Targeting Death Receptor TRAIL-R2 by Chalcones for TRAIL-Induced Apoptosis in Cancer Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cytotoxic and Apoptotic Activities of TRAIL in HeLa Cancer Cells
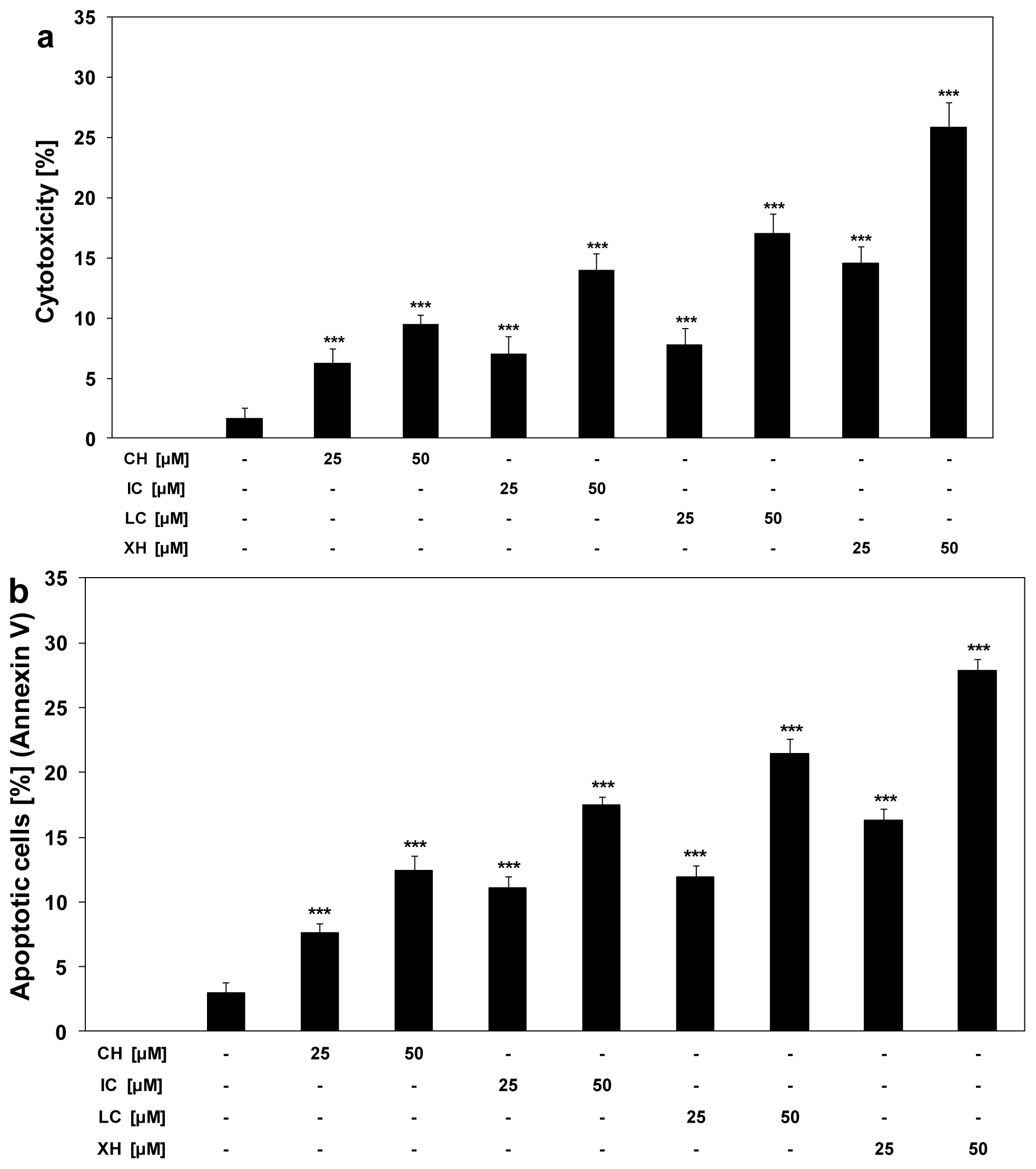
2.2. Cytotoxic and Apoptotic Activities of Chalcones in HeLa Cancer Cells
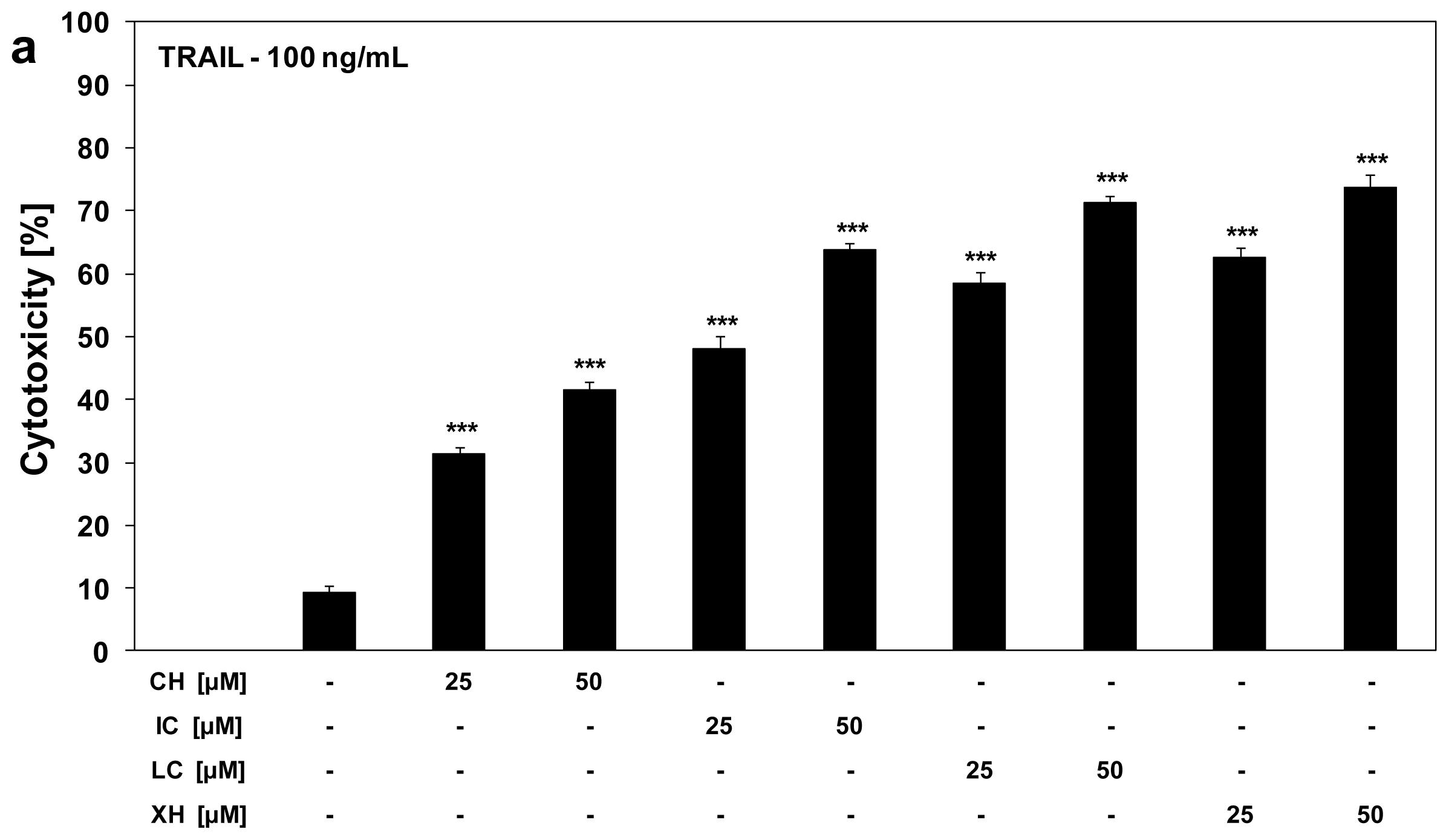
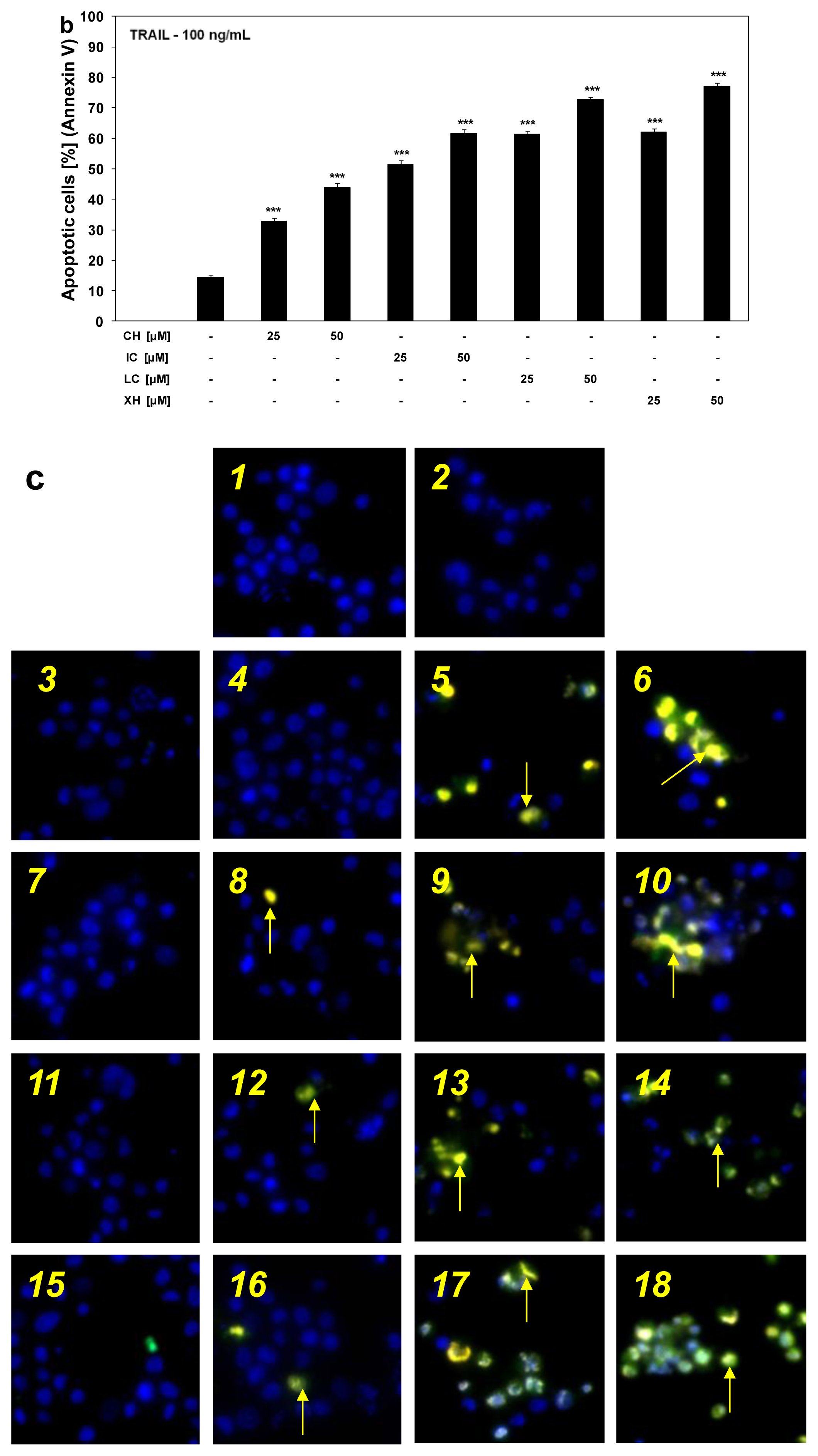
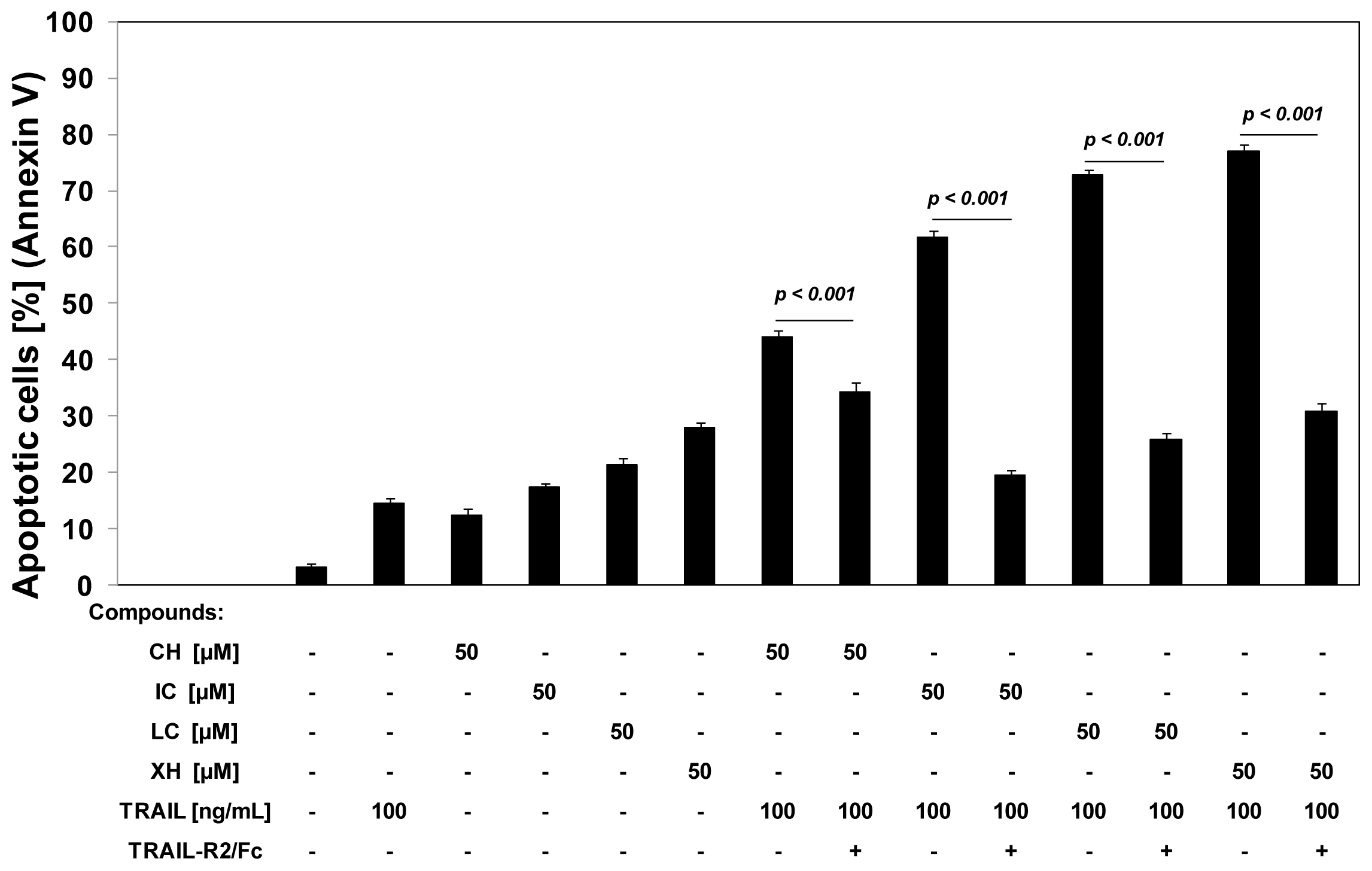
2.3. Cytotoxic and Apoptotic Activities of TRAIL in Combination with Chalcones in HeLa Cancer Cells
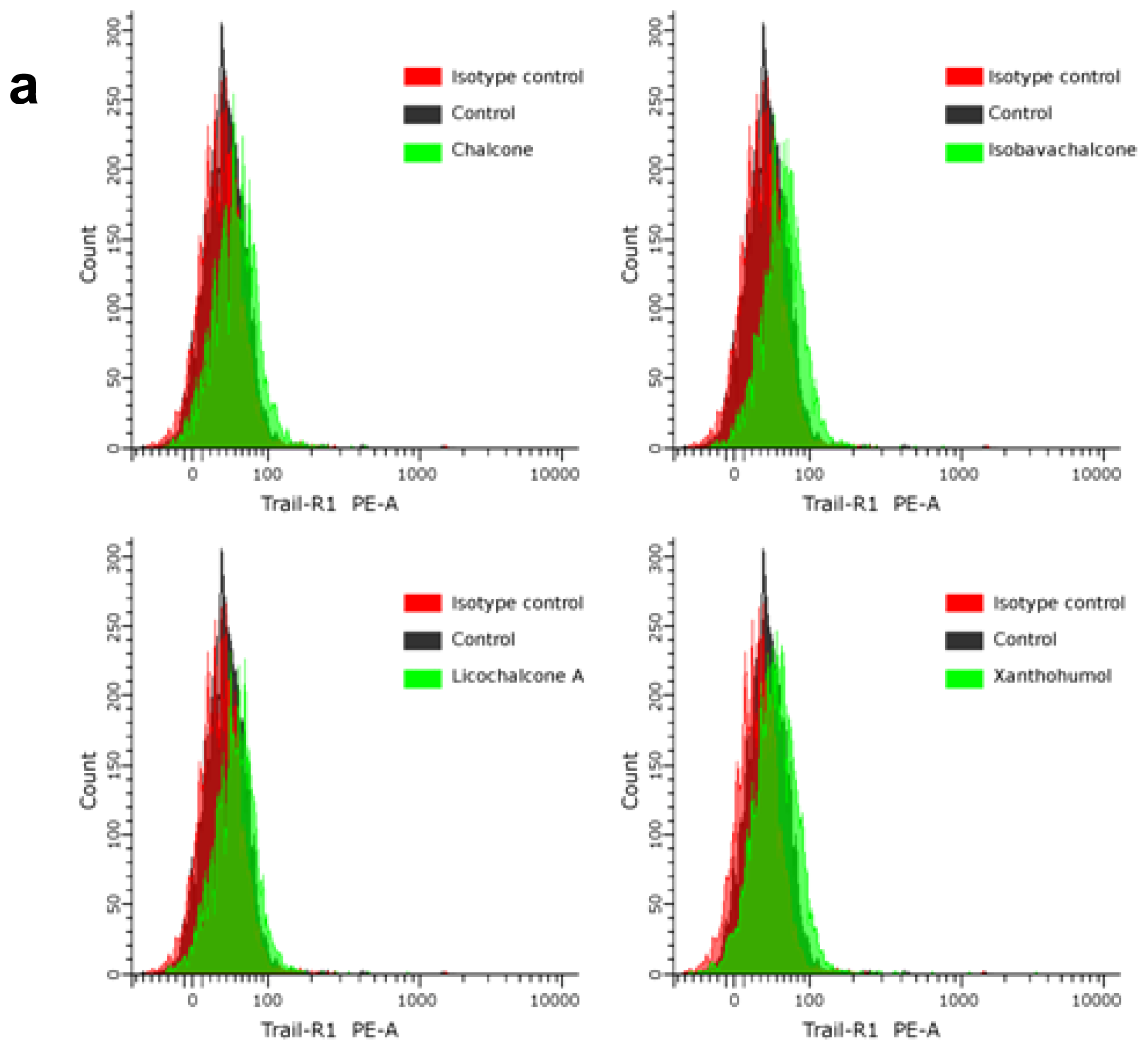
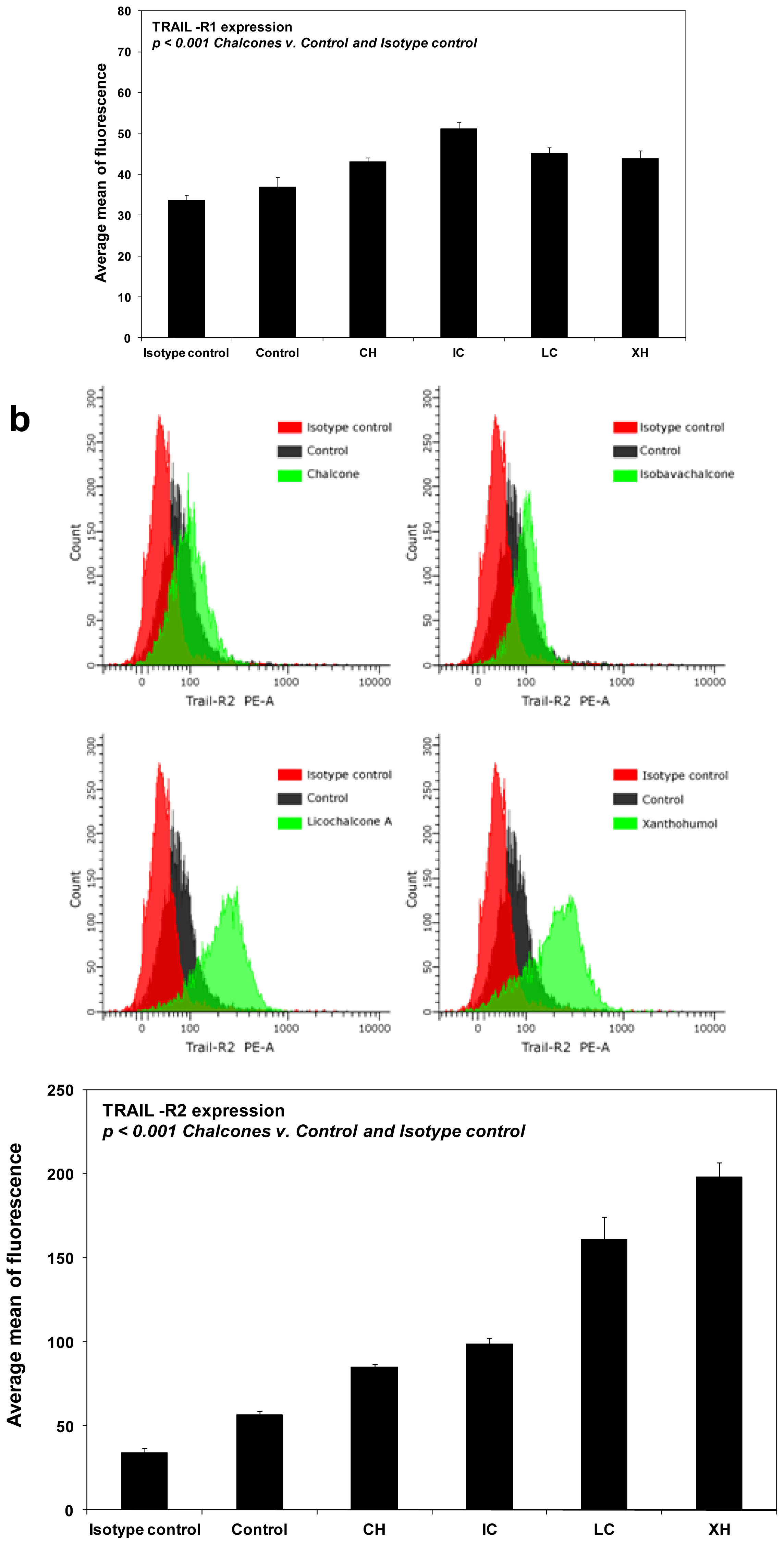
2.4. Chalcones Enhance TRAIL-Induced Apoptosis in HeLa Cancer Cells through Up-Regulation of TRAIL-R2
3. Experimental Section
3.1. Reagents
3.2. Cancer Cell Culture
3.3. Detection of Cell Death Using MTT Cytotoxicity Assay
3.4. Lactate Dehydrogenase Release Assay
3.5. Detection of Apoptotic Cell Death by Flow Cytometry
3.6. Detection of Apoptotic Cell Death by Fluorescence Microscopy
3.7. Analysis of Death Receptor Expression on the Cancer Cell Surface by Flow Cytometry
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Holoch, P.A.; Griffith, T.S. TNF-related apoptosis-inducing ligand (TRAIL): A new path to anti-cancer therapies. Eur. J.Pharmacol 2009, 625, 63–72. [Google Scholar]
- Lee, J.Y.; Huerta-Yepez, S.; Vega, M.; Baritaki, S.; Spandidos, D.A.; Bonavida, B. The NO TRAIL to YES TRAIL in cancer therapy. Int. J. Oncol 2007, 31, 685–691. [Google Scholar]
- Szliszka, E.; Zydowicz, G.; Janoszka, B.; Dobosz, C.; Kowalczyk-Ziomek, G.; Krol, W. Ethanolic extract of Brazilian green propolis sensitizes prostate cancer cells to TRAIL-induced apoptosis. Int. J. Oncol 2011, 38, 941–953. [Google Scholar]
- Mellier, G.; Huang, S.; Shenoy, K.; Pervaiz, S. TRAILing death in cancer. Mol. Aspects Med 2010, 31, 93–112. [Google Scholar]
- Szliszka, E.; Mazur, B.; Zydowicz, G.; Czuba, Z.P.; Krol, W. TRAIL-induced apoptosis and expression of death receptor TRAIL-R1 and TRAIL-R2 in bladder cancer cells. Folia Histochem. Cytobiol 2009, 47, 579–585. [Google Scholar]
- Mahalingam, D.; Szegezdi, E.; Keane, M.; de Jong, S.; Samali, A. TRAIL receptor signalling and modulation: Are we on the right TRAIL? Cancer Treat. Rev 2009, 35, 280–288. [Google Scholar]
- Qiu, B.; Sun, X.; Zhang, D.; Wang, Y.; Tao, J.; Ou, S. TRAIL and paclitaxel synergize to kill U87 cells and U87-derived stem-like cells in vitro. Int. J. Mol. Sci 2012, 13, 9142–9156. [Google Scholar]
- Stolfi, C.; Pallone, F.; Monteleone, G. Molecular targets of TRAIL-sensitizing agents in colorectal cancer. Int. J. Mol. Sci 2012, 13, 7886–7901. [Google Scholar]
- Zhang, L.; Fang, B. Mechanisms of resistance to TRAIL-induced apoptosis in cancer. Cancer Gene Ther 2005, 12, 228–237. [Google Scholar]
- Szliszka, E.; Bronikowska, J.; Majcher, A.; Miszkiewicz, J.; Krol, W. Enhanced sensitivity of hormone-refractory prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) mediated cytotoxicity by taxanes. Cent. Eur. J. Urol 2009, 62, 29–34. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Mertas, A.; Paradysz, A.; Krol, W. The dietary isoflavone biochanin-A sensitizes prostate cancer cells to TRAIL-induced apoptosis. Urol. Oncol. 2011. [Google Scholar] [CrossRef]
- Russo, M.; Mupo, A.; Spagnuolo, C.; Russo, G.L. Exploring death receptor pathways as selective targets in cancer therapy. Biochem. Pharmacol 2010, 80, 674–682. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Jernas, K.; Krol, W. Dietary flavonoids sensitize HeLa cells to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). Int. J. Mol. Sci 2008, 9, 56–64. [Google Scholar]
- Szliszka, E.; Gebka, J.; Bronikowska, J.; Krol, W. Dietary flavones enhance the effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) on bladder cancer cells. Cent. Eur. J. Urol 2010, 63, 138–143. [Google Scholar]
- Bronikowska, J.; Szliszka, E.; Czuba, Z.P.; Zwolinski, D.; Szmydki, B.; Krol, W. The combination of TRAIL and isoflavones enhances apoptosis in cancer cells. Molecules 2010, 15, 2000–2015. [Google Scholar]
- Szliszka, E.; Krol, W. The role of dietary polyphenols in tumor necrosis factor-related apoptosis inducing ligand (TRAIL)-induced apoptosis for cancer chemoprevention. Eur. J. Cancer Prev 2011, 20, 63–69. [Google Scholar]
- Go, M.L.; Wu, X.; Liu, X.L. Chalcones: An update on cytotoxic and chemopreventive properties. Curr. Med. Chem 2005, 12, 481–499. [Google Scholar]
- Orlikova, B.; Tasdemir, D.; Golais, F.; Dicato, M.; Dieterich, M. Dietary chalcones with chemopreventive and chemotherapeutic potential. Genes Nutr 2011, 6, 125–147. [Google Scholar]
- Rahman, R.N.; Zakaria, I.I.; Salleh, A.B.; Basri, M. Enzymatic properties and mutational studies of chalcone synthase from physcomitrella patens. Int. J. Mol. Sci 2012, 13, 9673–9691. [Google Scholar]
- Okuyama, T.; Takata, M.; Takayasu, J.; Hasegawa, T.; Tokuda, H.; Nishino, A.; Nishino, H.; Iwashima, A. Anti-tumor-promotion by principles obtained from Angelica keiskei. Planta Med 1991, 57, 242–246. [Google Scholar]
- Gerhauser, C.; Alt, A.; Heiss, E.; Gamal-Eldeen, A.; Klimo, K.; Knauft, J.; Neumann, I.; Scherf, H.R.; Frank, N.; Bartsch, H.; et al. Cancer chemopreventive activity of xanthohumol, a natural product derived from hop. Mol. Cancer Ther 2002, 1, 959–969. [Google Scholar]
- Haraguchi, H.; Inoue, J.; Tamura, Y.; Mizutani, K. Antioxidative components of Psoralea corylifolia (Leguminosae). Phytother. Res 2002, 16, 539–544. [Google Scholar]
- Fu, Y.; Hsieh, T.C.; Guo, J.; Kunicki, J.; Lee, M.Y.; Darzynkiewicz, Z.; Wu, J.M. Licochalcone-A, a novel flavonoid isolated from licorice root (Glycyrrhiza glabra), causes G2 and late-G1 arrests in androgen-independent PC-3 prostate cancer cells. Biochem. Biophys. Res. Commun 2004, 322, 263–270. [Google Scholar]
- Desmulle, L.; Bellahcene, A.; Dhooge, W.; Comhaire, F.; Roelens, F.; Huvaere, K.; Heyerick, A.; Castronovo, V.; Dekeukeleire, D. Antiproliferative properties of prenylated flavonoids from hops (Humulus lupulus L.) in prostate cancer cell lines. Phytomedicine 2006, 13, 732–734. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Sedek, L.; Paradysz, A.; Krol, W. Enhanced TRAIL-mediated apoptosis in prostate cancer cells by the bioactive compounds neobavaisoflavone and psoralidin isolated from Psoralea corylifolia. Pharmacol. Rep 2011, 63, 139–148. [Google Scholar]
- Boumendjel, A.; Boccard, J.; Carrupt, P.A.; Nicolle, E.; Blanc, M.; Geze, A.; Choisnard, L.; Wouessidjewe, D.; Matera, E.L.; Dumontet, C. Antimitotic and antiproliferative activities of chalcones: forward structure-activity relationship. J. Med. Chem 2008, 51, 2307–2310. [Google Scholar]
- Echeverria, C.; Santibanez, J.S.; Donoso-Tauda, O.; Escobar, C.A.; Ramirez-Tagle, R. Structural antitumoral activity relationships of synthetic chalcones. Int. J. Mol. Sci 2009, 10, 221–231. [Google Scholar]
- Vogel, S.; Barbic, M.; Jurgenliemk, G.; Heilmann, J. Synthesis, cytotoxicity, anti-oxidative and anti-inflammatory activity of chalcones and influence of A-ring modifications on the pharmacological effect. Eur. J. Med. Chem 2010, 45, 2206–2213. [Google Scholar]
- Syam, S.; Abdelwahab, S.I.; Al-Mamary, M.A.; Mohan, S. Synthesis of chalcones with anticancer activities. Molecules 2012, 17, 6179–6195. [Google Scholar]
- Eddouks, M.; Chattopadhyay, D.; De Feo, V.; Cho, W.C. Medicinal plants in the prevention and treatment of chronic diseases. Evid. Based Complement. Alternat. Med 2012, 2012, 458274. [Google Scholar]
- Thomasset, S.C.; Berry, T.P.; Garcea, G.; Marczylo, T.; Steward, W.P.; Gescher, A.J. Dietary polyphenolic phytochemicals – Promising cancer chemopreventive agents in humans? A review of their clinical properties. Int. J. Cancer 2007, 120, 451–458. [Google Scholar]
- Nowakowska, Z. A review of anti-infective and anti-inflammatory chalcones. Eur. J. Med. Chem 2007, 42, 125–137. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Mazur, B.; Sedek, L.; Paradysz, A.; Krol, W. Chalcones enhance TRAIL-induced apoptosis in prostate cancer cells. Int. J. Mol. Sci 2010, 11, 1–13. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Mazur, B.; Paradysz, A.; Krol, W. Chalcones and dihydrochalcones augment TRAIL-mediated apoptosis in prostate cancer cells. Molecules 2010, 15, 5336–5353. [Google Scholar]
- Szliszka, E.; Helewski, K.J.; Mizgala, E.; Krol, W. The dietary flavonol fisetin enhances the apoptosis-inducing potential of TRAIL in prostate cancer cells. Int. J. Oncol 2011, 39, 771–779. [Google Scholar]
- Cho, W.C. Targeting the signaling pathways in cancer therapy. Expert Opin. Ther. Targets. 2012, 16, 1–3. [Google Scholar]
- Szliszka, E.; Bronikowska, J.; Czuba, Z.P.; Krol, W. Isoflavones augment the effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) on prostate cancer cells. Cent. Eur. J. Urol 2010, 63, 182–186. [Google Scholar]
- Horinanka, M.; Yoshida, T.; Shiraishi, T.; Nakata, S.; Wakada, M.; Nakanishi, R.; Nishino, H.; Sakai, T. The combination of TRAIL and luteolin enhances apoptosis in human cervival cancer HeLa cells. Biochem. Biophys. Res. Commun 2005, 333, 833–838. [Google Scholar]
- Bronikowska, J.; Szliszka, E.; Jaworska, D.; Czuba, Z.P.; Krol, W. The coumarin psoralidin enhances anticancer effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). Molecules 2012, 17, 6449–6464. [Google Scholar]
- Shen, K.H.; Chang, J.K.; Hsu, Y.L.; Kuo, P.L. Chalcone arrests cell cycle progression and induces apoptosis through induction of mitochondrial pathway and inhibition of nuclear factor kappa-B signalling in human bladder cancer cells. Basic Clin. Pharmacol. Toxicol 2007, 101, 254–261. [Google Scholar]
- Hsu, Y.L.; Kuo, P.L.; Tzeng, W.S.; Lin, C.C. Chalcone inhibits the proliferation of human breast cancer cell by blocking cell cycle progression and inducing apoptosis. Food Chem. Toxicol 2006, 44, 704–713. [Google Scholar]
- Jing, H.; Zhou, X.; Dong, X.; Cao, J.; Zhu, H.; Lou, J.; Hu, Y.; He, Q.; Yang, B. Abrogation of Akt signaling by isobavachalcone contributes to its anti-proliferative effects towards human cancer cells. Cancer Lett 2010, 294, 167–177. [Google Scholar]
- Nishimura, R.; Tabata, K.; Arakawa, M.; Ito, Y.; Kimura, Y.; Akihisa, T.; Nagai, H.; Sakuma, A.; Kohno, H.; Suzuki, T. Isobavachalcone, a chalcone constituent of Angelica keiskei, induces apoptosis in neuroblastoma. Biol. Pharm. Bull 2007, 30, 1878–1883. [Google Scholar]
- Drenzek, J.G.; Seiler, N.L.; Jaskula-Sztul, R.; Rausch, M.M.; Rose, S.L. Xanthohumol decreases Notch1 expression and cell growth by cell cycle arrest and induction of apoptosis in epithelial ovarian cancer cell lines. Gynecol. Oncol 2011, 122, 396–401. [Google Scholar]
- Deeb, D.; Gao, X.; Jiang, H.; Arbab, A.S.; Dulchavsky, S.A.; Gautam, S.C. Growth inhibitory and apoptosis-inducing effects of xanthohumol, a prenylated chalone present in hops, in human prostate cancer cells. Anticancer Res 2010, 30, 3333–3339. [Google Scholar]
- Szliszka, E.; Krol, W. Soy isoflavones augment the effect of TRAIL-mediated apoptotic death in prostate cancer cells. Oncol. Rep 2011, 26, 533–541. [Google Scholar]
- Yoshida, T.; Horinaka, M.; Takara, M.; Tsuchihashi, M.; Mukai, N.; Wakada, M.; Sakai, T. Combination of isoliquiritigenin and tumor necrosis factor-related apoptosis-inducing ligand induces apoptosis in colon cancer HT29 cells. Environ. Health Prev. Med 2008, 13, 281–287. [Google Scholar]
- Kim, N. Butein sensitizes human leukemia cells to apoptosis induced by tumor necrosis factor-related apoptosis inducing ligand (TRAIL). Arch. Pharm. Res 2008, 31, 1179–1186. [Google Scholar]
- Moon, D.O.; Kim, M.O.; Choi, Y.H.; Kim, G.Y. Butein sensitizes human hepatoma cells to TRAIL-induced apoptosis via extracellular signal-regulated kinase/Sp1-dependent DR5 upregulation and NF-kappaB inactivation. Mol. Cancer Ther 2010, 9, 1583–1595. [Google Scholar]
- Tang, Y.; Li, X.; Liu, Z.; Simoneau, A.R.; Xie, J.; Zi, X. Flavokawain B, a kava chalcone, induces apoptosis via up-regulation of death-receptor 5 and Bim expression in androgen receptor negative, hormonal refractory prostate cancer cell lines and reduces tumor growth. Int. J. Cancer 2010, 127, 1758–1768. [Google Scholar]
- Szliszka, E.; Kostrzewa-Susłow, E.; Bronikowska, J.; Jaworska, D.; Janeczko, T.; Czuba, Z.P.; Krol, W. Synthetic flavanones augment the anticancer effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). Molecules 2012, 17, 11693–11711. [Google Scholar]
- Chen, W.; Wang, X.; Zhuang, J.; Zhang, L.; Lin, Y. Induction of death receptor 5 and suppression of surviving contribute to sensitization of TRAIL-induced cytotoxicity by quercetin in non-small lung cancer cells. Carcinogenesis 2007, 28, 2114–2121. [Google Scholar]
- Kim, J.Y.; Kim, E.H.; Park, S.S.; Lim, J.H.; Kwon, T.K.; Choi, K.S. Quercetin sensitizes human hepatoma cells to TRAIL-induced apoptosis via Sp1-mediated DR5 upregulation and proteasome-mediated c-FLIPS downregulation. J. Cell. Biochem 2008, 105, 1386–1398. [Google Scholar]
- Yoshida, T.; Konishi, M.; Horinaka, M.; Yasuda, T.; Goda, A.E.; Taniguchi, H.; Yano, K.; Wakada, M.; Sakai, T. Kaempferol sensitizes colon cancer cells to TRAIL-induced apoptosis. Biochem. Biophys. Res. Commun 2008, 375, 129–133. [Google Scholar]
- Jung, Y.H.; Heo, J.; Lee, Y.J.; Kwon, T.K.; Kim, Y.H. Quercetin enhances TRAIL-induced apoptosis in prostate cancer cells via increased protein stability of death receptor 5. Life Sci 2010, 86, 351–357. [Google Scholar]
- Jin, C.Y.; Park, C.; Hwang, H.J.; Kim, G.Y.; Choi, B.T.; Kim, W.J.; Choi, Y.H. Naringenin up-regulates the expression of death receptor 5 and enhances TRAIL-induced apoptosis in human lung cancer A549 cells. Mol. Nutr. Food Res 2011, 55, 300–309. [Google Scholar]
- Horinaka, M.; Yoshida, T.; Shiraishi, T.; Nakata, S.; Wakada, M.; Sakai, T. The dietary flavonoid apigenin sensitizes malignant tumor cells to tumor necrosis factor-related apoptosis-inducing ligand. Mol. Cancer Ther 2006, 5, 945–951. [Google Scholar]
- Tanaguchi, H.; Yoshida, T.; Horinaka, M.; Yasuda, T.; Goda, A.E.; Konishi, M.; Wakada, M.; Kataoka, K.; Yoshikawa, T.; Sakai, T. Baicalein overcomes tumor necrosis factor-related apoptosis-inducing ligand resistance via two different cell specific pathways in cancer cells but not in normal cells. Cancer Res 2008, 68, 8918–8927. [Google Scholar]
- Ding, J.; Polier, G.; Köhler, R.; Giaisi, M.; Krammer, P.H.; Li-Weber, M. Wogonin and related natural flavones overcome tumor necrosis factor-related apoptosis inducing ligand (TRAIL) protein resistance of tumors by down-regulation of c-FLIP protein and up-regulation of TRAIL receptor 2 expression. J. Biol. Chem 2012, 287, 641–649. [Google Scholar]
- Son, Y.G.; Kim, E.H.; Kim, J.Y.; Kim, S.U.; Kwon, T.K.; Yoon, A.R.; Yun, C.O.; Choi, K.S. Silibinin sensitizes human glioma cells to TRAIL-mediated apoptosis via DR5 upregulation and downregulation of cFLIP and surviving. Cancer Res 2007, 67, 8274–8284. [Google Scholar]
- Tsai, C.F.; Yeh, W.L.; Huang, S.M.; Tan, T.W.; Lu, D.Y. Wogonin induces reactive oxygen species production and cell apoptosis in human glioma cancer cells. Int. J. Mol. Sci 2012, 13, 9877–9892. [Google Scholar]
- Szliszka, E.; Majcher, A.; Domino, M.; Pietsz, G.; Krol, W. Cytotoxic activity of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) against bladder cancer cells after using chemotherapeutic drugs. Urol. Pol 2007, 60, 138–142. [Google Scholar]
- Szliszka, E.; Czuba, Z.P.; Kawczyk-Krupka, A.; Sieron-Stoltny, A.; Sieron, A.; Krol, W. Chlorin-based photodynamic therapy enhances the effect of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in bladder cancer cells. Med. Sci. Monit. 2012, 18, BR47–53. [Google Scholar]
- Szliszka, E.; Zydowicz, G.; Mizgala, E.; Krol, W. Artepillin C (3,5-diprenyl-4-hydroxycinnamic acid) sensitizes prostate cancer LNCaP cells to TRAIL-induced apoptosis. Int. J. Oncol 2012, 41, 818–828. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Szliszka, E.; Jaworska, D.; Ksek, M.; Czuba, Z.P.; Król, W. Targeting Death Receptor TRAIL-R2 by Chalcones for TRAIL-Induced Apoptosis in Cancer Cells. Int. J. Mol. Sci. 2012, 13, 15343-15359. https://doi.org/10.3390/ijms131115343
Szliszka E, Jaworska D, Ksek M, Czuba ZP, Król W. Targeting Death Receptor TRAIL-R2 by Chalcones for TRAIL-Induced Apoptosis in Cancer Cells. International Journal of Molecular Sciences. 2012; 13(11):15343-15359. https://doi.org/10.3390/ijms131115343
Chicago/Turabian StyleSzliszka, Ewelina, Dagmara Jaworska, Małgorzata Ksek, Zenon P. Czuba, and Wojciech Król. 2012. "Targeting Death Receptor TRAIL-R2 by Chalcones for TRAIL-Induced Apoptosis in Cancer Cells" International Journal of Molecular Sciences 13, no. 11: 15343-15359. https://doi.org/10.3390/ijms131115343




