Protein Profiling of Blood Samples from Patients with Hereditary Leiomyomatosis and Renal Cell Cancer by Surface-Enhanced Laser Desorption/Ionization Time-of-Flight Mass Spectrometry
Abstract
:1. Introduction
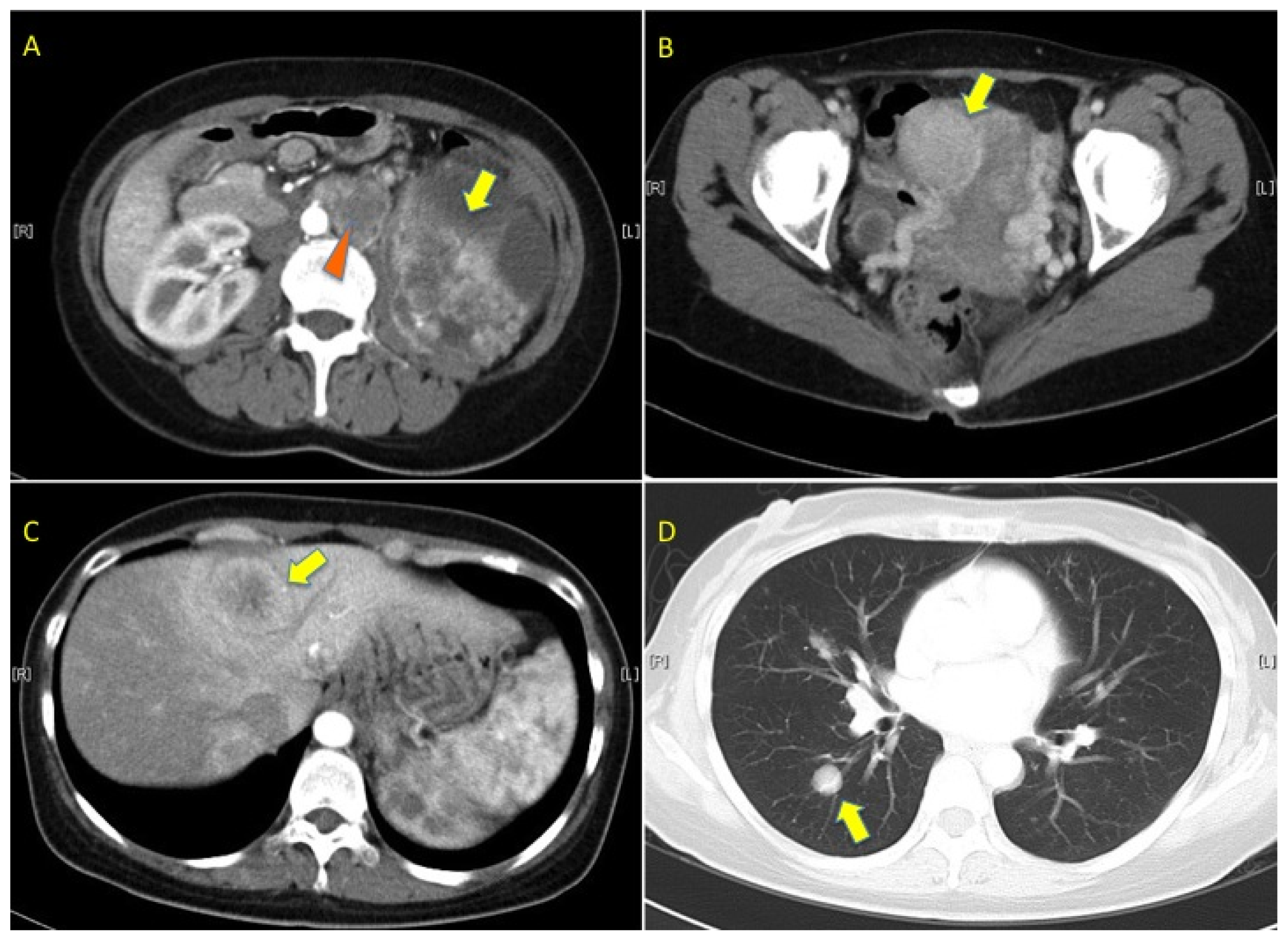
2. Case Presentation
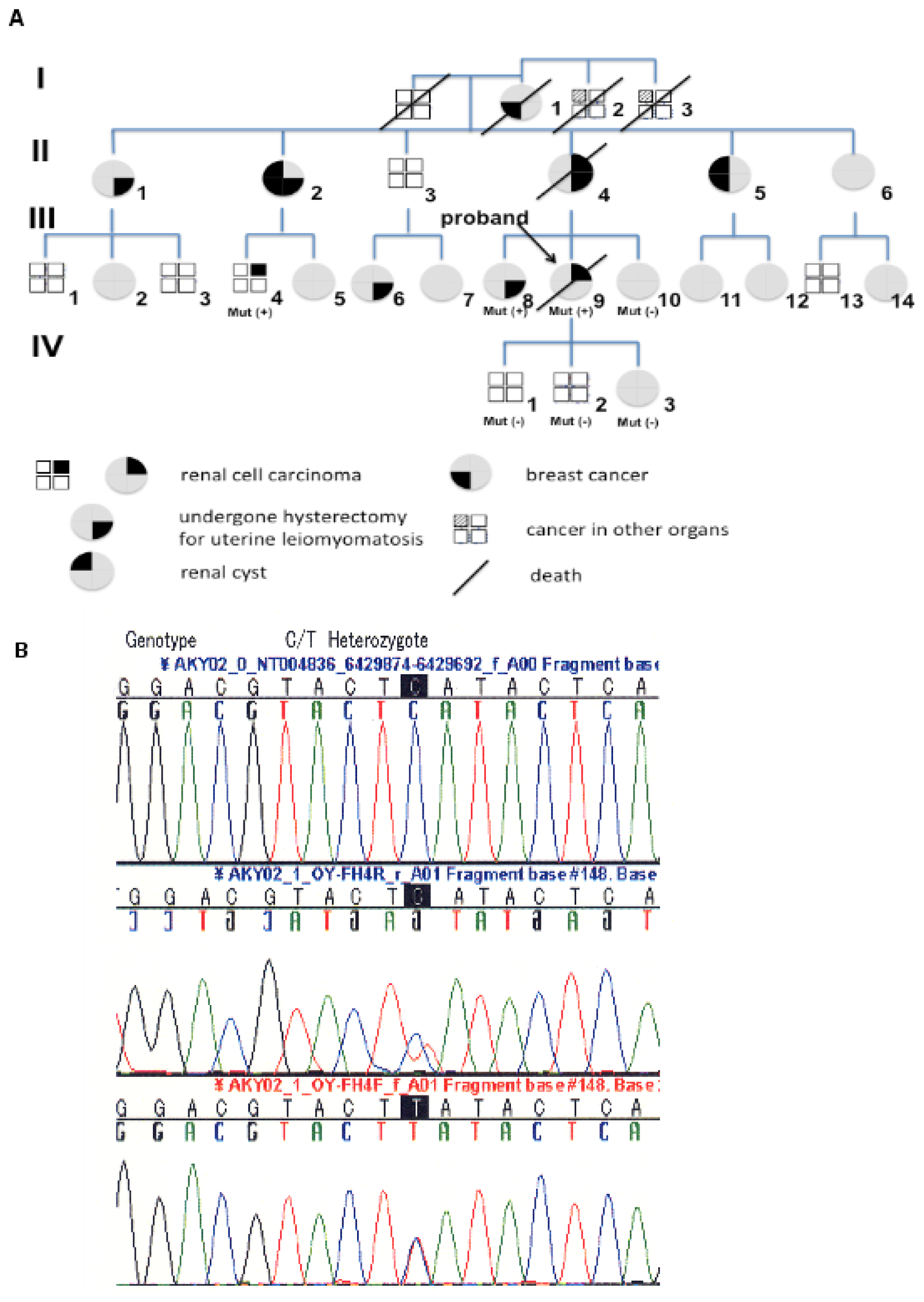
2.1. Pedigree 1
2.2. Pedigree 2
3. Results
3.1. Histological Examination
3.2. Mutation Screening
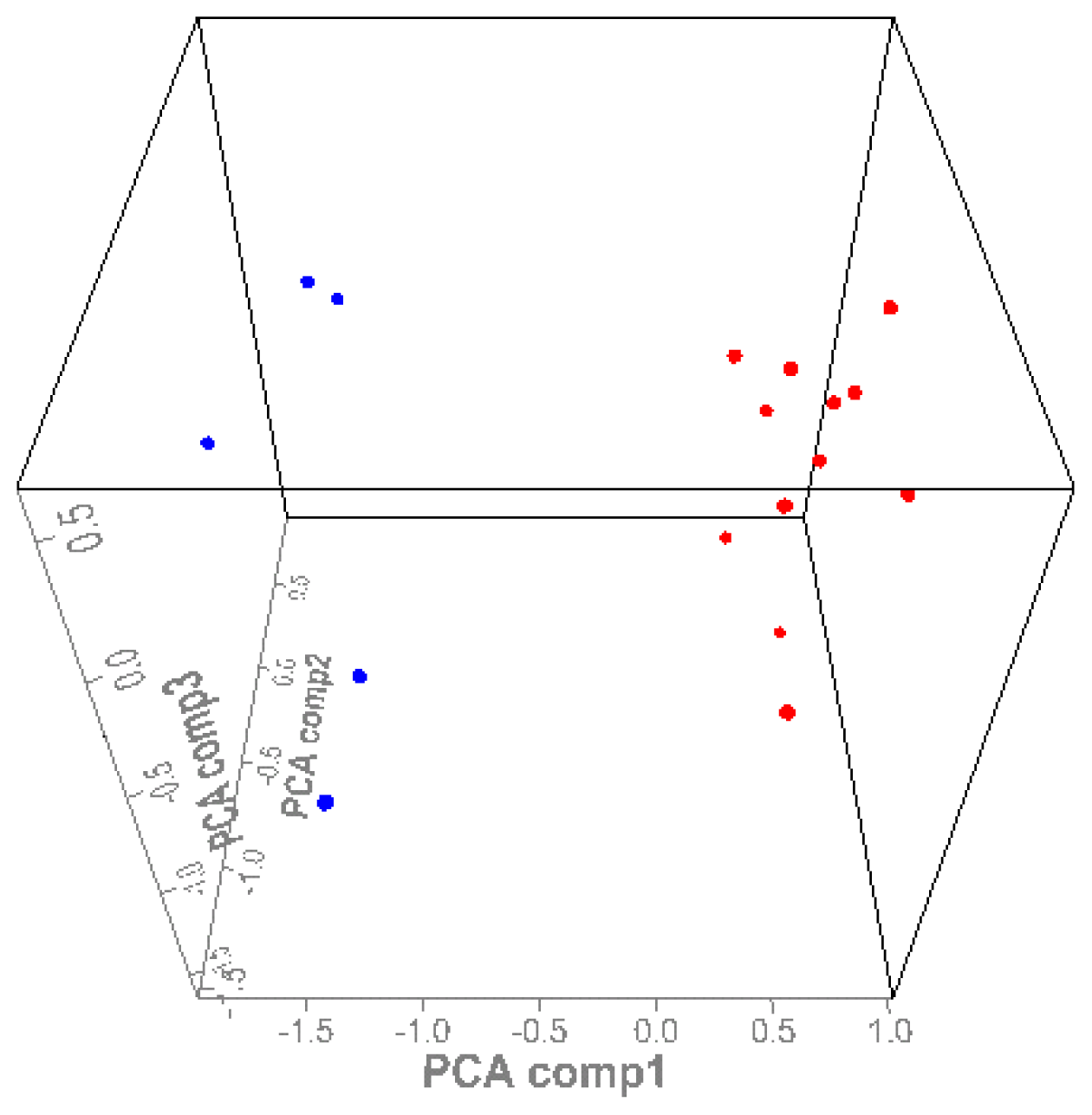
3.3. SELDI-TOF MS
4. Discussion
5. Materials and Methods
5.1. Mutation Screening
5.2. Serum Protein Profiling by SELDI-TOF/MS
5.3. Statistical Analysis
6. Conclusion
- ConsentWritten informed consent was obtained from the patient and the patient’s husband before death representatively of her family members for publication of this case report and accompanying images.
- Authors’ ContributionsTakao Kamai initiated the study, participated in its design and coordination, carried out the study, and drafted the manuscript. Naohisa Tomosugi carried out the mass spectrometry study. Takao Kamai, Hideyuki Abe, and Yasushi Kaji carried out the radiological study. Takao Kamai and Tetsunari Oyama carried out the pathological study. Ken-Ichiro Yoshida participated in the design of the study and helped to draft the manuscript. All authors read and approved the final manuscript.
- Conflict of InterestThe authors declare no conflict of interest.
Abbreviations
| HLRCC | Hereditary Leiomyomatosis and Renal Cell Cancer |
| FH | fumarate hydratase |
References
- Launonen, V.; Vierimaa, O.; Kiuru, M.; Isola, J.; Roth, S.; Pukkala, E.; Sistonen, P.; Herva, R.; Aaltonen, L.A. Inherited susceptibility to uterine leiomyomatosis and renal cell cancer. Proc. Natl. Acad. Sci. USA 2001, 98, 3387–3392. [Google Scholar]
- Kiuru, M.; Launonen, V.; Hietala, M.; Aittomaki, K.; Vierimaa, O.; Solovaara, R.; Arola, J.; Pukkala, E.; Sistonen, P.; Herva, R.; et al. Inherited familial cutaneous leiomyomatosis is a two-hit condition associated with renal cell cancer of characteristic histopathology. Am. J. Pathol 2001, 159, 825–829. [Google Scholar]
- Alam, N.A.; Rowan, A.J.; Wortham, N.C.; Pollard, P.J.; Mitchell, M.; Tyrer, J.P.; Barcley, E.; Calonje, E.; Manek, S.; Adams, S.J.; et al. Genetic and functional analyses of FH mutations in multiple cutaneous and uterin leiomyomatosis, hereditary leiomyomatosis and renal cancer, and fumarate hydratase deficiency. Hum. Mol. Geet 2003, 12, 1241–1252. [Google Scholar]
- Grubb, R.L.; Franks, M.E.; Toro, J.; Middelton, L.; Choyke, L.; Fowler, S.; Torres-Cabala, C.; Glenn, G.M.; Choyke, P.; Merino, M.J. Hereditary leiomyomatosis and renal cell cancer: A syndrome associated with an aggressive form of inherited renal cancer. J. Urol 2007, 177, 2074–2080. [Google Scholar]
- Tomlinson, I.P.; Alam, N.A.; Rowan, A.J.; Barclay, E.; Jaeger, E.E.; Kelsell, D.; Leigh, I.; Gorman, P.; Lamlum, H.; Rahman, S.; et al. Germline mutations in FH predispose to dominantly inherited uterine fibroids, skin leiomyomata and papillary renal cell cancer. Nat. Genet 2002, 30, 406–410. [Google Scholar]
- Alam, N.A.; Olpin, S.; Leigh, I.M. Inherited fumarate hydratase mutations and predisposition to cutaneous leiomyomas, uterine leiomyomas and renal cancer. Br. J. Dermatol 2005, 153, 11–17. [Google Scholar]
- Etzioni, R.; Urban, N.; Ramsey, S.; McIntosh, M.; Schwartz, S.; Reid, B.; Radich, L.; Anderson, G.; Hartwell, L. The case for early detection. Nat. Rev. Cancer 2003, 3, 243–252. [Google Scholar]
- Aebersold, R.; Mann, M. Mass spectrometry-based proteomics. Nature 2003, 422, 198–207. [Google Scholar]
- Linehan, W.M.; Pintooooo, P.A.; Srinivasan, R.; Merino, M.; Choyke, P.; Choyke, L.; Coleman, J.; Toro, J.; Glenn, G.; Vocke, C. Identification of the genes for kidney cancer: Opportunity for disease specific targeted therapeutics. Clin. Cancer Res 2007, 13, 671s–679s. [Google Scholar]
- Kiuru, M.; Lehtonen, R.; Arola, J.; Salovaara, R.; Jarvinen, H.; Aittomaki, K.; Sjoberg, J.; Visakorpi, T.; Knuutila, S.; Isola, J. Few FH mutations in sporadic counterparts of tumor types observed in hereditary leiomyomatosis and renal cell cancer families. Cancer Res 2002, 62, 4554–4557. [Google Scholar]
- Merino, M.J.; Torres-Cabala, C.; Pinto, P.; Linehan, W.M. The morphologic spectrum of kidney tumors in hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome. Am. J. Sur. Pathol 2007, 31, 1578–1585. [Google Scholar]
- Sudarshan, S.; Pinto, P.A.; Neckers, L.; Linehan, W.M. Mechanisms of disease: Hereditary leiomyomatosis and renal cell cancer—A distinct form of hereditary kidney cancer. Nat. Clin. Pract. Urol 2007, 4, 104–110. [Google Scholar]
- Sudarshan, S.; Linehan, W.M.; Neckers, L. HIF and fumarate hydratase in renal cancer. Br. J. Cancer 2007, 96, 403–407. [Google Scholar]
- Pollard, P.J.; Briere, J.J.; Alam, N.A.; Barwell, J.; Barclay, E.; Wortham, N.C.; Hunt, T.; Mitchell, M.; Olpin, S.; Moat, S.J. Accumulation of Krebs cycle intermediates and over-expression of HIF1 in tumours which result from germline FH and SDH mutations. Hum. Mol. Genet 2005, 14, 2231–2239. [Google Scholar]
- Isaacs, J.S.; Jung, Y.J.; Mole, D.R.; Lee, S.; Torres-Cabala, C.; Chung, Y.L.; Merino, M.; Trepel, J.; Zbar, B.; Toro, J. HIF overexpression correlates with biallelic loss of fumarate hydratase in renal cancer: Novel role of fumarate in regulation of HIF stability. Cancer Cell 2005, 8, 143–153. [Google Scholar]
- Mandriota, S.J.; Turner, K.J.; Davies, D.R.; Murray, P.G.; Morgan, N.V.; Sowter, H.M.; Wykoff, C.C.; Maher, E.R.; Harris, A.L.; Ratcliffe, P.J.; et al. HIF activation identifies early lesions in VHL kidneys: Evidence for site-specific tumor suppressor function in the nephron. Cancer Cell 2002, 1, 459–468. [Google Scholar]
- Pollard, P.J.; Spencer-Dene, B.; Shukla, D.; Howarth, K.; Nye, E.; El-Bahrawy, M.; Deheragoda, M.; Joannou, M.; McDonard, S.; Martin, A. Targeted inactivation of Fh1 causes prolirerative renal cyst development and activation of the hypoxia pathway. Cancer Cell 2007, 11, 311–319. [Google Scholar]
- Adam, J.; Hatipoglu, E.; O’Flaherty, L.; Temette, N.; Sahgal, N.; Lockstone, H.; Baban, D.; Nye, E.; Stamp, G.W.; Wolhuter, K.; et al. Renal cyst formation in Fh-1-Deficient mice is independent of the Hif/Phd pathway: Roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell 2011, 20, 524–537. [Google Scholar]
- Yogev, O.; Yogev, O.; Singer, E.; Shaulian, E.; Goldberg, M.; Fox, T.D.; Pines, O. Fumarase: A mitochondrial metabolic enzyme and a cytosolic/nuclear component of NA damage response. PLoS Biol 2010, 8, e1000328. [Google Scholar]
- Frezza, C.; Zheng, L.; Folger, O.; Rajagopalan, K.N.; MacKenzie, E.D.; Jerby, L.; Micaroni, M.; Chaneton, B.; Adam, J.; Hedley, A.; et al. Heam oxygenase is synthetically lethal with the tumour suppressor fumarate hydratase. Nature 2011, 477, 225–228. [Google Scholar]
- Raimundo, N.; Ahtinen, J.; Fumic, K.; Baric, I.; Remes, A.M.; Renkonen, R.; Lapatto, R.; Supmalainen, A. Differential metabolic consequences of fumarate hydratase and respiratory chain defects. Biochim. Biophys. Acta 2008, 1782, 287–294. [Google Scholar]
- Raimundo, N.; Vanharanta, S.; Aaltonen, L.A.; Hovatta, I.; Suomalainen, A. Downregulation of SRF-FOS-JUNB pathway in fumarate hydratase deficiency and in uterine leiomyomas. Oncogene 2009, 28, 1261–1273. [Google Scholar]
- Frezza, C.; Pollard, P.J.; Gottlieb, E. Inborn and acquired metabolic defects in cancer. J. Mol. Med 2011, 89, 213–220. [Google Scholar]
- Raimundo, N.; Baysal, B.E.; Shadel, G.S. Revisiting the TCA cycle: Signaling to tumor formation. Trends Mol. Med 2011, 11, 641–649. [Google Scholar]
- Toro, J.R.; Nickerson, M.L.; Wei, M.H.; Warren, M.B.; Glenn, G.M.; Turner, M.L.; Stewart, L.; Duray, P.; Tourre, O.; Sharma, N.; et al. Mutations in the fumarate hydratase gene cause hereditary leiomyomatosis and renal cell cancer in families in North America. Am. J. Hum. Genet 2003, 73, 95–106. [Google Scholar]
- Petricoin, E.F., III; Ardekani, A.M.; Hitt, B.A.; Levine, P.J.; Fusaro, V.A.; Steinberg, S.M.; Mills, G.B.; Simone, C.; Fishman, D.A.; Kohn, E.C.; et al. Use of proteomic patterns in serum to identify ovarian cancer. Lancet 2002, 359, 572–575. [Google Scholar]
- Isaaq, H.J.; Conrads, T.P.; Prieto, D.A.; Tirumalai, R.; Veenstra, T.D. SELDI-TOF MS for diagnostic proteomics. Anal. Chem 2003, 75, 149A–155A. [Google Scholar]
- Diamandis, E.P.; van der Merwe, D.E. Plasma protein profiling by mass spectrometry for cancer diagnosis: Opportunities and limitations. Clin. Cancer Res 2005, 11, 963–965. [Google Scholar]
- Engwegen, J.Y.; Gast, M.C.; Schellens, J.H.; Beijnen, J.H. Clinical proteomics: Searching for better tumour markers with SELDI-TOF mass spectrometry. Trends Pharmacol. Sci 2006, 27, 251–259. [Google Scholar]
- Kennedy, S. Proteomic profiling from human samples: The body fluid alternative. Toxicol. Lett 2001, 120, 379–384. [Google Scholar]
- Cho, W.C.; Cheng, C.H. Oncoproteomics: Current trends and future perspectives. Expert Rev. Proteomics 2007, 4, 401–410. [Google Scholar]
- Cho, W.C.; Yip, T.T.; Cheng, W.W.; Au, J.S. Serum amyloid A is elevated in the serum of lung cancer patients with poor prognosis. Br. J. Cancer 2010, 102, 1731–1735. [Google Scholar]
- Okamoto, A.; Yamamoto, H.; Amai, A.; Hatakeyama, S.; Iwabuchi, I.; Yoneyama, T.; Hashimoto, Y.; Koie, T.; Kamimura, N.; Mori, K.; et al. Protein profiling of post-prostatic massage urine specimens by surface-enhanced laser desorption/ionization time-of-flight mass spectrometry to discriminate between prostate cancer and benign lesions. Oncol Rep 2009, 21, 73–79. [Google Scholar]

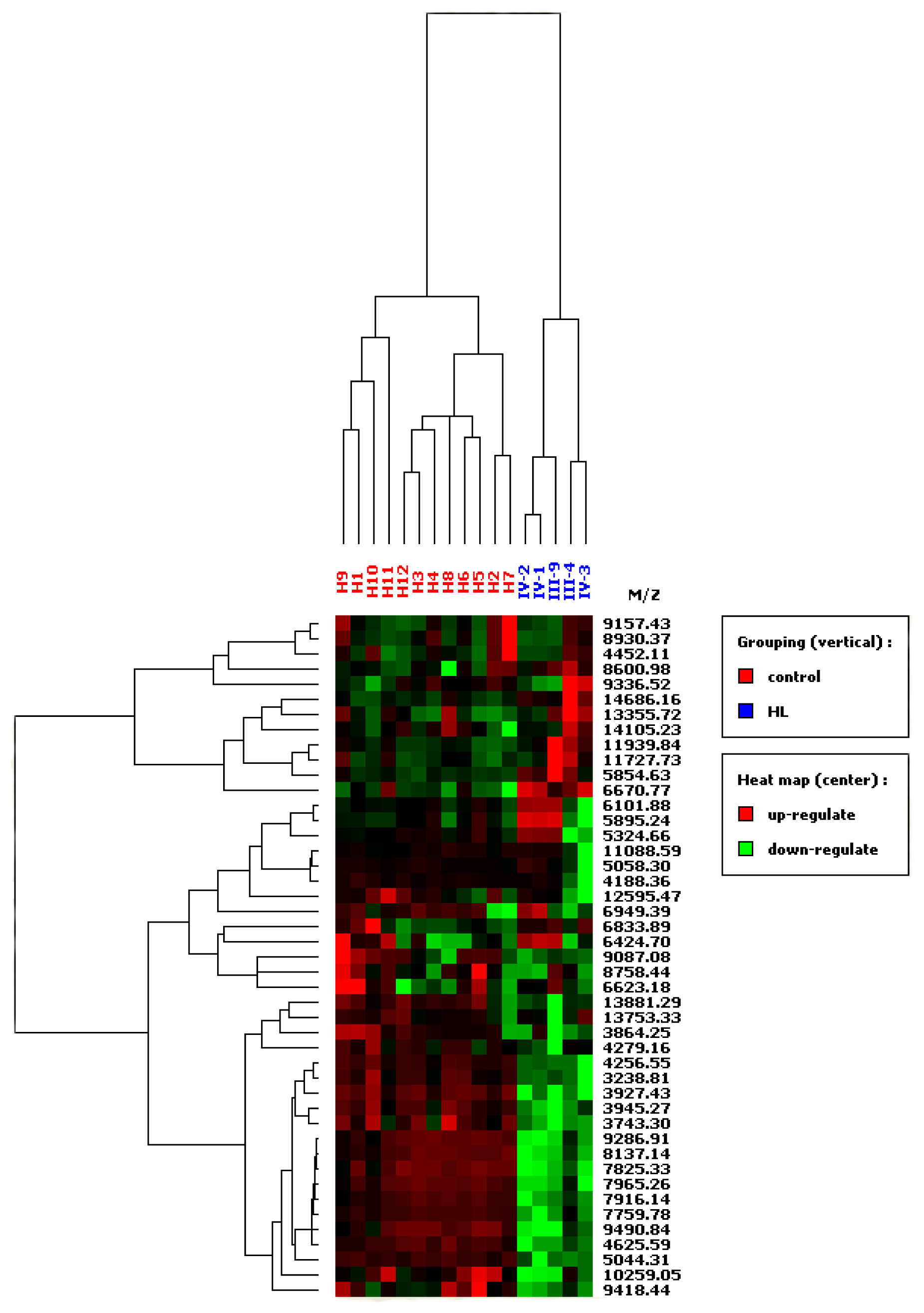

| Peaks in HLRCC | m/z | Healthy control (n = 2) | HLRCC of pedigree 1 (n = 5) | p value | ROC area | ||
|---|---|---|---|---|---|---|---|
| mean ± SD | 95% CI | mean ± SD | 95% CI | ||||
| up-regulated | 5,855 | 4.277 ± 0.520 | 0.330 | 8.517 ± 4.300 | 5.339 | 0.004 | 0.95 |
| 6,671 | 50623 ± 1.266 | 0.805 | 9.637 ± 1.761 | 2.187 | 0.003 | 0.95 | |
| 14,484 | 1.603 ± 0.222 | 0.141 | 2.714 ± 1.219 | 1.514 | 0.003 | 0.95 | |
| 11,940 | 1.840 ± 0.314 | 0.199 | 3.396 ± 1.679 | 2.084 | 0.011 | 0.85 | |
| down-regulated | 3239 | 6.589 ± 4.529 | 2.877 | 0.843 ± 0.434 | 0.538 | 0.002 | 0.98 |
| 3,743 | 3.785 ± 2.778 | 1.765 | 0.429 ± 0.184 | 0.229 | 0.002 | 0.98 | |
| 3,927 | 37.245 ± 26.749 | 16.996 | 0.175 ± 0.058 | 0.177 | 0.002 | 0.98 | |
| 3,945 | 15.475 ± 11.430 | 7.262 | 0.847 ± 0.641 | 0.859 | 0.002 | 0.98 | |
| 4,257 | 21.837 ± 12.996 | 8.257 | 0.297 ±0.320 | 0.398 | 0.002 | 0.98 | |
| 4,626 | 18.029 ± 3.370 | 2.141 | 3.620 ± 1.908 | 2.369 | 0.002 | 0.98 | |
| 5,044 | 20.584 ± 5.844 | 3.713 | 0.360 ± 0.338 | 0.419 | 0.002 | 0.98 | |
| 7,760 | 85.036 ± 21.119 | 13.418 | 6.305 ± 10.962 | 13.611 | 0.002 | 0.98 | |
| 7,825 | 4.868 ± 1.212 | 0.771 | 0.717 ± 0.432 | 0.536 | 0.002 | 0.98 | |
| 7,916 | 4.699 ±1.146 | 0.728 | 0.621 ± 0.511 | 0.635 | 0.002 | 0.98 | |
| 7,965 | 7.789 ± 1.861 | 1.182 | 1.002 ± 1.138 | 1.414 | 0.002 | 0.98 | |
| 8,137 | 17.275 ± 4.321 | 2.745 | 2.071 ± 1.848 | 2.295 | 0.002 | 0.98 | |
| 9,287 | 74.914 ± 17.311 | 10.999 | 12.774 ± 10.948 | 13.594 | 0.002 | 0.98 | |
| 9,491 | 10.193 ± 1.988 | 1.263 | 3.543 ± 1.764 | 2.190 | 0.002 | 0.98 | |
| 9,418 | 7.693 ± 2.733 | 1.762 | 4.088 ± 0.898 | 1.116 | 0.003 | 0.93 | |
| 13,881 | 5.568 ± 1.154 | 0.733 | 3.653 ± 0.873 | 1.085 | 0.006 | 0.93 | |
| 4,188 | 7.414 ± 2.517 | 1.599 | 3.472 ± 3.244 | 4.028 | 0.020 | 0.88 | |
| 9,087 | 9.829 ± 4.014 | 2.550 | 5.559 ± 0.827 | 1.027 | 0.015 | 0.88 | |
| 10,259 | 5.537 ± 3.738 | 2.375 | 1.370 ± 1.296 | 1.610 | 0.011 | 0.88 | |
| 3,864 | 11.781 ± 3.558 | 2.261 | 6.636 ± 2.746 | 3.410 | 0.035 | 0.83 | |
| 4,279 | 7.455 ± 4.752 | 3.019 | 2.944 ± 1.901 | 2.360 | 0.020 | 0.83 | |
| 13,881 | 5.568 ± 1.154 | 0.733 | 3.653 ± 0.873 | 1.085 | 0.006 | 0.93 | |
| 4,188 | 7.414 ± 2.517 | 1.599 | 3.472 ± 3.244 | 4.028 | 0.020 | 0.88 | |
| 9,087 | 9.829 ± 4.014 | 2.550 | 5.559 ± 0.827 | 1.027 | 0.015 | 0.88 | |
| 10,259 | 5.537 ± 3.738 | 2.375 | 1.370 ± 1.296 | 1.610 | 0.011 | 0.88 | |
| 3,864 | 11.781 ± 3.558 | 2.261 | 6.636 ± 2.746 | 3.410 | 0.035 | 0.83 | |
| 4,279 | 7.455 ± 4.752 | 3.019 | 2.944 ± 1.901 | 2.360 | 0.020 | 0.83 | |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kamai, T.; Tomosugi, N.; Abe, H.; Kaji, Y.; Oyama, T.; Yoshida, K.-I. Protein Profiling of Blood Samples from Patients with Hereditary Leiomyomatosis and Renal Cell Cancer by Surface-Enhanced Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Int. J. Mol. Sci. 2012, 13, 14518-14532. https://doi.org/10.3390/ijms131114518
Kamai T, Tomosugi N, Abe H, Kaji Y, Oyama T, Yoshida K-I. Protein Profiling of Blood Samples from Patients with Hereditary Leiomyomatosis and Renal Cell Cancer by Surface-Enhanced Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. International Journal of Molecular Sciences. 2012; 13(11):14518-14532. https://doi.org/10.3390/ijms131114518
Chicago/Turabian StyleKamai, Takao, Naohisa Tomosugi, Hideyuki Abe, Yasushi Kaji, Tetsunari Oyama, and Ken-Ichiro Yoshida. 2012. "Protein Profiling of Blood Samples from Patients with Hereditary Leiomyomatosis and Renal Cell Cancer by Surface-Enhanced Laser Desorption/Ionization Time-of-Flight Mass Spectrometry" International Journal of Molecular Sciences 13, no. 11: 14518-14532. https://doi.org/10.3390/ijms131114518




