenopteran mushroom body calyces by their afferent supply. J. Comp. Neurol 2001, 435, 474–489. [Google Scholar]
- Grünewald, B. Morphology of feedback neurons in the mushroom body of the honeybee, Apis mellifera. J. Comp. Neurol 1999, 404, 114–126. [Google Scholar]
- Rybak, J.; Menzel, R. Anatomy of the mushroom bodies in the honey bee brain: The neuronal connections of the alpha-lobe. J. Comp. Neurol 1993, 334, 444–465. [Google Scholar]
- Schroter, U.; Menzel, R. A new ascending sensory tract to the calyces of the honeybee mushroom body, the subesophageal-calycal tract. J. Comp. Neurol 2003, 465, 168–178. [Google Scholar]
- Timmusk, T.; Palm, K.; Metsis, M.; Reintam, T.; Paalme, V.; Saarma, M.; Persson, H. Multiple promoters direct tissue-specific expression of the rat BDNF gene. Neuron 1993, 10, 475–489. [Google Scholar]
- Chen, W.G.; Chang, Q.; Lin, Y.; Meissner, A.; West, A.E.; Griffith, E.C.; Jaenisch, R.; Greenberg, M.E. Derepression of BDNF transcription involves calcium-dependent phosphorylation of MeCP2. Science 2003, 302, 885–889. [Google Scholar]
- Niibori, Y.; Hayashi, F.; Hirai, K.; Matsui, M.; Inokuchi, K. Alternative poly(A) site-selection regulates the production of alternatively spliced vesl-1/homer1 isoforms that encode postsynaptic scaffolding proteins. Neurosci. Res 2007, 57, 399–410. [Google Scholar]
- Brakeman, P.R.; Lanahan, A.A.; O’Brien, R.; Roche, K.; Barnes, C.A.; Huganir, R.L.; Worley, P.F. Homer: A protein that selectively binds metabotropic glutamate receptors. Nature 1997, 386, 284–288. [Google Scholar]
- Xiao, B.; Tu, J.C.; Petralia, R.S.; Yuan, J.P.; Doan, A.; Breder, C.D.; Ruggiero, A.; Lanahan, A.A.; Wenthold, R.J.; Worley, P.F. Homer regulates the association of group 1 metabotropic glutamate receptors with multivalent complexes of homer-related, synaptic proteins. Neuron 1998, 21, 707–716. [Google Scholar]
- Prasanth, K.V.; Prasanth, S.G.; Xuan, Z.; Hearn, S.; Freier, S.M.; Bennett, C.F.; Zhang, M.Q.; Spector, D.L. Regulating gene expression through RNA nuclear retention. Cell 2005, 123, 249–263. [Google Scholar]
- Hall, L.L.; Smith, K.P.; Byron, M.; Lawrence, J.B. Molecular anatomy of a speckle. Anat. Rec. Part A 2006, 288, 664–675. [Google Scholar]
- Sone, M.; Hayashi, T.; Tarui, H.; Agata, K.; Takeichi, M.; Nakagawa, S. The mRNA-like noncoding RNA Gomafu constitutes a novel nuclear domain in a subset of neurons. J. Cell Sci 2007, 120, 2498–2506. [Google Scholar]
- Kavaliers, M.; Choleris, E. Antipredator responses and defensive behavior: Ecological and ethological approaches for the neurosciences. Neurosci. Biobehav. Rev 2001, 25, 577–586. [Google Scholar]
- Ono, M.; Igarashi, T.; Ohno, E.; Sasaki, M. Unusual thermal defence by a honeybee against mass attack by hornets. Nature 1995, 377, 334–336. [Google Scholar]
- Ono, M.; Terabe, H.; Hori, H.; Sasaki, M. Insect signalling: components of giant hornet alarm pheromone. Nature 2003, 424, 637–638. [Google Scholar]
- Sugahara, M.; Nishimura, Y.; Sakamoto, F. Differences in heat sensitivity between Japanese honeybees and hornets under high carbon dioxide and humidity conditions inside bee balls. Zool. Sci 2012, 29, 30–36. [Google Scholar]
- Hong, S.T.; Bang, S.; Hyun, S.; Kang, J.; Jeong, K.; Paik, D.; Chung, J.; Kim, J. cAMP signalling in mushroom bodies modulates temperature preference behaviour in Drosophila. Nature 2008, 454, 771–775. [Google Scholar]
- Viswanath, V.; Story, G.M.; Peier, A.M.; Petrus, M.J.; Lee, V.M.; Hwang, S.W.; Patapoutian, A.; Jegla, T. Opposite thermosensor in fruitfly and mouse. Nature 2003, 423, 822–823. [Google Scholar]
- Tracey, W.D., Jr; Wilson, R.I.; Laurent, G.; Benzer, S. Painless, a Drosophila gene essential for nociception. Cell 2003, 113, 261–273. [Google Scholar]
- Rosenzweig, M.; Brennan, K.M.; Tayler, T.D.; Phelps, P.O.; Patapoutian, A.; Garrity, P.A. The Drosophila ortholog of vertebrate TRPA1 regulates thermotaxis. Genes Dev 2005, 19, 419–424. [Google Scholar]
- Rosenzweig, M.; Kang, K.; Garrity, P.A. Distinct TRP channels are required for warm and cool avoidance in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2008, 105, 14668–14673. [Google Scholar]
- Hamada, F.N.; Rosenzweig, M.; Kang, K.; Pulver, S.R.; Ghezzi, A.; Jegla, T.J.; Garrity, P.A. An internal thermal sensor controlling temperature preference in Drosophila. Nature 2008, 454, 217–220. [Google Scholar]
- Lee, Y.; Lee, Y.; Lee, J.; Bang, S.; Hyun, S.; Kang, J.; Hong, S.T.; Bae, E.; Kaang, B.K.; Kim, J. Pyrexia is a new thermal transient receptor potential channel endowing tolerance to high temperatures in Drosophila melanogaster. Nat. Genet 2005, 37, 305–310. [Google Scholar]
- Xu, S.Y.; Cang, C.L.; Liu, X.F.; Peng, Y.Q.; Ye, Y.Z.; Zhao, Z.Q.; Guo, A.K. Thermal nociception in adult Drosophila: Behavioral characterization and the role of the painless gene. Genes Brain Behav 2006, 5, 602–613. [Google Scholar]
- Sokabe, T.; Tsujiuchi, S.; Kadowaki, T.; Tominaga, M. Drosophila painless is a Ca2+-requiring channel activated by noxious heat. J. Neurosci 2008, 28, 9929–9938. [Google Scholar]
- Gallio, M.; Ofstad, T.A.; Macpherson, L.J.; Wang, J.W.; Zuker, C.S. The coding of temperature in the Drosophila brain. Cell 2011, 144, 614–624. [Google Scholar]
- Kiya, T.; Hori, S.; Takeuchi, H.; Kubo, T. New approach toward understanding of neural basis of the honeybee “dance communication”. Tanpakushitsu Kakusan Koso 2008, 53, 1368–1374. [Google Scholar]
- Hammer, M.; Menzel, R. Learning and memory in the honeybee. J. Neurosci 1995, 15, 1617–1630. [Google Scholar]
- Sanjana, N.E.; Cong, L.; Zhou, Y.; Cunniff, M.M.; Feng, G.; Zhang, F. A transcription activator-like effector toolbox for genome engineering. Nat. Protoc 2012, 7, 171–192. [Google Scholar]
- Watanabe, T.; Ochiai, H.; Sakuma, T.; Horch, H.W.; Hamaguchi, N.; Nakamura, T.; Bando, T.; Ohuchi, H.; Yamamoto, T.; Noji, S.; et al. Non-transgenic genome modifications in a hemimetabolous insect using zinc-finger and TAL effector nucleases. Nat. Commun 2012, 3, 1017. [Google Scholar]
- Zhang, F.; Cong, L.; Lodato, S.; Kosuri, S.; Church, G.M.; Arlotta, P. Efficient construction of sequence-specific TAL effectors for modulating mammalian transcription. Nat. Biotechnol 2011, 29, 149–153. [Google Scholar] [Green Version]
- Venken, K.J.; Simpson, J.H.; Bellen, H.J. Genetic manipulation of genes and cells in the nervous system of the fruit fly. Neuron 2011, 72, 202–230. [Google Scholar]



© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kiya, T.; Ugajin, A.; Kunieda, T.; Kubo, T. Identification of kakusei, a Nuclear Non-Coding RNA, as an Immediate Early Gene from the Honeybee, and Its Application for Neuroethological Study. Int. J. Mol. Sci. 2012, 13, 15496-15509. https://doi.org/10.3390/ijms131215496
Kiya T, Ugajin A, Kunieda T, Kubo T. Identification of kakusei, a Nuclear Non-Coding RNA, as an Immediate Early Gene from the Honeybee, and Its Application for Neuroethological Study. International Journal of Molecular Sciences. 2012; 13(12):15496-15509. https://doi.org/10.3390/ijms131215496
Chicago/Turabian StyleKiya, Taketoshi, Atsushi Ugajin, Takekazu Kunieda, and Takeo Kubo. 2012. "Identification of kakusei, a Nuclear Non-Coding RNA, as an Immediate Early Gene from the Honeybee, and Its Application for Neuroethological Study" International Journal of Molecular Sciences 13, no. 12: 15496-15509. https://doi.org/10.3390/ijms131215496



